Bond-Line Notation Of Organic Compounds
Category : JEE Main & Advanced
Sometimes, the bonds between carbon atoms are represented by lines. For example, n-hexane has a continuous chain of six carbon atoms which may be represented as,
\[\begin{align} & C{{H}_{3}}-C{{H}_{2}}-C{{H}_{2}}-C{{H}_{2}}-C{{H}_{2}}-C{{H}_{3}} \\ & \,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,n-\text{Hexane} \\ \end{align}\]
![]()
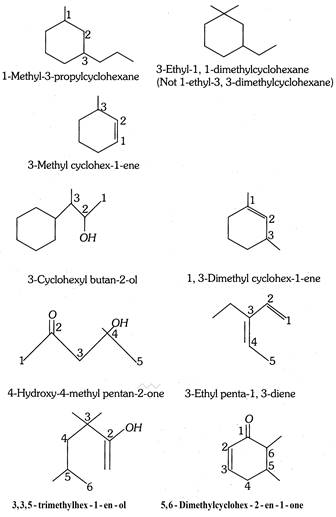
In this notation, the carbon atoms are represented by line ends and intersections. It is assumed that the required number of hydrogen atoms are present wherever they are necessary to satisfy the tetravalency of carbon. A single line represents a single bond (C - C) two parallel lines represent a double bond (C = C) and three parallel lines represent a triple bond \[(C\equiv C)\]. For example,
You need to login to perform this action.
You will be redirected in
3 sec
