question_answer 1)
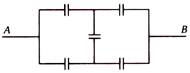
Each capacitor shown in figure is 2uF. Then the equivalent capacitance between A and B is
A)
2\[\mu \]F
done
clear
B)
4\[\mu \]F
done
clear
C)
6\[\mu \]F
done
clear
D)
8\[\mu \]F
done
clear
View Answer play_arrow
question_answer 2) What is the refractive index of a prism whose angle \[A=60{}^\circ \] and angle of minimum deviation \[dm=30{}^\circ \] ?
A)
\[\sqrt{2}\]
done
clear
B)
\[\frac{1}{\sqrt{2}}\]
done
clear
C)
1
done
clear
D)
\[\frac{1}{\sqrt{3}}\]
done
clear
View Answer play_arrow
question_answer 3) In a magnetic field of 0.05 T area of coil changes from 101 cm2 to 100 cm2 without changing the resistance which is 2\[\Omega \]. The amount of charge that flow during this period is
A)
\[2.5\times {{10}^{-6}}C\]
done
clear
B)
\[2\times {{10}^{-6}}C\]
done
clear
C)
\[{{10}^{-6}}C\]
done
clear
D)
\[8\times {{10}^{-6}}C\]
done
clear
View Answer play_arrow
question_answer 4) Which has more luminous efficiency?
A)
A 40 W bulb
done
clear
B)
A 40 W fluorescent tube
done
clear
C)
Both have same
done
clear
D)
Cannot say
done
clear
View Answer play_arrow
question_answer 5) A circular loop and a square loop are formed from the same wire and the same current is passed through them. Find the ratio of their dipole moments
A)
4\[\pi \]
done
clear
B)
\[\frac{4}{\pi }\]
done
clear
C)
\[\frac{2}{\pi }\]
done
clear
D)
2\[\pi \]
done
clear
View Answer play_arrow
question_answer 6) A jet of water with a cross-sectional area a is striking against a wall at an angle \[\theta \]to the horizontal and rebounds elastically. If the velocity of water jet is v and the density is \[\rho ,\] the normal force acting on the wall is
A)
\[2a{{v}^{2}}\rho \,\cos \theta \]
done
clear
B)
\[a{{v}^{2}}\rho \,\cos \theta \]
done
clear
C)
\[2av\rho \,\cos \theta \]
done
clear
D)
\[av\,\cos \theta \]
done
clear
View Answer play_arrow
question_answer 7) If a surface has a work function 4.0 eV, what is the maximum velocity of electrons liberated from the surface when it is irradiated with ultraviolet radiation of wavelength 0.2 \[\mu \]m?
A)
\[4.4\times {{10}^{5}}m/s\]
done
clear
B)
\[8.8\times {{10}^{7}}m/s\]
done
clear
C)
\[8.8\times {{10}^{5}}m/s\]
done
clear
D)
\[4.4\times {{10}^{7}}m/s\]
done
clear
View Answer play_arrow
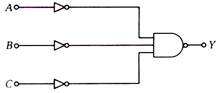
question_answer 8)
A proper combination of 3 NOT and 1 AND gates is shown. If A = 0, B = 1, C = 1 then the output of this combination is
A)
1
done
clear
B)
zero
done
clear
C)
not predictable
done
clear
D)
None of these
done
clear
View Answer play_arrow
question_answer 9) If \[\gamma \] denotes the ratio of the two specific heats of a gas, the ratio of the slopes of adiabatic and isothermal p-V curves at their point of intersection is
A)
1/\[\gamma \]
done
clear
B)
\[\gamma \]
done
clear
C)
\[\gamma \]- 1
done
clear
D)
\[\gamma \]+1
done
clear
View Answer play_arrow
question_answer 10) The weight of a body on surface of earth is 12.6 N. When it is raised to a height half the radius of earth its weight will be
A)
2.8 N
done
clear
B)
5.6 N
done
clear
C)
12.5 N
done
clear
D)
25.2 N
done
clear
View Answer play_arrow
question_answer 11) A particle having a mass 0.5 kg is projected under gravity with a speed of 98 m/s at an angle of 60°. The magnitude of the change in momentum in kg- m/s of the particle after 10 s is
A)
490
done
clear
B)
98
done
clear
C)
49
done
clear
D)
0.5
done
clear
View Answer play_arrow
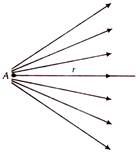
question_answer 12)
The given figure shows the electric lines of force emerging from a charged body. If the electric field at A and B are EA and EB respectively and if the displacement between A and B is r, then
A)
\[{{E}_{A}}>{{E}_{B}}\]
done
clear
B)
\[{{E}_{A}}<{{E}_{B}}\]
done
clear
C)
\[{{E}_{A}}=\frac{{{E}_{B}}}{r}\]
done
clear
D)
\[{{E}_{A}}=\frac{{{E}_{B}}}{{{r}^{2}}}\]
done
clear
View Answer play_arrow
question_answer 13) When the room temperature becomes equal to the dew point, the relative humidity of the room is
A)
100 %
done
clear
B)
zero %
done
clear
C)
70 %
done
clear
D)
85 %
done
clear
View Answer play_arrow
question_answer 14) A transistor is working in common emitter mode. Its amplification factor\[\left( \beta \right)\]is 80. If the base current is 250 \[\mu A\], the collector current will be
A)
\[1.25\,\mu A\]
done
clear
B)
\[\frac{250}{80}\,\mu A\]
done
clear
C)
\[430\,\mu A\]
done
clear
D)
\[250\times 80\,\mu A\]
done
clear
View Answer play_arrow
question_answer 15) You are marooned on a frictionless horizontal plane and cannot exert any horizontal force by pushing against the surface. How can you get off
A)
by jumping
done
clear
B)
by rolling your body on the surface
done
clear
C)
by splitting or sneezing or throwing any object
done
clear
D)
by throwing an object in opposite direction
done
clear
View Answer play_arrow
question_answer 16) A hollow cylinder with both sides open generates a frequency f in air. When the cylinder vertically immersed into water by half its length the frequency will be
A)
f
done
clear
B)
2f
done
clear
C)
f/2
done
clear
D)
f/4
done
clear
View Answer play_arrow
question_answer 17) If a shunt of \[\frac{1}{10}th\] of the coil resistance is applied to a moving coil galvanometer, its sensitivity becomes
A)
10 fold
done
clear
B)
\[\frac{1}{10}\] fold
done
clear
C)
11 fold
done
clear
D)
\[\frac{1}{11}\] fold
done
clear
View Answer play_arrow
question_answer 18) Solar radiation is
A)
transverse electromagnetic wave
done
clear
B)
longitudinal electromagnetic wave
done
clear
C)
stationary wave
done
clear
D)
None of the above
done
clear
View Answer play_arrow
question_answer 19) If 1 mg of U235 is completely annihilated, the energy liberated is
A)
\[9\times {{10}^{10}}J\]
done
clear
B)
\[9\times {{10}^{19}}J\]
done
clear
C)
\[9\times {{10}^{18}}J\]
done
clear
D)
\[9\times {{10}^{17}}J\]
done
clear
View Answer play_arrow
question_answer 20) Two stretched strings have lengths l and 2l while tensions are T and 4T respectively. If they are made of same material the ratio of their frequencies is
A)
2 : 1
done
clear
B)
1 : 2
done
clear
C)
1 : 1
done
clear
D)
1 : 4
done
clear
View Answer play_arrow
question_answer 21) An ideal gas expands along the path AB as shown in the p-V diagram. The work done is
A)
\[4\times {{10}^{4}}J\]
done
clear
B)
\[~1.2\times {{10}^{5}}J\]
done
clear
C)
\[2.4\times {{10}^{5}}J\]
done
clear
D)
None of these
done
clear
View Answer play_arrow
question_answer 22) When sound is produced in an aeroplane moving with a velocity of 200 m/s horizontal its echo is heard after 10\[\sqrt{5}\]. If velocity or sound in air is 300 m/s, the elevation of aircraft is
A)
250 m
done
clear
B)
250\[\sqrt{5}\] m
done
clear
C)
1250 m
done
clear
D)
2500 m
done
clear
View Answer play_arrow
question_answer 23) A solenoid 30 cm long is made by winding 2000 loops of wire on an iron rod whose cross-section is 1.5 cm2. If the relative permeability of the iron is 6000, what is the self-inductance of the solenoid?
A)
1.5 H
done
clear
B)
2.5 H
done
clear
C)
3.5 H
done
clear
D)
0.5 H
done
clear
View Answer play_arrow
question_answer 24) An experiment takes 10 min to raise temperature of water from 0°C and 100°C and another 55 min to convert it totally into steam by a stabilized heater. The latent heat of vaporization comes out to be
A)
530 cal/g
done
clear
B)
540 cal/g
done
clear
C)
550 cal/g
done
clear
D)
560 cal/g
done
clear
View Answer play_arrow
question_answer 25) Two points on a travelling wave having frequency 500 Hz and velocity 300 m/s are 60° out of phase, then the minimum distance between the two points is
A)
0.2
done
clear
B)
0.1
done
clear
C)
0.5
done
clear
D)
0.4
done
clear
View Answer play_arrow
question_answer 26) A resistor of 6 k\[\Omega \] with tolerance 10% and another of 4 k\[\Omega \] with tolerance 10% are connected in series. The tolerance of combination is about
A)
5%
done
clear
B)
10%
done
clear
C)
12%
done
clear
D)
15%
done
clear
View Answer play_arrow
question_answer 27) A cooking pot should have
A)
high specific heat and low conductivity
done
clear
B)
high specific heat and high conductivity
done
clear
C)
low specific heat and low conductivity
done
clear
D)
low specific heat and high conductivity
done
clear
View Answer play_arrow
question_answer 28) The phenomenon of Brownian movement may be taken as evidence of
A)
kinetic theory of matter
done
clear
B)
EMT of radiation
done
clear
C)
corpuscular theory of light
done
clear
D)
photoelectric phenomenon
done
clear
View Answer play_arrow
question_answer 29) In a simple cubic lattice, the co-ordination number is
A)
4
done
clear
B)
5
done
clear
C)
6
done
clear
D)
8
done
clear
View Answer play_arrow
question_answer 30) A proton and an \[\alpha \]-particle, accelerated through the same potential difference, enter a region of uniform magnetic field normally. If the radius of the proton orbit is 10 cm, the radius of \[\alpha \]-orbit is
A)
10 cm
done
clear
B)
10\[\sqrt{2}\] cm
done
clear
C)
20 cm
done
clear
D)
5\[\sqrt{2}\] cm
done
clear
View Answer play_arrow
question_answer 31)
Three identical thermal conductors are connected as shown in figure. Considering no heat loss due to radiation, temperature at the junction will be
A)
\[40{}^\circ \]
done
clear
B)
\[60{}^\circ \]
done
clear
C)
\[50{}^\circ \]
done
clear
D)
\[35{}^\circ \]
done
clear
View Answer play_arrow
question_answer 32) In a noiseless transformer an alternating current of 2 A is flowing in the primary coil. The number of turns on the primary and secondary coils are 100 and 20 respectively. The value of the current in the secondary coil is
A)
0.08 A
done
clear
B)
0.4 A
done
clear
C)
5 A
done
clear
D)
10 A
done
clear
View Answer play_arrow
question_answer 33) When the wavelength of sound changes from 1 m to 1.01 m, the number of beats heard are 4. The velocity of sound is
A)
404 m/s
done
clear
B)
4.04 m/s
done
clear
C)
414 m/s
done
clear
D)
400 m/s
done
clear
View Answer play_arrow
question_answer 34) A satellite with kinetic energy E revolving round the earth in a circular orbit. The minimum additional KE required for it to escape into outer space is
A)
\[\sqrt{2}\]E
done
clear
B)
2 E
done
clear
C)
E/\[\sqrt{2}\]
done
clear
D)
E
done
clear
View Answer play_arrow
question_answer 35) 1 Wb /m2 is equal to
A)
104 G
done
clear
B)
102 G
done
clear
C)
10-2 G
done
clear
D)
10-4 G
done
clear
View Answer play_arrow
question_answer 36) When a spring is extended by 2 cm energy stored is 100 J. When extended by further 2 cm, the energy increases by
A)
400 J
done
clear
B)
300 J
done
clear
C)
200 J
done
clear
D)
100 J
done
clear
View Answer play_arrow
question_answer 37) A mass M is split into two pans m and (M - m), which are, then separated by a certain distance. The ratio m/ M which maximizes the gravitational force between the pans is
A)
1 : 4
done
clear
B)
1 : 3
done
clear
C)
1 : 2
done
clear
D)
1 : 1
done
clear
View Answer play_arrow
question_answer 38) Two bulbs 40 W and 60 W and rated voltage 240 V are connected in series across a potential difference of 420 V. Which bulb will work at above its rated voltage?
A)
40 W bulb
done
clear
B)
60 W bulb
done
clear
C)
Both will work
done
clear
D)
None of these
done
clear
View Answer play_arrow
question_answer 39) A wheel of radius 5 cm rotates so, that the relationship between the angle of rotation of wheel radius and the time is described by the equation \[\phi =A+Bt+C{{t}^{2}}+D{{t}^{3}}\]where, D = 1 rad /s2. The change in tangential acceleration during each second of the motion for the point on the rim is
A)
0.3 ms-2
done
clear
B)
0.03 ms-2
done
clear
C)
3 ms-2
done
clear
D)
0.09 ms-2
done
clear
View Answer play_arrow
question_answer 40) A convex lens is placed between object and a screen. The size of object is 3 cm and an image of height 9 cm is obtained on the screen. When the lens is displaced to a new position, what will be the size of image on the screen?
A)
2 cm
done
clear
B)
6 cm
done
clear
C)
4 cm
done
clear
D)
1 cm
done
clear
View Answer play_arrow
question_answer 41) An ideal gas (\[\gamma \]= 1.5) is expanded adiabatically. How many times has the gas to be expanded to reduce the root mean square velocity of the molecules 2.0 times?
A)
8 times
done
clear
B)
16 times
done
clear
C)
\[\frac{1}{8}\] times
done
clear
D)
4 times
done
clear
View Answer play_arrow
question_answer 42) A simple pendulum with length L and mass of the both is vibrating with an amplitude a. Then the maximum tension in the string is
A)
mg
done
clear
B)
mg \[\left[ 1+{{\left( \frac{a}{L} \right)}^{2}} \right]\]
done
clear
C)
\[mg{{\left[ 1+\left( \frac{a}{2L} \right) \right]}^{2}}\]
done
clear
D)
\[mg{{\left[ 1+\left( \frac{a}{L} \right) \right]}^{2}}\]
done
clear
View Answer play_arrow
question_answer 43) A ball rolls off the top of stairway with a horizontal velocity of magnitude 1.8 m/s. The slops are 0.20 m high and 0.20 m wide. Which step will the ball hit first?
A)
First
done
clear
B)
Second
done
clear
C)
Third
done
clear
D)
Fourth
done
clear
View Answer play_arrow
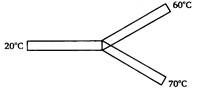
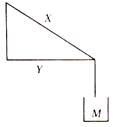
question_answer 44)
Two rods X and Y are attached to a weight of mass M as shown in figure, then
A)
both X and Y experience compression
done
clear
B)
both X and Y experience extension
done
clear
C)
X experiences extension and Y compression
done
clear
D)
Y experiences compression and X Extension
done
clear
View Answer play_arrow
question_answer 45) A disc of radius 0.1 m is rotating with a frequency 10 rev/s in a normal magnetic field of strength 0.1 T. Net induced emf is
A)
\[2\pi \times {{10}^{-2}}V\]
done
clear
B)
\[\pi \times {{10}^{-2}}V\]
done
clear
C)
\[\frac{\pi }{2}\times {{10}^{-2}}V\]
done
clear
D)
None of these
done
clear
View Answer play_arrow
question_answer 46) If force (F), length (L) and time (T) be considered fundamental units, then units of mass will be
A)
[FLT-2]
done
clear
B)
[FL-2T-1]
done
clear
C)
[FL-1T2]
done
clear
D)
[F2 L T-2]
done
clear
View Answer play_arrow
question_answer 47) The p-V diagram of 2 g of helium gas for a certain process A \[\to \]B is shown in the figure. What is the heat given to the gas during the process A \[\to \] B?
A)
4p0V0
done
clear
B)
6p0V0
done
clear
C)
4.5p0V0
done
clear
D)
2p0V0
done
clear
View Answer play_arrow
question_answer 48) Find the maximum velocity with which a train can be moved on a circular track of radius 200 m. The banking of the track is 5.71° (given, tan 5.71° = 0.1)
A)
1.4 m/s
done
clear
B)
14 m/s
done
clear
C)
140 m/s
done
clear
D)
0.14 m/s
done
clear
View Answer play_arrow
question_answer 49) The dimensions of Planck's constant is same as that of
A)
angular momentum
done
clear
B)
linear momentum
done
clear
C)
work
done
clear
D)
coefficient of viscosity
done
clear
View Answer play_arrow
question_answer 50) With what minimum acceleration can a fireman slide down a rope while breaking strength of the rope is \[\frac{2}{3}\]of the weight?
A)
\[\frac{2}{3}g\]
done
clear
B)
g
done
clear
C)
\[\frac{1}{3}g\]
done
clear
D)
Zero
done
clear
View Answer play_arrow
question_answer 51) The simplest way to check whether a system is colloidal is by
A)
electrodialysis
done
clear
B)
finding out particle size
done
clear
C)
tyndall effect
done
clear
D)
Brownian movement
done
clear
View Answer play_arrow
question_answer 52) In the reaction \[{{H}_{2}}(g)+C{{l}_{2}}(g)2HCl(g)\]
A)
\[{{K}_{p}}\ne {{K}_{c}}\]
done
clear
B)
\[{{K}_{p}}={{K}_{c}}\]
done
clear
C)
\[{{K}_{p}}>{{K}_{c}}\]
done
clear
D)
\[{{K}_{p}}<{{K}_{c}}\]
done
clear
View Answer play_arrow
question_answer 53) One mole of oxygen at 273 K and one mole of sulphur dioxide at 546 K are taken in two separate containers, then,
A)
kinetic energy of \[{{O}_{2}}>\] kinetic energy of \[S{{O}_{2}}\]
done
clear
B)
kinetic energy of \[{{O}_{2}}<\]kinetic energy of \[S{{O}_{2}}\]
done
clear
C)
kinetic energy of both are qual
done
clear
D)
None of the above
done
clear
View Answer play_arrow
question_answer 54) The complexes \[[PtC{{l}_{2}}{{(N{{H}_{3}})}_{4}}]B{{r}_{2}}\] and\[[PtB{{r}_{2}}{{(N{{H}_{3}})}_{4}}]C{{l}_{2}}\] are examples for isomerism
A)
geometrical
done
clear
B)
optical
done
clear
C)
ionisation
done
clear
D)
linkage
done
clear
View Answer play_arrow
question_answer 55) The electronic configuration of \[C{{r}^{3+}}\]is
A)
\[\text{ }\!\![\!\!\text{ }Ar\text{ }\!\!]\!\!\text{ }3{{d}^{4}}4{{s}^{2}}\]
done
clear
B)
\[\text{ }\!\![\!\!\text{ }Ar\text{ }\!\!]\!\!\text{ }3{{d}^{3}}4{{s}^{0}}\]
done
clear
C)
\[\text{ }\!\![\!\!\text{ }Ar\text{ }\!\!]\!\!\text{ }3{{d}^{2}}4{{s}^{1}}\]
done
clear
D)
\[\text{ }\!\![\!\!\text{ }Ar\text{ }\!\!]\!\!\text{ }3{{d}^{5}}4{{s}^{1}}\]
done
clear
View Answer play_arrow
question_answer 56) Angle strain in cyclopropane is
A)
\[{{24}^{o}}44'\]
done
clear
B)
\[{{9}^{o}}44'\]
done
clear
C)
44'
done
clear
D)
\[-{{5}^{o}}16'\]
done
clear
View Answer play_arrow
question_answer 57) Haloalkane in the presence of alcoholic KOH undergoes
A)
elimination
done
clear
B)
polymerization
done
clear
C)
dimerisation
done
clear
D)
substitution
done
clear
View Answer play_arrow
question_answer 58) Identify the ore not containing iron
A)
limonite
done
clear
B)
siderite
done
clear
C)
camallite
done
clear
D)
chalcopyrites
done
clear
View Answer play_arrow
question_answer 59) Ammonium ion is
A)
neither an acid nor base
done
clear
B)
both an acid and a base
done
clear
C)
a conjugate acid
done
clear
D)
a conjugate base
done
clear
View Answer play_arrow
question_answer 60) What is the equivalent weight of \[\text{SnC}{{\text{l}}_{\text{2}}}\]in the following reaction,\[\text{SnC}{{\text{l}}_{2}}\xrightarrow[{}]{{}}SnC{{l}_{4}}?\]
A)
95
done
clear
B)
45
done
clear
C)
60
done
clear
D)
30
done
clear
View Answer play_arrow
question_answer 61) During the formation of a chemical bond
A)
electron-electron repulsion becomes more than the nucleus-electron attraction
done
clear
B)
energy of the system does not change
done
clear
C)
energy increases
done
clear
D)
energy decreases
done
clear
View Answer play_arrow
question_answer 62) Nitration of nitrobenzene at \[125{{\,}^{o}}C\]with mixed acids gives
A)
meta-dinitrobenzene
done
clear
B)
ortho-dinitrobenzene
done
clear
C)
para-dinitrobenzene
done
clear
D)
1, 3, 5-trinitro benzene
done
clear
View Answer play_arrow
question_answer 63) Benzene reacts with chlorine in sunlight to give a final product
A)
\[CC{{l}_{4}}\]
done
clear
B)
\[{{C}_{6}}{{H}_{6}}C{{l}_{6}}\]
done
clear
C)
\[{{C}_{6}}C{{l}_{6}}\]
done
clear
D)
\[{{C}_{6}}{{H}_{5}}Cl\]
done
clear
View Answer play_arrow
question_answer 64) Hofmann's bromamide reaction is to convert
A)
acid to alcohol
done
clear
B)
alcohol to acid
done
clear
C)
amide to amine
done
clear
D)
amine to amide
done
clear
View Answer play_arrow
question_answer 65) What is the time (in sec) required for depositing all the silver present in 125 mL of \[\text{1 M AgN}{{\text{O}}_{\text{3}}}\] solution by passing a current of 241.25 A? (IF = 96500 coulombs)
A)
10
done
clear
B)
50
done
clear
C)
1000
done
clear
D)
100
done
clear
View Answer play_arrow
question_answer 66) The correct order in which the first ionisation potential increases is
A)
Na, K, Be
done
clear
B)
K, Na, Be
done
clear
C)
K, Be, Na
done
clear
D)
Be, Na, K
done
clear
View Answer play_arrow
question_answer 67) When cone. \[{{H}_{2}}S{{O}_{4}}\] is heated with \[{{P}_{2}}{{O}_{5}},\] the acid is converted into
A)
sulphur trioxide
done
clear
B)
sulphur dioxide
done
clear
C)
sulphur
done
clear
D)
a mixture of sulphur dioxide and sulphur trioxide
done
clear
View Answer play_arrow
question_answer 68) \[\text{M}{{\text{g}}^{\text{2+}}}\] is isoelectronic with
A)
\[C{{u}^{2+}}\]
done
clear
B)
\[Z{{n}^{2+}}\]
done
clear
C)
\[N{{a}^{+}}\]
done
clear
D)
\[C{{a}^{2+}}\]
done
clear
View Answer play_arrow
question_answer 69) Gram molecular volume of oxygen at STP is
A)
\[3200\text{ }c{{m}^{3}}\]
done
clear
B)
\[5600\text{ }c{{m}^{3}}\]
done
clear
C)
\[22400\,c{{m}^{3}}\]
done
clear
D)
\[11200\,c{{m}^{3}}\]
done
clear
View Answer play_arrow
question_answer 70) \[+I\]effect is shown by
A)
\[-C{{H}_{3}}\]
done
clear
B)
\[-Br\]
done
clear
C)
\[-Cl\]
done
clear
D)
\[-N{{O}_{2}}\]
done
clear
View Answer play_arrow
question_answer 71) The amino acid which is not optically active is
A)
lactic acid
done
clear
B)
serine
done
clear
C)
alanine
done
clear
D)
glycine
done
clear
View Answer play_arrow
question_answer 72) A condensation polymer among the following polymers is
A)
teflon
done
clear
B)
polysterene
done
clear
C)
PVC
done
clear
D)
decron
done
clear
View Answer play_arrow
question_answer 73) Dehydration of alcohol is an example of which type of reaction?
A)
Substitution
done
clear
B)
Elimination
done
clear
C)
Addition
done
clear
D)
Rearrangement
done
clear
View Answer play_arrow
question_answer 74) When sulphur dioxide is passed in an acidified \[{{K}_{2}}C{{r}_{2}}{{O}_{7}}\]solution, the oxidation state of sulphur is changed from
A)
+ 4 to 0
done
clear
B)
+ 4 to + 2
done
clear
C)
+ 4 to + 6
done
clear
D)
+ 6 to + 4
done
clear
View Answer play_arrow
question_answer 75) Which among the following statement is false?
A)
The adsorption may be monolayered or multilayered
done
clear
B)
Particle size of adsorbent will not affect the amount of adsorption
done
clear
C)
Increase of pressure increases amount of adsorption
done
clear
D)
All of the above
done
clear
View Answer play_arrow
question_answer 76) Molarity of a given orthophosphoric acid solution is 3 M. Its normality is
A)
9 N
done
clear
B)
0.3 N
done
clear
C)
3 N
done
clear
D)
1 N
done
clear
View Answer play_arrow
question_answer 77) Graphite is a soft solid lubricant extremely difficult to melt. The reason for this anomalous behaviour is that graphite
A)
is an allotropic form of carbon
done
clear
B)
is a non-crystalline substance
done
clear
C)
has carbon atoms arranged in large plates of rings of strongly bonded carbon atoms with weak inter plate bonds
done
clear
D)
has molecules of variable molecular masses like polymers
done
clear
View Answer play_arrow
question_answer 78) \[{{C}^{14}}\]is
A)
a natural non-radioactive isotope
done
clear
B)
an artificial non-radioactive isotope
done
clear
C)
an artificial radioactive isotope
done
clear
D)
a natural radioactive isotope
done
clear
View Answer play_arrow
question_answer 79) Presence of halogen in organic compounds can be detected using
A)
Leibig'stest
done
clear
B)
Duma's test
done
clear
C)
Kjeldahal test
done
clear
D)
Beilstien's test
done
clear
View Answer play_arrow
question_answer 80) Which one of the following is an example of non-reducing sugar?
A)
Sucrose
done
clear
B)
Lactose
done
clear
C)
Maltose
done
clear
D)
Cellobiose
done
clear
View Answer play_arrow
question_answer 81) Covalent compounds have low melting point because
A)
covalent molecules are held by van der Waals' force of attraction
done
clear
B)
covalent bond is less exothermic
done
clear
C)
covalent bond is weaker than ionic bond
done
clear
D)
covalent molecules have definate shape
done
clear
View Answer play_arrow
question_answer 82) Dalton's law of partial pressure is applicable to which one of the following systems?
A)
\[\text{N}{{\text{H}}_{\text{3}}}\text{+ HC1}\]
done
clear
B)
\[NO+{{O}_{2}}\]
done
clear
C)
\[{{H}_{2}}+C{{l}_{2}}\]
done
clear
D)
\[CO+{{H}_{2}}\]
done
clear
View Answer play_arrow
question_answer 83) Which of the following is an intensive property?
A)
Temperature
done
clear
B)
Viscosity
done
clear
C)
Surface tension
done
clear
D)
All of these
done
clear
View Answer play_arrow
question_answer 84) A metal present in vitamin \[{{\text{B}}_{\text{12}}}\] is
A)
aluminium
done
clear
B)
zinc
done
clear
C)
iron
done
clear
D)
cobalt
done
clear
View Answer play_arrow
question_answer 85) The oxidation state of iron in \[{{K}_{4}}[Fe{{(CN)}_{6}}]\]is
A)
1
done
clear
B)
4
done
clear
C)
3
done
clear
D)
2
done
clear
View Answer play_arrow
question_answer 86) Half-life of a reaction is found to be inversely proportional to the cube of initial concentration. The order of reaction is
A)
5
done
clear
B)
2
done
clear
C)
4
done
clear
D)
3
done
clear
View Answer play_arrow
question_answer 87) The most basic compound in the following is
A)
\[\text{N}{{\text{H}}_{\text{3}}}\]
done
clear
B)
\[\text{C}{{\text{H}}_{\text{3}}}\text{N}{{\text{H}}_{\text{2}}}\]
done
clear
C)
\[\text{HN(C}{{\text{H}}_{\text{3}}}{{\text{)}}_{\text{2}}}\]
done
clear
D)
\[N{{(C{{H}_{3}})}_{3}}\]
done
clear
View Answer play_arrow
question_answer 88) The reagent used in Clemmensen's reduction is
A)
cone. \[{{\text{H}}_{\text{2}}}\text{S}{{\text{O}}_{\text{4}}}\]
done
clear
B)
Zn - Hg/conc. \[\text{HCl}\]
done
clear
C)
\[aq.\text{ }KOH\]
done
clear
D)
\[alc.\text{ }KOH\]
done
clear
View Answer play_arrow
question_answer 89) Geometrical isomerism is shown by
A)
\[CC~\]
done
clear
B)
\[>C=C<\]
done
clear
C)
\[~C\equiv C\]
done
clear
D)
None of these
done
clear
View Answer play_arrow
question_answer 90) A compound that undergoes bromination easily is
A)
toluene
done
clear
B)
benzoic acid
done
clear
C)
phenol
done
clear
D)
benzene
done
clear
View Answer play_arrow
question_answer 91) Reaction of aniline with benzaldehyde is
A)
polymerization
done
clear
B)
condensation
done
clear
C)
addition
done
clear
D)
substitution
done
clear
View Answer play_arrow
question_answer 92) Entropy of the universe is
A)
constant
done
clear
B)
zero
done
clear
C)
continuously decreasing
done
clear
D)
continuously increasing
done
clear
View Answer play_arrow
question_answer 93) Aluminium oxide is not reduced by chemical reactions since
A)
reducing agent contaminate
done
clear
B)
the process pollute the environment
done
clear
C)
aluminium oxide is highly stable
done
clear
D)
aluminium oxide is reactive
done
clear
View Answer play_arrow
question_answer 94) The incorrect statement in respect of chromyi chloride test is
A)
formation of red vapours
done
clear
B)
formation of lead chromate
done
clear
C)
formation of chromyl chloride
done
clear
D)
liberation of chlorine
done
clear
View Answer play_arrow
question_answer 95) The set of compounds in which the reactivity of halogen atom in the ascending order is
A)
vinyl chloride, chloroethane, chlorobenzene
done
clear
B)
vinyl chloride, chlorobenzene, chloroethane
done
clear
C)
chloroethane, chlorobenzene, vinyl chloride
done
clear
D)
chlorobenzene, vinyl chloride, chloroethane
done
clear
View Answer play_arrow
question_answer 96) The IUPAC name for tertiary butyl iodide is
A)
4-iodo butane
done
clear
B)
2-iodo butane
done
clear
C)
1-iodo-3-methyl propane
done
clear
D)
2-iodo-2-methyl propane
done
clear
View Answer play_arrow
question_answer 97) Glacial acetic acid is obtained by
A)
chemically separating acetic acid
done
clear
B)
treating vinegar with dehydrating agents
done
clear
C)
crystallising, separating and melting acetic acid
done
clear
D)
distilling vinegar
done
clear
View Answer play_arrow
question_answer 98) Iron loses magnetic property at
A)
melting point
done
clear
B)
1000 K
done
clear
C)
Curie point
done
clear
D)
boiling point
done
clear
View Answer play_arrow
question_answer 99) The IUPAC name of \[{{K}_{2}}[Ni{{(CN)}_{4}}]\]is
A)
potassium tetracyanonickelate (II)
done
clear
B)
potassium tetracyanato nickelate (III)
done
clear
C)
potassium tetracyanatonickel (II)
done
clear
D)
potassium tetracyanonickel (III)
done
clear
View Answer play_arrow
question_answer 100) Which one of the following is aromatic?
A)
Cyclopentadienyl cation
done
clear
B)
Cyclooctatetraene
done
clear
C)
Cycloheptatriene
done
clear
D)
Cycloheptatrienylcation
done
clear
View Answer play_arrow
question_answer 101) Fear of dirt, disease and contaminants is.
A)
pathophobia
done
clear
B)
mysophobia
done
clear
C)
algophobia
done
clear
D)
None of these
done
clear
View Answer play_arrow
question_answer 102) The homeostatic regulation of an animal requires three basic components, ...... to detect changes,......to evaluate the changes and......to adjust the changes respectively
A)
receptoraffectors effectors
done
clear
B)
brain spinal cord, effectors
done
clear
C)
receptors integrators effectors
done
clear
D)
receptorsintegratons effectors
done
clear
View Answer play_arrow
question_answer 103) All plants and animals were created and perpetuated themselves without any change, is the theory of genesis given by
A)
Hulton
done
clear
B)
Cuvier
done
clear
C)
Haldane
done
clear
D)
Oparin
done
clear
View Answer play_arrow
question_answer 104) Which is not a vestigial organ in man?
A)
Wisdom teeth
done
clear
B)
Muscles of external ear-pinna
done
clear
C)
Fossa oval is
done
clear
D)
Ileum
done
clear
View Answer play_arrow
question_answer 105) ?Sexual selection? in Darwin theory was the
A)
wearest part
done
clear
B)
strong part
done
clear
C)
strongest part
done
clear
D)
None of these
done
clear
View Answer play_arrow
question_answer 106) Which of the following monkey has prehensile tail?
A)
Loris
done
clear
B)
Tarsiers
done
clear
C)
Spider monkey
done
clear
D)
Rhesus monkey
done
clear
View Answer play_arrow
question_answer 107) The odd one amongst following is
A)
Nitrococcus, Nitrobacter, Nitrosomonas
done
clear
B)
Rhizobiurn, Aulosira, Bacillus, Polymyxa
done
clear
C)
Clostridium, Rhizobiurn, Nostoc
done
clear
D)
Anabaena, Nostoc, Azotobacter, Clostridium
done
clear
View Answer play_arrow
question_answer 108) In algae, the bacteriological filter is
A)
Oscillatoria
done
clear
B)
Batrachospermum
done
clear
C)
Nostoc
done
clear
D)
Cosmarium
done
clear
View Answer play_arrow
question_answer 109) Schizogony of Plasmodium is also called as
A)
sporogony
done
clear
B)
gamogony
done
clear
C)
agamogony
done
clear
D)
binary fission
done
clear
View Answer play_arrow
question_answer 110) Lime-knots of slime moulds are
A)
swarm cells
done
clear
B)
spore
done
clear
C)
capillitia
done
clear
D)
fruit bodies
done
clear
View Answer play_arrow
question_answer 111) The total number of ascospores in Penicillium are
A)
8
done
clear
B)
16
done
clear
C)
4
done
clear
D)
2
done
clear
View Answer play_arrow
question_answer 112) One of the following is true moss.
A)
Irish moss
done
clear
B)
Bog moss
done
clear
C)
Club moss
done
clear
D)
Reindeer moss
done
clear
View Answer play_arrow
question_answer 113) How many meiosis occur in the sporangium of Nephrolepis?
A)
1
done
clear
B)
4
done
clear
C)
16
done
clear
D)
Uncertain and variable
done
clear
View Answer play_arrow
question_answer 114) Which of the following is categorised under living fossils?
A)
Pinus
done
clear
B)
Cycas
done
clear
C)
Selaginella
done
clear
D)
Fucus
done
clear
View Answer play_arrow
question_answer 115) Haustorial root is exceptional to
A)
Zea mays
done
clear
B)
Cactus
done
clear
C)
Cuscuta
done
clear
D)
Monotropa
done
clear
View Answer play_arrow
question_answer 116) Hydra can paralyse prey by
A)
nematocysts
done
clear
B)
tentacles
done
clear
C)
mouth
done
clear
D)
None of these
done
clear
View Answer play_arrow
question_answer 117) Chloragogen cells of earthworm are similar to......vertebrate organ
A)
spleen
done
clear
B)
liver
done
clear
C)
kidney
done
clear
D)
lung
done
clear
View Answer play_arrow
question_answer 118) Coelom in Echinodermata is
A)
pseudocoelic
done
clear
B)
haemocoelic
done
clear
C)
schizocoelic
done
clear
D)
enterocoelic
done
clear
View Answer play_arrow
question_answer 119) I sin glass is prepared from
A)
Ichthyophis
done
clear
B)
lungs
done
clear
C)
fishes
done
clear
D)
lizards
done
clear
View Answer play_arrow
question_answer 120) How can a tadpole be prevented to change into frog for long time?
A)
By giving very less food
done
clear
B)
By giving fat-rich food
done
clear
C)
By adding lot of thyroxine to the water in which they are kept
done
clear
D)
provide them an antithyroid substance like thiourea
done
clear
View Answer play_arrow
question_answer 121) The person with Turner?s syndrome has
A)
45 autosomes and X sex chromosome
done
clear
B)
44 autosomes and XYY sex chromosomes
done
clear
C)
45 autosomes and XYY sex chromosomes
done
clear
D)
44 autosomes and X sex chromosomes
done
clear
View Answer play_arrow
question_answer 122) Anticlinal or radial division mean division in
A)
transverse plane
done
clear
B)
at right angle to outer wall
done
clear
C)
plane parallel to outer wall
done
clear
D)
at any plane
done
clear
View Answer play_arrow
question_answer 123) Gaseous exchange in submerged hydrophytes takes place through
A)
lenticles
done
clear
B)
stomata
done
clear
C)
hydathodes
done
clear
D)
general surface of plants
done
clear
View Answer play_arrow
question_answer 124) Increase in circumference of stem is due to the activity of
A)
xylem
done
clear
B)
phloem
done
clear
C)
cambium
done
clear
D)
cortex
done
clear
View Answer play_arrow
question_answer 125) Histogens capping root apical meristem is
A)
dermatogen
done
clear
B)
calyptrogen
done
clear
C)
periblem
done
clear
D)
plerome
done
clear
View Answer play_arrow
question_answer 126) One of the following statement is false for heart wood.
A)
Made up of living cells
done
clear
B)
Forms central cylinder of wood
done
clear
C)
Solid and hard
done
clear
D)
Contains gums and resins
done
clear
View Answer play_arrow
question_answer 127) Hassal corpuscles are characteristic of
A)
thymus
done
clear
B)
adrenal
done
clear
C)
pineal
done
clear
D)
pancreas
done
clear
View Answer play_arrow
question_answer 128) Osteoblasts are found in
A)
blood
done
clear
B)
muscle
done
clear
C)
bone
done
clear
D)
cartilage
done
clear
View Answer play_arrow
question_answer 129) Translocation of sugars in flowering plants occur in the form of
A)
glucose
done
clear
B)
starch
done
clear
C)
sucrose
done
clear
D)
maltose
done
clear
View Answer play_arrow
question_answer 130) ?Die back? of citrus and ?reclamation? of legumes and cereals is due to deficiency of
A)
copper
done
clear
B)
zinc
done
clear
C)
sodium
done
clear
D)
molybdenum
done
clear
View Answer play_arrow
question_answer 131) Which fraction of the visible spectrum of solar radiations are primarily absorbed by carotenoids of the higher plants?
A)
Blue and green
done
clear
B)
Green and red
done
clear
C)
Red and violet
done
clear
D)
Violet and blue
done
clear
View Answer play_arrow
question_answer 132) Vitamin B! is present in one of these molecules
A)
TPP
done
clear
B)
FMN
done
clear
C)
NAD
done
clear
D)
ATP
done
clear
View Answer play_arrow
question_answer 133) Pacinian corpuscles occur in the skin of certain parts of body in mammals. These are
A)
type of glands
done
clear
B)
pain receptors
done
clear
C)
naked tactile receptors
done
clear
D)
encapsulated pressure receptors
done
clear
View Answer play_arrow
question_answer 134) Which of the following is an essential fatty acid in mammals?
A)
Stearic acid
done
clear
B)
Acetic acid
done
clear
C)
Palmitic acid
done
clear
D)
Gamma linolenic acid
done
clear
View Answer play_arrow
question_answer 135) If the skin of earthworm becomes dry, it dies due to
A)
asphyxia
done
clear
B)
starvation
done
clear
C)
intoxication
done
clear
D)
dehydration
done
clear
View Answer play_arrow
question_answer 136) Antigen antibody reactions are suppressed
A)
in liver dysfunction
done
clear
B)
in chronic inflammatory reactions
done
clear
C)
by\[\text{ACTH}\]
done
clear
D)
by\[\text{vitamin}-\text{K}\]
done
clear
View Answer play_arrow
question_answer 137) How many saddle joint can be traced in human body?
A)
2
done
clear
B)
4
done
clear
C)
8
done
clear
D)
10
done
clear
View Answer play_arrow
question_answer 138) Respiration is controlled by
A)
cerebellum
done
clear
B)
medulla oblongata
done
clear
C)
olfactory lobes
done
clear
D)
hypothalamus
done
clear
View Answer play_arrow
question_answer 139) In the following abnormalities of the eye, which one is a serious condition that leads to blindness?
A)
Presbyopia
done
clear
B)
Myopia
done
clear
C)
Hypermetropia
done
clear
D)
Glaucoma
done
clear
View Answer play_arrow
question_answer 140) The pigment responsible for flower induction is
A)
carotene
done
clear
B)
riboflavin
done
clear
C)
phytochrome
done
clear
D)
xanthophyll
done
clear
View Answer play_arrow
question_answer 141) Sage flower is pollinated by
A)
aphids
done
clear
B)
butterflies
done
clear
C)
bees
done
clear
D)
moths
done
clear
View Answer play_arrow
question_answer 142) Potato sprouting during storage can be prevented by
A)
gibberellins
done
clear
B)
auxins
done
clear
C)
maleichydrazide
done
clear
D)
cytokinin
done
clear
View Answer play_arrow
question_answer 143) The female hormone inhibin is secreted by
A)
zonapellucida
done
clear
B)
ovary
done
clear
C)
corpusluteum
done
clear
D)
uterine epithelium
done
clear
View Answer play_arrow
question_answer 144) Cleidoic eggs are found in
A)
fishes
done
clear
B)
amphibians
done
clear
C)
few reptiles
done
clear
D)
None of these
done
clear
View Answer play_arrow
question_answer 145) Even after disruption of all the hydrogens bonds, which structural level of a protein molecule still remains intact
A)
tertiary structure
done
clear
B)
primary structure
done
clear
C)
secondary structure
done
clear
D)
quaternary structure
done
clear
View Answer play_arrow
question_answer 146) The permanent decrease in population number occurs due to
A)
mortality
done
clear
B)
natality and migration
done
clear
C)
territoriality
done
clear
D)
emigration
done
clear
View Answer play_arrow
question_answer 147) Serule is
A)
hydrosere
done
clear
B)
biosere
done
clear
C)
microbial succession
done
clear
D)
lithosphere
done
clear
View Answer play_arrow
question_answer 148) Mycobiont and phycobiont association is
A)
symbiotic
done
clear
B)
commensal
done
clear
C)
parasitic
done
clear
D)
helotism
done
clear
View Answer play_arrow
question_answer 149) The conservation of natural resources within natural ecosystem is
A)
exsitu conservation
done
clear
B)
in situ conservation
done
clear
C)
Both (a) and (b)
done
clear
D)
None of the above
done
clear
View Answer play_arrow
question_answer 150) NMR is free from hazards as it does not use
A)
magnetic radiation
done
clear
B)
ionizing radiations
done
clear
C)
Both (a) and (b)
done
clear
D)
None of the above
done
clear
View Answer play_arrow
question_answer 151) Formalized system of trading agreements 10. with groups of countries is known as
A)
trading blocks
done
clear
B)
trade ventures
done
clear
C)
trade partners
done
clear
D)
trade organizations
done
clear
View Answer play_arrow
question_answer 152) Mahatma Gandhi was profoundly influenced by the writings of
A)
Bernard Shaw
done
clear
B)
Karl Marx
done
clear
C)
Lenin
done
clear
D)
Leo Tolstoy
done
clear
View Answer play_arrow
question_answer 153) Which country is the largest producer of Timber?
A)
United States
done
clear
B)
Brazil
done
clear
C)
Nigeria
done
clear
D)
Sweden
done
clear
View Answer play_arrow
question_answer 154) The Prime Minister of India is
A)
elected
done
clear
B)
appointed
done
clear
C)
nominated
done
clear
D)
selected
done
clear
View Answer play_arrow
question_answer 155) Which is not an All India Service?
A)
Indian Administrative Service
done
clear
B)
Indian Police Service
done
clear
C)
Indian Foreign Service
done
clear
D)
Indian Forest Service
done
clear
View Answer play_arrow
question_answer 156) Who is rightly called the ?Father of Local 15. Self Government in India?
A)
Lord Mayo
done
clear
B)
Lord Ripon
done
clear
C)
Lord Curzon
done
clear
D)
Lord Clive
done
clear
View Answer play_arrow
question_answer 157) Which Governor-General is associated with Doctrine of Lapse?
A)
Lord Ripon
done
clear
B)
Lord Dalhousie
done
clear
C)
Lord Bentinck
done
clear
D)
Lord Curzon
done
clear
View Answer play_arrow
question_answer 158) India attained ?Dominion Status? on
A)
15thJanuary, 1947
done
clear
B)
15thAugust, 1947
done
clear
C)
15thAugust, 1950
done
clear
D)
15thOctober, 1947
done
clear
View Answer play_arrow
question_answer 159) Which of the following causes Malaria?
A)
Insect
done
clear
B)
Bacteria
done
clear
C)
Protozoa
done
clear
D)
Virus
done
clear
View Answer play_arrow
question_answer 160) The largest tribal community in India is
A)
Bhils
done
clear
B)
Gonds
done
clear
C)
Santhals
done
clear
D)
Tharus
done
clear
View Answer play_arrow
question_answer 161) Teen Bigha Corridor links
A)
India and Pakistan
done
clear
B)
India and China
done
clear
C)
Bangladesh and Pakistan
done
clear
D)
Bangladesh and India
done
clear
View Answer play_arrow
question_answer 162) Who was the only Indian to be elected as President of the United Nations General Assembly?
A)
Vijayalakshmi Pandit
done
clear
B)
VK KrishanMenon
done
clear
C)
Jawaharlal Nehru
done
clear
D)
Rajeshwar Dayal
done
clear
View Answer play_arrow
question_answer 163) Which Indian State has the largest coastline?
A)
Andhra Pradesh
done
clear
B)
Maharashtra
done
clear
C)
Odisha
done
clear
D)
Tamil Nadu
done
clear
View Answer play_arrow
question_answer 164) RAF stands for
A)
Ready Action Force
done
clear
B)
Rapid Action Force
done
clear
C)
Reverse Action Force
done
clear
D)
Repeat Action Force
done
clear
View Answer play_arrow
question_answer 165) Bolometer is used to measure
A)
frequency
done
clear
B)
temperature
done
clear
C)
velocity
done
clear
D)
wavelength
done
clear
View Answer play_arrow
question_answer 166) Which of the following organizations prepares topographical maps of India?
A)
Geographical Survey of India
done
clear
B)
Survey of India
done
clear
C)
Geological Survey of India
done
clear
D)
Archaeological Survey of India
done
clear
View Answer play_arrow
question_answer 167) The most important ore of lead is
A)
Galena
done
clear
B)
Magnetite
done
clear
C)
Pyrolusite
done
clear
D)
Siderite
done
clear
View Answer play_arrow
question_answer 168) Who among the following had got the Bharat Ratna Award before becoming the President of India?
A)
Dr Zakir Hussian
done
clear
B)
Dr Rajendra Prasad
done
clear
C)
Dr S Radhakrishnan
done
clear
D)
VV Giri
done
clear
View Answer play_arrow
question_answer 169) On which of the following rivers is the Tehri Hydro Power Complex located?
A)
Alaknanda
done
clear
B)
Mandakini
done
clear
C)
Dhauli Ganga
done
clear
D)
Bhagirathi
done
clear
View Answer play_arrow
question_answer 170) Which of the following is not a Rabi crop in India?
A)
Wheat
done
clear
B)
Barley
done
clear
C)
Rapeseed
done
clear
D)
Jute
done
clear
View Answer play_arrow
question_answer 171) Who among the following built the famous Khajuraho Temple?
A)
The Chandella Kings
done
clear
B)
The Pala Kings
done
clear
C)
Vikramaditya
done
clear
D)
The Jaina Monks of Central India
done
clear
View Answer play_arrow
question_answer 172) Namdhapa National Park is in
A)
Mizoram
done
clear
B)
Manipur
done
clear
C)
Tripura
done
clear
D)
Arunachal Pradesh
done
clear
View Answer play_arrow
question_answer 173) Which one of the following is also called the power plants of the cell?
A)
Golgi body
done
clear
B)
Mitochondrion
done
clear
C)
Ribosome
done
clear
D)
Lysosome
done
clear
View Answer play_arrow
question_answer 174) In which of the Parliamentary Financial Committees is the RajyaSabha not represented?
A)
Public Accounts Committee
done
clear
B)
Estimates Committee
done
clear
C)
Committee on Public Undertakings
done
clear
D)
Expenditure Committee
done
clear
View Answer play_arrow
question_answer 175) National Income is the
A)
Net National Product at market price
done
clear
B)
Net National Product at factor cost
done
clear
C)
Net Domestic Product at market price
done
clear
D)
Net Domestic Product at factor cost
done
clear
View Answer play_arrow
question_answer 176) ?C? language is a
A)
low level language
done
clear
B)
high level language
done
clear
C)
machine level language
done
clear
D)
assembly level language
done
clear
View Answer play_arrow
question_answer 177) The density of population in an area is measured by the number of
A)
persons
done
clear
B)
children
done
clear
C)
families
done
clear
D)
houses
done
clear
View Answer play_arrow
question_answer 178) Which of the following schools is associated with Jawaharlal Nehru?
A)
Navyug School
done
clear
B)
Navodaya School
done
clear
C)
Sarvodaya School
done
clear
D)
None of these
done
clear
View Answer play_arrow
question_answer 179) Which nation is Mongia Port situated in?
A)
Sri Lanka
done
clear
B)
Bangladesh
done
clear
C)
Bhutan
done
clear
D)
India
done
clear
View Answer play_arrow
question_answer 180) ?Green House Effect? means
A)
pollution in houses in tropical regions
done
clear
B)
trapping of solar energy due to atmospheric oxygen
done
clear
C)
trapping of solar energy due to atmospheric carbon dioxide
done
clear
D)
cultivation in green houses so as to check pollution
done
clear
View Answer play_arrow
question_answer 181) 12 kg of rice costing ` 30 per kg is mixed with 8 kg of rice costing ` 40 per kg. The average per kg price of mixed rice is
A)
` 38
done
clear
B)
` 37
done
clear
C)
` 35
done
clear
D)
` 34
done
clear
View Answer play_arrow
question_answer 182) A train, 110 m long, is running at as peed of60 km/h. How many seconds does it take to cross another train, 170 m long, standing on parallel track?
A)
15.6
done
clear
B)
16.8
done
clear
C)
17.2
done
clear
D)
18
done
clear
View Answer play_arrow
question_answer 183) Three persons walk from place A to place B. Their speeds are in the ratio 4:3:5. The ratio of the times taken by them to reach B will be
A)
10 : 15 : 13
done
clear
B)
2 : 3 : 4
done
clear
C)
15 : 20 : 12
done
clear
D)
16 : 18 :15
done
clear
View Answer play_arrow
question_answer 184) \[{{\left( \frac{1}{2} \right)}^{-\frac{1}{2}}}\]is equal to
A)
\[\frac{1}{\sqrt{2}}\]
done
clear
B)
\[2\sqrt{2}\]
done
clear
C)
\[-\sqrt{2}\]
done
clear
D)
\[\sqrt{2}\]
done
clear
View Answer play_arrow
question_answer 185) How many\[\frac{1}{6}\], s together make \[41\frac{2}{3}\]?
A)
125
done
clear
B)
150
done
clear
C)
250
done
clear
D)
350
done
clear
View Answer play_arrow
question_answer 186) Who is fourth to the right of R?
A)
Q
done
clear
B)
P
done
clear
C)
A
done
clear
D)
B
done
clear
View Answer play_arrow
question_answer 187) Who is second to the left of D?
A)
B
done
clear
B)
W
done
clear
C)
Q
done
clear
D)
None of these
done
clear
View Answer play_arrow
question_answer 188) Which of the following pairs represents the first and second to the right of W?
A)
QB
done
clear
B)
DM
done
clear
C)
MR
done
clear
D)
None of these
done
clear
View Answer play_arrow
question_answer 189) Who is third to the left of P?
A)
M
done
clear
B)
R
done
clear
C)
D
done
clear
D)
None of these
done
clear
View Answer play_arrow
question_answer 190) In which of the following pairs is the second person sitting to the immediate right of the first person?
A)
RM
done
clear
B)
QB
done
clear
C)
MD
done
clear
D)
AB
done
clear
View Answer play_arrow
question_answer 191) In the phrase ?the birth of man in the consciousness of men?, man stands for
A)
noble human qualities
done
clear
B)
power and arrogance
done
clear
C)
an idealistic notion of the human self
done
clear
D)
egocentricity
done
clear
View Answer play_arrow
question_answer 192) People jeer at the ?birth of man? in the human consciousness when they
A)
restructure the social system
done
clear
B)
become power hungry
done
clear
C)
begin to think of themselves as God
done
clear
D)
becomes mentally deranged
done
clear
View Answer play_arrow
question_answer 193) In this passage, the phrase ?God in man? implies
A)
God having assumed the shape of man
done
clear
B)
neither fully godly nor fully human
done
clear
C)
man being transformed into God
done
clear
D)
the divine qualities in man
done
clear
View Answer play_arrow
question_answer 194) According to the author, ?salvation? of human beings lies in the
A)
orgy of national pride
done
clear
B)
extended trade relations
done
clear
C)
spiritual transformation of life
done
clear
D)
whole-hearted participation in political orgainzations
done
clear
View Answer play_arrow
question_answer 195) The author uses the expression ?ugly deformities? to show his indignation at
A)
the liberation of human consciousness
done
clear
B)
selfishness and materialism of the people
done
clear
C)
the drunken orgies of power
done
clear
D)
political organizations
done
clear
View Answer play_arrow
question_answer 196) In each of the following questions, choose the alternative which is opposite in meaning of the word given in capital letters. CELIBATE
A)
Profligate
done
clear
B)
Reprobate
done
clear
C)
Extravagant
done
clear
D)
Prodigal
done
clear
View Answer play_arrow
question_answer 197) In each of the following questions, choose the alternative which is opposite in meaning of the word given in capital letters. DITHER
A)
Cry
done
clear
B)
Refer
done
clear
C)
Decide
done
clear
D)
Defer
done
clear
View Answer play_arrow
question_answer 198) In each of the following questions, choose the alternative which is opposite in meaning of the word given in capital letters. CONSOLIDATE
A)
Isolate
done
clear
B)
Weaken
done
clear
C)
Divide
done
clear
D)
Identify
done
clear
View Answer play_arrow
question_answer 199) In each of the following questions, choose the alternative which is opposite in meaning of the word given in capital letters. INSOLENT
A)
Agreeable
done
clear
B)
Coward
done
clear
C)
Polite
done
clear
D)
Considerate
done
clear
View Answer play_arrow
question_answer 200) In each of the following questions, choose the alternative which is opposite in meaning of the word given in capital letters. LIABILITY
A)
Assumption
done
clear
B)
Exemption
done
clear
C)
Consumption
done
clear
D)
Presumption
done
clear
View Answer play_arrow