-
question_answer1)
The value of surface tension of a liquid at critical temperature is [AIIMS 1980]
A)
Zero done
clear
B)
Infinite done
clear
C)
Between 0 and \[\infty \] done
clear
D)
Cannot be determined done
clear
View Solution play_arrow
-
question_answer2)
The spherical shape of rain-drop is due to [CPMT 1976, 90; NCERT 1982; AIIMS 1998; MH CET 2000; DCE 1999; AFMC 1999; CPMT 2001; AFMC 2001]
A)
Density of the liquid done
clear
B)
Surface tension done
clear
C)
Atmospheric pressure done
clear
D)
Gravity done
clear
View Solution play_arrow
-
question_answer3)
Surface tension is due to
A)
Frictional forces between molecules done
clear
B)
Cohesive forces between molecules done
clear
C)
Adhesive forces between molecules done
clear
D)
Gravitational forces done
clear
View Solution play_arrow
-
question_answer4)
When there is no external force, the shape of a liquid drop is determined by [CPMT 1988, 86; DPMT 1982]
A)
Surface tension of the liquid done
clear
B)
Density of liquid done
clear
C)
Viscosity of liquid done
clear
D)
Temperature of air only done
clear
View Solution play_arrow
-
question_answer5)
Soap helps in cleaning clothes, because [DPMT 1983, 2001]
A)
Chemicals of soap change done
clear
B)
It increases the surface tension of the solution done
clear
C)
It absorbs the dirt done
clear
D)
It lowers the surface tension of the solution done
clear
View Solution play_arrow
-
question_answer6)
A pin or a needle floats on the surface of water, the reason for this is [MP PET/PMT 1988; CPMT 1975]
A)
Surface tension done
clear
B)
Less weight done
clear
C)
Upthrust of liquid done
clear
D)
None of the above done
clear
View Solution play_arrow
-
question_answer7)
Coatings used on raincoat are waterproof because
A)
Water is absorbed by the coating done
clear
B)
Cohesive force becomes greater done
clear
C)
Water is not scattered away by the coating done
clear
D)
Angle of contact decreases done
clear
View Solution play_arrow
-
question_answer8)
If temperature increases, the surface tension of a liquid [MP PMT 1994; EAMCET (Engg.) 1995; RPET 2003]
A)
Increases done
clear
B)
Decreases done
clear
C)
Remains the same done
clear
D)
Increases then decreases done
clear
View Solution play_arrow
-
question_answer9)
A drop of oil is placed on the surface of water. Which of the following statement is correct [NCERT 1976; DPMT 1982]
A)
It will remain on it as a sphere done
clear
B)
It will spread as a thin layer done
clear
C)
It will be partly as spherical droplets and partly as thin film done
clear
D)
It will float as a distorted drop on the water surface done
clear
View Solution play_arrow
-
question_answer10)
The temperature at which the surface tension of water is zero
A)
\[0{}^\circ C\] done
clear
B)
277 K done
clear
C)
\[370{}^\circ C\] done
clear
D)
Slightly less than 647 K done
clear
View Solution play_arrow
-
question_answer11)
A small air bubble is at the inner surface of the bottom of a beaker filled with cold water. Now water of the beaker is heated. The size of bubble increases. The reason for this may be
A)
Increase in the saturated vapour pressure of water done
clear
B)
Root mean square velocity of air molecules inside the bubble increases done
clear
C)
Decrease in surface tension of water done
clear
D)
All of the above done
clear
View Solution play_arrow
-
question_answer12)
The spiders and insects move and run about on the surface of water without sinking because
A)
Elastic membrane is formed on water due to property of surface tension done
clear
B)
Spiders and insects are lighter done
clear
C)
Spiders and insects swim on water done
clear
D)
Spider and insects experience upthrust done
clear
View Solution play_arrow
-
question_answer13)
Small droplets of a liquid are usually more spherical in shape than larger drops of the same liquid because [EAMCET 1988]
A)
Force of surface tension is equal and opposite to the force of gravity done
clear
B)
Force of surface tension predominates the force of gravity done
clear
C)
Force of gravity predominates the force of surface tension done
clear
D)
Force of gravity and force of surface tension act in the same direction and are equal done
clear
View Solution play_arrow
-
question_answer14)
Hairs of shaving brush cling together when it is removed from water due to
A)
Force of attraction between hair done
clear
B)
Surface tension done
clear
C)
Viscosity of water done
clear
D)
Characteristic property of hairs done
clear
View Solution play_arrow
-
question_answer15)
A square frame of side L is dipped in a liquid. On taking out, a membrane is formed. If the surface tension of the liquid is T, the force acting on the frame will be [MP PMT 1990; DPMT 2004]
A)
2 TL done
clear
B)
4 TL done
clear
C)
8 TL done
clear
D)
10 TL done
clear
View Solution play_arrow
-
question_answer16)
Water does not wet an oily glass because
A)
Cohesive force of oil>> adhesive force between oil and glass done
clear
B)
Cohesive force of oil > cohesive force of water done
clear
C)
Oil repels water done
clear
D)
Cohesive force for water > adhesive force between water and oil molecules done
clear
View Solution play_arrow
-
question_answer17)
A water drop takes the shape of a sphere in a oil while the oil drop spreads in water, because
A)
C.F. for water > A.F. for water and oil done
clear
B)
C.F. for oil > A.F. for water and oil done
clear
C)
C.F. for oil < A.F. for water and oil done
clear
D)
None of the above (A.F. = adhesive force C.F. = cohesive force) done
clear
View Solution play_arrow
-
question_answer18)
Which of the fact is not due to surface tension
A)
Dancing of a camphor piece over the surface of water done
clear
B)
Small mercury drop itself becomes spherical done
clear
C)
A liquid surface comes at rest after stirring done
clear
D)
Mercury does not wet the glass vessel done
clear
View Solution play_arrow
-
question_answer19)
In the glass capillary tube, the shape of the surface of the liquid depends upon [MP PMT 1989]
A)
Only on the cohesive force of liquid molecules done
clear
B)
Only on the adhesive force between the molecules of glass and liquid done
clear
C)
Only on relative cohesive and adhesive force between the atoms done
clear
D)
Neither on cohesive nor on adhesive force done
clear
View Solution play_arrow
-
question_answer20)
Force necessary to pull a circular plate of 5 cm radius from water surface for which surface tension is 75 dynes/cm, is [MP PMT 1991]
A)
\[30dyne\] done
clear
B)
60 dynes done
clear
C)
750 dynes done
clear
D)
750 \[\pi \]dynes done
clear
View Solution play_arrow
-
question_answer21)
The property of surface tension is obtained in
A)
Solids, liquids and gases done
clear
B)
Liquids done
clear
C)
Gases done
clear
D)
Matter done
clear
View Solution play_arrow
-
question_answer22)
The surface tension of a liquid [MNR 1990]
A)
Increases with area done
clear
B)
Decreases with area done
clear
C)
Increase with temperature done
clear
D)
Decrease with temperature done
clear
View Solution play_arrow
-
question_answer23)
If two glass plates are quite nearer to each other in water, then there will be force of
A)
Attraction done
clear
B)
Repulsion done
clear
C)
Attraction or repulsion done
clear
D)
None of the above done
clear
View Solution play_arrow
-
question_answer24)
On mixing the salt in water, the surface tension of water will
A)
Increase done
clear
B)
Decrease done
clear
C)
Remain unchanged done
clear
D)
None of the above done
clear
View Solution play_arrow
-
question_answer25)
The maximum force, in addition to the weight required to pull a wire of 5.0 cm long from the surface of water at temperature\[{{20}^{o}}C\], is 728 dynes. The surface tension of water is
A)
7.28 N/cm done
clear
B)
7.28 dyne/cm done
clear
C)
72.8 dyne/cm done
clear
D)
\[7.28\times {{10}^{2}}\]dyne/cm done
clear
View Solution play_arrow
-
question_answer26)
Consider a liquid contained in a vessel. The liquid solid adhesive force is very weak as compared to the cohesive force in the liquid. The shape of the liquid surface near the solid shall be [MNR 1994]
A)
Horizontal done
clear
B)
Almost vertical done
clear
C)
Concave done
clear
D)
Convex done
clear
View Solution play_arrow
-
question_answer27)
At which of the following temperatures, the value of surface tension of water is minimum [MP PMT/PET 1998]
A)
\[{{4}^{o}}C\] done
clear
B)
\[{{25}^{o}}C\] done
clear
C)
\[{{50}^{o}}C\] done
clear
D)
\[{{75}^{o}}C\] done
clear
View Solution play_arrow
-
question_answer28)
If a glass rod is dipped in mercury and withdrawn out, the mercury does not wet the rod because [MP PET 1995]
A)
Angle of contact is acute done
clear
B)
Cohesion force is more done
clear
C)
Adhesion force is more done
clear
D)
Density of mercury is more done
clear
View Solution play_arrow
-
question_answer29)
Mercury does not wet glass, wood or iron because [MP PET 1997]
A)
Cohesive force is less than adhesive force done
clear
B)
Cohesive force is greater than adhesive force done
clear
C)
Angle of contact is less than \[{{90}^{o}}\] done
clear
D)
Cohesive force is equal to adhesive force done
clear
View Solution play_arrow
-
question_answer30)
Surface tension of a liquid is found to be influenced by [ISM Dhanbad 1994]
A)
It increases with the increase of temperature done
clear
B)
Nature of the liquid in contact done
clear
C)
Presence of soap that increases it done
clear
D)
Its variation with the concentration of the liquid done
clear
View Solution play_arrow
-
question_answer31)
When a drop of water is dropped on oil surface, then [RPMT 1997]
A)
It will mix up with oil done
clear
B)
It spreads in the form of a film done
clear
C)
It will deform done
clear
D)
It remains spherical done
clear
View Solution play_arrow
-
question_answer32)
Two pieces of glass plate one upon the other with a little water in between them cannot be separated easily because of [JIPMER 1997]
A)
Inertia done
clear
B)
Pressure done
clear
C)
Surface tension done
clear
D)
Viscosity done
clear
View Solution play_arrow
-
question_answer33)
Small liquid drops assume spherical shape because [JIPMER 1997]
A)
Atmospheric pressure exerts a force on a liquid drop done
clear
B)
Volume of a spherical drop is minimum done
clear
C)
Gravitational force acts upon the drop done
clear
D)
Liquid tends to have the minimum surface area due to surface tension done
clear
View Solution play_arrow
-
question_answer34)
A thin metal disc of radius r floats on water surface and bends the surface downwards along the perimeter making an angle q with vertical edge of the disc. If the disc displaces a weight of water W and surface tension of water is T, then the weight of metal disc is [AMU (Med.) 1999]
A)
\[2\pi rT+W\] done
clear
B)
\[2\pi rT\cos \theta -W\] done
clear
C)
\[\text{2}\pi \text{rTcos}\theta +W\] done
clear
D)
\[W-2\pi rT\cos \theta \] done
clear
View Solution play_arrow
-
question_answer35)
A 10 cm long wire is placed horizontally on the surface of water and is gently pulled up with a force of \[2\text{ }\times {{10}^{-2}}N\]to keep the wire in equilibrium. The surface tension, in\[N{{m}^{-1}}\], of water is [AMU (Med.) 1999]
A)
0.1 done
clear
B)
0.2 done
clear
C)
0.001 done
clear
D)
0.002 done
clear
View Solution play_arrow
-
question_answer36)
It is easy to wash clothes in hot water because its [RPMT 2000]
A)
Surface tension is more done
clear
B)
Surface tension is less done
clear
C)
Consumes less soap done
clear
D)
None of these done
clear
View Solution play_arrow
-
question_answer37)
Due to which property of water, tiny particles of camphor dance on the surface of water [RPMT 1999]
A)
Viscosity done
clear
B)
Surface tension done
clear
C)
Weight done
clear
D)
Floating force done
clear
View Solution play_arrow
-
question_answer38)
The force required to separate two glass plates of area \[{{10}^{-2}}{{m}^{2}}\] with a film of water 0.05 mm thick between them, is (Surface tension of water is \[70\times {{10}^{-3}}\]N/m) [KCET 2000; Pb. PET 2001: RPET 2002]
A)
28 N done
clear
B)
14 N done
clear
C)
50 N done
clear
D)
38 N done
clear
View Solution play_arrow
-
question_answer39)
Oil spreads over the surface of water whereas water does not spread over the surface of the oil, due to [MH CET 2001]
A)
Surface tension of water is very high done
clear
B)
Surface tension of water is very low done
clear
C)
Viscosity of oil is high done
clear
D)
Viscosity of water is high done
clear
View Solution play_arrow
-
question_answer40)
Cohesive force is experienced between [MH CET 2001]
A)
Magnetic substances done
clear
B)
Molecules of different substances done
clear
C)
Molecules of same substances done
clear
D)
None of these done
clear
View Solution play_arrow
-
question_answer41)
The property utilized in the manufacture of lead shots is [AIIMS 2002]
A)
Specific weight of liquid lead done
clear
B)
Specific gravity of liquid lead done
clear
C)
Compressibility of liquid lead done
clear
D)
Surface tension of liquid lead done
clear
View Solution play_arrow
-
question_answer42)
The dimensions of surface tension are [MH CET 2002]
A)
\[[ML{{T}^{-1}}]\] done
clear
B)
\[[M{{L}^{2}}{{T}^{-2}}]\] done
clear
C)
\[[M{{L}^{0}}{{T}^{-2}}]\] done
clear
D)
\[[M{{L}^{-1}}{{T}^{-2}}]\] done
clear
View Solution play_arrow
-
question_answer43)
A wooden stick 2m long is floating on the surface of water. The surface tension of water 0.07 N/m. By putting soap solution on one side of the sticks the surface tension is reduced to 0.06 N/m. The net force on the stick will be [Pb. PMT 2002]
A)
0.07 N done
clear
B)
0.06 N done
clear
C)
0.01 N done
clear
D)
0.02 N done
clear
View Solution play_arrow
-
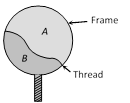
question_answer44)
A thread is tied slightly loose to a wire frame as in figure and the frame is dipped into a soap solution and taken out. The frame is completely covered with the film. When the portion A punctured with a pin, the thread. [KCET 2004]
A)
Becomes concave toward A done
clear
B)
Becomes convex towards A done
clear
C)
Remains in the initial position done
clear
D)
Either A or B depending on the size of A w.r.t. B done
clear
View Solution play_arrow
-
question_answer45)
The force required to take away a flat circular plate of radius 2 cm from the surface of water, will be (the surface tension of water is 70 dyne/cm) [Pb. PET 2001]
A)
\[280\pi \,dyne\] done
clear
B)
\[250\pi \,dyne\] done
clear
C)
\[140\pi \,dyne\] done
clear
D)
\[210\pi \,dyne\] done
clear
View Solution play_arrow
-
question_answer46)
Surface tension may be defined as [CPMT 1990]
A)
The work done per unit area in increasing the surface area of a liquid under isothermal condition done
clear
B)
The work done per unit area in increasing the surface area of a liquid under adiabatic condition done
clear
C)
The work done per unit area in increasing the surface area of a liquid under both isothermal and adiabatic conditions done
clear
D)
Free surface energy per unit volume done
clear
View Solution play_arrow
