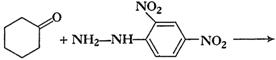
-
Write the structures of the following
compounds.
(i)
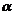
-methoxy
propionaldehyde
(ii) 3-hydroxy butanal
(iii) 2-hydroxy cyclopentane
carbaldehyde
(iv) 4-oxopentanal
(v) Di- sec. butyl ketone
(vi) 4-fluoroacetophenone
Recall the rules of IUPAC nomenclature. The suffix 'al'
represents the presence of CHO group and ?one? represents
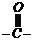
group.
-
Write the structures of products of the
following reactions:
(i)
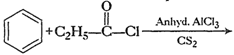
(ii)
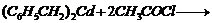
(iii)
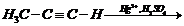
(iv)
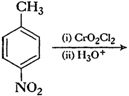
(i)Friedel Crafts reaction
(Acylation) i.e., introduction of acyl group inbenzene ring.
(ii) Formation of ketone.
(iii) Hydration of propyne i.e.,
addition of water to propyne andIsomerization,
(iv) Etard's reaction (Aldehyde is
formed i.e.,
group is
converted into-CHO group.).
-
View Answer play_arrow
-
Arrange the following compounds in increasing order oftheir reactivity in nucleophilic addition reactions.
(i) Ethanal, propanal, propanone, butanone.
(ii) Benzaldehyde, p-tolualdehyde, p-nitro benzaldehyde,acetophenone.
Reactivity depends on two factors: Steric effect and electronic effect.
Lesser the steric hindrance, higher is the reactivity.
View Answer play_arrow
-
Predict the products of the following
reactions:
(i)
(ii)
(iii)
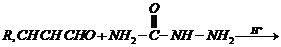
(iv)
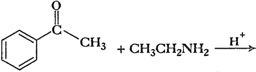
Elimination of
from
carbonyl and amine group and formation of
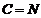
bond.
-
Give the IUPAC names of the following
compounds:
(i)
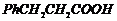
(ii)
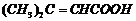
(iii)
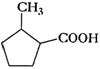
(iv)
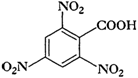
Recall the rules of IUPAC nomenclature
and use suffix ?oic acid? for
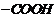
group.
-
Show how each
of the following compounds can be converted to benzoic acid?
(i)
Ethyl benzene (ii) Acetophenone
(iii)
Bromobenzene (iv) Phenylethene (styrene)
(i)
Oxidation by 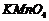 and alkaline
hydrolysis. (
and alkaline
hydrolysis. (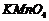 in alkalinemedium
converts complete carbon chain attached directly tobenzene nucleus into
?COOHgroup).
(ii)
lodoform reaction. (
in alkalinemedium
converts complete carbon chain attached directly tobenzene nucleus into
?COOHgroup).
(ii)
lodoform reaction. (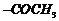 group is
converted into-COOK)
(iii)
Formation of Grignard reagent as intermediale.
(iv)
Oxidation, by
group is
converted into-COOK)
(iii)
Formation of Grignard reagent as intermediale.
(iv)
Oxidation, by 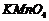 and alkaline
hydrolysis,
and alkaline
hydrolysis,
View Answer play_arrow
-
Winch acid -of each pan- shown here
would you expect to bestronger?
(i)
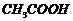
or
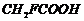
(ii)
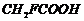
or
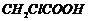
(iii)
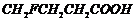
or
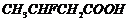
(iv)
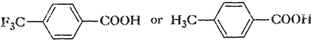
(a) Presence of an electron withdrawing
group tike F(- I showing group) makes a carboxyclic acid stronger.
(b) Higher the ?l effect,
stronger is the acid. The order of ?l effect is
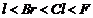
.
(c) As the distance between ?l showing
group and
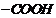
group
increases, acidity decreases.
-
What is meant
by the following terms? Give an example ofthe reaction in each case.
(i)
Cyanohydrin (ii) Acetal
(iii)
Semicarbazone (iv) Aldol
(v)
Hemiacetal (vi) Oxime
(vii)
Ketal (viii) lmine
(ix)
2,4-DNP derivative (x) Schiffs base
(i)
Product of addition reaction of carbonyl group with HCN.
(ii)
Reaction of aldehyde with monohydric alcohol.
(iii)
Reaction of carbonyl compound with semicarbazide,  (iv)
Carbonyl compounds with a-H atom react with dil, NaOH (catalyst)
(v)
Addition of monohydric alcohol with aldehyde.
(vi)
Reaction of carbonyl compound with hydroxylamine.
(vii)
Reaction of ketone with ethylene glycol.
(viii)
Reaction of carbonyl compound with ammonia derivatives.
(ix)
2,4-DNP derivative of carbonyl compound.
(x) Reaction
of carbonyl compound with primary amines.
(iv)
Carbonyl compounds with a-H atom react with dil, NaOH (catalyst)
(v)
Addition of monohydric alcohol with aldehyde.
(vi)
Reaction of carbonyl compound with hydroxylamine.
(vii)
Reaction of ketone with ethylene glycol.
(viii)
Reaction of carbonyl compound with ammonia derivatives.
(ix)
2,4-DNP derivative of carbonyl compound.
(x) Reaction
of carbonyl compound with primary amines.
View Answer play_arrow
-
Name the following compounds according
to IUPAC system of nomenclature:
(i)
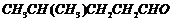
(ii)
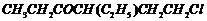
(iii)
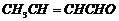
(iv)
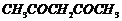
(v)
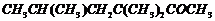
(vi)
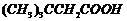
(vii)
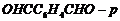
Write complete structure, choose
the longest chain and do the numbering.
Recall the rules of IUPAC, nomenclature
to give name. Use ?al? for
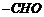
and ?one? for
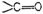
group.
-
Draw the structures of the following
compounds.
(i) 3-methyl butanal
(ii) p-nitro propiophenone
(in) p-methyl benzaldehyde
(iv) 4-methyl pent-3-en-2-one
(v) 4-chloro pentan-2-one
(vi) 3-bromo-4-phenyl pentanoic
acid
(vii) p, p-dihydroxy
benzophenone
(viii) Hex-2-en-4-ynoic acid
(i) First draw the main chain
with functional group and then add substituent groups,
(ii) ?al? represents
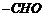
group
?one? represents
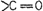
group and ?oic?
acid? represents
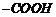
group.
-
Write the IUPAC names of the following
keynotes and aldehydes, wherever possible, give also common names.
(i)

(ii)
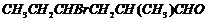
(iii)
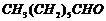
(iv)
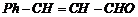
(v)
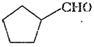
(vi)
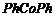
An in Q.2 of exercise.
-
Draw structures of the following
derivatives.
(i) The 2,4-dinitro phenyl
hydrazone of benzaldehyde
(ii) Cyclopropanone
oxime
(iii) Acetaldehyde dimethyl
acetal
(iv) The semicarbazone of
cyclobutanone
(v) The ethylene ketal of
hexan-3-one
(vi) The methyl herniacetal of
formaldehyde
(i) Product of the reaction of
benzaldehyde and 2,4-dinltropheriyi hydrazone (by removing one water molecule).
(ii) Product of the reaction of
hydroxyl amine
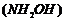
with
cyclopropanone (by removing one water molecule).
(iii) Product of the reaction of
acetaldehyde
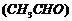
with two
molecules of methanol
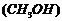
.
(iv) Product of the reaction of
cyclobutanone
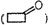
with semicarbazide
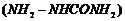
by
removing water molecule.
(iv) Product of the reaction of
hexan-3-one with epoxide
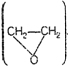
(vi) Product of the reaction of formaldehyde
(HCHO) with one molecule of methanol
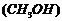
.
-
Predict the
products formed when cyclohexane carbaldehyde reacts with following reagents.
(i) 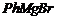 and
then
and
then 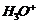 (ii)
Tollen's reagent
(iii)
Semicarbazide and weak acid
(iv)
Excess ethanel and acid
(v)
Zinc amalgam and dilute hydrofluoric acid
(i)
Nucleophilic addition of RMgBr (Grignard reagent) to
(ii)
Tollen's reagent
(iii)
Semicarbazide and weak acid
(iv)
Excess ethanel and acid
(v)
Zinc amalgam and dilute hydrofluoric acid
(i)
Nucleophilic addition of RMgBr (Grignard reagent) to 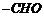 group.
Following by hydration. Carbinol is formed.
(ii)
Oxidation of ?CHO group to ?COOH and reduction of
group.
Following by hydration. Carbinol is formed.
(ii)
Oxidation of ?CHO group to ?COOH and reduction of 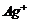 to Ag(silver),
i.e., silver mirror is formed.
(iii)
Removal of
to Ag(silver),
i.e., silver mirror is formed.
(iii)
Removal of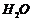 from
from 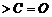 and
and
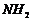 group so
semicarbazone is formed.
(iv)
Nucleophilic addition followed by substitution; Acetal is formed.
(v) Conversion
of ?CHO group to
group so
semicarbazone is formed.
(iv)
Nucleophilic addition followed by substitution; Acetal is formed.
(v) Conversion
of ?CHO group to 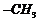 i.e.,
Cycloalkane is formed (Clemmnesen?s reduction).
i.e.,
Cycloalkane is formed (Clemmnesen?s reduction).
View Answer play_arrow
-
Which of the
following compounds would undergo aldol condensation, which the Cannizzaro
reaction and which neither? Write the structures of the expected products of
aldol condensation or Cannizzaro reaction.
(i)
Methanal (ii) 2-methyl pentanal
(iii)
Benzaldeliyde (iv) Benzophenone
(v)
Cyclohexanone (vi) 1-phenyl propanone
(vii)
Phenyl acetaldehyde (viii) Butan-1-ol
(ix)
2,2-dimethyl butanal
(a)
Compounds (aldehydes or ketones) having one or more  -H-atoms
undergo aldol condensation.
(b)
Compounds (aldehydes) without
-H-atoms
undergo aldol condensation.
(b)
Compounds (aldehydes) without 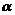 - H-atoms
undergo Cannizzaro reaction.
(c)
Compounds (ketones) without-
- H-atoms
undergo Cannizzaro reaction.
(c)
Compounds (ketones) without-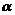 -H-atoms will
neither undergo aldol condensation nor Cannizzaro reaction.
So write the
structure of each compound to find the answer.
-H-atoms will
neither undergo aldol condensation nor Cannizzaro reaction.
So write the
structure of each compound to find the answer.
View Answer play_arrow
-
How will you convert ethanal into the following compounds?
(i) Butane- 1,3-diol
(ii) But-2-enal
(iii) But-2-enoic acid
Given conversions involve the following steps
(i) (a) Aldol condensation (b) Reduction
(ii) (a) Aldol condensation (b) Dehydration
(iii) (a) Aldol condensation (b) Dehydration
(c)Reaction with Token's reagent.
View Answer play_arrow
-
Write structural formulas and names of four possible aldol condensation products from propanal and butanal. In each case, indicate which aldehyde acts as nucleophile and which as electrophile?
View Answer play_arrow
-
An organic compound
with the molecular formula 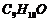 forms 2,4-DNP
derivative, reduces Tollen's reagent and undergoesCannizzaro reaction. On
vigorous oxidation, it gives 1,2-benzenedicarboxylic acid. Identify the
compound.
(i)
Identify the functional group from the reactions given by it.
(ii)
Find position of the functional group, from 1, 2-benzene dicarboxylicacid.
(iii)
Write the structure of actual compound.
(iv) Give all
reactions.
forms 2,4-DNP
derivative, reduces Tollen's reagent and undergoesCannizzaro reaction. On
vigorous oxidation, it gives 1,2-benzenedicarboxylic acid. Identify the
compound.
(i)
Identify the functional group from the reactions given by it.
(ii)
Find position of the functional group, from 1, 2-benzene dicarboxylicacid.
(iii)
Write the structure of actual compound.
(iv) Give all
reactions.
View Answer play_arrow
-
An organic compound (A) (molecular
formula
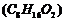
was
hydrolysed with dilute sulphuric acid to give a carhoxylic acid (B)and alcohol
(C). Oxidation of (C) with chromic acid produced (B). (C)on dehydration gives
but-1-ene. Write equations for the reactionsinvolved.
(i) Ester produces a carboxylic
acid and an alcohol on hydrolysis.
(ii) Ester (A) has 8 C-atoms, calculate
the number of carbon atoms in acid and alcohol.
(iii) Guess the alcohol and the
acid according to the given reactions.
(iv) Now guess the ester
(v) Write all the related equations,
-
Arrange the following compounds in
increasing order of their property as indicated:
(i) Acetaldehyde, acetone,
di-tert-butyl kentone, methyl tert-butyl ketone (reactivity towards HCN).
(ii)
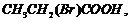
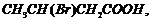
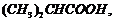
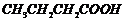
(acid
strength).
(iii) Benzoic acid, 4-nitrobenzoic
acid, 3,4-dinitro benzoic acid;4-methoxy benzoic acid (acid strength).
(i) Greater the steric
hindrance, lesser the reactivity of carbonyl compound towards nucleophilic
addition, i.e., towards HCN.
(ii) Presence of electron
donating group (EV^G) makes an acid more acidic. As the distance between EWG
and ?COOH increases, aciditydecreases.
(iii) Same as (ii).
-
How will you prepare the following
compounds frombenzene? You may use any inorganic reagent and any organic
reagenthaving not more than one carbon atom.
(i) Methyl benzoate (ii)
m-nitrobenzoic acid
(iii) p-nitrobenzoic
acid (iv) Phenyl acetic acid
(v) p-nitrobenzaldehyde.
The given conversions involve
the following processes
(i) Friedel Crafts alkylation,
oxidation, esterificaticn.
(ii) Introducing COOH as in (i)
then nitration.
(iii) Friedel Crafts alkylation;
then nitration;
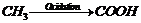
(iv) Friedel Crafts alkylation.
bromination inside chain, introducing CN in place of Br and hydrolysis.
(v) Formation of p-nitrotoluene as in
(iii) then oxidizing
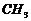
, to
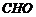
,
-
How will you bring about the following conversion in notmore than two steps?
(i) Propanone to propene
(ii) Benzoic acid to benzaldehyde
(iii) Ethanol to 3-hydroxy butanal
(iv) Benzene to m-nitroacetophenone
(v) Benzaldehyde to benzophenone
(vi) Bromobenzene to 1-phenyl ethanol
(vii) Benzaldehyde to 3-phenyl propan-1-ol
(viii) Benzaldehyde to 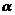 -hydroxy phenyl acetic acid
(ix) Benzoic acid to w-nitrobenzyl alcohol
(i) Alcohols readily gives alkene on dehydration, so first convert ketone into alcohol by reduction then do dehydration,
(ii) Benzaldehyde is easily obtained from benzoyl chloride so convert benzoic acid into benzoyl chloride and then do Rosenmund'sreduction.
(iii) Carbonyl compounds gives a compound having double carbon atomsthan the parent compounds, if undergo aldol condensation, so firstconvert alcohol to aldehyde by oxidation and then do Aldol condensation.
(iv)
-hydroxy phenyl acetic acid
(ix) Benzoic acid to w-nitrobenzyl alcohol
(i) Alcohols readily gives alkene on dehydration, so first convert ketone into alcohol by reduction then do dehydration,
(ii) Benzaldehyde is easily obtained from benzoyl chloride so convert benzoic acid into benzoyl chloride and then do Rosenmund'sreduction.
(iii) Carbonyl compounds gives a compound having double carbon atomsthan the parent compounds, if undergo aldol condensation, so firstconvert alcohol to aldehyde by oxidation and then do Aldol condensation.
(iv) 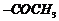 , is m-directing group, so do Friedel Craft acylation to introduce
, is m-directing group, so do Friedel Craft acylation to introduce 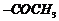 , group followed by nitration.
(v) (a) Oxidation to convert into acid.
(b) Heating of calcium salt.
(vi) (a) Formation of Grignard reagent,
(b) Treating with
, group followed by nitration.
(v) (a) Oxidation to convert into acid.
(b) Heating of calcium salt.
(vi) (a) Formation of Grignard reagent,
(b) Treating with 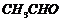 and hydrolysis.
(vii) Perkin's reaction followed by reduction.
(viii) Aldol condensation as it gives
and hydrolysis.
(vii) Perkin's reaction followed by reduction.
(viii) Aldol condensation as it gives 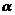 -hydroxyaldehyde which is oxidized to get acid.
(ix) Nitration followed by reduction.
-hydroxyaldehyde which is oxidized to get acid.
(ix) Nitration followed by reduction.
View Answer play_arrow
-
Describe the following.
(i) Acetylation
(ii) Cannizzaro reaction
(Hi) Cross aldol condensation
(iv) Decarboxylation
(i) Providing
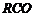
group.
(ii) Reaction of aldehydes
having no
-H-atom.
(iii) Two different aldehydes
are condensed.
(iv) Removal of
from
carboxyl group,
-
Complete each synthesis by giving missing starting material, reagent or products.
(i)
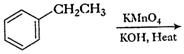
(ii)
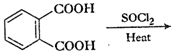
(iii)
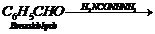
(iv)
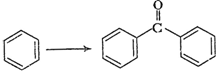
(v)
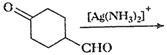
(vi)
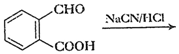
(vii)
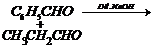
(viii)
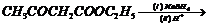
(ix)
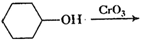
(x)
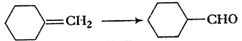
(xi)
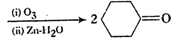
-
Give plausible explanation for each of
the following:
(i) Cyclohexanone forms
cyanohydrin in good yield but2,2,6-tnmethyl cyclohexanone does not.
(ii) These are two
groups
in semicarbazide. However, only one is involved in the formation of
semicarbazones.
(iii) During the preparation of
esters from a carboxylic acid and analcohol in the presence -of an acid
catalyst, the water or the ester should be removed as soon as it is formed.
(i)
effect
due to methyl groups: steric hindrance, make a carbonyl compound less reactive.
(ii) Lone pair of electrons
involved in conjugation with CO.
(iii) Consider the reversible nature of
the reaction.
-
An organic
compound contains 69.77% carbon, 11.63%hydrogen and rest oxygen. The molecular
mass of the compound is 86. Itdoes not reduce Tollen's reagent but forms an
addition compound withsodium hydrogen sulphite and give positive iodoform test.
On vigorousoxidation it gives ethanoic and propanoic acid. Write the
possiblestructure of the compound.
(i)
Derive empiricsl and molecular formula by first finding moles.
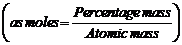 (ii)
According to the given chemical reactions, derive the structuralformula.
(iii) Write all
the related equations.
(ii)
According to the given chemical reactions, derive the structuralformula.
(iii) Write all
the related equations.
View Answer play_arrow
-
Although phenoxide ion has more number of resonatingstructures than carboxylate ion, carboxylate acid is a stronger acid thanphenol. Why?
Comparative effectiveness of delocalization of- negative charge isresponsible for the acidic strength. A resonating structure havingnegative charge on more electronegative atom, is more stable.
View Answer play_arrow
-
question_answer28)
Addition
of water to alkynes occurs in acidic
medium and in the presence of 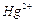 ions as a catalyst.
Which of the following products will be formed on addition of water to
but-1-yne under these conditions.
(a)
ions as a catalyst.
Which of the following products will be formed on addition of water to
but-1-yne under these conditions.
(a)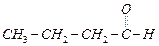 (b)
(b)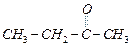 (c)
(c)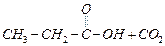 (d)
(d)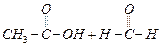
View Answer play_arrow
-
question_answer29)
Which
of the following compounds is most reactive towards nucleophilic addition reactions?
(a)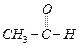 (b)
(b)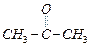 (c) (d)
(c) (d)
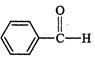
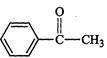
View Answer play_arrow
-
question_answer30)
The
correct order of increasing acidic strength is ?????..
(a) Phenol < Ethanol < Chloroacetic acid < Acetic acid
(b) Ethanol < Phenol < Chloroacetic acid < Acetic
acid
(c) Ethanol < Phenol < Acetic acid < Chloroacetic
acid
(d) Chloroacetic acid < Acetic acid < Phenol <
Ethanol
View Answer play_arrow
-
question_answer31)
Compound
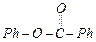 can be prepared
by the reaction of.............
(a) Phenol and benzoic acid in the presence of
NaOH
(b) Phenol and benzoyl chloride in the presence of pyridine
(c) Phenol and benzoyl chloride in the presence of
can be prepared
by the reaction of.............
(a) Phenol and benzoic acid in the presence of
NaOH
(b) Phenol and benzoyl chloride in the presence of pyridine
(c) Phenol and benzoyl chloride in the presence of 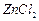 (d) Phenol and benzaldehyde in the presence of palladium
(d) Phenol and benzaldehyde in the presence of palladium
View Answer play_arrow
-
question_answer32)
The
reagent which does not react with both, acetone and benzaldehyde is
(a) Sodium hydrogensulphite
(b) Phenyl hydrazine
(c) Pehling's solution (d) Grignard reagent
View Answer play_arrow
-
question_answer33)
Cannizzaro's
reaction is not given by..............
(a) (b)
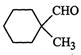
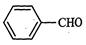 (c)HCHO (d)
(c)HCHO (d)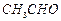
View Answer play_arrow
-
question_answer34)
Which
product is formed when the compound
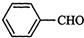 is treated with concentrated
aqueous KOH solution ?
(a)
is treated with concentrated
aqueous KOH solution ?
(a)
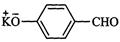 (b)
(b)
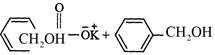 (c)
(c)
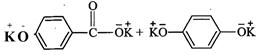 (d)
(d)
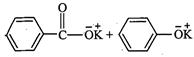
View Answer play_arrow
-
question_answer35)
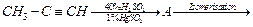
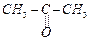 Structure of 'A' and type of isomerism in the
above reaction are respectively.
(a) Prop-l-en-2-ol, metamerism
(b) Prop-1-en-l-ol, tautomerism .
(c) Prop-2-en-2-ol, geometrical isomerism
(d) Prop-l-en-2-ol, tautomerism
Structure of 'A' and type of isomerism in the
above reaction are respectively.
(a) Prop-l-en-2-ol, metamerism
(b) Prop-1-en-l-ol, tautomerism .
(c) Prop-2-en-2-ol, geometrical isomerism
(d) Prop-l-en-2-ol, tautomerism
View Answer play_arrow
-
question_answer36)
Compounds
A and C in the following reaction
are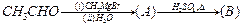
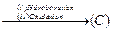 (a) identical (b) positional
isomers
(c) functional isomers (d) optical isomers
(a) identical (b) positional
isomers
(c) functional isomers (d) optical isomers
View Answer play_arrow
-
question_answer37)
Which
is the most suitable reagent for the following conversion ?
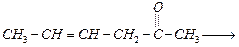
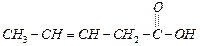 (a) Tollen's reagent (b) Benzoyl peroxide
(c)
(a) Tollen's reagent (b) Benzoyl peroxide
(c)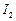 and
NaOH solution
(d) Sn and NaOH solution
and
NaOH solution
(d) Sn and NaOH solution
View Answer play_arrow
-
question_answer38)
Which
of the following compounds will give butanone on oxidation with alkaline 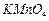 solution ?
(a) Butan-1-ol (b) Butan-2-ol
(c) Both of these (d) None of these
solution ?
(a) Butan-1-ol (b) Butan-2-ol
(c) Both of these (d) None of these
View Answer play_arrow
-
question_answer39)
In
Clemmensen Reduction, carbonyl compound is treated with..............
(a) Zinc amalgam + HCl
(b) Sodium amalgam + HCl
(c) Zinc amalgam + nitric acid
(d) Sodium amalgam + 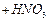
View Answer play_arrow
-
question_answer40)
Which
of the following compounds do not undergo aldol condensation ?
(a)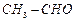 (b)
(c)
(b)
(c)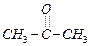 (d)
(d)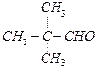 View Answer play_arrow
View Answer play_arrow
-
question_answer41)
Treatment
of compound 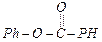 with NaOH solution yields
(a) Phenol (b) Sodium phenoxide
(c) Sodium benzoate (d) Benzophenone
with NaOH solution yields
(a) Phenol (b) Sodium phenoxide
(c) Sodium benzoate (d) Benzophenone
View Answer play_arrow
-
question_answer42)
Which
of the following conversions can be carried out by Clemmensen Reduction ?
(a) Benzaldehyde into benzyl alcohol
(b) Cyclohexanone into cyclohexane
(c) Benzoyl chloride into benzaldehyde
(d) Benzophenone into diphenylmethane
View Answer play_arrow
-
question_answer43)
Through
which of the following reactions number of carbon atoms can be increased in the
chain ?
(a) Grignard reaction
(b) Cannizzaro's reaction
(c) Aldol condensation (d) HVZ reaction
View Answer play_arrow
-
question_answer44)
Benzophenone
can be obtained by.............
(a) Benzoyl chloride + Benzene + 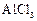 (b) Benzoyl chloride + Diphenylcadmium
(c) Benzoyl chloride + Phenylmagnesium chloride
(d) Benzene + Carbon monoxide +
(b) Benzoyl chloride + Diphenylcadmium
(c) Benzoyl chloride + Phenylmagnesium chloride
(d) Benzene + Carbon monoxide + 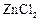
View Answer play_arrow
-
question_answer45)
Which
of the following is the correct representation for intermediate of
nucleophilic addition reaction to the given carbonyl compound (A):
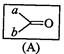 (a) (b)
(a) (b)
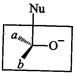
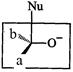 (c)?.. (d)
(c)?.. (d)
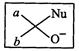
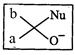
View Answer play_arrow
-
question_answer46)
Why
is there a large difference in the boiling points of butanal and butan-1-ol ?
View Answer play_arrow
-
question_answer47)
Write
a test to differentiate between pentan-2-one and pentan-3-one.
View Answer play_arrow
-
question_answer48)
Give
the IUPAC names of the following compounds
(i)??. (ii)
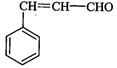
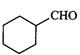 (iii)
(iii)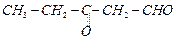 (iv)
(iv)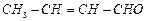
View Answer play_arrow
-
question_answer49)
Give
the structure of the following compounds.
(i) 4-Nitropropiophenone
(ii) 2-Hydroxycyclopentanecarbaldehyde
(iii) Phenylacetaldehyde
View Answer play_arrow
-
question_answer50)
Write
IUPAC names of the following structures.
(i)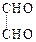 (ii) (iii)
(ii) (iii)
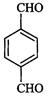
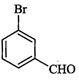
View Answer play_arrow
-
question_answer51)
Benzaldehyde
can be obtained from benzol chloride. Write reactions for obtaining benzal
chloride and then benzaldehyde from it.
View Answer play_arrow
-
question_answer52)
Name
the electrophile produced in the reaction of benzene with benzoyl chloride in
the presence of Anhydrous 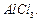 Name
the reaction also.
Name
the reaction also.
View Answer play_arrow
-
question_answer53)
Oxidation
ofketones involves carbon-carbon bond cleavage. Name the products formed on
oxidation of 2,5-dimethylhexan-3-one.
View Answer play_arrow
-
question_answer54)
Arrange
the following in decreasing order of their acidic strength and give reason for
your answer.
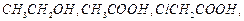
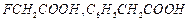
View Answer play_arrow
-
question_answer55)
What
product will be formed on reaction of propanal with 2-methylpropanal in the
presence of NaOH ? What products will be formed ? Write the name of the
reaction also.
View Answer play_arrow
-
question_answer56)
Compound
'A' was prepared by oxidation of compound 'B' with alkaline KMn04. Compound 'A'
on reduction with lithium aluminium hydride gets converted back to compound
'B'. When compound 'A' is heated with .compound B in the presence of 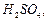 , it produces fruity
smell of compound C. To which family the compounds 'A', 'B' and 'C' belong to
?
, it produces fruity
smell of compound C. To which family the compounds 'A', 'B' and 'C' belong to
?
View Answer play_arrow
-
question_answer57)
Arrange
the following in decreasing order of their acidic strength. Give explanation
for the arrangement.
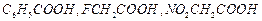
View Answer play_arrow
-
question_answer58)
Alkenes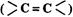 and carbonyl compounds
and carbonyl compounds 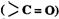 , both contain a n bond but
alkenes show lie addition reactions whereas carbonyl compounds show
nucleophilic addition reactions.
, both contain a n bond but
alkenes show lie addition reactions whereas carbonyl compounds show
nucleophilic addition reactions.
View Answer play_arrow
-
question_answer59)
Carboxylic
acids contain a carbonyl group but do not show the nucleophilic addition
reaction like or ketones. Why ?
View Answer play_arrow
-
question_answer60)
Identify
the compounds A, B and C in the following reaction.
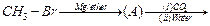
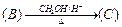
View Answer play_arrow
-
question_answer61)
Why
are carboxylic acids more acidic than alcohols or phenols although all of them
have hydrogen I atom attached to a oxygen atom (?O?H) ?
View Answer play_arrow
-
question_answer62)
Complete
the following reaction sequence.
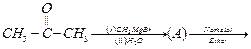
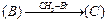
View Answer play_arrow
-
question_answer63)
Ethylbenzene
is generally prepared by acetylation of benzene followed by reduction and not
by direct alkylation. Think of a possible reason.
View Answer play_arrow
-
question_answer64)
Can
Gatterman-Koch reaction be considered similar to Friedel Craft's acylation ?
Discuss.
View Answer play_arrow
-
question_answer65)
Match
the common names given in Column I with the IUPAC names given in Column II.
|
|
Column I (Common names)
|
|
Column II (IUPAC
names)
|
|
(a)
|
Cinnamaldehyde
|
(i)
|
Pentanal
|
|
(b)
|
Acetophenone
|
(ii)
|
Prop-2-enal
|
|
(c)
|
Valeraldehyde
|
(iii)
|
4-Methylpent-3-en-2-one
|
|
(d)
|
Acrolein
|
(vi)
|
3-Phenylprop-2-enal
|
|
(e)
|
Mesityl oxide
|
(v)
|
1-Phenylethanone
|
View Answer play_arrow
-
question_answer66)
Match
the acids given in Column I with their correct IUPAC names given in Column II.
|
|
|
Column I (Acids)
|
|
Column II (IUPAC
names)
|
|
(a)
|
|
Phthalic acid
|
(i)
|
Hexane-1,
6-dioic acid
|
|
(b)
|
|
Oxalic acid
|
(ii)
|
Benzene-1,
2-dicarboxylic acid
|
|
(c)
|
|
Succinic acid
|
(iii)
|
Pentane-1,5-dioic
acid
|
|
(d)
|
|
Adipic acid
|
(vi)
|
Butane-1,4-dioic
acid
|
|
(e)
|
|
Glutaric acid
|
(v)
|
Ethane-1,2-dioic
acid
|
View Answer play_arrow
-
question_answer67)
Match
the reactions given in Column I with the suitable reagents given in Column
II.
|
|
Column I (Reactions)
|
|
Column II (Reagents)
|
|
(a)
|
Benzophenone  Diphenylmethane Diphenylmethane
|
(i)
|
|
|
(b)
|
Benzaldehyde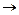 1-Phenylethanol
1-Phenylethanol
|
(ii)
|
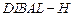
|
|
(c)
|
Cyclohexanone
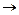 Cyclohexanol Cyclohexanol
|
(iii)
|
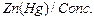 HCl HCl
|
|
(d)
|
Phenyl
benzoate 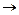 Benzaldehyde Benzaldehyde
|
(vi)
|
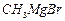
|
View Answer play_arrow
-
question_answer68)
Match
the example given in Column I with the name of the reaction in Column
II.
|
|
Column I (Example)
|
|
Column II (Reaction)
|
|
(a)
|
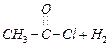 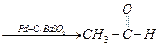
|
(i)
|
Friedel-Crafts
acylation
|
|
(b)
|
|
(ii)
|
HVZ
reaction
|
|
(c)
|
 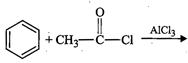
|
(iii)
|
Aldol
condensation
|
|
(d)
|
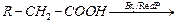 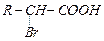
|
(iv)
|
Cannizzaro?s
reaction
|
|
(e)
|
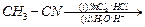 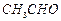
|
(v)
|
Rosenmund?s
reduction
|
|
(f)
|
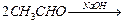 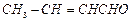
|
(vi)
|
Stephen?s
reaction
|
View Answer play_arrow
-
question_answer69)
Assertion
: Formaldehyde is a planar molecule.
View Answer play_arrow
-
question_answer70)
Assertion
: Compounds containing -CHO group are easily oxidised to corresponding
carboxylic acids.
View Answer play_arrow
-
question_answer71)
Assertion
: The 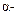 hydrogen atom in carbonyl
compounds is less acidic.
hydrogen atom in carbonyl
compounds is less acidic.
View Answer play_arrow
-
question_answer72)
Assertion
: Aromatic aldehydes and formaldehyde undergo Cannizzaro reaction.
View Answer play_arrow
-
question_answer73)
Assertion
: Aldehydes and ketones, both react with Tollens' reagent to form silver
mirror.
View Answer play_arrow
-
question_answer74)
An
alkene 'A' (Mol. formula 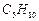 )
on ozonolysis gives a mixture of two compounds 'B' and 'C? Compound 'B' gives
positive Fehling's test and also forms iodo form on treatment with
)
on ozonolysis gives a mixture of two compounds 'B' and 'C? Compound 'B' gives
positive Fehling's test and also forms iodo form on treatment with 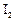 and NaOH, Compound
'C' does not give Fehling's test but forms iodoform. Identify the compounds A,
B and C, Write the reaction for ozonolysis and formation of iodoform from B and
C.
and NaOH, Compound
'C' does not give Fehling's test but forms iodoform. Identify the compounds A,
B and C, Write the reaction for ozonolysis and formation of iodoform from B and
C.
View Answer play_arrow
-
question_answer75)
An
aromatic compound 'A' (Molecular formula 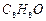 ) gives positive
2,4-DNP test. It gives a yellow precipitate of compound 'B' on treatment with
iodine and sodium hydroxide solution. Compound ?A? does not give Tollen's or
Fehling's test. On drastic oxidation with potassium permanganate it form a
carboxylic acid 'C' (Molecular formula
) gives positive
2,4-DNP test. It gives a yellow precipitate of compound 'B' on treatment with
iodine and sodium hydroxide solution. Compound ?A? does not give Tollen's or
Fehling's test. On drastic oxidation with potassium permanganate it form a
carboxylic acid 'C' (Molecular formula 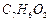 ), which is also
formed along with the yellow compound in the above reaction. Identify A, B and
C and write all the reactions involved.
), which is also
formed along with the yellow compound in the above reaction. Identify A, B and
C and write all the reactions involved.
View Answer play_arrow
-
question_answer76)
Write
down functional isomers of a carbonyl compound with molecular formula 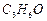 . Which isomer
will react faster with HCN and why ? Explain the mechanism of the reaction
also. Will the reaction lead to the completion with the conversion of whole
reactant into product at reaction conditions ? If a strong acid is added to the
reaction mixture what will be the effect on concentration of the product and
why ?
. Which isomer
will react faster with HCN and why ? Explain the mechanism of the reaction
also. Will the reaction lead to the completion with the conversion of whole
reactant into product at reaction conditions ? If a strong acid is added to the
reaction mixture what will be the effect on concentration of the product and
why ?
View Answer play_arrow
-
question_answer77)
When
liquid 'A' is treated with a freshly prepared ammonia cal silver nitrate
solution, it gives bright silver mirror. The liquid forms a white crystalline
solid on treatment with sodium hydrogensulphite.
Liquid 'B' also forms a white crystalline solid with sodium
hydrogensulphite but it does not give test with ammonia cal silver nitrate.
Which of the two liquids is aldehyde ? Write the chemical equations of these
reactions also.
View Answer play_arrow
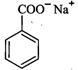
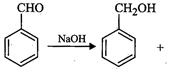
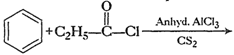 (ii)
(ii) (i)Friedel Crafts reaction
(Acylation) i.e., introduction of acyl group inbenzene ring.
(ii) Formation of ketone.
(iii) Hydration of propyne i.e.,
addition of water to propyne andIsomerization,
(iv) Etard's reaction (Aldehyde is
formed i.e.,
(i)Friedel Crafts reaction
(Acylation) i.e., introduction of acyl group inbenzene ring.
(ii) Formation of ketone.
(iii) Hydration of propyne i.e.,
addition of water to propyne andIsomerization,
(iv) Etard's reaction (Aldehyde is
formed i.e.,  (iii)
(iii)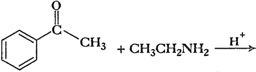 Elimination of
Elimination of  (iv)
(iv)
 Recall the rules of IUPAC nomenclature
and use suffix ?oic acid? for
Recall the rules of IUPAC nomenclature
and use suffix ?oic acid? for  (vi) Product of the reaction of formaldehyde
(HCHO) with one molecule of methanol
(vi) Product of the reaction of formaldehyde
(HCHO) with one molecule of methanol  (ii)
(ii)
 (iii)
(iii) (v)
(v)
 (vi)
(vi)
 (vii)
(vii)
![]() (c)HCHO (d)
(c)HCHO (d)![]()
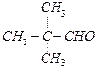 View Answer play_arrow
View Answer play_arrow

 (iii)
(iii)![]() (iv)
(iv)![]()
![]() (ii) (iii)
(ii) (iii)


![]()
![]()
![]()
![]()




![]() hydrogen atom in carbonyl
compounds is less acidic.
hydrogen atom in carbonyl
compounds is less acidic.
