-
question_answer1)
Which has zero dipole moment [NCERT 1990; BHU 2001]
A)
cis-2-butene done
clear
B)
trans-2-butene done
clear
C)
1-butene done
clear
D)
2-methyl-1-propene done
clear
View Solution play_arrow
-
question_answer2)
Dipole moment is shown by [DCE 1999]
A)
1, 4-dichloro benzene done
clear
B)
Cis-1, 2-dichloro ethane done
clear
C)
Trans-1, 2-dichloro, 2-pentene done
clear
D)
Trans-1, 2-dichloro ether done
clear
View Solution play_arrow
-
question_answer3)
Which compound shows dipole moment [RPMT 2002]
A)
1,4-di-chloro benzene done
clear
B)
1, 2-di-chloro benzene done
clear
C)
Trans-1, 2-di-chloro ethene done
clear
D)
Trans-2-butene done
clear
View Solution play_arrow
-
question_answer4)
Which of the following is a polar compound [MH CET 2003]
A)
\[{{C}_{2}}{{H}_{6}}\] done
clear
B)
\[CC{{l}_{4}}\] done
clear
C)
HCl done
clear
D)
\[C{{H}_{4}}\] done
clear
View Solution play_arrow
-
question_answer5)
The dipole moment is the highest for [AIIMS 2004]
A)
Trans-2-butene done
clear
B)
1, 3-Dimethylbenzene done
clear
C)
Acetophenone done
clear
D)
Ethanol done
clear
View Solution play_arrow
-
question_answer6)
Resonance structure of molecule does not have [IIT-JEE 1984]
A)
Identical arrangement of atoms done
clear
B)
Nearly the same energy content done
clear
C)
The same number of paired electrons done
clear
D)
Identical bonding done
clear
View Solution play_arrow
-
question_answer7)
All bonds in benzene are equal due to [Roorkee 1990; KCET 1998]
A)
Tautomerism done
clear
B)
Inductive effect done
clear
C)
Resonance done
clear
D)
Isomerism done
clear
View Solution play_arrow
-
question_answer8)
Aromatic properties of benzene are proved by [MP PMT 1994]
A)
Aromatic sextet theory done
clear
B)
Resonance theory done
clear
C)
Molecular orbital theory done
clear
D)
All of these done
clear
View Solution play_arrow
-
question_answer9)
Which of the following will show aromatic behaviour [KCET 1996]
A)
B)
C)
D)
View Solution play_arrow
-
question_answer10)
Which one of the following orders is correct regarding the inductive effect of the substituents [CBSE PMT 1998]
A)
\[-N{{R}_{2}}<-OR>-F\] done
clear
B)
\[-N{{R}_{2}}>-OR>-F\] done
clear
C)
\[-N{{R}_{2}}<-OR<-F\] done
clear
D)
\[-N{{R}_{2}}>-OR<-F\] done
clear
View Solution play_arrow
-
question_answer11)
Benzene is unreactive because [KCET 1998]
A)
It has double bonds done
clear
B)
It has carbon-carbon single bond done
clear
C)
Carbon are \[s{{p}^{2}}\] hybridised done
clear
D)
\[\pi \] electrons are delocalised done
clear
View Solution play_arrow
-
question_answer12)
Carboxylic acids are easily ionised. The main reason of this statement [UPSEAT 1999]
A)
Absence of a-hydrogen done
clear
B)
Resonance stabilisation of carboxylate ion done
clear
C)
Reactivity of a-hydrogen done
clear
D)
Hydrogen bond done
clear
View Solution play_arrow
-
question_answer13)
\['C-C'\] bond length in benzene lies between single and double bond. The reason is [RPET 1999]
A)
Resonance done
clear
B)
Isomerism done
clear
C)
Metamerism done
clear
D)
Inductive effect done
clear
View Solution play_arrow
-
question_answer14)
Credit for the ring structure of benzene goes to [RPET 1999]
A)
Wholer done
clear
B)
Faraday done
clear
C)
Kekule done
clear
D)
Baeyer done
clear
View Solution play_arrow
-
question_answer15)
Polarisation of electrons in acroline may be written as [DCE 2000]
A)
\[CH_{2}^{\delta -}=CH-C{{H}^{\delta +}}=O\] done
clear
B)
\[CH_{2}^{\delta -}=CH-CH={{O}^{\delta +}}\] done
clear
C)
\[CH_{2}^{\delta -}=C{{H}^{\delta +}}-CH=O\] done
clear
D)
\[CH_{2}^{\delta +}=CH-CH={{O}^{\delta -}}\] done
clear
View Solution play_arrow
-
question_answer16)
In the mixture of conc. \[{{H}_{2}}S{{O}_{4}}\] and \[HN{{O}_{3}}\]the nitrating species is [MP PMT 2000]
A)
\[{{N}_{2}}{{O}_{4}}\] done
clear
B)
\[NO_{2}^{+}\] done
clear
C)
\[N{{O}_{2}}\] done
clear
D)
\[NO_{2}^{-}\] done
clear
View Solution play_arrow
-
question_answer17)
Which of the following are not aromatic [DCE 2001]
A)
Benzene done
clear
B)
Cyclo-octatetrarenyl dianion done
clear
C)
Tropyllium cation done
clear
D)
Cyclopentadienyl cation done
clear
View Solution play_arrow
-
question_answer18)
Arrangement of \[{{(C{{H}_{3}})}_{3}}-C-{{(C{{H}_{2}})}_{3}}-CH-C{{H}_{3}}-C{{H}_{2}}-C{{H}_{3}}\] when attached to benzyl or an unsaturated group in increasing order of inductive effect is [AIEEE 2002]
A)
\[{{(C{{H}_{3}})}_{3}}-C-<{{(C{{H}_{3}})}_{2}}-CH-<C{{H}_{3}}-C{{H}_{2}}-\] done
clear
B)
\[C{{H}_{3}}-C{{H}_{2}}-<{{(C{{H}_{3}})}_{2}}-CH-<{{(C{{H}_{3}})}_{3}}-C-\] done
clear
C)
\[{{(C{{H}_{3}})}_{2}}-CH-<{{(C{{H}_{3}})}_{3}}-C-<C{{H}_{3}}-C{{H}_{2}}-\] done
clear
D)
\[{{(C{{H}_{2}})}_{3}}-C-<C{{H}_{3}}-C{{H}_{2}}-<{{(C{{H}_{3}})}_{2}}-CH-\] done
clear
View Solution play_arrow
-
question_answer19)
Which of the following is observed in ethylene molecule [MH CET 2002]
A)
Electromeric effect done
clear
B)
Inductive effect done
clear
C)
Homolytic fission done
clear
D)
None of these done
clear
View Solution play_arrow
-
question_answer20)
Cyclopentadienyl anion is [Orissa JEE 2003]
A)
Aromatic done
clear
B)
Non-aromatic done
clear
C)
Non-planar done
clear
D)
Aliphatic done
clear
View Solution play_arrow
-
question_answer21)
Orbital interaction between the sigma bonds of a substitutent group and a neighbouring pi orbital is known as [Kerala PMT 2004]
A)
Hyperconjugation done
clear
B)
Inductive effect done
clear
C)
Steric effect done
clear
D)
Dipole-dipole interactions done
clear
E)
Electric quadruple interactions done
clear
View Solution play_arrow
-
question_answer22)
Which of the following is the most stable compound [BHU 2004]
A)
\[P{{h}_{3}}\overset{+}{\mathop{C}}\,\] done
clear
B)
\[P{{h}_{2}}\overset{+}{\mathop{C}}\,H\] done
clear
C)
\[P{{h}_{3}}\overset{+}{\mathop{C}}\,{{H}_{2}}\] done
clear
D)
\[Ph\overset{+}{\mathop{C}}\,{{H}_{2}}\] done
clear
View Solution play_arrow
-
question_answer23)
Which of the following will be most easily attacked by an electrophile [MP PET 2004]
A)
B)
C)
D)
View Solution play_arrow
-
question_answer24)
Reactivity towards nucleophilic addition reaction of (I) HCHO, (II) \[C{{H}_{3}}CHO\], (III) \[C{{H}_{3}}COC{{H}_{3}}\] is [Orissa JEE 2004]
A)
II > III > I done
clear
B)
III > II > I done
clear
C)
I > II > III done
clear
D)
I > II < III done
clear
View Solution play_arrow
-
question_answer25)
Which of the following resonating structures of 1-methoxy-1, 3-butadiene is least stable [IIT Screening 2005]
A)
\[\overset{}{\mathop{C}}\,{{H}_{2}}-CH=CH-CH=\overset{\oplus }{\mathop{O}}\,-C{{H}_{3}}\] done
clear
B)
\[C{{H}_{2}}=C{{H}_{2}}-\overset{}{\mathop{C}}\,H-CH=\overset{\oplus }{\mathop{O}}\,-C{{H}_{3}}\] done
clear
C)
\[\overset{}{\mathop{C}}\,{{H}_{2}}-\overset{\oplus }{\mathop{C}}\,H-CH=CH-O-C{{H}_{3}}\] done
clear
D)
\[C{{H}_{2}}=CH-\overset{}{\mathop{C}}\,H-\overset{\oplus }{\mathop{C}}\,H-O-C{{H}_{3}}\] done
clear
View Solution play_arrow
-
question_answer26)
Which amongst the following is the most stable carbocation [CBSE PMT 2005]
A)
\[C{{H}_{3}}-\underset{C{{H}_{3}}}{\mathop{\underset{|}{\overset{+}{\mathop{C}}}\,\ \ \ \ }}\,\] done
clear
B)
\[C{{H}_{3}}-\underset{C{{H}_{3}}}{\overset{C{{H}_{3}}}{\mathop{\underset{|\ \ }{\overset{|\ \ }{\mathop{{{C}^{+}}}}}\,\ \ }}}\,\] done
clear
C)
\[\overset{+}{\mathop{C}}\,{{H}_{3}}\] done
clear
D)
\[C{{H}_{3}}\overset{+}{\mathop{C}}\,{{H}_{3}}\] done
clear
View Solution play_arrow
-
question_answer27)
Which is the decreasing order of stability [IIT-JEE (Screening) 1993] (i) \[C{{H}_{3}}-\overset{+}{\mathop{C}}\,H-C{{H}_{3}}\] (ii) \[C{{H}_{3}}-\overset{+}{\mathop{C}}\,H-O-C{{H}_{3}}\] (iii) \[C{{H}_{3}}-\overset{+}{\mathop{C}}\,H-CO-C{{H}_{3}}\]
A)
\[(i)<(ii)<(iii)\] done
clear
B)
\[(i)>(ii)>(iii)\] done
clear
C)
\[(iii)>(ii)>(i)\] done
clear
D)
\[(ii)>(iii)>(i)\] done
clear
View Solution play_arrow
-
question_answer28)
The order of decreasing stability of the carbanions (1) \[{{(C{{H}_{3}})}_{3}}\bar{\ddot{C}}\] (2) \[{{(C{{H}_{3}})}_{2}}\bar{\ddot{C}}H\] (3) \[C{{H}_{3}}\bar{\ddot{C}}{{H}_{2}}\] (4) \[{{C}_{6}}{{H}_{5}}\bar{\ddot{C}}{{H}_{2}}\] is [KCET 1996]
A)
1 > 2 > 3 > 4 done
clear
B)
4 > 3 > 2 > 1 done
clear
C)
4 > 1 > 2 > 3 done
clear
D)
1 > 2 > 4 > 3 done
clear
View Solution play_arrow
-
question_answer29)
Choose the chain terminating step (1) \[{{H}_{2}}\to {{H}^{\bullet }}+{{H}^{\bullet }}\] (2) \[B{{r}_{2}}\to B{{r}^{\bullet }}+B{{r}^{\bullet }}\] (3) \[B{{r}^{\bullet }}+HBr\to {{H}^{\bullet }}+B{{r}_{2}}\] (4) \[{{H}^{\bullet }}+B{{r}_{2}}\to HBr+B{{r}^{\bullet }}\] (5) \[B{{r}^{\bullet }}+B{{r}^{\bullet }}\to B{{r}_{2}}\] [RPET 2000]
A)
1 done
clear
B)
3 done
clear
C)
4 done
clear
D)
5 done
clear
View Solution play_arrow
-
question_answer30)
The compound, which gives the most stable carbonium on dehydrogenation [UPSEAT 2001]
A)
\[C{{H}_{3}}-\underset{C{{H}_{3}}}{\mathop{\underset{|\,\,\,\,\,\,\,\,}{\mathop{CH}}\,}}\,-C{{H}_{2}}OH\] done
clear
B)
\[C{{H}_{3}}-\underset{C{{H}_{3}}}{\overset{C{{H}_{3}}}{\mathop{\underset{|\,\,\,\,\,\,\,\,}{\overset{|\,\,\,\,\,\,\,\,\,}{\mathop{C\,\,\,-\,}}}\,}}}\,OH\] done
clear
C)
\[C{{H}_{3}}-C{{H}_{2}}-C{{H}_{2}}-C{{H}_{2}}OH\] done
clear
D)
\[C{{H}_{3}}-\underset{C{{H}_{3}}}{\mathop{\underset{|\,\,\,\,\,\,\,}{\mathop{CH}}\,}}\,-C{{H}_{2}}-C{{H}_{3}}\] done
clear
View Solution play_arrow
-
question_answer31)
Which of the following requires radical intermediate [Orissa JEE 2004]
A)
\[C{{H}_{3}}-CH=C{{H}_{2}}+HBr\to C{{H}_{3}}-\underset{Br\ }{\mathop{\underset{|}{\mathop{C}}\,H}}\,-C{{H}_{3}}\] done
clear
B)
\[C{{H}_{3}}-CHO+HCN\to C{{H}_{3}}-CH<\begin{matrix} CN \\ OH \\ \end{matrix}\] done
clear
C)
\[C{{H}_{3}}-CH=C{{H}_{2}}+HBr\to C{{H}_{3}}-C{{H}_{2}}-C{{H}_{2}}-Br\] done
clear
D)
\[C{{H}_{3}}CHO+N{{H}_{2}}OH\xrightarrow{{{H}^{+}}}C{{H}_{3}}-CH=N-OH\] done
clear
View Solution play_arrow
-
question_answer32)
Which of the following species is paramagnetic in nature [NCERT 1984]
A)
Free radical done
clear
B)
Carbonium ion done
clear
C)
Carbanion done
clear
D)
All the above done
clear
View Solution play_arrow
-
question_answer33)
In which of the following species the central C-atom is negatively charged [NCERT 1985]
A)
Carbanion done
clear
B)
Carbonium ion done
clear
C)
Carbocation done
clear
D)
Free radical done
clear
View Solution play_arrow
-
question_answer34)
Which of the following free radicals is most stable [NCERT 1982]
A)
Primary done
clear
B)
Methyl done
clear
C)
Secondary done
clear
D)
Tertiary done
clear
View Solution play_arrow
-
question_answer35)
Which of the following contains three pairs of electrons [BHU 1985]
A)
Carbocation done
clear
B)
Carbanion done
clear
C)
Free radical done
clear
D)
None of these done
clear
View Solution play_arrow
-
question_answer36)
Which of the following carbanion is most stable [NCERT 1983]
A)
Methyl done
clear
B)
Primary done
clear
C)
Secondary done
clear
D)
Tertiary done
clear
View Solution play_arrow
-
question_answer37)
Among the given cations, the most stable carbonium ion is [IIT-JEE 1981]
A)
sec-butyl done
clear
B)
ter-butyl done
clear
C)
n-butyl done
clear
D)
None of these done
clear
View Solution play_arrow
-
question_answer38)
In the compound given below
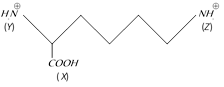
The correct order of the acidity of the positions (X), (Y) and (Z) is [IIT-JEE Screening 2004]
A)
(Z) > (X) > (Y) done
clear
B)
(X) > (Y) > (Z) done
clear
C)
(X) > (Z) > (Y) done
clear
D)
(Y) > (X) > (Z) done
clear
View Solution play_arrow
-
question_answer39)
C-C bond length in benzene is [MP PMT 1987; MP PMT 2001; AIIMS 2001]
A)
\[1.39\,\overset{\circ }{\mathop{\text{A}}}\,\] done
clear
B)
\[1.54\,\overset{\circ }{\mathop{\text{A}}}\,\] done
clear
C)
\[1.34\,\overset{\circ }{\mathop{\text{A}}}\,\] done
clear
D)
Different in different bonds done
clear
View Solution play_arrow
-
question_answer40)
Heterolysis of carbon-chlorine bond produces [MNR 1986; MP PET/PMT 1998]
A)
Two free radicals done
clear
B)
Two carbonium ions done
clear
C)
Two carbanions done
clear
D)
One cation and one anion done
clear
View Solution play_arrow
-
question_answer41)
In \[C{{H}_{3}}C{{H}_{2}}OH,\] the bond that undergoes heterolytic cleavage most readily is [IIT-JEE 1988]
A)
C - C done
clear
B)
C - O done
clear
C)
C - H done
clear
D)
O - H done
clear
View Solution play_arrow
-
question_answer42)
Which of the following intermediate have the complete octet around the carbon atom [Orissa JEE 2003]
A)
Carbonium ion done
clear
B)
Carbanion ion done
clear
C)
Free radical done
clear
D)
Carbene done
clear
View Solution play_arrow
-
question_answer43)
A solution of D (+) - 2-chloro-2-phenylethane in toluene racemises slowly in the presence of small amount of \[SbC{{l}_{5}},\] due to the formation of [IIT-JEE 1999]
A)
Carbanion done
clear
B)
Carbene done
clear
C)
Free radical done
clear
D)
Carbocation done
clear
View Solution play_arrow
-
question_answer44)
The reagent in Friedel Craft's reaction is [DPMT 2005]
A)
Pyridine done
clear
B)
\[RCOCl\] done
clear
C)
\[RCOOH\] done
clear
D)
\[HCl\] done
clear
View Solution play_arrow
-
question_answer45)
Which gives monosubstituted product [DPMT 2005]
A)
\[o\]-dinitrobenzene done
clear
B)
\[m\]-dinitrobenzene done
clear
C)
\[p\]-dinitrobenzene done
clear
D)
Nitrobenzene done
clear
View Solution play_arrow
-
question_answer46)
An aromatic compounds among other things should have a \[\pi \]-electron cloud containing electrons where \[n\] can't be [J & K 2005]
A)
1/2 done
clear
B)
3 done
clear
C)
2 done
clear
D)
1 done
clear
View Solution play_arrow
-
question_answer47)
Which of the following is an electrophile [J & K 2005]
A)
\[{{H}_{2}}O\] done
clear
B)
\[S{{O}_{3}}\] done
clear
C)
\[N{{H}_{3}}\] done
clear
D)
\[ROR\] done
clear
View Solution play_arrow
-
question_answer48)
The presence of the chlorine atom on benzene ring makes the second substituent enter at a position [J & K 2005]
A)
ortho done
clear
B)
meta done
clear
C)
para done
clear
D)
ortholpara done
clear
View Solution play_arrow
-
question_answer49)
Which is the most stable carbocation [J & K 2005]
A)
iso-propyl done
clear
B)
Triphenylmethyl cation done
clear
C)
Ethyl cation done
clear
D)
\[\pi \]-propyl cation done
clear
View Solution play_arrow
