-
question_answer1)
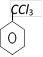
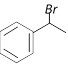
\[\xrightarrow{1\text{ eqv}\text{. of }B{{r}_{2}}/Fe}\]A. Compounds A is [Orissa JEE 2005]
A)
B)
C)
D)
View Solution play_arrow
-
question_answer2)
Ethyl bromide can be converted into ethyl alcohol by [KCET 1989]
A)
Heating with dilute hydrochloric acid and zinc done
clear
B)
Boiling with an alcoholic solution of KOH done
clear
C)
The action of moist silver oxide done
clear
D)
Refluxing methanol done
clear
View Solution play_arrow
-
question_answer3)
Reaction of ethyl chloride with sodium leads to [NCERT 1984]
A)
Ethane done
clear
B)
Propane done
clear
C)
n-butane done
clear
D)
n-pentane done
clear
View Solution play_arrow
-
question_answer4)
Treatment of ammonia with excess of ethyl chloride will yield [AIIMS 1992]
A)
Diethyl amine done
clear
B)
Ethane done
clear
C)
Tetraethyl ammonium chloride done
clear
D)
Methyl amine done
clear
View Solution play_arrow
-
question_answer5)
\[2CHC{{l}_{3}}+{{O}_{2}}\xrightarrow{X}2COC{{l}_{2}}+2HCl\] In the above reaction, X stands for [CPMT 1985]
A)
An oxidant done
clear
B)
A reductant done
clear
C)
Light and air done
clear
D)
None of these done
clear
View Solution play_arrow
-
question_answer6)
Phosgene is the common name for [DPMT 1983; CPMT 1993; MP PMT 1994; Kurukshetra CEE 1998; RPMT 2000, 02]
A)
\[C{{O}_{2}}\] and \[P{{H}_{3}}\] done
clear
B)
Phosphoryl chloride done
clear
C)
Carbonyl chloride done
clear
D)
Carbon tetrachloride done
clear
View Solution play_arrow
-
question_answer7)
When chloroform is treated with amine and KOH, we get [CPMT 1979]
A)
Rose odour smell done
clear
B)
Sour almond like smell done
clear
C)
Offensive odour done
clear
D)
Sour oil of winter green like smell done
clear
View Solution play_arrow
-
question_answer8)
A mixture of two organic chlorine compounds was treated with sodium metal in ether solution. Isobutane was obtained as a product. The two chlorine compounds are [KCET 1988]
A)
Methyl chloride and propyl chloride done
clear
B)
Methyl chloride and ethyl chloride done
clear
C)
Isopropyl chloride and methyl chloride done
clear
D)
Isopropyl chloride and ethyl chloride done
clear
View Solution play_arrow
-
question_answer9)
Alkyl halides can be converted into Grignard reagents by [KCET 1989]
A)
Boiling them with Mg ribbon in alcoholic solution done
clear
B)
Warming them with magnesium powder in dry ether done
clear
C)
Refluxing them with \[MgC{{l}_{2}}\] solution done
clear
D)
Warming them with \[MgC{{l}_{2}}\] done
clear
View Solution play_arrow
-
question_answer10)
Which is not present in Grignard reagent [CBSE PMT 1991]
A)
Methyl group done
clear
B)
Magnesium done
clear
C)
Halogen done
clear
D)
\[-COOH\] group done
clear
View Solution play_arrow
-
question_answer11)
The reactivity of ethyl chloride is [KCET 1986]
A)
More or less equal to that of benzyl chloride done
clear
B)
More than that of benzyl chloride done
clear
C)
More or less equal to that of chlorobenzene done
clear
D)
Less than that of chlorobenzene done
clear
View Solution play_arrow
-
question_answer12)
The reactivity of halogen atom is minimum in [KCET 1985]
A)
Propyl chloride done
clear
B)
Propyl iodide done
clear
C)
Isopropyl chloride done
clear
D)
Isopropyl bromide done
clear
View Solution play_arrow
-
question_answer13)
Chlorobenzene is
A)
Less reactive than benzyl chloride done
clear
B)
More reactive than ethyl bromide done
clear
C)
Nearly as reactive as methyl chloride done
clear
D)
More reactive than isopropyl chloride done
clear
View Solution play_arrow
-
question_answer14)
The reactivities of methyl chloride, propyl chloride and chlorobenzene are in the order [KCET 1988]
A)
Methyl chloride > propyl chloride > chlorobenzene done
clear
B)
Propyl chloride > methyl chloride > chlorobenzene done
clear
C)
Methyl chloride > chlorobenzene > propyl chloride done
clear
D)
Chlorobenzene > propyl chloride > methyl chloride done
clear
View Solution play_arrow
-
question_answer15)
Which of the following compound will make precipitate most readily with \[AgN{{O}_{3}}\] [CPMT 1992]
A)
\[CC{{l}_{3}}CHO\] done
clear
B)
\[CHC{{l}_{3}}\] done
clear
C)
\[{{C}_{6}}{{H}_{5}}C{{H}_{2}}Cl\] done
clear
D)
\[CH{{I}_{3}}\] done
clear
View Solution play_arrow
-
question_answer16)
Carbylamine is liberated when..... is heated with chloroform and alcoholic potash [KCET 1992]
A)
An aldehyde done
clear
B)
A primary amine done
clear
C)
A secondary amine done
clear
D)
A phenol done
clear
View Solution play_arrow
-
question_answer17)
Salicylic acid can be prepared using Reimer-Tiemann's reaction by treating phenol with [KCET 1989]
A)
Methyl chloride in the presence of anhydrous aluminium chloride done
clear
B)
Carbon dioxide under pressure in sodium hydroxide solution done
clear
C)
Carbon tetrachloride and concentrated sodium hydroxide done
clear
D)
Sodium nitrite and a few drops of concentrated sulphuric acid done
clear
View Solution play_arrow
-
question_answer18)
Grignard reagent is prepared by the reaction between [CBSE PMT 1994; DPMT 1996; Pb. PMT 1999; MH CET 1999]
A)
Zinc and alkyl halide done
clear
B)
Magnesium and alkyl halide done
clear
C)
Magnesium and alkane done
clear
D)
Magnesium and aromatic hydrocarbon done
clear
View Solution play_arrow
-
question_answer19)
Reaction of t-butyl bromide with sodium methoxide produces [CBSE PMT 1994]
A)
Isobutane done
clear
B)
Isobutylene done
clear
C)
Sodium t-butoxide done
clear
D)
t-butyl methyl ether done
clear
View Solution play_arrow
-
question_answer20)
War gas is formed from [BHU 1995]
A)
\[P{{H}_{3}}\] done
clear
B)
\[{{C}_{2}}{{H}_{2}}\] done
clear
C)
Zinc phosphate done
clear
D)
Chloropicrin done
clear
View Solution play_arrow
-
question_answer21)
What happens when \[CC{{l}_{4}}\] is treated with \[AgN{{O}_{3}}\] [EAMCET 1987; CBSE PMT 1988; MP PET 2000]
A)
\[N{{O}_{2}}\] will be evolved done
clear
B)
A white ppt. of \[AgCl\] will be formed done
clear
C)
\[CC{{l}_{4}}\] will dissolve in \[AgN{{O}_{3}}\] done
clear
D)
Nothing will happen done
clear
View Solution play_arrow
-
question_answer22)
If we use pyrene \[(CC{{l}_{4}})\] in the Riemer-Tiemann reaction in place of chloroform, the product formed is [CBSE PMT 1989; MP PMT 1990; MH CET 1999]
A)
Salicylaldehyde done
clear
B)
Phenolphthalein done
clear
C)
Salicylic acid done
clear
D)
Cyclohexanol done
clear
View Solution play_arrow
-
question_answer23)
\[{{C}_{6}}{{H}_{5}}C{{H}_{2}}Cl+KCN(aq.)\to X+Y\] Compounds X and Y are [BHU 1979]
A)
\[{{C}_{6}}{{H}_{6}}+KCl\] done
clear
B)
\[{{C}_{6}}{{H}_{5}}C{{H}_{2}}CN+KCl\] done
clear
C)
\[{{C}_{6}}{{H}_{5}}C{{H}_{3}}+KCl\] done
clear
D)
None of these done
clear
View Solution play_arrow
-
question_answer24)
The bad smelling substance formed by the action of alcoholic caustic potash on chloroform and aniline is [MP PMT 1971, 92, 2001; CPMT 1971, 86; AFMC 2002; RPMT 1999]
A)
Phenyl isocyanide done
clear
B)
Nitrobenzene done
clear
C)
Phenyl cyanide done
clear
D)
Phenyl isocyanate done
clear
View Solution play_arrow
-
question_answer25)
Ethylidene chloride on treatment with aqueous KOH gives [MP PMT 1986]
A)
Ethylene glycol done
clear
B)
Acetaldehyde done
clear
C)
Formaldehyde done
clear
D)
None done
clear
View Solution play_arrow
-
question_answer26)
Reaction \[{{C}_{2}}{{H}_{5}}I+{{C}_{5}}{{H}_{11}}I+2Na\to {{C}_{2}}{{H}_{5}}-{{C}_{5}}{{H}_{11}}+2NaI\] is called [MP PMT 1992]
A)
Hoffmann's reaction done
clear
B)
Dow's reaction done
clear
C)
Wurtz's reaction done
clear
D)
Riemer-Tiemann's reaction done
clear
View Solution play_arrow
-
question_answer27)
In presence of \[AlC{{l}_{3}},\] benzene and n-propyl bromide react in Friedal-Craft's reaction to form [MP PMT 1991]
A)
n-propyl benzene done
clear
B)
1, 2-dinormal propyl benzene done
clear
C)
1, 4-dinormal propyl benzene done
clear
D)
Isopropyl benzene done
clear
View Solution play_arrow
-
question_answer28)
The dehydrobromination of 2-bromobutane gives \[C{{H}_{3}}CH=CHC{{H}_{3}}.\] The product is
A)
Hofmann product done
clear
B)
Saytzeff product done
clear
C)
Hoffmann-Saytzeff product done
clear
D)
Markownikoff product done
clear
View Solution play_arrow
-
question_answer29)
Ethylene difluoride on hydrolysis gives
A)
Glycol done
clear
B)
Fluoroethanol done
clear
C)
Difluoroethanol done
clear
D)
Freon done
clear
View Solution play_arrow
-
question_answer30)
Benzyl chloride when oxidised by \[pb{{(N{{O}_{3}})}_{2}}\] gives [MP PMT 1989]
A)
Benzoic acid done
clear
B)
Benzaldehyde done
clear
C)
Benzene done
clear
D)
None done
clear
View Solution play_arrow
-
question_answer31)
Which of the following statements about chloroform is false [Manipal MEE 1995]
A)
It is a colourless, sweet-smelling liquid done
clear
B)
It is almost insoluble in water done
clear
C)
It is highly inflammable done
clear
D)
It can be used as an inhalational anaesthetic agent done
clear
View Solution play_arrow
-
question_answer32)
\[CC{{l}_{4}}\] cannot give precipitate with \[AgN{{O}_{3}}\] due to [CPMT 1979]
A)
Formation of complex with \[AgN{{O}_{3}}\] done
clear
B)
Evolution of \[C{{l}_{2}}\] gas done
clear
C)
Chloride ion is not formed done
clear
D)
\[AgN{{O}_{3}}\] does not give silver ion done
clear
View Solution play_arrow
-
question_answer33)
On heating \[CHC{{l}_{3}}\] with aq. NaOH, the product is [CPMT 1971, 78; BHU 1997; EAMCET 1998; JIPMER (Med.) 2002]
A)
\[C{{H}_{3}}COONa\] done
clear
B)
\[HCOONa\] done
clear
C)
Sodium oxalate done
clear
D)
\[C{{H}_{3}}OH\] done
clear
View Solution play_arrow
-
question_answer34)
Ethyl bromide reacts with lead-sodium alloy to form [MP PMT/PET 1988; MP PET 1997]
A)
Tetraethyl lead done
clear
B)
Tetraethyl bromide done
clear
C)
Both A and B done
clear
D)
None of the above done
clear
View Solution play_arrow
-
question_answer35)
Iodoform heated with Ag powder to form [DPMT 1985]
A)
Acetylene done
clear
B)
Ethylene done
clear
C)
Methane done
clear
D)
Ethane done
clear
View Solution play_arrow
-
question_answer36)
Ethyl bromide reacts with silver nitrite to form [DPMT 1985; IIT-JEE 1991]
A)
Nitroethane done
clear
B)
Nitroethane and ethyl nitrite done
clear
C)
Ethyl nitrite done
clear
D)
Ethane done
clear
View Solution play_arrow
-
question_answer37)
Which of the following reactions leads to the formation of chloritone [RPMT 2003]
A)
\[CHC{{l}_{3}}+C{{H}_{3}}COC{{H}_{3}}\] done
clear
B)
\[CC{{l}_{4}}+\] Acetone done
clear
C)
\[CHC{{l}_{3}}+KOH\] done
clear
D)
\[CHC{{l}_{3}}+HN{{O}_{3}}\] done
clear
View Solution play_arrow
-
question_answer38)
\[C{{H}_{3}}-C{{H}_{2}}-C{{H}_{2}}Br+KOH\text{ (alc}\text{.)}\to \text{Product}\] Product in above reaction is [RPMT 2003]
A)
\[C{{H}_{3}}-CH=C{{H}_{2}}\] done
clear
B)
\[C{{H}_{3}}-C{{H}_{2}}-C{{H}_{3}}\] done
clear
C)
A and B both done
clear
D)
None of these done
clear
View Solution play_arrow
-
question_answer39)
\[A+CC{{l}_{4}}+KOH\to \] Salicylic acid 'A' in above reaction is [RPMT 2003]
A)
B)
C)
D)
View Solution play_arrow
-
question_answer40)
Identify \[X\] and \[Y\] in the following sequence \[{{C}_{2}}{{H}_{5}}Br\xrightarrow{X}\text{product}\xrightarrow{\text{Y}}{{C}_{3}}{{H}_{7}}N{{H}_{2}}\] [Orissa JEE 2005]
A)
\[X=KCN,\,Y=LiAl{{H}_{4}}\] done
clear
B)
\[X=KCN,\,Y={{H}_{3}}{{O}^{+}}\] done
clear
C)
\[X=C{{H}_{3}}Cl,\,Y=AlC{{l}_{3}}/HCl\] done
clear
D)
\[X=C{{H}_{3}}N{{H}_{2}},Y=HN{{O}_{2}}\] done
clear
View Solution play_arrow
-
question_answer41)
1-chlorobutane reacts with alcoholic KOH to form [IIT-JEE 1991; AFMC 1998]
A)
1-butene done
clear
B)
2-butane done
clear
C)
1-butanol done
clear
D)
2-butanol done
clear
View Solution play_arrow
-
question_answer42)
Which of the following reactions gives \[{{H}_{2}}C=C=C=C{{H}_{2}}\] [Roorkee Qualifying 1998]
A)
\[C{{H}_{2}}Br-CBr=C{{H}_{2}}\xrightarrow{Zn/C{{h}_{3}}OH}\] done
clear
B)
\[HC\equiv C-C{{H}_{2}}-COOH\underset{{{40}^{o}}C}{\mathop{\xrightarrow{Aq.{{K}_{2}}C{{O}_{3}}}}}\,\] done
clear
C)
\[C{{H}_{2}}Br-C\equiv C-C{{H}_{2}}Br\underset{\text{Heat}}{\mathop{\xrightarrow{Zn}}}\,\] done
clear
D)
\[2C{{H}_{2}}=CH-C{{H}_{2}}I\underset{{}}{\mathop{\xrightarrow{{}}}}\,\] done
clear
View Solution play_arrow
-
question_answer43)
When ethyl amine is heated with chloroform and alcoholic KOH, a compound with offensive smell is obtained. This compound is [CPMT 1983, 84; RPMT 2002]
A)
A secondary amine done
clear
B)
An isocyanide done
clear
C)
A cyanide done
clear
D)
An acid done
clear
View Solution play_arrow
-
question_answer44)
Chlorobenzene on fusing with solid NaOH gives [DPMT 1981; CPMT 1990]
A)
Benzene done
clear
B)
Benzoic acid done
clear
C)
Phenol done
clear
D)
Benzene chloride done
clear
View Solution play_arrow
-
question_answer45)
DDT can be prepared by reacting chlorobenzene (in the presence of conc. \[{{H}_{2}}S{{O}_{4}}\]) with
A)
\[C{{l}_{2}}\] in ultraviolet light done
clear
B)
Chloroform done
clear
C)
Trichloroacetone done
clear
D)
Chloral hydrate done
clear
View Solution play_arrow
-
question_answer46)
When phenol reacts with \[CHC{{l}_{3}}\] and KOH, the product obtained would be [RPMT 1997]
A)
Salicylaldehyde done
clear
B)
p-hydroxy benzaldehyde done
clear
C)
Both A and B done
clear
D)
Chloretone done
clear
View Solution play_arrow
-
question_answer47)
Ethyl chloride on heating with silver cyanide forms a compound X. The functional isomer of X is [EAMCET 1997; KCET 2005]
A)
\[{{C}_{2}}{{H}_{5}}NC\] done
clear
B)
\[{{C}_{2}}{{H}_{5}}CN\] done
clear
C)
\[{{H}_{3}}C-NH-C{{H}_{3}}\] done
clear
D)
\[{{C}_{2}}{{H}_{5}}N{{H}_{2}}\] done
clear
View Solution play_arrow
-
question_answer48)
Which of the following statements is incorrect [CPMT 1977]
A)
\[{{C}_{2}}{{H}_{5}}Br\] reacts with alco. KOH to form \[{{C}_{2}}{{H}_{5}}OH\] done
clear
B)
\[{{C}_{2}}{{H}_{5}}Br\] when treated with metallic sodium gives ethane done
clear
C)
\[{{C}_{2}}{{H}_{5}}Br\] when treated with sodium ethoxide forms diethyl ether done
clear
D)
\[{{C}_{2}}{{H}_{5}}Br\] with AgCN forms ethyl isocyanide done
clear
View Solution play_arrow
-
question_answer49)
When chloroform is exposed to air and sunlight, it gives [NCERT 1984; CPMT 1978, 87; CBSE PMT 1990; EAMCET 1993; MNR 1994; MP PET 1997, 2000; BHU 2001; AFMC 2002]
A)
Carbon tetrachloride done
clear
B)
Carbonyl chloride done
clear
C)
Mustard gas done
clear
D)
Lewsite done
clear
View Solution play_arrow
-
question_answer50)
An organic halide is shaken with aqueous \[NaOH\] followed by the addition of dil. \[HN{{O}_{3}}\] and silver nitrate solution gave white ppt. The substance can be [JIPMER 1997]
A)
\[{{C}_{6}}{{H}_{4}}(C{{H}_{3}})Br\] done
clear
B)
\[{{C}_{6}}{{H}_{5}}C{{H}_{2}}Cl\] done
clear
C)
\[{{C}_{6}}{{H}_{5}}Cl\] done
clear
D)
None of these done
clear
View Solution play_arrow
-
question_answer51)
A compound A has a molecular formula \[{{C}_{2}}C{{l}_{3}}OH.\] It reduces Fehling solution and on oxidation gives a monocarboxylic acid B. A is obtained by action of chlorine on ethyl alcohol. A is [CBSE PMT 1994; MP PET 1997; KCET 2005]
A)
Chloral done
clear
B)
\[CHC{{l}_{3}}\] done
clear
C)
\[C{{H}_{3}}Cl\] done
clear
D)
Chloroacetic acid done
clear
View Solution play_arrow
-
question_answer52)
Following equation illustrates \[{{C}_{6}}{{H}_{5}}Cl+2NaOH\underset{200\,atm}{\mathop{\xrightarrow{200-{{250}^{o}}C}}}\,{{C}_{6}}{{H}_{5}}ONa+NaCl+{{H}_{2}}O\] [Bihar CEE 1995]
A)
Dow's process done
clear
B)
Kolbe's process done
clear
C)
Carbylamine test done
clear
D)
Haloform reaction done
clear
View Solution play_arrow
-
question_answer53)
One of the following that cannot undergo dehydro-halogenation is [J & K 2005]
A)
Iso-propyl bromide done
clear
B)
Ethanol done
clear
C)
Ethyl bromide done
clear
D)
None of these done
clear
View Solution play_arrow
-
question_answer54)
A compound X on reaction with chloroform and NaOH gives a compound with a very unpleasant odour. X is [MP PMT 1999]
A)
\[{{C}_{6}}{{H}_{5}}CON{{H}_{2}}\] done
clear
B)
\[{{C}_{6}}{{H}_{5}}N{{H}_{2}}\] done
clear
C)
\[{{C}_{6}}{{H}_{5}}C{{H}_{2}}NHC{{H}_{3}}\] done
clear
D)
\[{{C}_{6}}{{H}_{5}}NHC{{H}_{3}}\] done
clear
View Solution play_arrow
-
question_answer55)
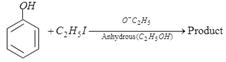
In the above reaction product is
A)
\[{{C}_{6}}{{H}_{5}}O{{C}_{2}}{{H}_{5}}\] done
clear
B)
\[{{C}_{2}}{{H}_{5}}O{{C}_{2}}{{H}_{5}}\] done
clear
C)
\[{{C}_{6}}{{H}_{5}}O{{C}_{6}}{{H}_{5}}\] done
clear
D)
\[{{C}_{6}}{{H}_{5}}I\] done
clear
View Solution play_arrow
-
question_answer56)
\[{{C}_{2}}{{H}_{5}}Cl+KCN\xrightarrow{{}}X\xrightarrow{\text{Hydrolysis}}Y.\] 'X' and 'Y' are [MP PET 1995]
A)
\[{{C}_{2}}{{H}_{6}}\] and \[{{C}_{2}}{{H}_{5}}CN\] done
clear
B)
\[C{{H}_{3}}C{{H}_{2}}C{{H}_{2}}OH,\] and \[C{{H}_{3}}-CHOH-C{{H}_{3}}\] done
clear
C)
\[C{{H}_{3}}-C(C{{H}_{3}})(OH)-C{{H}_{3}}\] and \[{{C}_{2}}{{H}_{5}}C{{H}_{2}}N{{H}_{2}}\] done
clear
D)
\[{{C}_{2}}{{H}_{5}}CN\] and \[{{C}_{2}}{{H}_{5}}COOH\] done
clear
View Solution play_arrow
-
question_answer57)
Iodoform is formed on warming \[{{I}_{2}}\] and NaOH with [MP PET 1995; DCE 1999; RPET 1999; RPMT 2002]
A)
\[{{C}_{2}}{{H}_{5}}OH\] done
clear
B)
\[C{{H}_{3}}OH\] done
clear
C)
HCOOH done
clear
D)
\[{{C}_{6}}{{H}_{6}}\] done
clear
View Solution play_arrow
-
question_answer58)
Which of the following reacts with phenol to give salicylaldehyde after hydrolysis [MP PMT 1995]
A)
Dichloromethane done
clear
B)
Trichloromethane done
clear
C)
Methyl chloride done
clear
D)
None of these done
clear
View Solution play_arrow
-
question_answer59)
Dehydrohalogenation in monohaloalkanes produces
A)
A single bond done
clear
B)
A double bond done
clear
C)
A triple bond done
clear
D)
Fragmentation done
clear
View Solution play_arrow
-
question_answer60)
When chloroform is treated with conc. \[HN{{O}_{3}}\] it gives [CPMT 1986; MP PMT 1989; AFMC 1998, 99; EAMCET 1991; BHU 1999]
A)
\[CHC{{l}_{2}}N{{O}_{2}}\] done
clear
B)
\[CC{{l}_{3}}N{{O}_{2}}\] done
clear
C)
\[CHC{{l}_{2}}HN{{O}_{3}}\] done
clear
D)
None of these done
clear
View Solution play_arrow
-
question_answer61)
A sample of chloroform being used as anaesthetic is tested by [AIIMS 1980; CPMT 1983]
A)
Fehling solution done
clear
B)
Ammoniacal \[C{{u}_{2}}C{{l}_{2}}\] done
clear
C)
\[AgN{{O}_{3}}\] solution done
clear
D)
\[AgN{{O}_{3}}\] solution after boiling with alcoholic \[KOH\] solution done
clear
View Solution play_arrow
-
question_answer62)
Dehydrohalogenation of an alkyl halide is [MP PMT 1996]
A)
An addition reaction done
clear
B)
A substitution reaction done
clear
C)
An elimination reaction done
clear
D)
An oxidation reaction done
clear
View Solution play_arrow
-
question_answer63)
Reaction of aqueous sodium hydroxide on (i) ethyl bromide and (ii) chlorobenzene gives
A)
(i) Ethene and (ii) o-chlorophenol done
clear
B)
(i) Ethyl alcohol and (ii) o-chlorophenol done
clear
C)
(i) Ethyl alcohol and (ii) phenol done
clear
D)
(i) Ethyl alcohol and (ii) no reaction done
clear
View Solution play_arrow
-
question_answer64)
2-bromopentane is heated with potassium ethoxide in ethanol. The major product obtained is [CBSE PMT 1998]
A)
Pentene-1 done
clear
B)
cis pentene-2 done
clear
C)
trans pentene-2 done
clear
D)
2-ethoxypentane done
clear
View Solution play_arrow
-
question_answer65)
What is the product formed in the following reaction \[{{C}_{6}}{{H}_{5}}OH+CC{{l}_{4}}\underset{(2)\,\,{{H}^{+}}}{\mathop{\xrightarrow{(1)\,\,NaOH}}}\,\] [KCET 1998]
A)
p-hydroxybenzoic acid done
clear
B)
o-hydroxybenzoic acid done
clear
C)
Benzaldehyde done
clear
D)
Salicylaldehyde done
clear
View Solution play_arrow
-
question_answer66)
When chloroform is treated with excess oxygen it forms [MH CET 1999]
A)
\[COC{{l}_{2}}+HCl\] done
clear
B)
\[COC{{l}_{2}}+C{{l}_{2}}+{{H}_{2}}\] done
clear
C)
\[COC{{l}_{2}}+C{{l}_{2}}+{{H}_{2}}O\] done
clear
D)
No product will be formed done
clear
View Solution play_arrow
-
question_answer67)
Which isomer of cyclohexane hexachloride is a very strong insecticide [MP PET 2003]
A)
\[\alpha \] done
clear
B)
\[\beta \] done
clear
C)
\[\gamma \] done
clear
D)
\[\delta \] done
clear
View Solution play_arrow
-
question_answer68)
Haloalkane in the presence of alcoholic KOH undergoes [KCET (Engg/Med.) 2002]
A)
Elimination done
clear
B)
Polymerisation done
clear
C)
Dimerisation done
clear
D)
Substitution done
clear
View Solution play_arrow
-
question_answer69)
The set of compounds in which the reactivity of halogen atom in the ascending order is [KCET (Engg.) 2002]
A)
Vinyl chloride, chlorethane, chlorobenzene done
clear
B)
Vinyl chloride, chlorobenzene, chloroethane done
clear
C)
Chloroethane, chlorobenzene, vinyl chloride done
clear
D)
Chlorobenzene, vinyl chloride chloroethane done
clear
View Solution play_arrow
-
question_answer70)
Alkyl halides react with Mg in dry ether to form [DPMT 2000; MP PET 2001]
A)
Magnesium halide done
clear
B)
Grignard's reagent done
clear
C)
Alkene done
clear
D)
Alkyne done
clear
View Solution play_arrow
-
question_answer71)
In the following sequence of reactions \[C{{H}_{3}}C{{H}_{2}}C{{H}_{2}}Br\xrightarrow{KOH(alc)}(A)\xrightarrow{HBr}(B)\xrightarrow{KOH\,(aq.)}(C),\]The product is [JIPMER 2001]
A)
Propan - 2 - ol done
clear
B)
Propan - l - ol done
clear
C)
Propyne done
clear
D)
Propene done
clear
View Solution play_arrow
-
question_answer72)
Alkyl halide on heating with alc. \[N{{H}_{3}}\]in a sealed tube results? [Orissa JEE 2002]
A)
\[1{}^\circ \] amine done
clear
B)
\[2{}^\circ \] amine done
clear
C)
\[3{}^\circ \] amine done
clear
D)
All of these done
clear
View Solution play_arrow
-
question_answer73)
When \[C{{H}_{3}}C{{H}_{2}}CHC{{l}_{2}}\] is treated with \[NaN{{H}_{2,}}\] the product formed is [CBSE PMT 2002]
A)
\[C{{H}_{3}}-CH=C{{H}_{2}}\] done
clear
B)
\[C{{H}_{3}}-C\equiv CH\] done
clear
C)
\[C{{H}_{3}}C{{H}_{2}}CH(N{{H}_{2}})(Cl)\] done
clear
D)
\[C{{H}_{3}}C{{H}_{2}}C{{(N{{H}_{2}})}_{2}}\] done
clear
View Solution play_arrow
-
question_answer74)
By heating a mixture of \[CHC{{l}_{3}}\]with silver powder, the compound formed is [Kurukshetra CET 2002]
A)
Acetylene done
clear
B)
Silver acetate done
clear
C)
Methanol done
clear
D)
None of these done
clear
View Solution play_arrow
-
question_answer75)
Chloropicrin is [Kurukshetra CET 2002]
A)
Trichloro acetaldehyde done
clear
B)
Nitrochloroform done
clear
C)
2, 4, 6-trinitro phenol done
clear
D)
None of these done
clear
View Solution play_arrow
-
question_answer76)
Which of the following are correct statements about \[{{C}_{2}}{{H}_{5}}Br\] [Roorkee 1999]
A)
It reacts with metallic Na to give ethane done
clear
B)
It gives nitroethane on heating with aqueous ethanolic solution of \[AgN{{O}_{2}}\] done
clear
C)
It gives \[{{C}_{2}}{{H}_{5}}OH\]on boiling with alcoholic potash done
clear
D)
It forms ethylacetate on heating with silver acetate done
clear
View Solution play_arrow
-
question_answer77)
Aryl halide is less reactive than alkyl halide towards nucleophilic substitution because [RPMT 2002]
A)
Less stable carbonium ion done
clear
B)
Due to large \[C-Cl\]bond energy done
clear
C)
Inductive effect done
clear
D)
Resonance stabilization and \[s{{p}^{2}}\]- hybridisation of C attached to halide done
clear
View Solution play_arrow
-
question_answer78)
Methyl chloride reacts with silver acetate to yield [BVP 2003]
A)
Acetaldehyde done
clear
B)
Acetyl chloride done
clear
C)
Methyl acetate done
clear
D)
Acetic acid done
clear
View Solution play_arrow
-
question_answer79)
Chloroform for anesthetic purposes is tested for its purity with the reagent [DPMT 2001]
A)
Silver nitrate done
clear
B)
Lead nitrate done
clear
C)
Ammoniacal \[C{{u}_{2}}C{{l}_{2}}\] done
clear
D)
Lead nitrate done
clear
View Solution play_arrow
-
question_answer80)
2, 6 - Dimethylheptane on monochlorination produces??. derivatives [DPMT 2001]
A)
5 done
clear
B)
6 done
clear
C)
3 done
clear
D)
4 done
clear
View Solution play_arrow
-
question_answer81)
The less reactivity of chlorine atom in \[C{{H}_{2}}=CH-Cl\] is due to [DCE 2001]
A)
Inductive effect done
clear
B)
Resonance stabilization done
clear
C)
Electromeric effect done
clear
D)
Electronegativity done
clear
View Solution play_arrow
-
question_answer82)
\[C{{H}_{3}}-C{{H}_{2}}-Br\xrightarrow{\text{alc}\text{.KCN}}\]\[C{{H}_{3}}C{{H}_{2}}CN\]\[\xrightarrow{\text{HOH}}X\] In this reaction, product X is [MH CET 2002]
A)
Acetic acid done
clear
B)
Propionic acid done
clear
C)
Butyric acid done
clear
D)
Formic acid done
clear
View Solution play_arrow
-
question_answer83)
In alkaline hydrolysis of a tertiary alkyl halide by aqueous alkali if concentration of alkali is doubled, then the reaction [MH CET 2002]
A)
Will be doubled done
clear
B)
Will be halved done
clear
C)
Will remain constant done
clear
D)
Can?t say done
clear
View Solution play_arrow
-
question_answer84)
\[AgN{{O}_{3}}\]does not give precipitate with \[CHC{{l}_{3}}\] because [MP PET 1999; CPMT 2002]
A)
\[CHC{{l}_{3}}\]does not ionise in water done
clear
B)
\[AgN{{O}_{3}}\]does not reacts with \[CHC{{l}_{3}}\] done
clear
C)
\[CHC{{l}_{3}}\] is chemically inert done
clear
D)
None of these done
clear
View Solution play_arrow
-
question_answer85)
The reaction between chlorobenzene and chloral in the presence of concentrated sulphuric acid produces [Pb. PMT 2001]
A)
Gammexane done
clear
B)
p,p-dichloro diphenyl trichloro ethane done
clear
C)
Chloropicrin done
clear
D)
Benzene hexachloride done
clear
View Solution play_arrow
-
question_answer86)
False statement is [RPET 1999]
A)
Chloroform is heavier than water done
clear
B)
\[CC{{l}_{4}}\] is non-inflammable done
clear
C)
Vinyl chloride is more reactive than allyl chloride done
clear
D)
\[B{{r}^{-}}\] is a good nucleophile as compared to \[{{I}^{-}}\] done
clear
View Solution play_arrow
-
question_answer87)
Chloroform is slowly oxidise by air in presence of light to form [MH CET 1999; UPSEAT 2001, 02; RPMT 2003]
A)
Formyl chloride done
clear
B)
Phosgene done
clear
C)
Trichloroacetic acid done
clear
D)
Formic acid done
clear
View Solution play_arrow
-
question_answer88)
Alcoholic potash is used to bring about [KCET (Engg.) 2001]
A)
Dehydrogenation done
clear
B)
Dehydration done
clear
C)
Dehydrohalogenation done
clear
D)
Dehalogenation done
clear
View Solution play_arrow
-
question_answer89)
Vinyl chloride reacts with \[HCl\] to form [JIPMER 2000]
A)
1, 1- dichloro ethane done
clear
B)
1, 2- dichloro ethane done
clear
C)
Tetrachloro ethylene done
clear
D)
Mixture of 1, 2 and 1, 1 - dichloro ethane done
clear
View Solution play_arrow
-
question_answer90)
\[R-X+NaOH\xrightarrow{{}}ROH+NaX\] The above reaction is classified as [BHU 1982; CBSE PMT 1991; RPET 2000]
A)
Nucleophilic substitution done
clear
B)
Electrophilic substitution done
clear
C)
Reduction done
clear
D)
Oxidation done
clear
View Solution play_arrow
-
question_answer91)
Reduction of acetyl chloride with \[{{H}_{2}}\] in presence of Pd gives [MP PMT 2001]
A)
\[C{{H}_{3}}COC{{H}_{3}}\] done
clear
B)
\[{{C}_{2}}{{H}_{5}}OH\] done
clear
C)
\[C{{H}_{3}}COOH\] done
clear
D)
\[C{{H}_{3}}CHO\] done
clear
View Solution play_arrow
-
question_answer92)
When methyl bromide is heated with\[Zn\]it gives [MP PMT 2001]
A)
\[C{{H}_{4}}\] done
clear
B)
\[{{C}_{2}}{{H}_{6}}\] done
clear
C)
\[{{C}_{2}}{{H}_{4}}\] done
clear
D)
\[C{{H}_{3}}OH\] done
clear
View Solution play_arrow
-
question_answer93)
Phenol reacts with \[CHC{{l}_{3}}\] and NaOH (at \[340K\]) to give [MP PMT 1997; CBSE PMT 2002]
A)
o-chlorophenol done
clear
B)
Salicylaldehyde done
clear
C)
Benzaldehyde done
clear
D)
Chlorobenzene done
clear
View Solution play_arrow
-
question_answer94)
Iodoform on heating with \[KOH\]gives [MP PMT 2000]
A)
\[C{{H}_{3}}CHO\] done
clear
B)
\[C{{H}_{3}}COOK\] done
clear
C)
\[HCOOK\] done
clear
D)
\[HCHO\] done
clear
View Solution play_arrow
-
question_answer95)
Which reaction is correct in the conversion of chloroform to acetylene [Pb. PMT 2000]
A)
\[CHC{{l}_{3}}+AgN{{O}_{3}}\] done
clear
B)
\[CHC{{l}_{3}}+{{O}_{2}}\] done
clear
C)
\[CHC{{l}_{3}}+HN{{O}_{3}}\] done
clear
D)
\[CHC{{l}_{3}}+Ag\] done
clear
View Solution play_arrow
-
question_answer96)
Which of the following gases are poisonous [Pb. PMT 2000]
A)
\[CHC{{l}_{3}}\] done
clear
B)
\[C{{O}_{2}}\] done
clear
C)
None of these done
clear
D)
\[CO\] done
clear
View Solution play_arrow
-
question_answer97)
Which of the following alkyl halide is used as a methylating agent [KCET (Med.) 2000; MP PET 1999]
A)
\[C{{H}_{3}}I\] done
clear
B)
\[{{C}_{2}}{{H}_{5}}Br\] done
clear
C)
\[{{C}_{2}}{{H}_{5}}Cl\] done
clear
D)
\[{{C}_{6}}{{H}_{5}}Cl\] done
clear
View Solution play_arrow
-
question_answer98)
\[{{C}_{6}}{{H}_{6}}C{{l}_{6}}\], on treatment with alcoholic \[KOH\], yields [AFMC 2000]
A)
\[{{C}_{6}}{{H}_{6}}\] done
clear
B)
\[{{C}_{6}}{{H}_{3}}C{{l}_{3}}\] done
clear
C)
\[({{C}_{6}}{{H}_{6}})\ OH\] done
clear
D)
\[{{C}_{6}}{{H}_{6}}C{{l}_{4}}\] done
clear
View Solution play_arrow
-
question_answer99)
When ethyl iodide is heated with silver nitrate, the product obtained is [CPMT 2000]
A)
\[{{C}_{2}}{{H}_{5}}Ag\] done
clear
B)
\[Ag-O-N{{O}_{2}}\] done
clear
C)
\[{{C}_{2}}{{H}_{5}}O-N{{O}_{2}}\] done
clear
D)
\[{{C}_{2}}{{H}_{5}}I-N{{O}_{2}}\] done
clear
View Solution play_arrow
-
question_answer100)
\[CHC{{l}_{3}}\] and HF lead to the formation of a compound of fluorine of molecular weight 70. The compound is [RPET 2000]
A)
Fluoroform done
clear
B)
Fluorine monoxide done
clear
C)
Fluorine dioxide done
clear
D)
Fluromethanol done
clear
View Solution play_arrow
-
question_answer101)
Chloroform with zinc dust in water gives [UPSEAT 2000]
A)
\[C{{H}_{4}}\] done
clear
B)
Chloropicrin done
clear
C)
\[CC{{l}_{4}}\] done
clear
D)
\[C{{H}_{2}}C{{l}_{2}}\] done
clear
View Solution play_arrow
-
question_answer102)
Which of the following is used as a catalyst for preparing Grignard reagent [KCET 1998]
A)
Iron powder done
clear
B)
Iodine powder done
clear
C)
Activated charcoal done
clear
D)
Manganese dioxide done
clear
View Solution play_arrow
-
question_answer103)
For a given alkyl group the densities of the halides follow the order [MP PMT 1997]
A)
\[RI<RBr<RCl\] done
clear
B)
\[RI<RCl<RBr\] done
clear
C)
\[RBr<RI<RCl\] done
clear
D)
\[RCl<RBr<RI\] done
clear
View Solution play_arrow
-
question_answer104)
Which halide will be least reactive in respect to hydrolysis [MP PET 2003]
A)
Vinyl chloride done
clear
B)
Allyl chloride done
clear
C)
Ethyl chloride done
clear
D)
t-Butyl chloride done
clear
View Solution play_arrow
-
question_answer105)
In nucleophilic aliphatic substitution, the nucleophiles are generally
A)
Acids done
clear
B)
Bases done
clear
C)
Salts done
clear
D)
Neutral molecules done
clear
View Solution play_arrow
-
question_answer106)
Which one of the following compounds does not react with bromine [DPMT 1983]
A)
Ethylamine done
clear
B)
Propene done
clear
C)
Phenol done
clear
D)
Chloroform done
clear
View Solution play_arrow
-
question_answer107)
Allyl chloride on dehydro chlorination gives [Kerala (Med.) 2003]
A)
Propadiene done
clear
B)
Propylene done
clear
C)
Acetylchloride done
clear
D)
Acetone done
clear
View Solution play_arrow
-
question_answer108)
Toluene reacts with excess of \[C{{l}_{2}}\] in presence of sunlight to give a product which on hydrolysis followed by reaction with NaOH gives [Orissa JEE 2004]
A)
B)
C)
D)
None of these done
clear
View Solution play_arrow
-
question_answer109)
An alkyl bromide produces a single alkene when it reacts with sodium ethoxide and ethanol. This alkene undergoes hydrogenation and produces 2-methyl butane. What is the identity of the alkyl bromide [Kerala PMT 2004]
A)
1-bromo-2, 2-dimethylpropane done
clear
B)
1-bromobutane done
clear
C)
1-bromo-2-methylbutane done
clear
D)
2-bromo-2-methylbutane done
clear
E)
2-bromopentane done
clear
View Solution play_arrow
-
question_answer110)
On treating a mixture of two alkyl halides with sodium metal in dry ether, 2-methyl propane was obtained. The alkyl halides are [KCET 2004]
A)
2-chloropropane and chloromethane done
clear
B)
2-chloropropane and chloroethane done
clear
C)
Chloromethane and chloroethane done
clear
D)
Chloromethane and 1-chloropropane done
clear
View Solution play_arrow
-
question_answer111)
In which case formation of butane nitrile is possible [Orissa JEE 2004]
A)
\[{{C}_{3}}{{H}_{7}}Br+KCN\] done
clear
B)
\[{{C}_{4}}{{H}_{9}}Br+KCN\] done
clear
C)
\[{{C}_{3}}{{H}_{7}}OH+KCN\] done
clear
D)
\[{{C}_{4}}{{H}_{9}}OH+KCN\] done
clear
View Solution play_arrow
-
question_answer112)
The reaction of an aeromatic halogen compound with an alkyl halides in presence of sodium and ether is called [MP PMT 2004]
A)
Wurtz reaction done
clear
B)
Sandmeyer's reaction done
clear
C)
Wurtz-fittig reaction done
clear
D)
Kolbe reaction done
clear
View Solution play_arrow
-
question_answer113)
The compound added to prevent chloroform to form phosgene gas is [MP PET 2004]
A)
\[{{C}_{2}}{{H}_{5}}OH\] done
clear
B)
\[C{{H}_{3}}COOH\] done
clear
C)
\[C{{H}_{3}}COC{{H}_{3}}\] done
clear
D)
\[C{{H}_{3}}OH\] done
clear
View Solution play_arrow
-
question_answer114)
Among the following, the one which reacts most readily with ethanol is [AIIMS 2004]
A)
p-nitrobenzyl bromide done
clear
B)
p-chlorobenzyl bromide done
clear
C)
p-methoxybenzyl bromide done
clear
D)
p-methylbenzyl bromide done
clear
View Solution play_arrow
-
question_answer115)
Chloropicrin is obtained by the reaction of [CBSE PMT 2004]
A)
Chlorine on picric acid done
clear
B)
Nitric acid on chloroform done
clear
C)
Steam on carbon tetrachloride done
clear
D)
Nitric acid on chlorobenzene done
clear
View Solution play_arrow
-
question_answer116)
In Wurtz reaction alkyl halide react with [MH CET 2004]
A)
Sodium in ether done
clear
B)
Sodium in dry ether done
clear
C)
Sodium only done
clear
D)
Alkyl halide in ether done
clear
View Solution play_arrow
-
question_answer117)
Chloroform, when kept open, is oxidised to [CPMT 2004]
A)
\[C{{O}_{2}}\] done
clear
B)
\[COC{{l}_{2}}\] done
clear
C)
\[C{{O}_{2}},\ C{{l}_{2}}\] done
clear
D)
None of these done
clear
View Solution play_arrow
-
question_answer118)
Chloroform reacts with concentrated \[HN{{O}_{3}}\] to give [Pb. CET 2000]
A)
Water gas done
clear
B)
Tear gas done
clear
C)
Laughing gas done
clear
D)
Producer gas done
clear
View Solution play_arrow
-
question_answer119)
When ethyl chloride and alcoholic KOH are heated, the compound obtained is [MH CET 2003]
A)
\[{{C}_{2}}{{H}_{4}}\] done
clear
B)
\[{{C}_{2}}{{H}_{2}}\] done
clear
C)
\[{{C}_{6}}{{H}_{6}}\] done
clear
D)
\[{{C}_{2}}{{H}_{6}}\] done
clear
View Solution play_arrow
-
question_answer120)
Chloroform, on warming with Ag powder, gives [MH CET 2003]
A)
\[{{C}_{2}}{{H}_{6}}\] done
clear
B)
\[{{C}_{3}}{{H}_{8}}\] done
clear
C)
\[{{C}_{2}}{{H}_{4}}\] done
clear
D)
\[{{C}_{2}}{{H}_{2}}\] done
clear
View Solution play_arrow
-
question_answer121)
When alkyl halide is heated with dry \[A{{g}_{2}}O\], it produces [CPMT 1997; BHU 2004]
A)
Ester done
clear
B)
Ether done
clear
C)
Ketone done
clear
D)
Alcohol done
clear
View Solution play_arrow
-
question_answer122)
Reaction of alkyl halides with aromatic compounds in presence of anhydrous \[AlC{{l}_{3}}\] is known as [UPSEAT 2004]
A)
Friedal-Craft reaction done
clear
B)
Hofmann degradation done
clear
C)
Kolbe's synthesis done
clear
D)
Beckmann rearrangement done
clear
View Solution play_arrow
-
question_answer123)
Two percent of ethanol is added during the oxidation of chloroform to stop the formation of carbonyl chloride. In this reaction ethanol acts as [Pb. CET 2001]
A)
Auto catalyst done
clear
B)
Negative catalyst done
clear
C)
Positive catalyst done
clear
D)
None of these done
clear
View Solution play_arrow
-
question_answer124)
When benzene is heated with chlorine in the presence of sunlight, it forms [Pb. CET 2000]
A)
B.H.C. done
clear
B)
Cyclopropane done
clear
C)
p-dichlorobenzene done
clear
D)
None of these done
clear
View Solution play_arrow
-
question_answer125)
Ethylene di bromide on heating with metallic sodium in ether solution yields [Pb. CET 2004]
A)
Ethene done
clear
B)
Ethyne done
clear
C)
2-butene done
clear
D)
1-butene done
clear
View Solution play_arrow
-
question_answer126)
The reaction, \[C{{H}_{3}}Br+Na\to \text{Product}\], is called [Pb. CET 2003]
A)
Perkin reaction done
clear
B)
Levit reaction done
clear
C)
Wurtz reaction done
clear
D)
Aldol condensation done
clear
View Solution play_arrow
-
question_answer127)
At normal temperature iodoform is [MP PET 2004]
A)
Thick viscous liquid done
clear
B)
Gas done
clear
C)
Volatile liquid done
clear
D)
Solid done
clear
View Solution play_arrow
-
question_answer128)
Which of the following statements about benzyl chloride is incorrect [KCET 2004]
A)
It is less reactive than alkyl halides done
clear
B)
It can be oxidised to benzaldehyde by boiling with copper nitrate solution done
clear
C)
It is a lachrymatory liquid and answers Beilstein?s test done
clear
D)
It gives a white precipitate with alcoholic silver nitrate done
clear
View Solution play_arrow
-
question_answer129)
Ethylene dichloride and ethylidine chloride are isomeric compounds. The false statement about these isomers is that they [DCE 2003]
A)
React with alcoholic potash and give the same product done
clear
B)
Are position isomers done
clear
C)
Contain the same percentage of chlorine done
clear
D)
Are both hydrolysed to the same product done
clear
View Solution play_arrow
-
question_answer130)
An alkyl bromide (X) reacts with Na to form 4, 5-diethyloctane. Compound X is [Roorkee 1999]
A)
\[C{{H}_{3}}{{(C{{H}_{2}})}_{3}}Br\] done
clear
B)
\[C{{H}_{3}}{{(C{{H}_{2}})}_{5}}Br\] done
clear
C)
\[C{{H}_{3}}{{(C{{H}_{2}})}_{3}}CH.Br.C{{H}_{3}}\] done
clear
D)
\[C{{H}_{3}}{{(C{{H}_{2}})}_{2}}CH.Br.C{{H}_{2}}C{{H}_{3}}\] done
clear
View Solution play_arrow
-
question_answer131)
In the following reaction X is \[C{{H}_{3}}N{{H}_{2}}+X+KOH\to C{{H}_{3}}NC\,\,\text{(highly offensive odour)}\] [MP PET 1994]
A)
\[C{{H}_{2}}C{{l}_{2}}\] done
clear
B)
\[CHC{{l}_{3}}\] done
clear
C)
\[C{{H}_{3}}Cl\] done
clear
D)
\[CC{{l}_{4}}\] done
clear
View Solution play_arrow
-
question_answer132)
Which metal is used in Wurtz synthesis [CPMT 1986; DPMT 1979; MP PET 2002]
A)
Ba done
clear
B)
Al done
clear
C)
Na done
clear
D)
Fe done
clear
View Solution play_arrow
-
question_answer133)
Which of the following is boiled with ethyl chloride to form ethyl alcohol [MNR 1982]
A)
Alcoholic KOH done
clear
B)
Aqueous KOH done
clear
C)
\[{{H}_{2}}O\] done
clear
D)
\[{{H}_{2}}{{O}_{2}}\] done
clear
View Solution play_arrow
-
question_answer134)
Why is chloroform put into dark coloured bottles [MP PET 2002]
A)
To prevent evaporation done
clear
B)
To prevent from moisture done
clear
C)
To prevent it from oxidation to form phosgene done
clear
D)
To prevent its reaction with glass done
clear
View Solution play_arrow
-
question_answer135)
DDT is
A)
A solid done
clear
B)
A liquid done
clear
C)
A gas done
clear
D)
A solution done
clear
View Solution play_arrow
-
question_answer136)
Bottles containing \[{{C}_{6}}{{H}_{5}}I\] and \[{{C}_{6}}{{H}_{5}}C{{H}_{2}}I\]lost their original labels. They were labelled A and B for testing. A and B were separately taken in test tubes and boiled with \[NaOH\]solution. The end solution in each tube was made acidic with dilute \[HN{{O}_{3}}\]and then some \[AgN{{O}_{3}}\] solution was added. Substance B give a yellow precipitate. Which one of the following statements is true for this experiment [AIEEE 2003]
A)
A was \[{{C}_{6}}{{H}_{5}}I\] done
clear
B)
A was \[{{C}_{6}}{{H}_{5}}C{{H}_{2}}I\] done
clear
C)
B was \[{{C}_{6}}{{H}_{5}}I\] done
clear
D)
Addition of \[HN{{O}_{3}}\]was unnecessary done
clear
View Solution play_arrow
-
question_answer137)
Which of the following statements is incorrect regarding benzyl chloride [KCET 2003]
A)
It gives white precipitate with alcoholic \[AgN{{O}_{3}}\] done
clear
B)
It is an aromatic compound with substitution in the side chain done
clear
C)
It undergoes nucleophilic substitution reaction done
clear
D)
It is less reactive than vinyl chloride done
clear
View Solution play_arrow
-
question_answer138)
Alkyl halide can be converted into alkene by [BCECE 2005]
A)
Nucleophilic substitution reaction done
clear
B)
Elimination reaction done
clear
C)
Both nucleophilic substitution and elimination reaction done
clear
D)
Rearrangement done
clear
View Solution play_arrow
-
question_answer139)
The major product formed in the following reaction is \[\underset{H}{\overset{\,\,\,\,\,\,\,\,\,C{{H}_{3}}}{\mathop{C{{H}_{3}}-\underset{|}{\overset{|}{\mathop{C}}}\,-C{{H}_{2}}}}}\,\ Br\ \ \underset{C{{H}_{3}}OH}{\mathop{\xrightarrow{C{{H}_{3}}O}}}\,\] [AIIMS 2005]
A)
\[\underset{H}{\overset{\,\,\,\,\,\,\,\,\,C{{H}_{3}}}{\mathop{C{{H}_{3}}-\underset{|}{\overset{|}{\mathop{C}}}\,-C{{H}_{2}}}}}\,OC{{H}_{3}}\] done
clear
B)
\[C{{H}_{3}}\underset{\,\,\,\,\,OC{{H}_{3}}}{\mathop{-\underset{|}{\mathop{C}}\,H-}}\,C{{H}_{2}}\ C{{H}_{3}}\] done
clear
C)
\[\overset{\,\,\,\,\,\,\,C{{H}_{3}}}{\mathop{C{{H}_{3}}-\overset{|}{\mathop{C}}\,=C{{H}_{2}}}}\,\] done
clear
D)
\[\underset{\,\,\,\,\,\,\,\,\,\,\,OC{{H}_{3}}}{\overset{\,\,\,\,\,\,\,\,C{{H}_{3}}}{\mathop{C{{H}_{3}}-\underset{|}{\overset{|}{\mathop{C}}}\,-C{{H}_{3}}}}}\,\] done
clear
View Solution play_arrow
-
question_answer140)
The major product obtained on treatment of \[C{{H}_{3}}C{{H}_{2}}CH(F)C{{H}_{3}}\] with \[C{{H}_{3}}{{O}^{-}}/C{{H}_{3}}OH\] is [AIIMS 2005]
A)
\[C{{H}_{3}}C{{H}_{2}}CH(OC{{H}_{3}})C{{H}_{3}}\] done
clear
B)
\[C{{H}_{3}}CH=CHC{{H}_{3}}\] done
clear
C)
\[C{{H}_{3}}C{{H}_{2}}CH=C{{H}_{2}}\] done
clear
D)
\[C{{H}_{3}}C{{H}_{2}}C{{H}_{2}}C{{H}_{2}}OC{{H}_{3}}\] done
clear
View Solution play_arrow
-
question_answer141)
When phenyl magnesium bromide reacts with t-butanol, the product would be [IIT 2005]
A)
Benzene done
clear
B)
Phenol done
clear
C)
t-butyl benzene done
clear
D)
t-butyl phenyl ether done
clear
View Solution play_arrow
-
question_answer142)
Alkyl halides react with dialkyl copper reagents to give [AIEEE 2005]
A)
Alkenes done
clear
B)
Alkyl copper halides done
clear
C)
Alkanes done
clear
D)
Alkenyl halides done
clear
View Solution play_arrow
-
question_answer143)
Which of the following is liquid at room temperature [AFMC 2005]
A)
\[C{{H}_{3}}I\] done
clear
B)
\[C{{H}_{3}}Br\] done
clear
C)
\[{{C}_{2}}{{H}_{5}}Cl\] done
clear
D)
\[C{{H}_{3}}F\] done
clear
View Solution play_arrow
-
question_answer144)
Which of the following haloalkanes is most reactive [KCET 2005]
A)
1-chloropropane done
clear
B)
1-bromopropane done
clear
C)
2-chloropropane done
clear
D)
2-bromopropane done
clear
View Solution play_arrow
-
question_answer145)
Grignard reagent adds to [KCET 2005]
A)
\[>C=O\] done
clear
B)
\[-C\equiv N\] done
clear
C)
\[>C=S\] done
clear
D)
All of the above done
clear
View Solution play_arrow
-
question_answer146)
Analyse the following reaction and identify the nature of \[A\] and \[B\] [Kerala CET 2005] \[B\underset{hv}{\mathop{\xleftarrow{HBr}}}\,\]
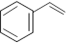
\[\xrightarrow{HBr}A\]
A)
B)
C)
D)
E)
View Solution play_arrow
 done
clear
done
clear
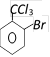 done
clear
done
clear
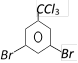 done
clear
done
clear
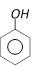 done
clear
done
clear
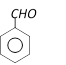 done
clear
done
clear
 In the above reaction product is
In the above reaction product is 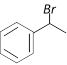 done
clear
done
clear
 & B is
& B is 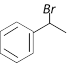 done
clear
done
clear
