question_answer 1) Simple pendulum is executing simple harmonic motion with time period T. If the length of the pendulum is increased by 21%, then the increase in the time period of the pendulum of the increased length is:
A)
22%
done
clear
B)
13%
done
clear
C)
50%
done
clear
D)
10%
done
clear
View Answer play_arrow
question_answer 2) The function of heavy water in a nuclear reactor is to:
A)
slow down the neutrons
done
clear
B)
increase the neutrons
done
clear
C)
stop the electrons
done
clear
D)
none of the above
done
clear
View Answer play_arrow
question_answer 3) Which one of the following has the highest neutrons ratio?
A)
\[_{92}{{U}^{235}}\]
done
clear
B)
\[_{8}{{O}^{16}}\]
done
clear
C)
\[_{2}H{{e}^{4}}\]
done
clear
D)
\[_{26}F{{e}^{56}}\]
done
clear
View Answer play_arrow
question_answer 4) A concave mirror having the focal length 15 cm, forms an image having twice of the linear dimensions of the object. If the image is virtual, then the position of the object will be:
A)
7.5cm
done
clear
B)
22.5cm
done
clear
C)
40 cm
done
clear
D)
30 cm
done
clear
View Answer play_arrow
question_answer 5) The temperature of the cold junction of a thermocouple is \[0{}^\circ C\] and the temperature of the hot junction is \[T{}^\circ C\]. The relation for the thermo emf is given by, \[E=AT-\frac{1}{2}B{{T}^{2}}\] (when A = 16 and B - 0.08). The temperature of inversion will be:
A)
\[500{}^\circ C\]
done
clear
B)
\[460{}^\circ C\]
done
clear
C)
\[600{}^\circ C\]
done
clear
D)
\[400{}^\circ C\]
done
clear
View Answer play_arrow
question_answer 6) Two material having the dielectric constants \[{{k}_{1}}\] and \[{{k}_{2}}\] are filled between two parallel plates of a capacitor. Where area of each plate is A and the distance between the plates is d. The capacity of the capacitor is:
A)
\[\frac{A{{\varepsilon }_{0}}({{k}_{1}}\times {{k}_{2}})}{d({{k}_{1}}+{{k}_{2}})}\]
done
clear
B)
\[\frac{A{{\varepsilon }_{0}}({{k}_{1}}-{{k}_{2}})}{d}\]
done
clear
C)
\[\frac{A{{\varepsilon }_{0}}{{k}_{1}}{{k}_{2}}}{({{k}_{1}}+{{k}_{2}})}\]
done
clear
D)
\[\frac{A{{\varepsilon }_{0}}({{k}_{1}}+{{k}_{2}})}{d}\]
done
clear
View Answer play_arrow
question_answer 7) A closely wound flat circular coil of 25 rums of wire has diameter of 10 cm which carries current of 4 A, the flux density at the centre of a coil will be:
A)
\[1.256\times {{10}^{-3}}T\]
done
clear
B)
\[1.679\times {{10}^{-5}}T\]
done
clear
C)
\[1.512\times {{10}^{-5}}T\]
done
clear
D)
\[2.28\times {{10}^{-4}}T\]
done
clear
View Answer play_arrow
question_answer 8) Four lenses having the focal length of +15 cm, 20 cm, + 150 cm, and -+- 250 cm respectively are provided to make an astronomical telescope. The focal length of the eyepiece to produce the largest magnification, should be:
A)
+ 250 cm
done
clear
B)
+ 155 cm
done
clear
C)
25 cm
done
clear
D)
+ 15 cm
done
clear
View Answer play_arrow
question_answer 9) When radioactive substance emits an \[\alpha \]-particle, then its position in the periodic table is lowered by :
A)
two places
done
clear
B)
three places
done
clear
C)
five places
done
clear
D)
one place
done
clear
View Answer play_arrow
question_answer 10) The colour of a star indicates its:
A)
velocity
done
clear
B)
temperature
done
clear
C)
size
done
clear
D)
length
done
clear
View Answer play_arrow
question_answer 11) Fraunhofer line of the solar system is an example of:
A)
line absorption spectrum
done
clear
B)
line emission spectrum
done
clear
C)
emission of band spectrum
done
clear
D)
none of the above
done
clear
View Answer play_arrow
question_answer 12) When added an impurity into the silicon which one of the following produces n-type of semiconductors?
A)
Iron
done
clear
B)
Magnesium
done
clear
C)
Aluminium
done
clear
D)
Phosphorus
done
clear
View Answer play_arrow
question_answer 13) What is electric flux associated with one of faces of the cube, when a charge (q) is enclosed in a cube?
A)
\[\frac{6q}{{{\varepsilon }_{0}}}\]
done
clear
B)
\[\frac{q}{6{{\varepsilon }_{0}}}\]
done
clear
C)
\[\frac{q}{3{{\varepsilon }_{0}}}\]
done
clear
D)
\[\frac{3q}{{{\varepsilon }_{0}}}\]
done
clear
View Answer play_arrow
question_answer 14) The point charges Q and - 2Q are placed at some distance apart. If the electric field at the location of Q is E, the electric field at the location of - 2Q will be:
A)
\[-\frac{3E}{2}\]
done
clear
B)
\[-E\]
done
clear
C)
\[-\frac{E}{2}\]
done
clear
D)
\[-2E\]
done
clear
View Answer play_arrow
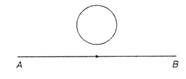
question_answer 15)
The current flows from A to B as shown in figure, then the direction of the induced current in the loop will be:
A)
straight line
done
clear
B)
anti-clockwise
done
clear
C)
clockwise
done
clear
D)
none of the above
done
clear
View Answer play_arrow
question_answer 16) The cell has an emf of 2V and the internal resistance of this cell is 0.1\[\Omega \], it is connected to resistance of 3.9\[\Omega \] the voltage across the cell will be:
A)
1.95V
done
clear
B)
1.5V
done
clear
C)
2V
done
clear
D)
1.8V
done
clear
View Answer play_arrow
question_answer 17) A gun fires a bullet of mass 50 g with a velocity of 30 m/s. Due to this, the gun is pushed back with a velocity of 1 m/s, then the mass of the gun is:
A)
1.5kg
done
clear
B)
5.5kg
done
clear
C)
0.5 kg
done
clear
D)
3.5kg
done
clear
View Answer play_arrow
question_answer 18)
The frequency of oscillator of the springs as shown in figure will be:
A)
\[\frac{1}{2\pi }\sqrt{\frac{({{k}_{1}}+{{k}_{2}})m}{{{k}_{1}}{{k}_{2}}}}\]
done
clear
B)
\[\frac{1}{2\pi }\sqrt{\frac{{{k}_{1}}{{k}_{2}}}{({{k}_{1}}+{{k}_{2}})m}}\]
done
clear
C)
\[\frac{1}{2\pi }\sqrt{\frac{k}{m}}\]
done
clear
D)
\[2\pi \sqrt{\frac{k}{m}}\]
done
clear
View Answer play_arrow
question_answer 19) Which of the following paics does not have similar dimensions?
A)
Tension and surface tension
done
clear
B)
Stress and pressure
done
clear
C)
Plancks constant and angular momentum
done
clear
D)
Angle and strain
done
clear
View Answer play_arrow
question_answer 20) How does the red shift confirm that the universe is expanding?
A)
Wavelength of light emitted by galaxies appears to decrease
done
clear
B)
Wavelength of light emitted by galaxies appears to be the same
done
clear
C)
Wavelength of light emitted by galaxies appears to increase
done
clear
D)
None of the above
done
clear
View Answer play_arrow
question_answer 21) The current gain for a transistor works if common-base amplifier is 0.96. If the emitter current is 7.2 mA, the base current will be;
A)
0.42mA
done
clear
B)
0.49mA
done
clear
C)
0.29 mA
done
clear
D)
0.35 mA
done
clear
View Answer play_arrow
question_answer 22) In an atom bomb, the energy is released because of the :
A)
chain reaction of neutrons and \[_{92}{{U}^{238}}\]
done
clear
B)
chain reaction of neutrons and \[_{92}{{U}^{235}}\]
done
clear
C)
chain reaction of neutrons and \[_{92}{{U}^{236}}\]
done
clear
D)
chain reaction of neutrons and \[_{92}{{U}^{240}}\]
done
clear
View Answer play_arrow
question_answer 23) What change occurs, if the monochromatic light used in Youngs double slit experiment replaced by white light ?
A)
Only the central fringe is white and all other fringes are observed coloured
done
clear
B)
No fringes are observed
done
clear
C)
All the bright fringes become white
done
clear
D)
All the bright fringes are coloured between violet and red
done
clear
View Answer play_arrow
question_answer 24) In a circuit the coil of a choke:
A)
decreases the current
done
clear
B)
increases the current
done
clear
C)
has high resistance to DC circuit
done
clear
D)
no effect with the current
done
clear
View Answer play_arrow
question_answer 25) The internal resistance of a cell is the resistance of;
A)
electrolyte used in the cell
done
clear
B)
electrodes of the cell
done
clear
C)
vessel of the cell
done
clear
D)
none of the above
done
clear
View Answer play_arrow
question_answer 26) The escape velocity from the earth is 11.2 km/s. The escape velocity from a planet having twice the radius and the same mean density as the earth, is:
A)
11.2 km/s
done
clear
B)
22.4 km/s
done
clear
C)
15.00 km/s
done
clear
D)
5.8 km/s
done
clear
View Answer play_arrow
question_answer 27) A metal ball of mass 2 kg moving with speed of 36 km/h has a collision with a stationary ball of mass 3 kg. If after collision, both the bait move together, the loss in kinetic energy due to collision is :
A)
80 J
done
clear
B)
40 J
done
clear
C)
60 J
done
clear
D)
160 J
done
clear
View Answer play_arrow
question_answer 28) A constant torque of 31.4 N-m is exerted on a pivoted wheel. If the angular acceleration of the wheel is \[4\pi \,\,rad/{{s}^{2}},\] then the moment of inertia, will be:
A)
\[5.8\,kg-{{m}^{2}}\]
done
clear
B)
\[4.5\,kg-{{m}^{2}}\]
done
clear
C)
\[5.6\,kg-{{m}^{2}}\]
done
clear
D)
\[2.5\,kg-{{m}^{2}}\]
done
clear
View Answer play_arrow
question_answer 29) When the two surface are coated with the lubricant, then they will :
A)
slide upon each other
done
clear
B)
stick to each other
done
clear
C)
roll upon each other
done
clear
D)
none of the above
done
clear
View Answer play_arrow
question_answer 30) In a circuit, the current lags behind the voltage by a phase difference of \[\pi /2,\] the circuit will contain which of the following:
A)
only R
done
clear
B)
only C
done
clear
C)
R and C
done
clear
D)
only L
done
clear
View Answer play_arrow
question_answer 31) The length and breadth of a metal sheet are 3.124 m and 3.002 m respectively, the area of this sheet up to four correct significant figure is:
A)
\[9.378\,{{m}^{2}}\]
done
clear
B)
\[9.37\,{{m}^{2}}\]
done
clear
C)
\[9.378248\,{{m}^{2}}\]
done
clear
D)
\[9.3782\,{{m}^{2}}\]
done
clear
View Answer play_arrow
question_answer 32) Two equal vectors have a resultant equal to either of them, then the angle between them will be:
A)
\[110{}^\circ C\]
done
clear
B)
\[120{}^\circ C\]
done
clear
C)
\[60{}^\circ C\]
done
clear
D)
\[150{}^\circ C\]
done
clear
View Answer play_arrow
question_answer 33) A stone tied to the end of string of 80 cm long, is whirled in a horizontal circle with a constant speed. If the stone makes 25 revolutions in 14 s. Then, magnitude of acceleration of the same will be:
A)
\[990\,cm/{{s}^{2}}\]
done
clear
B)
\[680\,cm/{{s}^{2}}\]
done
clear
C)
\[750\,cm/{{s}^{2}}\]
done
clear
D)
\[650\,cm/{{s}^{2}}\]
done
clear
View Answer play_arrow
question_answer 34) The tension in a piano wire is 10 N. The tension in a piano wire to produce a node of double frequency is :
A)
20 N
done
clear
B)
40 N
done
clear
C)
10N
done
clear
D)
120 N
done
clear
View Answer play_arrow
question_answer 35) Two sound waves have phase difference of 60°, then they will have the path difference of:
A)
\[3\lambda \]
done
clear
B)
\[\frac{\lambda }{3}\]
done
clear
C)
\[\frac{\lambda }{6}\]
done
clear
D)
\[\lambda \]
done
clear
View Answer play_arrow
question_answer 36) A sings with a frequency n and B sings with a frequency 1/8 that of A. If the energy remains the same and the amplitude of A is a, then amplitude of B will be :
A)
\[2a\]
done
clear
B)
\[8a\]
done
clear
C)
\[4a\]
done
clear
D)
\[a\]
done
clear
View Answer play_arrow
question_answer 37) The velocity of a bullet is reduced from 200 m/s to 100 m/s, while travelling through a wooden block of thickness 10 cm. Assuming it to be uniform, the retardation will be:
A)
\[15\times {{10}^{4}}m/{{s}^{2}}\]
done
clear
B)
\[10\times {{10}^{4}}m/{{s}^{2}}\]
done
clear
C)
\[12\times {{10}^{4}}m/{{s}^{2}}\]
done
clear
D)
\[14.5m/{{s}^{2}}\]
done
clear
View Answer play_arrow
question_answer 38) Two projectiles are projected with the same velocity. If one is projected at an angle of 30° and the other at 60° to the horizontal. The ratio of maximum heights reached, is:
A)
1 : 3
done
clear
B)
2 : 1
done
clear
C)
3 : 1
done
clear
D)
1 : 4
done
clear
View Answer play_arrow
question_answer 39) A force \[(3\mathbf{\hat{i}}+4\mathbf{\hat{j}})\] N acts on a body and displaced it by\[(3\mathbf{\hat{i}}+4\mathbf{\hat{j}})\] m. The work done by the force is :
A)
5 J
done
clear
B)
25 J
done
clear
C)
10 J
done
clear
D)
30 J
done
clear
View Answer play_arrow
question_answer 40) In an adiabatic change, the pressure and temperature of a monoatomic gas are related with relation as\[P\alpha {{T}^{C}}\], where C is equal to:
A)
\[\frac{5}{4}\]
done
clear
B)
\[\frac{5}{3}\]
done
clear
C)
\[\frac{5}{2}\]
done
clear
D)
\[\frac{3}{5}\]
done
clear
View Answer play_arrow
question_answer 41) If in a wire of Youngs modulus Y, longitudinal strain X is produced then the potential energy stored in its unit volume will be:
A)
\[0.5y{{x}^{2}}\]
done
clear
B)
\[0.5{{y}^{2}}x\]
done
clear
C)
\[2y{{x}^{2}}\]
done
clear
D)
\[y{{x}^{2}}\]
done
clear
View Answer play_arrow
question_answer 42) Which one of the following is not a thermodynamical co-ordinate?
A)
V
done
clear
B)
R
done
clear
C)
T
done
clear
D)
P
done
clear
View Answer play_arrow
question_answer 43) According to Hookes law of elasticity,, if stress is increased, then the ratio of stress 10 strain :
A)
becomes zero
done
clear
B)
remains constant
done
clear
C)
decreases
done
clear
D)
increases
done
clear
View Answer play_arrow
question_answer 44) A stone tied to a string is rotated with a uniform speed in a vertical plane. If mass of the stone is m, the length of the string is r and the linear speed of the stone is v, when the stone is at its lowest point, then the tension in the string will be : (g = acceleration due to gravity)
A)
\[\frac{m{{v}^{2}}}{r}+mg\]
done
clear
B)
\[\frac{m{{v}^{2}}}{r}-mg\]
done
clear
C)
\[\frac{mv}{r}\]
done
clear
D)
mg
done
clear
View Answer play_arrow
question_answer 45) A black body is heated from 27°C to \[{{127}^{o}}C\]. The ratio of their energies of radiation emitted will be :
A)
9 : 16
done
clear
B)
27 : 64
done
clear
C)
81 : 256
done
clear
D)
3 : 4
done
clear
View Answer play_arrow
question_answer 46) cyclotron is used accelerate:
A)
positive ion
done
clear
B)
negative ion
done
clear
C)
electron
done
clear
D)
none of these
done
clear
View Answer play_arrow
question_answer 47) A spherical drop of water has 1 mm radius. If the surface tension of water is \[70\times {{10}^{-3}}\]N/m, then the difference of pressures between inside and outside of the spherical drop is:
A)
\[140\,N/{{m}^{2}}\]
done
clear
B)
\[14\,N/{{m}^{2}}\]
done
clear
C)
\[35\,N/{{m}^{2}}\]
done
clear
D)
none of these
done
clear
View Answer play_arrow
question_answer 48) A body A starts from rest with an acceleration\[{{a}_{1}}\]. After 2 s another body B starts from rest with an acceleration\[{{a}_{2}}\]. If they travel equal distances in 5 s, after the starts of A, the ratio \[{{a}_{1}}:{{a}_{2}}\] will be equal to:
A)
9 : 5
done
clear
B)
5 : 7
done
clear
C)
5 : 9
done
clear
D)
7: 9
done
clear
View Answer play_arrow
question_answer 49) In a sinusoidal wave the time required for a particular point to move from equilibrium pasition to maximum displacement is 0.17 s, then the frequency of wave is:
A)
1.47 Hz
done
clear
B)
0.36 Hz
done
clear
C)
2.94 Hz
done
clear
D)
2.48 Hz
done
clear
View Answer play_arrow
question_answer 50) Bernoullis principle is based on the law of conservation of:
A)
mass
done
clear
B)
energy
done
clear
C)
angular momentum
done
clear
D)
linear momentum
done
clear
View Answer play_arrow
question_answer 51) Assertion: A metallic shield in form of a hoi; shell may be built to block an electric field. Reason: In a hollow spherical shield, t: electric Held inside it is zero at every point.
A)
A
done
clear
B)
B
done
clear
C)
C
done
clear
D)
D
done
clear
E)
E
done
clear
View Answer play_arrow
question_answer 52) Assertion: A rocket moves forward by push" the surrounding air backwards. Reason: It derives the necessary thrust to move forward according to Newtons third Lav. motion.
A)
A
done
clear
B)
B
done
clear
C)
C
done
clear
D)
D
done
clear
E)
E
done
clear
View Answer play_arrow
question_answer 53) Assertion: In a movie, ordinarily 24 frames are projected per second from one end lo die other of the complete film. Reason: The image formed on retina of eye is sustained up to 1/10 s after the removal of stimulus.
A)
A
done
clear
B)
B
done
clear
C)
C
done
clear
D)
D
done
clear
E)
E
done
clear
View Answer play_arrow
question_answer 54) Assertion: In adiabatic compression, the internal energy and temperature of the system pet decreased. Reason: The adiahatic compression is a slow process.
A)
A
done
clear
B)
B
done
clear
C)
C
done
clear
D)
D
done
clear
E)
E
done
clear
View Answer play_arrow
question_answer 55) Assertion: In Youngs experiment, the fringe width tor dark fringes is different from that for white fringes. Reason: In Youngs double slit experiment when the fringes are performed with a source of white light, then only black and bright fringe are observed.
A)
A
done
clear
B)
B
done
clear
C)
C
done
clear
D)
D
done
clear
E)
E
done
clear
View Answer play_arrow
question_answer 56) Assertion: The isothermal curves intersect each other at a certain point. Reason: The isothermal change takes place slowly so, the isothermal curves have very little slope.
A)
A
done
clear
B)
B
done
clear
C)
C
done
clear
D)
D
done
clear
E)
E
done
clear
View Answer play_arrow
question_answer 57) Assertion: Blue colour of sky appears due to scattering of blue colour. Reason: Blue colour has shortest wave length in visible spectrum.
A)
A
done
clear
B)
B
done
clear
C)
C
done
clear
D)
D
done
clear
E)
E
done
clear
View Answer play_arrow
question_answer 58) Assertion: X-rays travel with the speed of light. Reason: X-rays are electromagnetic rays.
A)
A
done
clear
B)
B
done
clear
C)
C
done
clear
D)
D
done
clear
E)
E
done
clear
View Answer play_arrow
question_answer 59) Assertion: When the speed of an electron increases its specific charge decreases. Reason: Specific charge is ratio of the charge to mass.
A)
A
done
clear
B)
B
done
clear
C)
C
done
clear
D)
D
done
clear
E)
E
done
clear
View Answer play_arrow
question_answer 60) Assertion :\[_{Z}{{X}^{A}}\] undergoes 2\[\alpha \]-decays, 2\[\beta \]-decays and 2\[\gamma \]-decays and the daughter product is \[_{Z-2}{{Y}^{A-B}}\] Reason: In a-decay the mass number decreases by 4 and atomic number decreases by 2. In \[\beta \]-decay the mass number remains unchanged, but atomic number increases by 1 only.
A)
A
done
clear
B)
B
done
clear
C)
C
done
clear
D)
D
done
clear
E)
E
done
clear
View Answer play_arrow
question_answer 61) Water glass is:
A)
\[N{{a}_{2}}Si{{O}_{3}}\]
done
clear
B)
\[M{{g}_{2}}Si\]
done
clear
C)
\[SiC{{l}_{4}}\]
done
clear
D)
\[Ca{{({{H}_{2}}P{{O}_{4}})}_{2}}\]
done
clear
View Answer play_arrow
question_answer 62) In diamond crystal each carbon atom is linked with carbon atoms. The number of carbon atoms linked is:
A)
2
done
clear
B)
4
done
clear
C)
3
done
clear
D)
1
done
clear
View Answer play_arrow
question_answer 63) With ammoniacal cuprous chloride solution a reddish brown precipitate is obtained on treating with:
A)
\[C{{H}_{4}}\]
done
clear
B)
\[{{C}_{2}}{{H}_{4}}\]
done
clear
C)
\[{{C}_{2}}{{H}_{2}}\]
done
clear
D)
\[{{C}_{3}}{{H}_{6}}\]
done
clear
View Answer play_arrow
question_answer 64) The boiling points of four saturated hydro- carbons are given below. Which boiling point suggests maximum number of carbon atoms in its molecule?
A)
\[-{{162}^{o}}C\]
done
clear
B)
\[-{{88.6}^{o}}C\]
done
clear
C)
\[-{{0.5}^{o}}C\]
done
clear
D)
\[-{{42.2}^{o}}C\]
done
clear
View Answer play_arrow
question_answer 65) The molarity of \[{{H}_{2}}S{{O}_{4}}\] solution, which has a density 1.84 g/cc at \[{{35}^{o}}C\] and contains 98% by weight is:
A)
1.84M
done
clear
B)
18.4 M
done
clear
C)
20.6 M
done
clear
D)
24.5 M
done
clear
View Answer play_arrow
question_answer 66) The composition of duralumin is:
A)
\[Al\,\,94%,\,\,Mg\,6%\]
done
clear
B)
\[Cu56%,Zn24%,Ni\text{ }20%\]
done
clear
C)
\[Cu\,\,95%,Al\,\,5%\]
done
clear
D)
\[Al\text{ }95%,\text{ }Cu\text{ }4%,\text{ }Mn\text{ }0.5%,\text{ }Mg\text{ }0.5%\]
done
clear
View Answer play_arrow
question_answer 67) \[{{10}^{21}}\] molecules are removed from 200 mg of \[C{{O}_{2}}\]. The moles of \[C{{O}_{2}}\] left are:
A)
\[2.88\times {{10}^{-3}}\]
done
clear
B)
\[28.8\times {{10}^{-3}}\]
done
clear
C)
\[288\times {{10}^{-3}}\]
done
clear
D)
\[28.8\times {{10}^{3}}\]
done
clear
View Answer play_arrow
question_answer 68) Which one of the following is not a Green house gase?
A)
\[C{{O}_{2}}\]
done
clear
B)
\[{{H}_{2}}O\]
done
clear
C)
\[{{N}_{3}}\]
done
clear
D)
\[{{O}_{3}}\]
done
clear
View Answer play_arrow
question_answer 69) The velocity of electron in second shell of hydrogen atom is:
A)
\[10.94\times {{10}^{6}}m{{s}^{-1}}\]
done
clear
B)
\[18.88\times {{10}^{6}}m{{s}^{-1}}\]
done
clear
C)
\[1.888\times {{10}^{6}}m{{s}^{-1}}\]
done
clear
D)
\[1.094\times {{10}^{6}}m{{s}^{-1}}\]
done
clear
View Answer play_arrow
question_answer 70) The weight of \[NaCl\] decomposed by 4.9 g of \[{{H}_{2}}S{{O}_{4}}\], if 6 g of sodium hydrogen sulphate and 1.825 of \[HCl\] were produced in the reaction:
A)
6.921 g
done
clear
B)
4.65 g
done
clear
C)
2.925 g
done
clear
D)
1.4 g
done
clear
View Answer play_arrow
question_answer 71) The oxidation number of \[Cr\] in \[{{K}_{2}}C{{r}_{2}}{{O}_{7}}\] is:
A)
\[+2\]
done
clear
B)
\[+4\]
done
clear
C)
\[+6\]
done
clear
D)
\[+7\]
done
clear
View Answer play_arrow
question_answer 72) The electrical conductivity of semi-conductors:
A)
decrease with temperature
done
clear
B)
increase with temperature
done
clear
C)
remain constant on heating
done
clear
D)
none of the above
done
clear
View Answer play_arrow
question_answer 73) 20 g of a substance were dissolved in 500 mL of water and the osmotic pressure I of the solution was found to be 600 mm of mercury at \[{{15}^{o}}C\]. The molecular weight of substance is:
A)
998
done
clear
B)
1028
done
clear
C)
1098
done
clear
D)
1198
done
clear
View Answer play_arrow
question_answer 74) The decreasing order of stability of alkyl carbonium ion is in the order of:
A)
\[R-\underset{R}{\overset{R}{\mathop{\underset{|}{\overset{|}{\mathop{{{C}^{+}}}}}\,}}}\,>R-\underset{H}{\overset{R}{\mathop{\underset{|}{\overset{|}{\mathop{{{C}^{+}}}}}\,}}}\,>R-\underset{H}{\overset{H}{\mathop{\underset{|}{\overset{|}{\mathop{{{C}^{+}}}}}\,}}}\,>H-\underset{H}{\overset{H}{\mathop{\underset{|}{\overset{|}{\mathop{{{C}^{+}}}}}\,}}}\,\]
done
clear
B)
\[R-\underset{H}{\overset{H}{\mathop{\underset{|}{\overset{|}{\mathop{{{C}^{+}}}}}\,}}}\,>R-\underset{H}{\overset{H}{\mathop{\underset{|}{\overset{|}{\mathop{{{C}^{+}}}}}\,}}}\,>R-\underset{H}{\overset{R}{\mathop{\underset{|}{\overset{|}{\mathop{{{C}^{+}}}}}\,}}}\,>H-\underset{R}{\overset{R}{\mathop{\underset{|}{\overset{|}{\mathop{{{C}^{+}}}}}\,}}}\,\]
done
clear
C)
\[R-\underset{H}{\overset{R}{\mathop{\underset{|}{\overset{|}{\mathop{{{C}^{+}}}}}\,}}}\,>R-\underset{R}{\overset{R}{\mathop{\underset{|}{\overset{|}{\mathop{{{C}^{+}}}}}\,}}}\,>R-\underset{H}{\overset{H}{\mathop{\underset{|}{\overset{|}{\mathop{{{C}^{+}}}}}\,}}}\,>H-\underset{H}{\overset{n}{\mathop{\underset{|}{\overset{|}{\mathop{{{C}^{+}}}}}\,}}}\,\]
done
clear
D)
\[R-\underset{H}{\overset{H}{\mathop{\underset{|}{\overset{|}{\mathop{{{C}^{+}}}}}\,}}}\,>R-\underset{H}{\overset{R}{\mathop{\underset{|}{\overset{|}{\mathop{{{C}^{+}}}}}\,}}}\,>R-\underset{R}{\overset{R}{\mathop{\underset{|}{\overset{|}{\mathop{{{C}^{+}}}}}\,}}}\,>H-\underset{H}{\overset{n}{\mathop{\underset{|}{\overset{|}{\mathop{{{C}^{+}}}}}\,}}}\,\]
done
clear
View Answer play_arrow
question_answer 75) Zwitter ion contains:
A)
-ve charge
done
clear
B)
+ve charge
done
clear
C)
both +ve and -ve charge
done
clear
D)
none of the above
done
clear
View Answer play_arrow
question_answer 76) Water gas is produced by:
A)
passing steam over red hot coke
done
clear
B)
passing steam and air over red hot coke
done
clear
C)
burning coke in excess of air
done
clear
D)
burning coke in limited supply of air
done
clear
View Answer play_arrow
question_answer 77) Which of the following element is represented by electronic configuration\[1{{s}^{2}}2{{s}^{2}}2p_{x}^{1}2p_{y}^{1}2p_{z}^{1}\]?
A)
Nitrogen
done
clear
B)
Oxygen
done
clear
C)
Fluorine
done
clear
D)
Sulphur
done
clear
View Answer play_arrow
question_answer 78) Which one of the following hydroxide is insoluble in water?
A)
\[Ca{{(OH)}_{2}}\]
done
clear
B)
\[Ba{{(OH)}_{2}}\]
done
clear
C)
\[Be{{(OH)}_{2}}\]
done
clear
D)
\[Mg{{(OH)}_{2}}\]
done
clear
View Answer play_arrow
question_answer 79) Reduction of nitrobenzene with \[Sn/HCl\]produce:
A)
azobenzene
done
clear
B)
azoxybenzene
done
clear
C)
nitrobenzene
done
clear
D)
aniline
done
clear
View Answer play_arrow
question_answer 80) The normality of orthophosphoric acid having purity of 70% by weight and specific gravity 1.54 is:
A)
11N
done
clear
B)
22 N
done
clear
C)
33 N
done
clear
D)
44 N
done
clear
View Answer play_arrow
question_answer 81) Hinsbergs reagent is:
A)
\[\underset{COO{{C}_{2}}{{H}_{5}}}{\overset{COO{{C}_{2}}{{H}_{5}}}{\mathop{|}}}\,\]
done
clear
B)
\[{{C}_{6}}{{H}_{5}}S{{O}_{2}}Cl\]
done
clear
C)
\[{{C}_{6}}{{H}_{5}}S{{O}_{2}}N{{H}_{2}}\]
done
clear
D)
\[C{{H}_{3}}COC{{H}_{2}}COO{{C}_{2}}{{H}_{5}}\]
done
clear
View Answer play_arrow
question_answer 82) The pH value of N/10 \[NaOH\] is:
A)
9
done
clear
B)
10
done
clear
C)
12
done
clear
D)
13
done
clear
View Answer play_arrow
question_answer 83) Salol is:
A)
acetyl salicylic acid
done
clear
B)
phenyl salicylate
done
clear
C)
methyl salicylate
done
clear
D)
none of the above
done
clear
View Answer play_arrow
question_answer 84) Aldehyde and ketones can be distinguished by:
A)
ammonia
done
clear
B)
\[{{H}_{2}}S{{O}_{4}}\]
done
clear
C)
alkaline \[KMn{{O}_{4}}\]
done
clear
D)
Fehling solution
done
clear
View Answer play_arrow
question_answer 85) Among the following, insecticide is:
A)
BHC
done
clear
B)
phosphine
done
clear
C)
chloral
done
clear
D)
aspirin
done
clear
View Answer play_arrow
question_answer 86) The strongest oxidising agent is:
A)
\[{{F}_{2}}\]
done
clear
B)
\[C{{l}_{2}}\]
done
clear
C)
\[{{I}_{2}}\]
done
clear
D)
\[B{{r}_{2}}\]
done
clear
View Answer play_arrow
question_answer 87) The colloid is:
A)
urea
done
clear
B)
blood
done
clear
C)
cane sugar
done
clear
D)
\[NaCl\]
done
clear
View Answer play_arrow
question_answer 88) The tribasic acid is:
A)
\[{{H}_{3}}P{{O}_{4}}\]
done
clear
B)
\[{{H}_{3}}P{{O}_{3}}\]
done
clear
C)
\[{{H}_{3}}P{{O}_{2}}\]
done
clear
D)
\[HP{{O}_{3}}\]
done
clear
View Answer play_arrow
question_answer 89) The de-Broglie wavelength associated with a particle of mass 10kg moving with a velocity of \[10m{{s}^{-1}}\] is:
A)
\[6.63\times {{10}^{-7}}\]
done
clear
B)
\[6.63\times {{10}^{-16}}\]
done
clear
C)
\[6.63\times {{10}^{-21}}\]
done
clear
D)
\[6.63\times {{10}^{-29}}\]
done
clear
View Answer play_arrow
question_answer 90) The number of water molecules in Mohrs salt is:
A)
2
done
clear
B)
4
done
clear
C)
6
done
clear
D)
8
done
clear
View Answer play_arrow
question_answer 91) One gram equimolecular mixture of \[N{{a}_{2}}C{{O}_{3}}\] and \[NaHC{{O}_{3}}\] is reacted with \[0.1\text{ }NHCl\]. The millilitres of \[0.1\text{ }NHCl\] required to react completely with the above mixture is:
A)
\[15.78\text{ }mL\]
done
clear
B)
\[157.8\text{ }mL\]
done
clear
C)
\[198.4\text{ }mL\]
done
clear
D)
\[295.5\text{ }mL\]
done
clear
View Answer play_arrow
question_answer 92) The product formed by the reaction of acetamide with bromine in presence of \[NaOH\] is:
A)
\[C{{H}_{3}}CN\]
done
clear
B)
\[C{{H}_{3}}CHO\]
done
clear
C)
\[C{{H}_{3}}C{{H}_{2}}OH\]
done
clear
D)
\[C{{H}_{3}}N{{H}_{2}}\]
done
clear
View Answer play_arrow
question_answer 93) In lake test of \[A{{l}^{3+}}\]ion, there is formation of coloured floating. It is due to:
A)
absorption of litmus by \[{{H}_{2}}O\]
done
clear
B)
adsorption of litmus of \[Al{{(OH)}_{3}}\]
done
clear
C)
adsorption of litmus \[Al(OH)_{4}^{-}\]
done
clear
D)
none of the above
done
clear
View Answer play_arrow
question_answer 94) The colligative property is not represented by:
A)
elevation in boiling point
done
clear
B)
osmotic pressure
done
clear
C)
optical activity
done
clear
D)
relative lowering of vapour pressure
done
clear
View Answer play_arrow
question_answer 95) The internal energy of a substance is:
A)
increase with increase in temperature
done
clear
B)
decrease with increase in temperature
done
clear
C)
remain unaffected with temperature
done
clear
D)
can be calculated by the reaction \[E=m{{c}^{2}}\]
done
clear
View Answer play_arrow
question_answer 96) The movement of colloidal particle under applied electric current is known as:
A)
electro dialysis
done
clear
B)
dialysis
done
clear
C)
electrophoresis
done
clear
D)
none of the above
done
clear
View Answer play_arrow
question_answer 97) Both oxidation and reduction takes place in:
A)
\[NaBr+HCl\xrightarrow{{}}NaCl+HBr\]
done
clear
B)
\[HBr+AgN{{O}_{3}}\xrightarrow{{}}AgBr+HN{{O}_{3}}\]
done
clear
C)
\[{{H}_{2}}+B{{r}_{2}}\xrightarrow{{}}2HBr\]
done
clear
D)
\[CaO+{{H}_{2}}S{{O}_{4}}\xrightarrow{{}}CaS{{O}_{4}}+{{H}_{2}}O\]
done
clear
View Answer play_arrow
question_answer 98) The heat of reaction for: \[{{C}_{10}}{{H}_{g}}(s)+12{{O}_{2}}(g)\xrightarrow{{}}10\,C{{O}_{2}}(g)\] \[+4{{H}_{2}}O(l)\] at constant volume is \[-1228.2\text{ }kcal\] at \[{{25}^{o}}C\]. The heat of reaction at constant pressure and same temperature is:
A)
\[-1228.2\text{ }kcal\]
done
clear
B)
\[-1229.3\text{ }kcal\]
done
clear
C)
\[-1232.9\text{ }kcal\]
done
clear
D)
\[-1242.6\text{ }kcal\]
done
clear
View Answer play_arrow
question_answer 99) A solution having hydrogen ion concentration is \[0.0005\text{ }g-equi/L,\]its \[pOH\] is:
A)
\[8.2798\]
done
clear
B)
\[10.6990\]
done
clear
C)
\[12.7854\]
done
clear
D)
\[13.3344\]
done
clear
View Answer play_arrow
question_answer 100) Which of the following is unaffected by temperature?
A)
Normality
done
clear
B)
Molarity
done
clear
C)
Molality
done
clear
D)
Formality
done
clear
View Answer play_arrow
question_answer 101) Philosophers wool on heating with \[BaO\] at \[{{1100}^{o}}C\] produce:
A)
\[Ba+ZnC{{l}_{2}}\]
done
clear
B)
\[BaCd{{O}_{2}}\]
done
clear
C)
\[BaZn{{O}_{2}}\]
done
clear
D)
\[Ba{{O}_{2}}+Zn\]
done
clear
View Answer play_arrow
question_answer 102) Primary amine reacts with carbon disulphide and \[HgC{{l}_{2}}\] to produce alkyl isothiocyanate. This reaction is:
A)
Carbylamine reaction
done
clear
B)
Hofmann bromide reaction
done
clear
C)
Perkin reaction
done
clear
D)
Hofmann mustard oil reaction
done
clear
View Answer play_arrow
question_answer 103) The number of sigma electrons in toluene is:
A)
6
done
clear
B)
9
done
clear
C)
15
done
clear
D)
30
done
clear
View Answer play_arrow
question_answer 104) Sucrose on treatment with cone. \[HCl\] produce:
A)
glucose
done
clear
B)
fructose
done
clear
C)
glucose + fructose
done
clear
D)
laevulinic acid
done
clear
View Answer play_arrow
question_answer 105)
A)
Clemmensen reduction
done
clear
B)
Rosenmund reduction
done
clear
C)
Birch reduction
done
clear
D)
Wolff-Kishner reduction
done
clear
View Answer play_arrow
question_answer 106) The shape of \[N{{H}_{3}}\] molecule is:
A)
tetrahedral
done
clear
B)
trigonal planar
done
clear
C)
trigonal pyramidal
done
clear
D)
linear
done
clear
View Answer play_arrow
question_answer 107) Sodium forms \[N{{a}^{+}}\] ion but it does not form \[N{{a}^{2+}}\] because:
A)
very low value of 1st and II nd IE
done
clear
B)
very high value of 1st and II nd IE
done
clear
C)
high value of 1st IE and low value of II nd IE
done
clear
D)
low value of 1st IE and high value of II nd IE
done
clear
View Answer play_arrow
question_answer 108) Two moles of an ideal gas are compressed isothermally \[({{100}^{o}}C)\] and reversibly from a pressure of 10 atm to 25 atm, then the free energy change is:
A)
\[+15.482\text{ }kJ\]
done
clear
B)
\[+10.464\text{ }kJ\]
done
clear
C)
\[+5.684kJ\]
done
clear
D)
\[+3.364kJ\]
done
clear
View Answer play_arrow
question_answer 109) The most suitable method for removing water traces from ethanol is:
A)
distillation
done
clear
B)
passing dry \[HCl\]
done
clear
C)
reacting it with \[Mg\]
done
clear
D)
heat with sodium metal
done
clear
View Answer play_arrow
question_answer 110) In the exothermic reaction the enthalpy of reaction is always:
A)
zero
done
clear
B)
positive
done
clear
C)
negative
done
clear
D)
none of these
done
clear
View Answer play_arrow
question_answer 111) Assertion: Anhydrous \[Ba{{O}_{2}}\] is used for preparing \[{{H}_{2}}{{O}_{2}}\]. Reason: Hydrated \[Ba{{O}_{2}}\] is not available.
A)
A
done
clear
B)
B
done
clear
C)
C
done
clear
D)
D
done
clear
E)
E
done
clear
View Answer play_arrow
question_answer 112) Assertion: Fluorine exists only in -1 oxidation state. Reason: Fluorine has \[2{{s}^{2}}2{{p}^{5}}\]configuration.
A)
A
done
clear
B)
B
done
clear
C)
C
done
clear
D)
D
done
clear
E)
E
done
clear
View Answer play_arrow
question_answer 113) Assertion: Neopentane forms one mono- substituted compound. Reason: Neopentane is isomer of pentane.
A)
A
done
clear
B)
B
done
clear
C)
C
done
clear
D)
D
done
clear
E)
E
done
clear
View Answer play_arrow
question_answer 114) Assertion: Glycerol is purified by distillation under reduced pressure. Reason: Glycerol is a trihydric alcohol.
A)
A
done
clear
B)
B
done
clear
C)
C
done
clear
D)
D
done
clear
E)
E
done
clear
View Answer play_arrow
question_answer 115) Assertion: Magnesium continue to bum in nitric oxide. Reason: During burning heat evolved do not decompose NO.
A)
A
done
clear
B)
B
done
clear
C)
C
done
clear
D)
D
done
clear
E)
E
done
clear
View Answer play_arrow
question_answer 116) Assertion: All molecules in a gas have same speed. Reason: Gas contains molecules of different size and shape.
A)
A
done
clear
B)
B
done
clear
C)
C
done
clear
D)
D
done
clear
E)
E
done
clear
View Answer play_arrow
question_answer 117) Assertion: Iron is found in free state in nature. Reason: Iron is highly reactive element.
A)
A
done
clear
B)
B
done
clear
C)
C
done
clear
D)
D
done
clear
E)
E
done
clear
View Answer play_arrow
question_answer 118) Assertion: A solution of \[FeC{{l}_{3}}\] in water produces brown precipitate on standing. Reason: Hydrolysis of \[FeC{{l}_{3}}\] takes place in water.
A)
A
done
clear
B)
B
done
clear
C)
C
done
clear
D)
D
done
clear
E)
E
done
clear
View Answer play_arrow
question_answer 119) Assertion: During test for nitrogen with Lassaigne extract on adding \[FeC{{l}_{3}}\] solution sometimes a red precipitate is obtained. Reason: Sulphur is also present
A)
A
done
clear
B)
B
done
clear
C)
C
done
clear
D)
D
done
clear
E)
E
done
clear
View Answer play_arrow
question_answer 120) Assertion: \[C{{H}_{4}}\] does not react with \[C{{l}_{2}}\] in dark. Reason: Chlorination of \[C{{H}_{4}}\] takes place in sunlight.
A)
A
done
clear
B)
B
done
clear
C)
C
done
clear
D)
D
done
clear
E)
E
done
clear
View Answer play_arrow
question_answer 121) Sex organs in Funaria develop:
A)
in protonema
done
clear
B)
outside capsule
done
clear
C)
in the axil of leaf
done
clear
D)
at the tip of gametophore
done
clear
View Answer play_arrow
question_answer 122) In which portion of Cycas diploxylic vascular bundles are found?
A)
Root
done
clear
B)
Stem
done
clear
C)
Leaflet
done
clear
D)
Rachis and leaflet
done
clear
View Answer play_arrow
question_answer 123) Curling of tendrils is due to:
A)
thigmotropism
done
clear
B)
phototropism
done
clear
C)
chemotrophism
done
clear
D)
nyctinasty
done
clear
View Answer play_arrow
question_answer 124) Trophic level are formed by:
A)
plants
done
clear
B)
animals
done
clear
C)
organisms linked in food chain
done
clear
D)
carnivores
done
clear
View Answer play_arrow
question_answer 125) In bacteria site of respiration is:
A)
mesosome
done
clear
B)
episome
done
clear
C)
plasmid
done
clear
D)
cytoplasm
done
clear
View Answer play_arrow
question_answer 126) The usage of binomial names, for plant species was accepted by all after the publication of the work by:
A)
Hooker
done
clear
B)
Linnaeus
done
clear
C)
Bentham
done
clear
D)
Darwin
done
clear
View Answer play_arrow
question_answer 127) Funaria gametophyte is:
A)
dioecious
done
clear
B)
heteroecious
done
clear
C)
autoecious
done
clear
D)
monoecious and autoecious
done
clear
View Answer play_arrow
question_answer 128) I Sometimes, the fern plant arises from fern prothallus without fertilization. This is an example of:
A)
apospory
done
clear
B)
apogamy
done
clear
C)
parthenocarpy
done
clear
D)
gametogenesis
done
clear
View Answer play_arrow
question_answer 129) Zygospore of Spirogyra at the time of meiosis is divided into four nuclei. How many nuclei degenerate out of these four?
A)
One
done
clear
B)
Two
done
clear
C)
Three
done
clear
D)
Four
done
clear
View Answer play_arrow
question_answer 130) Cycas is:
A)
monoecious
done
clear
B)
bisexual
done
clear
C)
dioecious
done
clear
D)
hermaphrodite
done
clear
View Answer play_arrow
question_answer 131) Clove is:
A)
flower bud
done
clear
B)
axillary bud
done
clear
C)
thalamus
done
clear
D)
ovule
done
clear
View Answer play_arrow
question_answer 132) In which family (9) + 1 androecium condition; is found?
A)
Malvaceae
done
clear
B)
Papilionaceae
done
clear
C)
Solanaceae
done
clear
D)
Poaceae
done
clear
View Answer play_arrow
question_answer 133) Powdery mildews of crops are caused by:
A)
bacteria
done
clear
B)
ascomycetes
done
clear
C)
basidiomycetes
done
clear
D)
phycomycetes
done
clear
View Answer play_arrow
question_answer 134) Abundance of a species in a population, within habitat is called :
A)
niche density
done
clear
B)
absolute density
done
clear
C)
relative density
done
clear
D)
geographic density
done
clear
View Answer play_arrow
question_answer 135) Desert can be converted into green land by planting:
A)
oxylophytes
done
clear
B)
psammophytes
done
clear
C)
halophytes
done
clear
D)
trees
done
clear
View Answer play_arrow
question_answer 136) Deforestation causes:
A)
soil erosion
done
clear
B)
soil pollution
done
clear
C)
noise pollution
done
clear
D)
air pollution
done
clear
View Answer play_arrow
question_answer 137) Porous wood contains:
A)
vessels
done
clear
B)
tracheids
done
clear
C)
fibres
done
clear
D)
parenchyma
done
clear
View Answer play_arrow
question_answer 138) In rainy season, door get swelled due to:
A)
imbibition
done
clear
B)
diffusion
done
clear
C)
transpiration
done
clear
D)
respiration
done
clear
View Answer play_arrow
question_answer 139) Blackmans law of limiting factor is applied to:
A)
respiration
done
clear
B)
transpiration
done
clear
C)
photorespiration
done
clear
D)
photosynthesis
done
clear
View Answer play_arrow
question_answer 140) The drug bellodona is obtained from:
A)
Atropa
done
clear
B)
Opium
done
clear
C)
Rauwolffia
done
clear
D)
Solanwn
done
clear
View Answer play_arrow
question_answer 141) Pollinia are found in:
A)
wheat
done
clear
B)
madar
done
clear
C)
mango
done
clear
D)
banana
done
clear
View Answer play_arrow
question_answer 142) Which part of embryo comes out first during seed germination?
A)
Radicle
done
clear
B)
Plumule
done
clear
C)
Hypocotyl
done
clear
D)
Epicotyl
done
clear
View Answer play_arrow
question_answer 143) Minimata disease is pollution related disease. It result from:
A)
oil spills in sea
done
clear
B)
DDT pollution
done
clear
C)
release of industrial waste containing mercury in fishing water
done
clear
D)
accumulation of arsenic
done
clear
View Answer play_arrow
question_answer 144) Monocarpic plants flower:
A)
once
done
clear
B)
twice
done
clear
C)
many times
done
clear
D)
never
done
clear
View Answer play_arrow
question_answer 145) A prokaryotic cell lacks:
A)
true nucleus
done
clear
B)
nuclear membrane
done
clear
C)
membrane bound organelles
done
clear
D)
all of the above
done
clear
View Answer play_arrow
question_answer 146) Induction of cell division and delay in senescence is done by:
A)
cytokinins
done
clear
B)
auxins
done
clear
C)
GA
done
clear
D)
CoA
done
clear
View Answer play_arrow
question_answer 147) Mirabilis jalapa shows:
A)
codominance
done
clear
B)
incomplete dominance
done
clear
C)
dominance
done
clear
D)
complementary genes
done
clear
View Answer play_arrow
question_answer 148) In genetic engineering, which of following is used?
A)
Plasmid
done
clear
B)
Plastid
done
clear
C)
Mitochondria
done
clear
D)
ER
done
clear
View Answer play_arrow
question_answer 149) The maintenance of internal favourable conditions, by a self-regulated mechanisms inspite of the fact that there are changes in environment, is known as:
A)
entropy
done
clear
B)
enthalpy
done
clear
C)
homeostasis
done
clear
D)
steady state
done
clear
View Answer play_arrow
question_answer 150) Which of the following is a single membranous structure?
A)
Lysosome
done
clear
B)
Nucleus
done
clear
C)
Mitochondria
done
clear
D)
Chloroplast
done
clear
View Answer play_arrow
question_answer 151) Brunners glands are present in:
A)
duodenum
done
clear
B)
oesophagus
done
clear
C)
ileum
done
clear
D)
stomach
done
clear
View Answer play_arrow
question_answer 152) 5th cranial nerve of frog is called :
A)
vagus
done
clear
B)
trigeminal
done
clear
C)
optic
done
clear
D)
ophthalmic
done
clear
View Answer play_arrow
question_answer 153) Which of the following is made up of a single bone in mammal?
A)
Dentary
done
clear
B)
Hyoid
done
clear
C)
Upper jaw
done
clear
D)
All of these
done
clear
View Answer play_arrow
question_answer 154) Spindle fibers of mitotic cell are made up of:
A)
tubulin
done
clear
B)
actin
done
clear
C)
myosin
done
clear
D)
collagen
done
clear
View Answer play_arrow
question_answer 155) Gemmule formation in sponges are useful:
A)
asexual reproduction
done
clear
B)
sexual reproduction
done
clear
C)
parthenogenesis
done
clear
D)
parthenocarpy
done
clear
View Answer play_arrow
question_answer 156) Which of the following is not a mental disorder?
A)
Epilepsy
done
clear
B)
Neurosis
done
clear
C)
Psychosis
done
clear
D)
Plague
done
clear
View Answer play_arrow
question_answer 157) Pollorum disease of poultry is caused by:
A)
Mycobacterium,
done
clear
B)
Salmonella
done
clear
C)
Clostridium
done
clear
D)
Hemophilus
done
clear
View Answer play_arrow
question_answer 158) Discontinuous variations are:
A)
essential features
done
clear
B)
acquired character
done
clear
C)
non-essential changes
done
clear
D)
mutations
done
clear
View Answer play_arrow
question_answer 159) Cessation of menstrual cycle in women is called:
A)
menopause
done
clear
B)
lactation
done
clear
C)
ovulation
done
clear
D)
parturition
done
clear
View Answer play_arrow
question_answer 160) Barr-body in mammals represents:
A)
one of the two X-chromosomes in cells of female
done
clear
B)
all heterochromatin of male and female cells
done
clear
C)
Y-chromosome of male
done
clear
D)
all heterochromatin of female cells
done
clear
View Answer play_arrow
question_answer 161) If frogs brain is crushed, even then moves on pinpointing. It is called:
A)
simple reflex
done
clear
B)
conditional reflex
done
clear
C)
neurotransmitter function
done
clear
D)
autonomic nerve conditions
done
clear
View Answer play_arrow
question_answer 162) Acromegaly is due to hypersecretion of: 3
A)
insulin
done
clear
B)
throxin
done
clear
C)
growth hormone
done
clear
D)
none of these
done
clear
View Answer play_arrow
question_answer 163) Sella turcica is found:
A)
near pituitary
done
clear
B)
in bone
done
clear
C)
in joints
done
clear
D)
near thyroid
done
clear
View Answer play_arrow
question_answer 164) Major protein of connective tissue is:
A)
myosin
done
clear
B)
collagen
done
clear
C)
melanin
done
clear
D)
keratin
done
clear
View Answer play_arrow
question_answer 165) Toxic substances are detoxified in human in:
A)
kidney
done
clear
B)
lungs
done
clear
C)
liver
done
clear
D)
stomach
done
clear
View Answer play_arrow
question_answer 166) Outer covering of cartilage is called:
A)
perichondrium
done
clear
B)
periostaeum
done
clear
C)
endosternum
done
clear
D)
perilonettum
done
clear
View Answer play_arrow
question_answer 167) Steroid hormones are similar in structure
A)
tryosine
done
clear
B)
cholesterol
done
clear
C)
coenzyme A
done
clear
D)
glycerol
done
clear
View Answer play_arrow
question_answer 168) The protoplasmic segment of a striated ran fiber is termed as:
A)
sarcoplasm
done
clear
B)
sarcomere
done
clear
C)
neuromere
done
clear
D)
metamere
done
clear
View Answer play_arrow
question_answer 169) Meroblastic cleavage refers to which type of division of egg?
A)
Complete
done
clear
B)
Spiral
done
clear
C)
Incomplete
done
clear
D)
Horizontal
done
clear
View Answer play_arrow
question_answer 170) Nodules with nitrogen fixing bacteria are found in:
A)
Cotton
done
clear
B)
gram
done
clear
C)
Mustard
done
clear
D)
wheat
done
clear
View Answer play_arrow
question_answer 171) Assertion: TMV is a virus which causes mosaic disease. Reason: TMV has RNA as genetic material,
A)
A
done
clear
B)
B
done
clear
C)
C
done
clear
D)
D
done
clear
E)
E
done
clear
View Answer play_arrow
question_answer 172) Assertion: Mosses are evolved from algae. Reason: Protonema of mosses is similar to some green algae.
A)
A
done
clear
B)
B
done
clear
C)
C
done
clear
D)
D
done
clear
E)
E
done
clear
View Answer play_arrow
question_answer 173) Assertion: Many plants are propagated vegetatively even though they bear seeds. Reason: Potatos multiply by tubers, apple by cutting etc.
A)
A
done
clear
B)
B
done
clear
C)
C
done
clear
D)
D
done
clear
E)
E
done
clear
View Answer play_arrow
question_answer 174) Assertion: Power house of cell is mitochondria. Reason: ATP is produced in mitochondria,
A)
A
done
clear
B)
B
done
clear
C)
C
done
clear
D)
D
done
clear
E)
E
done
clear
View Answer play_arrow
question_answer 175) Assertion: Scurvy is caused by deficiency of vitamin. Reason: Deficiency of ascorbic acid causes scurvy.
A)
A
done
clear
B)
B
done
clear
C)
C
done
clear
D)
D
done
clear
E)
E
done
clear
View Answer play_arrow
question_answer 176) Assertion: Cell wall is not found in animal cell. Reason: Animal cells are covered by cell membrane.
A)
A
done
clear
B)
B
done
clear
C)
C
done
clear
D)
D
done
clear
E)
E
done
clear
View Answer play_arrow
question_answer 177) Assertion: During physiology of excretion, deamination does not take place in liver. Reason: Deamination is a process to make use of excess of amino acids which cannot be incorporated into protoplasm.
A)
A
done
clear
B)
B
done
clear
C)
C
done
clear
D)
D
done
clear
E)
E
done
clear
View Answer play_arrow
question_answer 178) Assertion: Cartilage and bone are rigid connective tissues. Reason: Blood is a connective tissue,
A)
A
done
clear
B)
B
done
clear
C)
C
done
clear
D)
D
done
clear
E)
E
done
clear
View Answer play_arrow
question_answer 179) Assertion: Plasmids are extra chromosomal DNA. Reason: Plasmids are found in bacteria and are useful in genetic engineering,
A)
A
done
clear
B)
B
done
clear
C)
C
done
clear
D)
D
done
clear
E)
E
done
clear
View Answer play_arrow
question_answer 180) Assertion: Plasmodium vivax is responsible for malaria. Reason: Malaria is caused by polluted water,
A)
A
done
clear
B)
B
done
clear
C)
C
done
clear
D)
D
done
clear
E)
E
done
clear
View Answer play_arrow
question_answer 181) National Anthem ?Jana Gana Mana? was adopted on:
A)
26 January, 1950
done
clear
B)
26 July, 1947
done
clear
C)
15 August, 1947
done
clear
D)
24 January, 1950
done
clear
View Answer play_arrow
question_answer 182) Who is the chairman of Rajya Sabha?
A)
Speaker of Lok Sabha
done
clear
B)
Home Minister
done
clear
C)
President
done
clear
D)
Vice President
done
clear
View Answer play_arrow
question_answer 183) Miss Universe event 2000 was held at:
A)
Peuotro Rico
done
clear
B)
London
done
clear
C)
Paris
done
clear
D)
Nicosia (Cyprus)
done
clear
View Answer play_arrow
question_answer 184) The earthquake is measured by:
A)
Lactometer
done
clear
B)
Seismograph
done
clear
C)
Hygrometer
done
clear
D)
Barometer
done
clear
View Answer play_arrow
question_answer 185) The writer of ?Daughter of East? is:
A)
Indira Gandhi
done
clear
B)
Benazir Bhutto
done
clear
C)
Amrita Pritam
done
clear
D)
Marget Thatcher
done
clear
View Answer play_arrow
question_answer 186) Which one of the following is the argest steel arch bridge?
A)
Seawise
done
clear
B)
Petronas
done
clear
C)
Strahov
done
clear
D)
George
done
clear
View Answer play_arrow
question_answer 187) AIDS is caused by:
A)
Helminth
done
clear
B)
Protozoa
done
clear
C)
Virus
done
clear
D)
Bacteria
done
clear
View Answer play_arrow
question_answer 188) Which one of the following function of the 14. platlets occurs in our body ?
A)
It helps in breathing
done
clear
B)
It helps in strengthening of gums
done
clear
C)
It hels in circulation of blood
done
clear
D)
It helps in clotting of blood
done
clear
View Answer play_arrow
question_answer 189) At the first time, the song Vande Mataram was sung in:
A)
Indian National Congress session
done
clear
B)
Indian National Congress session
done
clear
C)
Quit India Movement 1942
done
clear
D)
Congress session 1911
done
clear
View Answer play_arrow
question_answer 190) Teachers day is celebrated on
A)
5th September
done
clear
B)
16 August
done
clear
C)
21st September
done
clear
D)
1st April
done
clear
View Answer play_arrow
question_answer 191) Which one of the following determines the salary of Attorney General ?
A)
Speaker of Lok Sabha
done
clear
B)
Home Minister
done
clear
C)
President of India
done
clear
D)
Prime Minister
done
clear
View Answer play_arrow
question_answer 192) Which one of the following vitamin can be most easily synthesised in the human body?
A)
Vitamin B
done
clear
B)
Vitamin C
done
clear
C)
Vitamin A
done
clear
D)
Vitamin D
done
clear
View Answer play_arrow
question_answer 193) From where did Mahatma Gandhi start the famous Dandi March?
A)
Surat
done
clear
B)
Mumbai
done
clear
C)
Bardoli
done
clear
D)
Ahmedabad
done
clear
View Answer play_arrow
question_answer 194) In which year was English recommi medium of instruction for higher India by Lord Macaulay?
A)
1833
done
clear
B)
1835
done
clear
C)
1859
done
clear
D)
1825
done
clear
View Answer play_arrow
question_answer 195) Who discovered the sea route to India?
A)
Vasco de Gama
done
clear
B)
Columbus
done
clear
C)
Magellan
done
clear
D)
Hopkins
done
clear
View Answer play_arrow
question_answer 196) The oath of office to a Supreme Court administered by:
A)
The Chief Justice
done
clear
B)
The President of India
done
clear
C)
The Chief Justice of India
done
clear
D)
The Law minister
done
clear
View Answer play_arrow
question_answer 197) Garba dance is a dance style of:
A)
Gujrat
done
clear
B)
Uttar Pradesh
done
clear
C)
Nagaland
done
clear
D)
Bihar
done
clear
View Answer play_arrow
question_answer 198) Who addressed the U.N. General Assi the first time in Hindi ?
A)
Rajendra Prasad
done
clear
B)
Atal BihariVajps
done
clear
C)
Jawahar Lai Nehru
done
clear
D)
Swam Singh
done
clear
View Answer play_arrow
question_answer 199) A former cricketer after whose name no championship has been started in India:
A)
C.K. Naidu
done
clear
B)
Daleep Singh
done
clear
C)
Lala Amarnath
done
clear
D)
Vijay Merchant
done
clear
View Answer play_arrow
question_answer 200) Currency note bears the signature of the:
A)
Finance Minister
done
clear
B)
Governor, Reserve Bank of India
done
clear
C)
Cabinet Secretary
done
clear
D)
President
done
clear
View Answer play_arrow
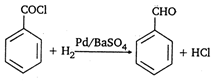 The above reaction is:
The above reaction is:
