-
What would be the IUPA C name and symbol for the element
with atomic number 120?
View Answer play_arrow
-
How would you justify the presence of 18 elements in the 5th
period of the periodic table?
View Answer play_arrow
-
The elements Z = 117 and 120 have not yet been discovered.
In which family/group would you place these elements and also give the
electronic configuration in each case.
View Answer play_arrow
-
Considering the
atomic number and position in the periodic table arrange the following elements
in the increasing order of metallic character:
\[Si,Be,Mg,Na,P\]
View Answer play_arrow
-
Which of the
following species will have the largest and smallest size?
\[Mg,M{{g}^{2+}},Al,A{{l}^{3+}}\]
View Answer play_arrow
-
The first ionisation enthalpy (\[{{\Delta }_{i}}H\]) values
of third period elements, Na, Mg and Si are respectively 496, 737 and \[786KJ\,mo{{l}^{-1}}\].
Predict whether the first (\[{{\Delta }_{i}}H\]) value for \[Al\] will be more
close to 575 or \[760KJmo{{l}^{-1}}\] ? Justify your answer.
View Answer play_arrow
-
Which of the following will
have the most negative electron gain enthalpy and which the least negative?
\[P,S,Cl,F\]
Explain your answer.
View Answer play_arrow
-
Predict the formulae
of compounds which might be formed by the following pairs of elements:
(a) silicon
and bromine
(b) aluminium and
sulphur
View Answer play_arrow
-
Are the oxidation state and covalency of \[Al\] in \[{{\left[
AlCl{{({{H}_{2}}O)}_{5}} \right]}^{2+}}\] same?
View Answer play_arrow
-
Show by a chemical reaction with water that \[N{{a}_{2}}O\]
is a basic oxide and is an
acidic oxide.
View Answer play_arrow
-
What is the basic theme of organisation in the periodic table?
View Answer play_arrow
-
Which important property did Mendeleev use to classify the
elements in the periodic table and did he stick to that?
View Answer play_arrow
-
What is the basic difference in approach between Mendeleev's
periodic law and the modem periodic law?
View Answer play_arrow
-
On the basis of quantum numbers, justify that sixth period
of the periodic table should have 32 elements.
View Answer play_arrow
-
In terms of period and group where would you locate the element with Z = 114?
View Answer play_arrow
-
Write the atomic number of the element present in the third period and seventeenth group of the periodic table.
View Answer play_arrow
-
Which element do you think would have been named by: (i) Lawrence Berkeley laboratory (ii) Seaborg's group?
View Answer play_arrow
-
Why do elements in the same group have similar physical and chemical properties?
View Answer play_arrow
-
What does atomic radius or ionic radius really mean to you?
View Answer play_arrow
-
How do atomic radius vary in a
period and in a group?
How do you explain the variation?
View Answer play_arrow
-
What do you
understand by isoelectronic species? Name a species that will be isoelectronic
with each of the following atoms or ions:
(i) \[{{F}^{-}}\]
(ii) \[Ar\]
(iii) \[M{{g}^{2+}}\]
(iv) \[R{{b}^{+}}\]
View Answer play_arrow
-
Consider the fallowing species:
\[{{N}^{3-}},{{O}^{2-}},{{F}^{-}},N{{a}^{+}},M{{g}^{2+}}\]
and \[A{{l}^{3+}}\]
(a) What is common
in them?
(b) Arrange them in order of increasing ionic radii.
View Answer play_arrow
-
Explain why cations are smaller and onions larger in radii
than their parent atoms?
View Answer play_arrow
-
What is significance of terms- 'isolated gaseous atom and
'ground state' while defining the ionisation enthalpy and electron gain
enthalpy?
View Answer play_arrow
-
The energy of an electron in the ground state of the
hydrogen atom is\[-2.18\times {{10}^{-18}}J\]. Calculate the ionisation energy
(enthalpy) of atomic hydrogen in J \[mo{{l}^{-1}}.\]
View Answer play_arrow
-
Among the second period
elements, the actual ionisation energies are in the order
\[Li\text{
}
View Answer play_arrow
-
How would you explain the fact
that the first ionisation enthalpy of sodium is lower than
that of magnesium but its second ionisation enthalpy is higher than that of
magnesium?
View Answer play_arrow
-
What are the factors due to which ionisation enthalpy of the
main group elements tends to decrease down the group?
View Answer play_arrow
-
The first ionisation
enthalpy values (in \[KJ\,\,mo{{l}^{-1}}\]) of group 13 elements are :
\[\underset{801}{\mathop{B}}\,\,\,\,\,\,\,\,\,\,\underset{577}{\mathop{Al}}\,\,\,\,\,\,\,\,\,\underset{579}{\mathop{Ga}}\,\,\,\,\,\,\,\,\,\underset{558}{\mathop{\ln
}}\,\,\,\,\,\,\underset{589}{\mathop{Tl}}\,\]
How will you explain this deviation from the general trend?
View Answer play_arrow
-
Which of the following pairs of
elements would have a more negative electron gain enthalpy? Explain.
(i) O or F (ii) F or Cl
View Answer play_arrow
-
Would you expect the second
electron gain enthalpy of O as positive, more negative or less negative than
the first?
Justify your answer.
View Answer play_arrow
-
What is the basic difference between the terms electron gain
enthalpy and electronegativity?
View Answer play_arrow
-
How would you react to the statement that the electronegativity
of N on Pauling scale is 3.0 in all the nitrogen compounds?
View Answer play_arrow
-
Describe the theory associated
with the radius of an atom as it :
(a) gains an electron
(b) loses an electron
View Answer play_arrow
-
Would you expect the ionisation
enthalpies of two isotopes of the same element to be same or different?
Justify your answer.
View Answer play_arrow
-
What are the major differences between metals and non-metals?
View Answer play_arrow
-
Use the periodic
table to answer the following questions:
(a) Identify
an element with five electrons in the outer subshell.
(b) Identify
the element that would tend to lose two electrons.
(c) Identify the
element that would tend to gain two electrons.
(d) Identify the group having metal, non-metal, liquid as
well as gas at room temperature.
View Answer play_arrow
-
The increasing order of reactivity among group 1 elements is
\[Li\text{ }
View Answer play_arrow
-
Write the general outer
electronic configuration of s-, p-,
\[d\]- and \[f\]- block elements.
View Answer play_arrow
-
Assign the position of the element having outer electronic
configuration (i) \[n{{s}^{2}}n{{p}^{4}}\] for \[n=3\] (ii) \[(n-1){{d}^{2}}n{{s}^{2}}\]
for \[n=4\] and (iii) \[(n-2){{f}^{7}}(n-1){{d}^{1}}n{{s}^{2}}\] for \[n=6,\] in
the periodic table.
View Answer play_arrow
-
The first \[({{\Delta
}_{i}}{{H}_{1}})\] and the second \[({{\Delta }_{i}}{{H}_{2}})\]ionisation
enthalpies\[(in\,kJ\,mo{{l}^{-1}})\] and the \[({{\Delta }_{eg}}H)\] electron
gain enthalpy of a few elements are given below:
Elements
\[{{\Delta
}_{\mathbf{i}}}{{\mathbf{H}}_{\mathbf{1}}}\]
\[{{\Delta
}_{\mathbf{i}}}{{\mathbf{H}}_{\mathbf{2}}}\]
\[{{\Delta
}_{eg}}\mathbf{H}\]
1.
520
7300
60
2.
419
3051
48
3.
1681
3374
328
4.
1008
1846
295
5.
2372
5251
+ 48
6.
738
1451
40
Which of the
above elements is likely to be:
(a) the
least reactive element.
(b) the most
reactive metal.
(c) the most
reactive non-metal.
(d) the
least reactive non-metal.
(e) the metal which
conform a stable binary halide of the formula \[M{{X}_{2}}\] (X = halogen).
(f) the metal which can form a predominantly stable covalent
halide of the formula MX (X = halogen).
View Answer play_arrow
-
Predict the formulae
of the stable binary compounds that would be formed by the combination of the following
pairs of elements:
(a) Lithium
and oxygen
(b) Magnesium
and nitrogen
(c) Aluminium
and iodine
(d) Silicon
and oxygen
(e) Phosphorus and
fluorine
(f) Element 71 and fluorine
View Answer play_arrow
-
In the modem
periodic table, the period indicates the value of :
(a) atomic
mass
(b) atomic
number
(c) valence
principal quantum number
(d) azimuthal quantum number
View Answer play_arrow
-
Which of the following
statements related to the modem periodic table is incorrect?
(a) The
block indicates value of azimuthal quantum number \[(l)\] for the last subshell
that received electrons in building up the electronic configuration.
(b) Each
block contains a number of columns equal to the number of electrons that can
occupy that subshell.
(c) The \[p\]-block
has 6 columns, because a maximum of 6 electrons can occupy all the orbitals in
a p-shell.
(d) The \[d\]-block has 8 columns, because a maximum of 8 electrons
can occupy all the orbitals in a d-subshell.
View Answer play_arrow
-
Anything that
influences the valence electrons mil affect the chemistry of the element. Which
one of the following factors does not affect the valence shell?
(a) Valence
principal quantum number
(b) Nuclear
charge
(c) Nuclear mass
(d) Number of core electrons
View Answer play_arrow
-
The size of
isoelectronic species \[{{F}^{-}}\], Ne and \[N{{a}^{+}}\] is affected by :
(a) Nuclear
charge (z)
(b) Valence
principal quantum number (n)
(c) Electron-electron
interaction in the outer orbitals
(d) None of the factors because their size is same
View Answer play_arrow
-
Which one of the
following statements is incorrect in relation to ionisation enthalpy?
(a) Ionisation
enthalpy increases for each successive electron.
(b) The
greatest increase in ionisation enthalpy is experienced on removal of electron
from core noble gas configuration.
(c) End of valence
electrons is worked by a big jump in ionisation enthalpy.
(d) Removal of electron from orbitals bearing lower n value
is easier than orbital having higher n value.
View Answer play_arrow
-
Considering the
elements,
\[B,Al,Mg\]and K
the correct
order of their metallic character is:
(a) B >
Al > Mg > K (b) Al > Mg > B > K
(c) Mg > Al >
K > B (d) K > Mg > Al > B
View Answer play_arrow
-
Considering the
elements,
B, C, N, F and Si
the correct
order of their non-metallic character is:
(a) B > C > Si
> N > F (b) Si > C > B > N> F
(c) F > N > C > B > Si (d) F > N >
C > Si > B
View Answer play_arrow
-
Considering the
elements,
F, Cl, O and N
the correct
order of their chemical reactivity in terms of oxidising property is:
(a) F >
Cl > O > N (b) F> O > Cl > N
(c) Cl > F > O
> N (d) O > F > N > Cl
View Answer play_arrow
-
question_answer51)
In the following Questions only one
option is correct.
Consider the isoelectronic
species, \[N{{a}^{+}},M{{g}^{2+}},{{F}^{-}}\] and \[{{O}^{2-}}\]. The correct
order of increasing length of their radii is :
(a)\[{{F}^{-}}<{{O}^{2-}}<M{{g}^{2+}}<N{{a}^{+}}\]
(b) \[M{{g}^{2+}}<N{{a}^{+}}<{{F}^{-}}<{{O}^{2-}}\]
(c) \[{{O}^{2-}}<{{F}^{-}}<N{{a}^{+}}<M{{g}^{2+}}\] (d)
\[{{O}^{2-}}<{{F}^{-}}<M{{g}^{2+}}<N{{a}^{+}}\]
View Answer play_arrow
-
question_answer52)
Which of the following is not an
actinoid?
(a) Curium, (Z = 96) (b)
Californium, (Z = 98)
(c) Uranium, (Z = 92) (d)
Terbium, (Z = 65)
View Answer play_arrow
-
question_answer53)
The order of screening effect of
electrons of s, p, d and \[f-\]orbitals of a given shell of an atom on its
outer shell electrons is :
(a) \[s>p>d>f\] (b) \[f>d>p>s\] (c)
\[p<d<s>f\] (d) \[f>p>s>d\]
View Answer play_arrow
-
question_answer54)
The first ionisation enthalpies
of Na, Mg, Al and Si are in the order:
(a) \[Na<Mg>Al<Si\] (b)\[Na>Mg>Al>Si\] (c)
\[Na<Mg<Al<Si~~\] (d)\[Na>Mg>Al<Si\]
View Answer play_arrow
-
question_answer55)
The electronic configuration of
gadolinium (Z = 64) is :
(a) \[\left[ Xe \right]\text{
}4{{f}^{3}}5{{d}^{5}}\text{ }6{{s}^{2}}\] (b) \[\left[ Xe
\right]\text{ }4{{f}^{7}}5{{d}^{2}}6{{s}^{1}}\] (c) \[[Xe]4{{f}^{7}}5{{d}^{1}}6{{s}^{2}}\]
(d) \[[Xe]4{{f}^{8}}5{{d}^{6}}6{{s}^{2}}\]
View Answer play_arrow
-
question_answer56)
The statement that is not correct for
periodic classification of elements is :
(a)
The properties of elements are periodic function of their atomic numbers.
(b) Non-metallic elements are less in
number than metallic elements.
(c) For transition elements, the
\[3d\]-orbitals are filled with electrons after \[3p\]-orbitals and before \[4s\]-orbitals.
(d) The first ionisation
enthalpies of elements generally increase with increase in atomic number as we
go along a period.
View Answer play_arrow
-
question_answer57)
Among halogens, the correct
order of amount of energy released in electron gain (electron gain enthalpy)
is:
(a) \[F>Cl>Br>I\] (b)
\[F<Cl<Br<I\] (c) \[F<Cl>Br>I\] (d)
\[F<Cl<Br>I\]
View Answer play_arrow
-
question_answer58)
The period number in the long
form of the periodic table is equal to :
(a) magnetic quantum number of
any element of the period.
(b) atomic number of any element
of the period.
(c) maximum principal quantum
number of any element of the period.
(d) maximum azimuthal quantum
number of any element of the period.
View Answer play_arrow
-
question_answer59)
The elements in which electrons
are progressively filled in \[4f\]-orbitals are called :
(a) actinoids (b)
transition elements
(c) lanthanides (d)
halogens
View Answer play_arrow
-
question_answer60)
Which of the following is the
correct order of size of the given species?
(a) \[I>{{I}^{-}}>{{I}^{+}}\]
(b) \[{{I}^{+}}>{{I}^{-}}>I\] (c)
\[I>{{I}^{+}}>{{I}^{-}}\] (d) \[{{I}^{-}}>I>{{I}^{+}}\]
View Answer play_arrow
-
question_answer61)
The formation of the oxide ion, \[{{O}^{2-}}\],
from oxygen atom requires first an exothermic and then an endothermic step as shown
below :
\[O(g)+e\to
{{O}^{-}}(g);\,\,\,\,\,\,\,\,\,\Delta {{H}^{O-}}=-141\,kJ\,mo{{l}^{-1}}\]
\[{{O}^{-}}(g)+e\to
{{O}^{2-}}(g);\,\,\,\,\,\,\,\,\,\Delta {{H}^{O-}}=+780\,kJ\,mo{{l}^{-1}}\]
Thus, process of formation of \[{{O}^{2-}}\]
in gas phase is unfavourable even though \[{{O}^{2-}}\] is isoelectronic with
neon. It is due to the fact that:
(a) oxygen is more
electronegative
(b) addition of an electron in
oxygen results in larger size of the ion
(c) electron repulsion outweighs
the stability gained by achieving noble gas configuration
(d) \[{{O}^{-}}\] ion has
comparatively smaller size than oxygen atom
View Answer play_arrow
-
question_answer62)
Comprehension given below is
followed by some multiple questions. Each question has one correct option.
Choose the correct option:
In the modem periodic table, elements
are arranged in order of increasing atomic numbers which is related to the electronic
configuration. Depending upon the type of orbitals receiving the last electron,
the elements in the periodic table have been divided into four blocks, viz., \[s,\text{
}p,\text{ }d\]and\[f\], the modem periodic table consists 7 periods and 18
groups. Each period begins with filling of a new energy shell. In accordance
with the Aufbau principle, the seven periods (1 to 7) have 2, 8, 8, 18, 18, 32
and 32 elements respectively. The seventh period is still incomplete. To avoid
the periodic table being too long, the two series of f-block elements, called
lanthanoids and actinoids are placed at the bottom of the main body of the periodic
table.
[A] The element with atomic number 57
belongs to:
(a) s-block (b)
p-block (c) d-block (d) f-block
[B] The last element of the p-block in 6th
period is represented by the outermost electronic configuration:
(a) \[7{{s}^{2}}7{{p}^{6}}\]
(b) \[5{{f}^{14}}6{{d}^{10}}7{{s}^{2}}7{{p}^{6}}\] (c)
\[4{{f}^{14}}5{{d}^{10}}6{{s}^{2}}6{{p}^{6}}\] (d) \[4{{f}^{14}}5{{d}^{10}}6{{s}^{2}}6{{p}^{4}}\]
[C] Which of the elements whose atomic
numbers are given below, cannot be accommodated in the present set up of the
long form of the periodic table?
(a) 107 (b)
118 (c) 126 (d)
102
[D] The electronic configuration of the
element which is just above the element with atomic number 43 in the same group
is:
(a) \[1{{s}^{2}}2{{s}^{2}}2{{p}^{6}}3{{s}^{2}}3{{p}^{6}}3{{d}^{5}}4{{s}^{2}}\] (b)
\[1{{s}^{2}}2{{s}^{2}}2{{p}^{6}}3{{s}^{2}}3{{p}^{6}}3{{d}^{5}}4{{s}^{2}}4{{p}^{6}}\]
(c) \[1{{s}^{2}}2{{s}^{2}}2{{p}^{6}}3{{s}^{2}}3{{p}^{6}}3{{d}^{5}}4{{s}^{2}}\] (d)
\[1{{s}^{2}}2{{s}^{2}}2{{p}^{6}}3{{s}^{2}}3{{p}^{6}}3{{d}^{7}}4{{s}^{2}}\]
[E] The elements with atomic numbers 35, 53
and 85 are all:
(a) noble gases (b)
halogens (c) heavy metals (d) light metals
View Answer play_arrow
-
question_answer63)
Electronic configuration of four
elements A, B, C and D are given below :
A. \[1{{s}^{2}}2{{s}^{2}}2{{p}^{6}}\]
B. \[1{{s}^{2}}2{{s}^{2}}2{{p}^{4}}\] C. \[1{{s}^{2}}2{{s}^{2}}2{{p}^{6}}3{{s}^{1}}\]
D. \[1{{s}^{2}}2{{s}^{2}}2{{p}^{5}}\]
Which of the following is the
correct order of increasing tendency to gain electron?
(a) A < C < B <
D (b) A < B < C < D (c) D
< B < C < A (d) D < A < B < C
View Answer play_arrow
-
question_answer64)
In the following questions two or
more options may be correct.
Which of the following elements can show
covalency greater than 4?
(a) Be (b)
P (c) S (d)
B
View Answer play_arrow
-
question_answer65)
Those elements impart colour to
the flame on heating in it, the atoms of which require low energy for
ionisation (i.e., absorb energy in the visible region of spectrum). The elements
of which of the following groups will impart colour to the flame?
(a) 2 (b)
13 (c) 1 (d)
17
View Answer play_arrow
-
question_answer66)
Which of the following sequences
contain atomic numbers of only representative elements?
(a) 3, 33, 53, 87 (b)
2, 10, 22, 36 (c) 7, 17, 25, 37, 48 (d) 9, 35, 51,
88
View Answer play_arrow
-
question_answer67)
Which of the following elements
will gain one electron more readily in comparison to other elements of their group?
(a) S(g) (b)
Na(g) (c) O(g) (d)
Cl(g)
View Answer play_arrow
-
question_answer68)
Which of the following
statements are correct?
(a) Helium has the highest first
ionisation enthalpy in the periodic table
(b) Chlorine has less negative
electron gain enthalpy than fluorine
(c) Mercury and bromine are
liquids at room temperature
(d) In any period, atomic radius
of alkali metal is the highest
View Answer play_arrow
-
question_answer69)
Which of the following sets
contain only isoelectronic ions?
(a) \[\text{Z}{{\text{n}}^{\text{2+}}}\text{,C}{{\text{a}}^{\text{2+}}}\text{,G}{{\text{a}}^{\text{3+}}}\text{,A}{{\text{l}}^{\text{3+}}}\] (b)
\[{{\text{K}}^{\text{+}}}\text{,C}{{\text{a}}^{\text{2+}}}\text{,S}{{\text{c}}^{\text{3+}}}\text{,C}{{\text{l}}^{\text{-}}}\]
(c) \[{{P}^{3-}},{{S}^{2-}},C{{l}^{-}},{{K}^{+}}\] (d)
\[T{{i}^{4+}},Ar,C{{r}^{3+}},{{V}^{5+}}\]
View Answer play_arrow
-
question_answer70)
In which of the following
options, order of arrangement does not agree the variation of property
indicated against it?
(a) \[A{{l}^{3+}}<M{{g}^{2+}}<N{{a}^{+}}<{{F}^{-}}\]
(increasing ionic size)
(b) \[B\text{ }<\text{
}C\text{ }<\text{ }N\text{ }<\text{ O}\]
(increasing first ionisation
enthalpy)
(c) \[I\text{ }<\text{
}Br\text{ }<\text{ }Cl\text{ }<\text{ }F\]
(increasing electron gain
enthalpy)
(d) \[I\text{ }<\text{
}Br\text{ }<\text{ }Cl\text{ }<\text{ }F\]
(increasing metallic radius)
View Answer play_arrow
-
question_answer71)
Which of the following have no
unit?
(a) Electronegativity (b)
Electron gain enthalpy
(c) Ionisation enthalpy (d)
Metallic character
View Answer play_arrow
-
question_answer72)
Ionic radii vary in :
(a) inverse proportion to the
effective nuclear charge
(b) inverse proportion to the
square of effective nuclear charge
(c) direct proportion to the
screening effect
(d) direct proportion to the
square of screening effect
View Answer play_arrow
-
question_answer73)
An element belongs to 3rd period
and group 13 of the periodic table. Which of the following properties will be shown
by the element?
(a) Good conductor of
electricity (b) Liquid, metallic
(c) Solid, metallic (d)
Solid, non-metallic
View Answer play_arrow
-
question_answer74)
Explain why the electron gain enthalpy
of fluorine is less negative than that of chlorine?
View Answer play_arrow
-
question_answer75)
All transition elements are d-block
elements, but all the d-block elements are not transition elements. Explain.
View Answer play_arrow
-
question_answer76)
Identify the group and valency
of the element having atomic number 119. Also predict the outermost electronic configuration
and write the general formula of its oxide.
View Answer play_arrow
-
question_answer77)
Ionisation enthalpies of
elements of second period are given below :
Ionisation enthalpy (kcal\[mo{{l}^{-1}}\]):
520, 899, 801, 1086, 1402, 1314, 1681, 2080
Match the correct enthalpy with
the elements and complete the graph given in the following figure. Also symbols
of elements with their atomic numbers.
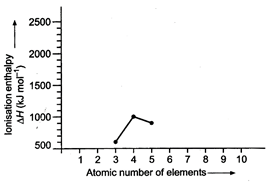
View Answer play_arrow
-
question_answer78)
Among the elements B, Al, C and
Si:
(i) Which element has the
highest first ionisation enthalpy?
(ii) Which element has the most
metallic character? Justify your answer in each case.
View Answer play_arrow
-
question_answer79)
Write four characteristics of p-block
elements.
View Answer play_arrow
-
question_answer80)
Choose the correct order of
atomic radii of fluorine neon (in pm) out of the options given below and
justify your answer.
(i) 72, 160 (ii)
160, 160
(iii) 72, 72 (iv)
160, 72
View Answer play_arrow
-
question_answer81)
Illustrate by taking examples of
transition elements non-transition elements that oxidation states are largely based
on electronic configuration.
View Answer play_arrow
-
question_answer82)
Nitrogen has positive electron
gain enthalpy whereas oxygen has negative. However, oxygen has lower ionisation
enthalpy than nitrogen. Explain.
View Answer play_arrow
-
question_answer83)
First member of each group of
representative elements (i.e., s and p-block elements) shows anomalous
behaviour. Illustrate with two examples.
View Answer play_arrow
-
question_answer84)
p-block elements form acidic,
basic and amphoteric oxides. Explain each property by giving two examples also
write the reactions of these oxides with water.
View Answer play_arrow
-
question_answer85)
How would you explain the fact
that first ionisation enthalpy of sodium is lower than that of magnesium but
its second ionisation enthalpy is higher than that of magnesium?
View Answer play_arrow
-
question_answer86)
What do you understand by
exothermic reaction endothermic reaction? Give one example of each type.
View Answer play_arrow
-
question_answer87)
Arrange the elements N, P, O and
S in the order of:
(i) increasing first ionisation
enthalpy.
(ii) increasing non-metallic
character
View Answer play_arrow
-
question_answer88)
Explain from general trend by
using the following figure:
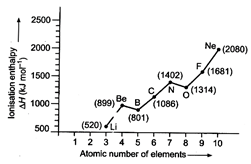
View Answer play_arrow
-
question_answer89)
Explain the following :
(a) Electronegativity of
elements increase on moving from left to right in the periodic table.
(b) Ionisation enthalpy decrease in a
group from top to bottom.
View Answer play_arrow
-
question_answer90)
How does the metallic and non-metallic
character vary on moving from left to right in a period?
View Answer play_arrow
-
question_answer91)
The radius of \[N{{a}^{+}}\]
cation is less than that of Na atom. Give reason
View Answer play_arrow
-
question_answer92)
Among alkali metals which element do you
expect to be least electronegative and why?
View Answer play_arrow
-
question_answer93)
Match the correct radius with the
element.
Element Atomic
radius (pm)
(i) Be (a)
74
(i) C (b)
88
(iii) O (c) 111
(iv) b (d) 77
(v) N (e)
66
View Answer play_arrow
-
question_answer94)
Match the correct ionisation
enthalpies and electron gain enthalpies of the following elements :
|
Elements
|
\[\Delta {{\mathbf{H}}_{\mathbf{1}}}\]
|
\[\Delta {{\mathbf{H}}_{\mathbf{2}}}\]
|
\[{{\Delta }_{eg}}\mathbf{H}\]
|
|
(i) Most
reactive non-metal
|
(a) 419
|
3051
|
-48
|
|
(ii) Most
reactive metal
|
(b) 1681
|
3374
|
-328
|
|
(iii) Least
reactive element
|
(c) 738
|
1451
|
-40
|
|
(iv) Metal
forming binary halide
|
(d) 2372
|
5251
|
+48
|
View Answer play_arrow
-
question_answer95)
Electronic configuration of some
elements is given in column I and their electron gain enthalpies are given in column
II. Match the electronic configuration with electron gain enthalpy.
|
Column l
Electronic
configuration
|
Column ll
Electron gain enthalpy/ \[\mathbf{kJ}\,\mathbf{mo}{{\mathbf{l}}^{\mathbf{-1}}}\]
|
|
(1) \[1{{s}^{2}}2{{s}^{2}}2{{p}^{6}}\]
|
(a) ? 53
|
|
(2) \[1{{s}^{2}}2{{s}^{2}}2{{p}^{6}}3{{s}^{1}}\]
|
(b) ? 328
|
|
(3) \[1{{s}^{2}}2{{s}^{2}}2{{p}^{6}}2{{p}^{5}}\]
|
(c) ? 141
|
|
(4) \[1{{s}^{2}}2{{s}^{2}}2{{p}^{4}}\]
|
(d) + 48
|
View Answer play_arrow
-
question_answer96)
In the following questions a
statement of Assertion (A) followed by a statement of Reason (R) is given.
Choose the correct option out of the choices given below each question.
Assertion (A): Generally
ionisation enthalpy increases from left to right in a period.
Reason (R): When
successive electrons are added to the orbitals in the same principal quantum
level, the shielding effect of inner electrons (core of electrons) does not increase
very much to compensate for the increased attraction of the electron to the
nucleus.
(a) Assertion is correct
statement and Reason is wrong statement.
(b) Assertion and Reason both
are correct statements and Reason is correct explanation of Assertion.
(c) Assertion and Reason both
are wrong statements.
(d) Assertion is wrong statement
and Reason is correct statement.
View Answer play_arrow
-
question_answer97)
Assertion (A): Boron has
a small first ionisation enthalpy than beryllium.
Reason (R): The
penetration of a 2s-electron to the nucleus is more than 2p-electron, hence 2p
is more shielded by the inner core of electrons than the 2s-electrons.
(a) Assertion and Reason both
are correct statements but
Reason is not correct
explanation for Assertion.
(b) Assertion is correct
statement but Reason is wrong statement.
(c) Assertion and Reason both
are correct statements and
Reason is correct explanation
for Assertion.
(d) Assertion and Reason both
are wrong statements.
View Answer play_arrow
-
question_answer98)
Assertion (A): Electron
gain enthalpy becomes less negative as we go down the group.
Reason (R): Size of the
atom increases on going down the group and the added electron would be farther
from the nucleus.
(a) Assertion and Reason both
are correct statments but Reason is not correct explanation for Assertion.
(b) Assertion and Reason both
are correct statements and Reason is correct explanation for Assertion.
(c) Assertion and Reason both
are wrong statements.
(d) Assertion is wrong statement
but Reason is correct statement.
View Answer play_arrow
-
question_answer99)
Discuss the factors affecting electron
gain enthalpy and trend in its variation in the periodic table.
View Answer play_arrow
-
question_answer100)
Define ionisation enthalpy.
Discuss the factors affecting ionisation enthalpy of the elements and its
trends in periodic table.
View Answer play_arrow
-
question_answer101)
Justify the given statement with
suitable examples?"the properties of the elements are a periodic function
of their atomic numbers."
View Answer play_arrow
-
question_answer102)
Write down the outermost
electronic configuration of alkali metals. How will you justify their placement
in group I of the periodic table?
View Answer play_arrow
-
question_answer103)
Write the drawbacks in
Mendeleev's periodic table that led to its modifications.
View Answer play_arrow
-
question_answer104)
In what manner is the long form
of periodic table better than Mendeleev's periodic table? Explain with
examples.
View Answer play_arrow
-
question_answer105)
Discuss and compare the trends in
ionisation enthalpy of elements of group I with those of group 17 elements.
View Answer play_arrow

