-
question_answer1)
Asma lit a perfume stick in the corner of a room. When she stood at the other corner, she smelled the perfume after sometime. What can we conclude from this?
A)
Rooms produce perfume when a perfume stick is lit in a corner. done
clear
B)
All the air present in a room gives perfume. done
clear
C)
Perfumes do not spread from one place to another. done
clear
D)
All gases, including perfumes, have the property of spreading from one place to another. done
clear
View Solution play_arrow
-
question_answer2)
Ramu dissolved some salt in water. He obtained salty water. What is the solute in this experiment?
A)
Salt done
clear
B)
Water done
clear
C)
Salty water done
clear
D)
All of these done
clear
View Solution play_arrow
-
question_answer3)
Ramu dissolved some salt in water. He obtained salty water. What is the solvent in the salty solution?
A)
Salt done
clear
B)
Water done
clear
C)
Salty Water done
clear
D)
All of these done
clear
View Solution play_arrow
-
question_answer4)
The following table contains some substances placed under the headings: solids, liquids and gases.
| Solids | Liquids | Gases |
| Sugar | Water | Steam |
| Salt | Petrol | Oxygen |
| Iron | Milk | Kerosene |
Which of the following is placed in the wrong group?
A)
Salt done
clear
B)
Petrol done
clear
C)
Oxygen done
clear
D)
Kerosene done
clear
View Solution play_arrow
-
question_answer5)
Match the following different states of matter in Column - I with their properties in Column - II.
| Column-I | Column-II |
| p. | Solids | i. | They have no fixed shape and no fixed volume. |
| q. | Liquids | ii. | They have fixed shape and fixed volume. |
| r. | Gases | iii. | They have fixed volume but not fixed shape. |
A)
\[p-i,q-iii,r-ii\] done
clear
B)
\[p-iii.q-i,\text{ }r-ii\] done
clear
C)
\[p-ii,q-iii,\text{ }r-i\] done
clear
D)
\[p-ii,q-i,r-iii\] done
clear
View Solution play_arrow
-
question_answer6)
A student took an unknown substance into a beaker. The substance took the shape of the beaker. Later he transferred the substance into a measuring cylinder. The substance took the shape of the cylinder but its volume remained the same. What is the state of this substance?
A)
Solid done
clear
B)
Gas done
clear
C)
Liquid done
clear
D)
All done
clear
View Solution play_arrow
-
question_answer7)
Study the following figure and answer the questions from 7.
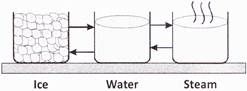
The state of water in the first beaker is
A)
liquid. done
clear
B)
solid. done
clear
C)
gas. done
clear
D)
All of these done
clear
View Solution play_arrow
-
question_answer8)
Study the following figure and answer the questions from 8.

The ice present in the first beaker becomes water upon
A)
cooling. done
clear
B)
heating. done
clear
C)
evaporation. done
clear
D)
condensation. done
clear
View Solution play_arrow
-
question_answer9)
Study the following figure and answer the questions from 9.

To obtain water from water vapour, we should _____ the water vapour.
A)
heat done
clear
B)
evaporate done
clear
C)
condense done
clear
D)
do nothing done
clear
View Solution play_arrow
-
question_answer10)
Which of the following is wrong?
A)
\[Solid~\xrightarrow{heat}Liquid\] done
clear
B)
\[Solid\xrightarrow{Cool}Gas\] done
clear
C)
\[Liquid\xrightarrow{Cool}Solid\] done
clear
D)
\[Liquid~\xrightarrow{heat}Gas\] done
clear
View Solution play_arrow
-
question_answer11)
Which of the following figures shows the arrangement of molecules for solids?
A)
B)
C)
D)
View Solution play_arrow
-
question_answer12)
Ankur has two glasses. One glass is filled with ice cubes and the other is filled with water. Which of the following states that ice is different from water?
A)
The glass with ice cubes is colder than that with water. done
clear
B)
Water is liquid and the ice cubes are solid. done
clear
C)
Water fills more of the glass than ice cubes. done
clear
D)
All of these done
clear
View Solution play_arrow
-
question_answer13)
On cooling water becomes ice (a solid state) liquids take the shape of the container and it fills more of the glass. Study the following table and answer the questions from 13.
| Substance | Solvent | Remark |
| Sand | Water | does not dissolve |
| Sugar | Water | dissolves |
| Salt | Water | dissolves |
| Water | kerosene | does not dissolve |
| Oxygen | Water | dissolves |
Name the solutes which are soluble in water?
A)
Sand and sugar done
clear
B)
Sand and salt done
clear
C)
Salt and kerosene done
clear
D)
Sugar and salt done
clear
View Solution play_arrow
-
question_answer14)
On cooling water becomes ice (a solid state) liquids take the shape of the container and it fills more of the glass. Study the following table and answer the questions from 14.
| Substance | Solvent | Remark |
| Sand | Water | does not dissolve |
| Sugar | Water | dissolves |
| Salt | Water | dissolves |
| Water | kerosene | does not dissolve |
| Oxygen | Water | dissolves |
Which of the following is insoluble substance in water?
A)
Kerosene done
clear
B)
Sugar done
clear
C)
Salt done
clear
D)
Oxygen done
clear
View Solution play_arrow
-
question_answer15)
On cooling water becomes ice (a solid state) liquids take the shape of the container and it fills more of the glass. Study the following table and answer the questions from 15.
| Substance | Solvent | Remark |
| Sand | Water | does not dissolve |
| Sugar | Water | dissolves |
| Salt | Water | dissolves |
| Water | kerosene | does not dissolve |
| Oxygen | Water | dissolves |
Which of the following is the gaseous substance that get dissolved in water?
A)
Sugar in water done
clear
B)
Water in kerosene done
clear
C)
Sand in kerosene done
clear
D)
Oxygen in water done
clear
View Solution play_arrow
-
question_answer16)
Identify the substance that does not get dissolved in water?
A)
Sand done
clear
B)
Oxygen done
clear
C)
Water done
clear
D)
Sugar done
clear
View Solution play_arrow
-
question_answer17)
Which of the following processes involves a decrease in the inter-molecular force between the molecules?
A)
Condensation done
clear
B)
Crystallization done
clear
C)
Evaporation done
clear
D)
All of the above done
clear
View Solution play_arrow
-
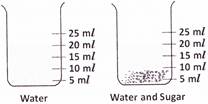
question_answer18)
A student took 20 milliliters of water in a beaker and dissolved two teaspoonful of sugar in it. When he checked the volume, to his surprise, the volume remained same at 20 milliliters. Can you guess the reason?
A)
Sugar did not actually dissolve in the water. done
clear
B)
Sugar evaporated. done
clear
C)
Sugar occupied the empty spaces in between the molecules of water. done
clear
D)
Sugar has zero volume. done
clear
View Solution play_arrow
-
question_answer19)
We can separate sand from water by
A)
filtration. done
clear
B)
evaporation. done
clear
C)
boiling. done
clear
D)
all of these. done
clear
View Solution play_arrow
-
question_answer20)
A student took water and dissolved some salt in it. Later she placed the salty water under sunlight for three days. She found only crystals of salt but no water. The process demonstrated in the above experiment is
A)
filtration. done
clear
B)
boiling. done
clear
C)
condensation. done
clear
D)
evaporation. done
clear
View Solution play_arrow
-
question_answer21)
Which of the following states of matter has the strongest forces of attraction between its molecules?
A)
Solid done
clear
B)
Liquid done
clear
C)
Gas done
clear
D)
None of these done
clear
View Solution play_arrow
-
question_answer22)
The bubbles that come out rapidly when we open soda water bottle are
A)
nitrogen bubbles. done
clear
B)
oxygen bubbles. done
clear
C)
water bubbles. done
clear
D)
carbon dioxide bubbles. done
clear
View Solution play_arrow
-
question_answer23)
Which of the following is/are in a liquid state normally?
A)
Water done
clear
B)
Petrol done
clear
C)
Mercury done
clear
D)
All of them done
clear
View Solution play_arrow
-
question_answer24)
A cube of ice kept on a table melts and becomes water. On heating, the water gets
A)
liquid. done
clear
B)
ice, done
clear
C)
melted. done
clear
D)
evaporated. done
clear
View Solution play_arrow
-
question_answer25)
In which of the following substances is intermolecular space more?
A)
Mercury done
clear
B)
Water done
clear
C)
Hydrogen done
clear
D)
Iron done
clear
View Solution play_arrow
-
question_answer26)
Which of the following are properties of sugar?
| P - Definite shape |
| Q - Definite volume |
| R - Can be compressed |
A)
Only P and Q done
clear
B)
Only Q and R done
clear
C)
Only P and R done
clear
D)
P. Q and R done
clear
View Solution play_arrow
-
question_answer27)
Which of the following sequences is correct about the changes in state of water in the water cycle?
A)
Solid \[\to \] Gas \[\to \] Liquid done
clear
B)
Gas \[\to \] Solid \[\to \] Liquid done
clear
C)
Liquid \[\to \] Gas \[\to \] Liquid done
clear
D)
Liquid \[\to \] Solid \[\to \] Gas done
clear
View Solution play_arrow
-
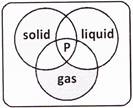
question_answer28)
Which of the following can be placed at '?' in the Venn diagram given below?
A)
Oxygen done
clear
B)
Water done
clear
C)
Clay done
clear
D)
Paper done
clear
View Solution play_arrow
-
question_answer29)
Lucky left a cup of hot coffee on his table. He noticed something rising out of the surface of the coffee.
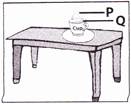
Which of the following describes correctly how the particles are arranged at P and Q?
A)
B)
C)
D)
View Solution play_arrow
-
question_answer30)
Mary filled an empty beaker to the brim with water and dropped some marbles too as shown below.
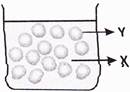
What are the states of substances labelled as X and Y?
A)
X-solid, Y-liquid done
clear
B)
X-liquid, Y-solid done
clear
C)
X-solid, Y-gas done
clear
D)
X-Liquid, Y-gas done
clear
View Solution play_arrow
-
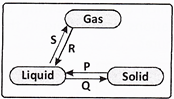
question_answer31)
Which of the following labelled arrows in the given figure represents the process of evaporation?
A)
P done
clear
B)
Q done
clear
C)
R done
clear
D)
S done
clear
View Solution play_arrow
-
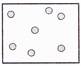
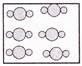
question_answer32)
Study the properties of X given below.
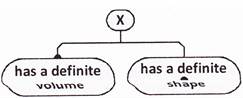
Which of the following can X represent?
A)
Water done
clear
B)
Sugar done
clear
C)
Oxygen done
clear
D)
Steam done
clear
View Solution play_arrow
-
question_answer33)
Which of these substances has definite volume but no fixed shape?
A)
Book done
clear
B)
Oxygen done
clear
C)
Water vapour done
clear
D)
Lemon juice done
clear
View Solution play_arrow
-
question_answer34)
A Which of the following has more amount of air?
A)
B)
C)
D)
View Solution play_arrow
-
question_answer35)
Which of the following can increase in volume when air is blown into it?
A)
B)
C)
D)
View Solution play_arrow
-
question_answer36)
Which of the following transformations is occurring in the given figure?
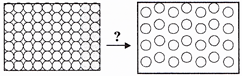
A)
Condensation done
clear
B)
Melting done
clear
C)
Evaporation done
clear
D)
Boiling done
clear
View Solution play_arrow
-
question_answer37)
Which of the following is an outcome of condensation?
A)
Wind done
clear
B)
Storm done
clear
C)
Rain done
clear
D)
Gale done
clear
View Solution play_arrow
-
question_answer38)
Equal amounts of water were taken in four containers in the following manner.
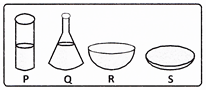
In which of the containers will water evaporate the fastest?
A)
P done
clear
B)
Q done
clear
C)
R done
clear
D)
S done
clear
View Solution play_arrow
-
question_answer39)
Which of the following conversions is correct?
A)
\[\text{Water }\xrightarrow{\text{heat}}\text{Steam }\xrightarrow{\text{heat}}\text{ Ice}\] done
clear
B)
\[\text{Steam }\xrightarrow{cool}\text{Water }\xrightarrow{cool}\text{ Ice}\] done
clear
C)
\[\text{Water}\xrightarrow{cool}\text{Ice }\xrightarrow{cool}Steam\] done
clear
D)
\[\text{Steam }\xrightarrow{heat}\text{Ice }\xrightarrow{cool}Water\] done
clear
View Solution play_arrow
-
question_answer40)
The figures given below shows the outcome when X was added to Y.
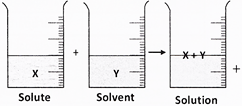
This shows that ___
A)
X is completely soluble in Y. done
clear
B)
X is completely insoluble in Y. done
clear
C)
Y does not dissolves X. done
clear
D)
Y is partially insoluble in X. done
clear
View Solution play_arrow
 done
clear
done
clear
 done
clear
done
clear
 done
clear
done
clear
 done
clear
done
clear

 Which of the following can X represent?
Which of the following can X represent? 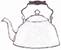 done
clear
done
clear
 done
clear
done
clear

