Aromatic Carboxylic Acids
Category : JEE Main & Advanced
Aromatic acid contain one or more carboxyl group (COOH) attached directly to aromatic nucleus.
Examples
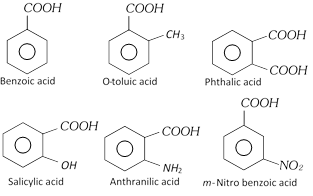
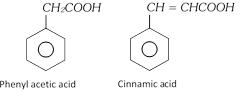
Aromatic acid containing –COOH group in the side chain, they are considered as aryl substituted aliphatic acid.
Examples
Benzoic Acid
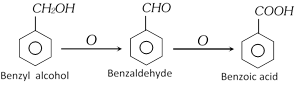
(1) Methods of Preparation
(i) From oxidation of Benzyl alcohol [Laboratory method]
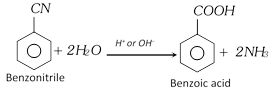
(ii) From hydrolysis of nitriles or cyanides
(iii) From Grignard reagent
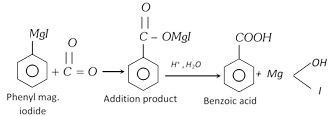
(iv) By hydrolysis of esters
\[\underset{\text{Methyl benzoate}}{\mathop{{{C}_{6}}{{H}_{5}}COOC{{H}_{3}}}}\,+{{H}_{2}}O\xrightarrow{{{H}^{+}}orO{{H}^{-}}}\underset{\text{Benzoic acid}}{\mathop{{{C}_{6}}{{H}_{5}}COOH}}\,+\underset{\text{Methanol}}{\mathop{C{{H}_{3}}OH}}\,\]
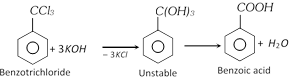
(v) From trihalogen derivatives of hydrocarbons
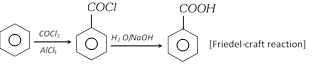
(vi) From benzene
(vii) From Toluene
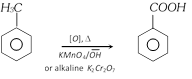
(viii) From o-xylene [Industrial method]
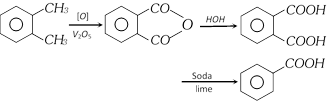
(ix) From naphthalene [Industrial method]
![]()
(2) Physical Properties
(i) It is a white crystalline solid.
(ii) It has m.p. 394 K.
(iii) It is sparingly soluble in cold water but fairly soluble in hot water, alcohol and ether.
(iv) It has a faint aromatic odour and readily sublimes and is volatile in steam.
(3) Acidity of Aromatic Carboxylic Acid : Aromatic acid dissociates to give a carboxylate anion and proton.
\[{{C}_{6}}{{H}_{5}}COOH\rightleftharpoons {{C}_{6}}{{H}_{5}}CO\overset{-}{\mathop{O}}\,\,+{{H}^{+}}\]
Since the carboxylate anion \[(ArCO\overset{-}{\mathop{O}}\,)\] is resonance stabilised to a greater extent than the carboxylic acid (ArCOOH).
\[Ar-\overset{O}{\mathop{\overset{|\,|}{\mathop{C}}\,}}\,-OH\leftrightarrow Ar-\overset{{{O}^{-}}}{\mathop{\overset{|\,\,\,}{\mathop{C\,}}\,}}\,=\overset{\,+}{\mathop{O}}\,H\] \[\overset{\text{Resonance in carboxylic acid}}{\mathop{\left[ \begin{align} & \text{Non-equivalent structure and} \\ & \text{hence less stable} \\ \end{align} \right]}}\,\]
\[Ar-\overset{O}{\mathop{\overset{|\,|}{\mathop{C}}\,}}\,-{{O}^{-}}\leftrightarrow Ar-\overset{{{O}^{-}}}{\mathop{\overset{|\,}{\mathop{C\,}}\,}}\,=O\] \[\overset{\text{Resonance in carboxylate anion}}{\mathop{\left[ \begin{align} & \text{Equivalent structure and hence} \\ & \text{more stable} \\ \end{align} \right]}}\,\]
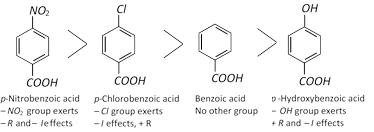
Effect of Substituents on Acidity : The overall influence of a substituent on acidity of substituted benzoic acids is due to two factors.
(i) Inductive effect : If the substituent exerts–I effect, it increases the acidity of carboxylic acids, while if it exerts + I effect it decreases the acidity. Inductive effect affects all positions, i.e., o–, m– and p–.
(ii) Resonance effect : Like inductive effect, if the resonance producing group exerts minus effect i.e., if it withdraws electrons, it increases the strength of the benzoic acid. Similarly, if the group causes +R effect it decreases the acidity of benzoic acid. However, remember that resonance effect affects only o- and p-positions. Thus if resonance producing group is present in the m-position it will not exert its effect.
In case resonance and inductive effects both operate in the molecule, resonance effect being stronger overpowers the inductive effect.
Thus on the above basis, the following order of acidity can be explained.
Similarly:
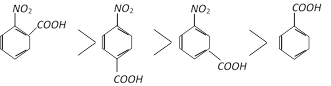
Acidity is only due to electron withdrawing inductive effect of the \[-N{{O}_{2}}\] group (resonance does not affect the m-position) while in the p-isomer acidity is due to electron withdrawing inductive as well as resonance effect.
The acidity of the three isomers of hydroxybenzoic acids follows the following order.
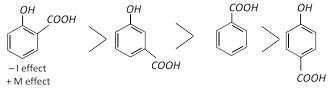
Resonance effect cannot operate and hence only the acid-strengthening –I effect takes part with the result m-hydroxybenzoic acid is stronger acid than benzoic acid. Like other substituted benzoic acid.
Acidic character among benzoic acids having different electron releasing group.
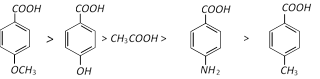
(4) Chemical Properties :
(i) Reactions of carboxylic group
(ii) Reactions of aromatic ring
(i) Reactions of Carboxylic Group
(a) Reaction with metals
![]()
(b) Reaction with Alkalies Or \[NaHC{{O}_{3}}\] Or \[N{{a}_{2}}C{{O}_{3}}\]:
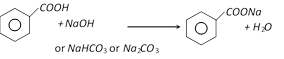
(c) Formation of Esters :
Aromatic acid (benzoic acid) having no group in its ortho positions can be readily esterified with alcohol in presence of a mineral acid.
![]()
In presence of ortho substituent the rate of esterification is greatly decreased due to steric effect.
The esterification of the various benzoic acids :
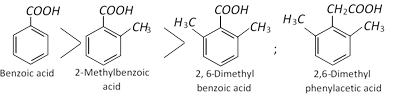
The substituted phenylacetic acid is easily esterified because \[-\,COOH\] group is separated from benzene ring by \[-\,C{{H}_{2}}-\,\]part.
The ortho-substituted benzoic acids can be easily esterified by treating the silver salt of the acid with alkyl halides, i.e.,
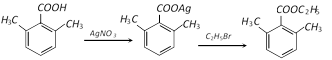
This is due to the fact that in such cases the attack of the alkyl group of the alkyl halides is on the oxygen atom of the \[-COOH\] group but not on the sterically hindered carbon atom.
(d) Formation of acid chloride
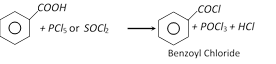
(e) Reaction with \[{{N}_{3}}H\] [Schmidt reaction]
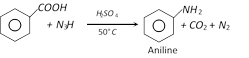
(f) Reaction with sodalime
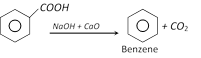
(g) Reaction with anhydride

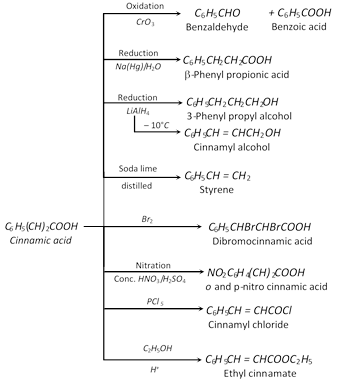
(h) Reduction

(i) Decarboxylation
![]()
(j) Hunsdiecker reaction :
\[\underset{\text{Silver benzoate}}{\mathop{{{C}_{6}}{{H}_{5}}COOAg}}\,\underset{(B{{r}_{2}}\text{ or }C{{l}_{2}})}{\mathop{+{{X}_{2}}}}\,\underset{\text{heat}}{\mathop{\xrightarrow{\text{in }CC{{l}_{4}}}}}\,\underset{\text{Phenyl halide}}{\mathop{{{C}_{6}}{{H}_{5}}-X+C{{O}_{2}}}}\,\uparrow +AgX\]
(ii) Reactions of Aromatic Ring
(a) Nitration
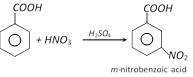
(b) Sulphonation
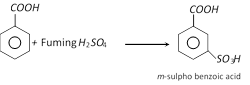
(c) Chlorination
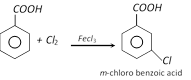
(d) Reduction

(5) Uses : Benzoic acid is used,
(5) Uses : Benzoic acid is used,
(i) in medicine in the form of its salts especially as urinary antiseptic.
(ii) As sodium benzoate for preservation of food such as fruit juices, tomato ketchup, pickles etc.
(iii) In the preparation of aniline blue.
(iv) In treatment of skin diseases like eczema.
(6) General Tests
(i) Benzoic acid dissolves in hot water but separates out in the form of white shining flakes on cooling.
(ii) It evolves \[C{{O}_{2}}\] with sodium bicarbonate, i.e., it gives effervescence with sodium carbonate.
(iii) Neutral ferric chloride gives a buff coloured precipitate.
(iv) When warmed with ethyl alcohol and a little conc. \[{{H}_{2}}S{{O}_{4}},\] a fragrant odour of ethyl benzoate is obtained.
(v) When heated strongly with soda lime, benzene vapours are evolved which are inflammable.
Cinnamic Acid [\[\beta -\]Phenyl acrylic acid]
(1) Methods of Preparation
(i) By Perkin's reaction
\[{{C}_{6}}{{H}_{5}}CHO+{{(C{{H}_{3}}CO)}_{2}}O\underset{180{}^\circ C}{\mathop{\xrightarrow{C{{H}_{3}}COONa}}}\,\]
\[{{C}_{6}}{{H}_{5}}CH=CHCOOH+C{{H}_{3}}COOH\]
(ii) By Claisen condensation
\[{{C}_{6}}{{H}_{5}}CHO+C{{H}_{3}}COO{{C}_{2}}{{H}_{5}}\xrightarrow{{{C}_{2}}{{H}_{5}}ONa'}\]
\[{{C}_{6}}{{H}_{5}}CH\underset{\text{Ester}}{\mathop{=CHCOO{{C}_{2}}{{H}_{5}}}}\,\underset{{{H}^{+}}}{\mathop{\xrightarrow{{{H}_{2}}O}}}\,\]
\[{{C}_{6}}{{H}_{5}}CH=CHCOOH+{{C}_{2}}{{H}_{5}}OH\]
(iii) By knoevenagel reaction
\[{{C}_{6}}{{H}_{5}}CHO+C{{H}_{2}}{{(COOH)}_{2}}\underset{\text{heat}}{\mathop{\xrightarrow{N{{H}_{3}}}}}\,\] \[{{C}_{6}}{{H}_{5}}CH=CHCOOH+C{{O}_{2}}+{{H}_{2}}O\]
(iv) Industrial method
\[\underset{\text{Benzal chloride}}{\mathop{{{C}_{6}}{{H}_{5}}CHC{{l}_{2}}}}\,+\underset{\text{Sodium acetate}}{\mathop{{{H}_{2}}CHCOONa}}\,\xrightarrow{200{}^\circ C}{{C}_{6}}{{H}_{5}}CH=CHCOOH+NaCl+HCl\]
(2) Physical Properties
(i) It is a white crystalline solid and its melting point \[133{}^\circ C\].
(ii) It is sparingly soluble in water.
(iii) It exhibits geometrical isomerism.
\[\underset{\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\underset{\begin{smallmatrix} \text{ Trans-form} \\ \text{(Cinnamic acid)} \end{smallmatrix}}{\mathop{H-C-COOH}}\,}{\mathop{{{C}_{6}}{{H}_{5}}-\underset{|\,|}{\mathop{C}}\,-H}}\,\]
\[\underset{\begin{smallmatrix} \,\,\,\,\,\,\,\,\,\,\,Cis\text{-form} \\ \text{(Allo cinnamic acid)} \end{smallmatrix}}{\mathop{\underset{HOOC-C-H}{\mathop{{{C}_{6}}{{H}_{5}}-\underset{|\,|}{\mathop{C}}\,-H}}\,}}\,\]
Cinnamic acid (stable form) occurs in nature both free and as esters in balsams and resins.
(3) Chemical properties
Salicylic acid [O-Hydroxy benzoic acid]; 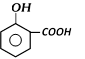
Salicylic acid is present in many essential oils in the form of esters. Oil of winter green is a methyl ester of salicylic acid.
(1) Methods of preparation
(i) Kolbe Schmidt reaction
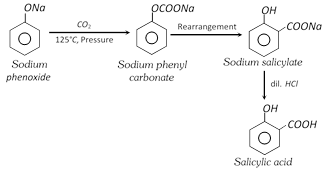
It is a commercial method. The reaction yields both o- and p- isomers. Salicylic acid is more volatile and separated by steam distillation.
(ii) Reimer-Tiemann reaction
![]()
(iii) From benzene derivatives
(a) ![]()
(b) 
(c) 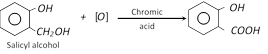
(d) 
(e) 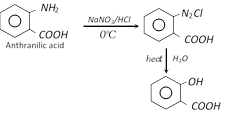
(2) Physical properties
(i) It is a colourless needle shaped crystalline compound.
(ii) Its m.p. is \[{{156}^{o}}C\].
(iii) It is sparingly soluble in cold water but readily soluble in hot water, alcohol, ether and chloroform.
(iv) It is steam volatile.
(v) It is poisonous in nature. However, its derivative used in medicine internally and externally as antipyretic and antiseptic.
(3) Chemical properties
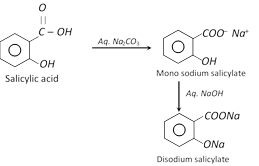
(i) Reaction with \[N{{a}_{2}}C{{O}_{3}},\] \[NaHC{{O}_{3}}\] or \[NaOH\]
(ii) Reaction with alcohols or phenols

Methyl salicylate is an oily liquid (oil of winter green) with pleasant material. It is also used in medicine in the treatment of rheumatic pain and as a remedy for aches, sprains and bruises. It is used in perfumery and as a flavouring. It is used for making of iodex.
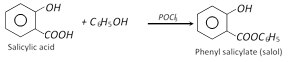
Salol is a white solid m.pt. \[{{43}^{o}}C\]. It is a good internal antiseptic. It is used in making of toothpastes. Salol absorbs ultraviolet light and its main use now is sun-screening agent and stabiliser of plastics.
(iii) Decarboxylation

(iv) Acetylation
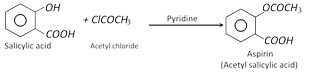
(v) Reaction with ferric chloride solution
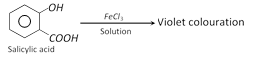
(vi) Reaction with PCl5
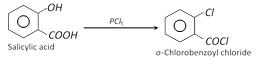
(vii) Bromination
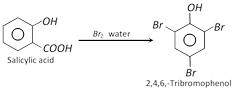
(viii) Nitration

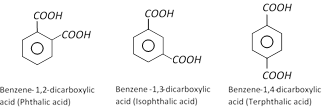
Phthalic acid [1,2,-Benzene dicarboxylic acid]
![]()
There are three isomer (ortho, meta, para) of benzene dicarboxylic acid.
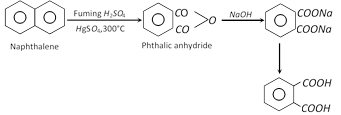
(1) Methods of preparation
(i) By the oxidation of o-xylene :

(ii) From naphthalene (Industrial method) : It is known as aerial oxidation.
(2) Physical properties
(i) It is colourless crystalline compound.
(2) Physical properties
(i) It is colourless crystalline compound.
(ii) Its melting point is not sharp \[(195213{}^\circ C)\].
(iii) It is sparingly soluble in cold water but soluble in hot water, alcohol, ether, benzene etc.
(3) Chemical properties
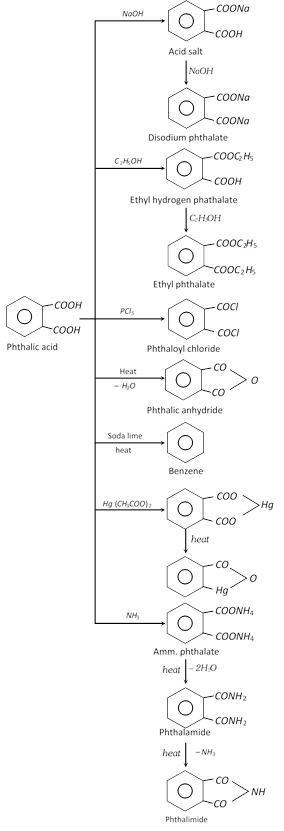
(4) Uses : It is used in the manufacture of plastics, dyes and other compounds such as phthalic anhydride, phthalimide, anthraquinone and fluorescein etc.
You need to login to perform this action.
You will be redirected in
3 sec
