question_answer 1) Deuteron and alpha particle in air are at separation \[1\overset{\text{o}}{\mathop{\text{A}}}\,\] The magnitude of electric field intensity on a-particle due to deuteron is
A)
\[\text{5}.\text{76}\times \text{l}{{0}^{\text{11}}}\text{ N}/\text{C}\]
done
clear
B)
\[\text{1}.\text{44}\times \text{l}{{0}^{\text{11}}}\text{ N}/\text{C}\]
done
clear
C)
\[\text{2}.\text{828}\times \text{l}{{0}^{\text{11}}}\text{ N}/\text{C}\]
done
clear
D)
zero
done
clear
View Answer play_arrow
question_answer 2) If \[{{E}_{1}}\] be the electric field strength of a short dipole at a point on its axial line and \[{{E}_{2}}\]that on the equatorial line at the same distance, then
A)
\[{{E}_{1}}={{E}_{2}}\]
done
clear
B)
\[{{E}_{1}}=2{{E}_{2}}\]
done
clear
C)
\[{{E}_{2}}=2{{E}_{1}}\]
done
clear
D)
None of the above
done
clear
View Answer play_arrow
question_answer 3) 20 A current is flowing in a long straight wire. The intensity of magnetic field at a distance of 10 m from the wire, will be
A)
\[\text{4}\times \text{l}{{\text{0}}^{-\text{5}}}\text{Wb}/{{\text{m}}^{\text{2}}}\]
done
clear
B)
\[\text{2}\times \text{l}{{\text{0}}^{-\text{5}}}\text{Wb}/{{\text{m}}^{\text{2}}}\]
done
clear
C)
\[3\times {{10}^{-5}}\,Wb/{{m}^{2}}\]
done
clear
D)
\[\text{8}\times \text{l}{{\text{0}}^{-5}}\text{Wb}/{{\text{m}}^{\text{2}}}\]
done
clear
View Answer play_arrow
question_answer 4) An electron having charge \[1.6\times {{10}^{-19}}C\]and mass \[\text{9}\times \text{1}{{0}^{-31}}\text{kg}\]is moving with \[\text{4}\times \text{1}{{0}^{\text{6}}}\text{ m}/\text{s}\] speed in a magnetic field of \[\text{2}\times \text{1}{{0}^{-\text{1}}}\] T in a circular orbit. The force acting on an electron and the radius of circular orbit will be
A)
\[1.28\times {{10}^{-14}}N,1.1\times {{10}^{-3}}m\]
done
clear
B)
\[\text{1}.\text{28}\times \text{1}{{0}^{\text{15}}}\text{ N},\text{ 1}.\text{2}\times \text{1}{{0}^{-\text{12}}}\text{ m}\]
done
clear
C)
\[\text{1}.\text{28}\times \text{l}{{0}^{-\text{13}}}\text{ N},\text{ 1}.\text{1}\times \text{l}{{0}^{-\text{4}}}\text{ m}\]
done
clear
D)
None of the above
done
clear
View Answer play_arrow
question_answer 5) The capacity of a condenser is \[\text{4}\times \text{1}{{0}^{-6}}\] F and its potential is 100 V. The energy released on discharging it fully will be
A)
0.04 J
done
clear
B)
0.02 J
done
clear
C)
0.025 J
done
clear
D)
0.05 J
done
clear
View Answer play_arrow
question_answer 6) The electric potential at the surface of an atomic nucleus \[\left( \text{Z }=\text{ 5}0. \right)\] of radius 9.0 x 10-13 cm is
A)
\[\text{9}\times \text{1}{{0}^{5}}\text{ V}\]
done
clear
B)
\[\text{8}\times \text{1}{{0}^{\text{6}}}\text{ V}\]
done
clear
C)
80 V
done
clear
D)
9 V
done
clear
View Answer play_arrow
question_answer 7) The half-life of radium is 1620 yr and its atomic weight is 226 kg per kilomol. The number of atoms that will decay from its 1 g sample per second will be (Avogadros number \[\text{N}=\text{6}.0\text{23}\times \text{1}{{0}^{\text{23}}}\text{ atoms}/\text{ mol})\]
A)
\[\text{3}.\text{61}\times \text{l}{{0}^{\text{1}0}}\]
done
clear
B)
\[\text{3}.\text{61}\times \text{l}{{0}^{\text{12}}}\]
done
clear
C)
\[\text{3}.\text{11}\times \text{l}{{0}^{\text{15}}}\]
done
clear
D)
\[\text{31}.\text{1}\times \text{l}{{0}^{\text{15}}}\]
done
clear
View Answer play_arrow
question_answer 8) One milligram of matter converted into energy, will give
A)
9 J
done
clear
B)
\[9\times {{10}^{3}}J\]
done
clear
C)
\[9\times {{10}^{5}}J\]
done
clear
D)
\[9\times {{10}^{10}}J\]
done
clear
View Answer play_arrow
question_answer 9) In a simple microscope, if the final image is located at 25 cm from the eye placed close to the lens, then the magnifying power is
A)
\[\frac{25}{f}\]
done
clear
B)
\[1+\frac{25}{f}\]
done
clear
C)
\[\frac{f}{25}\]
done
clear
D)
\[\frac{f}{25}+1\]
done
clear
View Answer play_arrow
question_answer 10) A black body at 200 K is found to exit maximum energy at a wavelength 14 \[\mu \]m. When its temperature is raised to 1000 K, then wavelength at which maximum energy emitted is
A)
14 mm
done
clear
B)
7 \[\mu m\]
done
clear
C)
2.8\[\mu m\]
done
clear
D)
28mm
done
clear
View Answer play_arrow
question_answer 11) The ratio of thermal conductivity of two rods of different material is 5 4. The two rods of same area of cross-section and same thermal resistance will have the lengths in the ratio
A)
45
done
clear
B)
91
done
clear
C)
19
done
clear
D)
54
done
clear
View Answer play_arrow
question_answer 12) Joule second is the unit of
A)
momentum
done
clear
B)
angular momentum
done
clear
C)
work
done
clear
D)
pressure
done
clear
View Answer play_arrow
question_answer 13) The two slits at a distance of 1 mm are illuminated by the light of wavelength \[\text{6}.5\times \text{l}{{\text{0}}^{-\text{7}}}\text{m}.\] the interference fringes are observed on a serene placed at a distance of 1 m. The distance between third dark fringe and fifth bright fringe will be
A)
0.65 cm
done
clear
B)
4.8 mm
done
clear
C)
1.63 mm
done
clear
D)
3.25 cm
done
clear
View Answer play_arrow
question_answer 14) Two thin lenses, one of focal length + 60 cm and the other of focal length - 20 cm are put in contact, the combined focal length is
A)
15 cm
done
clear
B)
-15 cm
done
clear
C)
- 30 cm
done
clear
D)
30 cm
done
clear
View Answer play_arrow
question_answer 15) When light travels from one medium to the other medium of which the refractive index is different, then which of the following will change?
A)
Frequency, wavelength and velocity
done
clear
B)
Frequency and wavelength
done
clear
C)
Frequency and velocity
done
clear
D)
Wavelength and velocity
done
clear
View Answer play_arrow
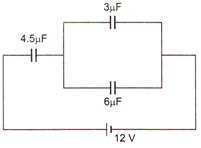
question_answer 16)
In the circuit shown in the figure, the potential difference across the 4.5\[\mu F\] capacitor is
A)
\[\frac{8}{3}V\]
done
clear
B)
\[4\,V\]
done
clear
C)
\[6\,V\]
done
clear
D)
\[8\,V\]
done
clear
View Answer play_arrow
question_answer 17) When \[_{\text{92}}{{\text{U}}^{\text{235}}}\] undergoes fission, 0.1% its original mass is changed into energy. How much energy is released if 1 kg of \[_{\text{92}}\text{ }{{\text{U}}^{\text{235}}}\] undergoes fission?
A)
\[\text{9}\times \text{1}{{0}^{\text{1}0}}\text{ J}\]
done
clear
B)
\[\text{9}\times \text{1}{{0}^{\text{11}}}\text{ J}\]
done
clear
C)
\[\text{9}\times \text{1}{{0}^{\text{12}}}\text{ J}\]
done
clear
D)
\[\text{9}\times \text{1}{{0}^{13}}\text{J}\]
done
clear
View Answer play_arrow
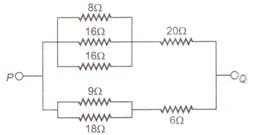
question_answer 18)
The equivalent resistance of the arrangement of resistances shown in the given figure between points P and Q is
A)
60
done
clear
B)
80
done
clear
C)
240
done
clear
D)
160
done
clear
View Answer play_arrow
question_answer 19) A cricketer hits a ball with a velocity 25 m/s at \[60{}^\circ \] above the horizontal. How far above the ground it passes over a fielder 50 m from the bat? (assume the ball is struck very close to the ground)
A)
8.2 m
done
clear
B)
9.0 m
done
clear
C)
11.6 m
done
clear
D)
12.7 m
done
clear
View Answer play_arrow
question_answer 20) It is easier to roll a barrel than pull it along the road. This statement is
A)
not possible
done
clear
B)
uncertain
done
clear
C)
false
done
clear
D)
true
done
clear
View Answer play_arrow
question_answer 21) The diameter of brass road is 4 mm. Youngs modulus of brass is 9 xl010 N/m2. The force required to stretch 0.1% of its length is
A)
360 \[\pi \]N
done
clear
B)
36 N
done
clear
C)
\[\text{36}\pi \times \text{1}{{0}^{5}}\text{ N}\]
done
clear
D)
\[\text{14471}\times \text{1}{{0}^{\text{3}}}\text{ N}\]
done
clear
View Answer play_arrow
question_answer 22) A beam of ions enters normally into a uniform magnetic field of \[\text{4}\times \text{1}{{0}^{-2}}\] T with velocity \[\text{2}\times \text{1}{{0}^{\text{5}}}\text{ m}/\text{s}.\]If the specific charge of the ion - \[\text{5}\times \text{l}{{0}^{\text{7}}}\text{ C}/\text{kg},\]Then the radius of the circular path described will be
A)
0.10 m
done
clear
B)
0.06 m
done
clear
C)
0.20 m
done
clear
D)
0.25 m
done
clear
View Answer play_arrow
question_answer 23) Parsec is a unit of
A)
angle
done
clear
B)
velocity
done
clear
C)
time
done
clear
D)
distance
done
clear
View Answer play_arrow
question_answer 24) If a given mass of gas occupies a volume 10 cc at 1 atm pressure and temperature \[100{}^\circ C\]. What will be its volume at 4 ac= pressure, the temperature being the same?
A)
100 cc
done
clear
B)
400 cc
done
clear
C)
104 cc
done
clear
D)
2.5 cc
done
clear
View Answer play_arrow
question_answer 25) Ultraviolet radiation of 6.2 eV falls on an aluminium foil surface. Work function s 4.2 eV. The kinetic energy of the fastes electron emitted approximately
A)
\[\text{3}.\text{2}\times \text{I}{{\text{0}}^{\text{-21}}}\text{ J}\]
done
clear
B)
\[\text{3}.\text{2}\times \text{1}{{0}^{-19}}\text{ J}\]
done
clear
C)
\[\text{3}.\text{2}\times \text{1}{{0}^{-\text{15}}}\text{ J}\]
done
clear
D)
\[\text{3}.\text{2}\times \text{l}{{\text{O}}^{-17}}\text{ J}\]
done
clear
View Answer play_arrow
question_answer 26) Two gases of equal masses are in thermal equilibrium. If \[{{\text{p}}_{a}},\]\[{{\text{p}}_{b}}\]and \[{{V}_{a}},\]\[{{V}_{b}}\] are their respective pressures and volumes, then which relation is true?
A)
\[2{{p}_{a}}{{V}_{a}}={{p}_{b}}{{V}_{b}}\]
done
clear
B)
\[{{p}_{a}}\ne {{p}_{b}},{{V}_{a}}={{V}_{b}}\]
done
clear
C)
\[\frac{{{p}_{a}}}{{{V}_{a}}}=\frac{{{p}_{b}}}{{{V}_{b}}}\]
done
clear
D)
\[{{p}_{a}}{{V}_{a}}={{p}_{b}}{{V}_{b}}\]
done
clear
View Answer play_arrow
question_answer 27) A thin uniform rod of mass m and length is hinged at the lower end to a level floor and strands vertically. It is now allowed to fall, then its upper end will strike the floor with the velocity
A)
\[\sqrt{2gl}\]
done
clear
B)
\[\sqrt{5gl}\]
done
clear
C)
\[\sqrt{3gl}\]
done
clear
D)
\[\sqrt{mgl}\]
done
clear
View Answer play_arrow
question_answer 28) The magnitude of acceleration of particle executing SHM at the position of maximum displacement is
A)
zero
done
clear
B)
minimum
done
clear
C)
maximum
done
clear
D)
None of these
done
clear
View Answer play_arrow
question_answer 29) Mud houses are cooler in summer and warmer in winter because
A)
mud is a good conductor of heat
done
clear
B)
mud is a super conductor of heat
done
clear
C)
mud is a bad conductor of heat
done
clear
D)
None of the above
done
clear
View Answer play_arrow
question_answer 30) Drift velocity varies with the velocity of electric field as per the relation
A)
\[{{v}_{d}}={{E}^{2}}\]
done
clear
B)
\[{{v}_{d}}\propto E\]
done
clear
C)
\[{{v}_{d}}\propto \frac{1}{E}\]
done
clear
D)
\[{{v}_{d}}\propto {{E}^{2}}\]
done
clear
View Answer play_arrow
question_answer 31) If the critical angle for total internal reflection from a medium to vacuum is \[30{}^\circ \], then velocity of light in medium will be
A)
\[\text{1}.\text{5}\times \text{1}{{0}^{\text{8}}}\text{ m}/\text{s}\]
done
clear
B)
\[\text{2}\times \text{l}{{0}^{\text{8}}}\text{ m}/\text{s}\]
done
clear
C)
\[\text{3}\times \text{l}{{0}^{\text{8}}}\text{m}/\text{s}\]
done
clear
D)
\[\text{5}\times \text{l}{{0}^{\text{8}}}\text{m}/\text{s}\]
done
clear
View Answer play_arrow
question_answer 32) What is the momentum of a 10000 kg truck whose velocity is 20 m/s?
A)
\[\text{2}\times \text{1}{{0}^{\text{5}}}\text{ kg m}/\text{s}\]
done
clear
B)
\[\text{1}\times \text{1}{{0}^{8}}\text{ kg m}/\text{s}\]
done
clear
C)
4 kg m/s
done
clear
D)
None of these
done
clear
View Answer play_arrow
question_answer 33) A 10 kg ball moving with velocity 2 m/s collides with a 20 kg mass initially at rest. If both of them coalesce, the final velocity of combined mass is
A)
\[\frac{3}{4}m/s\]
done
clear
B)
\[\frac{1}{3}m/s\]
done
clear
C)
\[\frac{3}{2}m/s\]
done
clear
D)
\[\frac{2}{3}m/s\]
done
clear
View Answer play_arrow
question_answer 34) A 30 g bullet travelling initially at 500 m/s penetrates 12 cm into wooden block. The average force exerted will be
A)
31250 N
done
clear
B)
41250 N
done
clear
C)
31750 N
done
clear
D)
30450 N
done
clear
View Answer play_arrow
question_answer 35) With what minimum acceleration can a fireman slide down a rope whose breaking strength is two third of his weight?
A)
\[\frac{g}{m}\]
done
clear
B)
\[\frac{2}{3}g\]
done
clear
C)
\[\frac{3}{2}g\]
done
clear
D)
\[\frac{g}{2}\]
done
clear
View Answer play_arrow
question_answer 36) Calculate the force required to separate the glass plate of area \[\text{1}{{0}^{-2}}\text{ }{{\text{m}}^{\text{2}}}\]with a film of water 0.05 mm thick, [surface tension of water is \[\text{7}0\times \text{1}{{0}^{-3}}\text{ N}/\text{m}]\]
A)
25 N
done
clear
B)
20 N
done
clear
C)
14 N
done
clear
D)
28 N
done
clear
View Answer play_arrow
question_answer 37) At which of the following temperature would the molecules of gas have twice the average kinetic energy they have at \[20{}^\circ C\]?
A)
\[40{}^\circ C\]
done
clear
B)
\[80{}^\circ C\]
done
clear
C)
\[586{}^\circ C\]
done
clear
D)
\[313{}^\circ C\]
done
clear
View Answer play_arrow
question_answer 38) Air is expanded from 50 L to 150 L at 2 atm pressure. The external work done is \[\left( \text{1 atm }=\text{1}\times \text{l}{{0}^{\text{5}}}\text{ N}/{{\text{m}}^{\text{2}}} \right)\]
A)
\[\text{2}\times \text{l}{{\text{0}}^{-\text{8}}}\text{J}\]
done
clear
B)
\[2\times {{10}^{4}}J\]
done
clear
C)
200 J
done
clear
D)
2000 J
done
clear
View Answer play_arrow
question_answer 39) The mass of moon is 1% of mass of earth. The ratio of gravitational pull of earth on moon and that of moon on earth will be
A)
1 : 1
done
clear
B)
1 : 10
done
clear
C)
1 : 100
done
clear
D)
2 : 1
done
clear
View Answer play_arrow
question_answer 40) At what temperature, the speed of sound in air will become double of its value at \[27{}^\circ C\]?
A)
\[54{}^\circ C\]
done
clear
B)
\[627{}^\circ C\]
done
clear
C)
\[327{}^\circ C\]
done
clear
D)
\[927{}^\circ C\]
done
clear
View Answer play_arrow
question_answer 41) A wave is expressed by the equation \[\text{y}=\text{ }0.\text{5 sin }\pi \left( 0.0\text{1x}-\text{3t} \right)\] Where x, y are in metres and f in second. The speed of propagation will be
A)
150 m/s
done
clear
B)
300 m/s
done
clear
C)
350 m/s
done
clear
D)
250 m/s
done
clear
View Answer play_arrow
question_answer 42) A uniform rope of mass 0.1 kg and length 2.5 m hangs from ceiling. The speed of transverse wave in the rope at upper end and at a point 0.5 m distance from lower end will be
A)
\[\text{5 m}/\text{s},\text{ 2}.\text{24 m}/\text{s}\]
done
clear
B)
\[\text{1}0\text{ m}/\text{s},\text{ 3}.\text{23 m}/\text{s}\]
done
clear
C)
\[\text{7}.\text{5 m}/\text{s},\text{ 1}.\text{2 m}/\text{s}\]
done
clear
D)
None of the above
done
clear
View Answer play_arrow
question_answer 43) If the horizontal and vertical component of earths magnetic field are equal at a certain place, the angle of dip is
A)
\[90{}^\circ \]
done
clear
B)
\[60{}^\circ \]
done
clear
C)
\[45{}^\circ \]
done
clear
D)
\[0{}^\circ \]
done
clear
View Answer play_arrow
question_answer 44) A wire of length L and cross-sectional area A is made of a material of Youngs modulus Y. If the wire is stretched by the amount x. The work done is
A)
\[\frac{YA{{x}^{2}}}{2L}\]
done
clear
B)
\[\frac{YA{{x}^{2}}}{L}\]
done
clear
C)
\[YA{{x}^{2}}L\]
done
clear
D)
\[\frac{YAx}{2L}\]
done
clear
View Answer play_arrow
question_answer 45) The length of an astronomical telescope for normal vision (relaxed eye) will be
A)
\[{{f}_{o}}-{{f}_{e}}\]
done
clear
B)
\[{{f}_{o}}/{{f}_{e}}\]
done
clear
C)
\[{{f}_{o}}\times {{f}_{e}}\]
done
clear
D)
\[{{f}_{o}}+{{f}_{e}}\]
done
clear
View Answer play_arrow
question_answer 46) A biconvex lens is made of material of refractive index 1.5 each of radius of curvature of lens surface is 20 cm. The focal length of lens is
A)
20cm
done
clear
B)
10cm
done
clear
C)
25cm
done
clear
D)
30cm
done
clear
View Answer play_arrow
question_answer 47) An equiconvex lens of glass of focal length 0.1m is cut along a plane perpendicular to principle axis into two equal parts. The ratio of focal length of new lenses formed is
A)
1 : 1
done
clear
B)
1: 2
done
clear
C)
2 : 1
done
clear
D)
\[2:\frac{1}{2}\]
done
clear
View Answer play_arrow
question_answer 48) In an X-ray tube, electrons bombarded the target produce X-rays of minimum wavelength 1Å. The energy of bombarding electron will be
A)
100 eV
done
clear
B)
14375 eV
done
clear
C)
12000 eV
done
clear
D)
12375 eV
done
clear
View Answer play_arrow
question_answer 49) In producing chlorine through electrolysis.. 100 kW power at 125 V is being consumed. The mass of chlorine per minute liberated, will be \[(z=0.367\times {{10}^{-6}}kg/C)\]
A)
\[\text{17}.\text{61}\times \text{l}{{\text{0}}^{\text{-3}}}\text{ kg}\]
done
clear
B)
\[10.61\times {{10}^{-3}}kg\]
done
clear
C)
\[\text{12}.\text{61}\times \text{1}{{0}^{-3}}\text{kg}\]
done
clear
D)
None of the above
done
clear
View Answer play_arrow
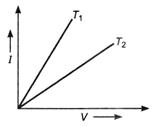
question_answer 50)
The current-voltage graph for a given men. wire at two different temperatures \[{{T}_{1}}\]and are shown in given figure, which of the relation is correct from the following?
A)
\[{{T}_{2}}>{{T}_{1}}\]
done
clear
B)
\[{{T}_{1}}>{{T}_{2}}\]
done
clear
C)
\[{{T}_{1}}={{T}_{2}}\]
done
clear
D)
None of these
done
clear
View Answer play_arrow
question_answer 51) When \[C{{H}_{3}}COOH\] reacts with \[C{{H}_{3}}-MgX\]
A)
\[C{{H}_{3}}COX\] is formed
done
clear
B)
hydrocarbon is formed
done
clear
C)
acetone is formed
done
clear
D)
alcohol is formed
done
clear
View Answer play_arrow
question_answer 52) A cyclic hydrocarbon molecule has all the carbon and hydrogen in a single plane. All the carbon-carbon bonds are of same length, less than \[1.54\,\overset{o}{\mathop{A}}\,\], but more than \[1.34\,\overset{o}{\mathop{A}}\,\]. The C?C bond angle will be
A)
\[{{109}^{o}}28\]
done
clear
B)
\[{{100}^{o}}\]
done
clear
C)
\[{{180}^{o}}\]
done
clear
D)
\[{{120}^{o}}\]
done
clear
View Answer play_arrow
question_answer 53) Which will reduce zinc oxide to zinc?
A)
Mg
done
clear
B)
Pb
done
clear
C)
Cu
done
clear
D)
Fe
done
clear
View Answer play_arrow
question_answer 54) Some chemists at ISRO wished to prepare a saturated solution of a silver compound and they wanted it to have the highest concentration of silver ion possible. Which of the following compounds, would they use? \[{{K}_{sp}}(AgCl)=1.8\times {{10}^{-10}}\] \[{{K}_{sp}}(AgBr)=5.0\times {{10}^{-13}}\] \[{{K}_{sp}}(A{{g}_{2}}Cr{{O}_{4}})=2.4\times {{10}^{-12}}\]
A)
AgCl
done
clear
B)
AgBr
done
clear
C)
\[A{{g}_{2}}Cr{{O}_{4}}\]
done
clear
D)
Any of these
done
clear
View Answer play_arrow
question_answer 55) By Wurtz reaction, a mixture of methyl iodide and ethyl iodide gives
A)
butane
done
clear
B)
ethane
done
clear
C)
propane
done
clear
D)
a mixture of the above three
done
clear
View Answer play_arrow
question_answer 56) Addition of \[SnC{{l}_{2}}\] to \[HgC{{l}_{2}}\] gives precipitate
A)
white turning to red
done
clear
B)
white turning to grey
done
clear
C)
black turning to white
done
clear
D)
None of the above
done
clear
View Answer play_arrow
question_answer 57) In fermentation by zymase, alcohol and \[C{{O}_{2}}\]are obtained from
A)
invert sugar
done
clear
B)
glucose
done
clear
C)
fructose
done
clear
D)
All of these
done
clear
View Answer play_arrow
question_answer 58) The stability of ferric ion is due to
A)
half filled \[f\]-orbitals
done
clear
B)
half filled d-orbitals
done
clear
C)
"completely filled \[f\]-orbitals
done
clear
D)
completely filled d-orbitals
done
clear
View Answer play_arrow
question_answer 59) Electron affinity is positive, when
A)
O changes into \[{{O}^{-}}\]
done
clear
B)
\[{{O}^{-}}\] changes into \[{{O}^{2-}}\]
done
clear
C)
O changes into \[{{O}^{+}}\]
done
clear
D)
electron affinity is always negative
done
clear
View Answer play_arrow
question_answer 60) lonisation potential for a noble gas is
A)
maximum in a period
done
clear
B)
minimum in a period
done
clear
C)
either minimum or maximum
done
clear
D)
constant
done
clear
View Answer play_arrow
question_answer 61) Ethyl amine on acetylation gives
A)
N-ethyl acetamide
done
clear
B)
acetamide
done
clear
C)
methyl acetamide
done
clear
D)
None of the above
done
clear
View Answer play_arrow
question_answer 62) Strongest oxidising agent among halogen is
A)
\[{{I}_{2}}\]
done
clear
B)
\[B{{r}_{2}}\]
done
clear
C)
\[C{{l}_{2}}\]
done
clear
D)
\[{{F}_{2}}\]
done
clear
View Answer play_arrow
question_answer 63) Which reagent can convert acetic acid into ethanol?
A)
Na + alcohol
done
clear
B)
\[LiAl{{H}_{4}}\] + ether
done
clear
C)
\[{{H}_{2}}\] + Pt
done
clear
D)
Sn + HCl
done
clear
View Answer play_arrow
question_answer 64) In presence of moisture, \[S{{O}_{2}}\] can
A)
act as oxidant
done
clear
B)
act as reductant
done
clear
C)
gain electron
done
clear
D)
not act as reductant
done
clear
View Answer play_arrow
question_answer 65) Acetals are
A)
ketones
done
clear
B)
diethers
done
clear
C)
aldehyde
done
clear
D)
hydroxy aldehydes
done
clear
View Answer play_arrow
question_answer 66) The principle involved in the classification of basic radicals, is
A)
common ion effect
done
clear
B)
solubility product
done
clear
C)
valency of radicals
done
clear
D)
strength of salt
done
clear
View Answer play_arrow
question_answer 67) Formation of diethyl ether from ethanol is based on a
A)
dehydration reaction
done
clear
B)
dehydrogenation reaction
done
clear
C)
hydrogenation reaction
done
clear
D)
homolytic fission reaction
done
clear
View Answer play_arrow
question_answer 68) Hypo-phosphorus acid, \[{{H}_{3}}P{{O}_{2}}\] is
A)
a monobasic acid
done
clear
B)
atribasicacid
done
clear
C)
a dibasic acid
done
clear
D)
not acidic at all
done
clear
View Answer play_arrow
question_answer 69) What is obtained when acetyl chlorids is heated with benzene in presence of anhydrous is \[AlC{{l}_{3}}\]?
A)
Acetyl benzoic acid
done
clear
B)
Anisol
done
clear
C)
Acetophenone
done
clear
D)
Chlorobenzene
done
clear
View Answer play_arrow
question_answer 70) Which gas is used in airated ware:
A)
\[C{{O}_{2}}\]
done
clear
B)
\[S{{O}_{2}}\]
done
clear
C)
CO
done
clear
D)
Water vapours
done
clear
View Answer play_arrow
question_answer 71) The refluxing of \[{{(C{{H}_{3}})}_{2}}NCO{{H}_{3}}\], with acid gives
A)
\[{{(C{{H}_{3}})}_{2}}NH+C{{H}_{3}}COOH\]
done
clear
B)
\[{{(C{{H}_{3}})}_{2}}NCOOH+C{{H}_{4}}\]
done
clear
C)
\[2C{{H}_{3}}OH+C{{H}_{3}}CON{{H}_{2}}\]
done
clear
D)
\[2C{{H}_{3}}N{{H}_{2}}+C{{H}_{3}}COOH\]
done
clear
View Answer play_arrow
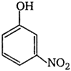
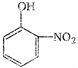
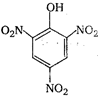
question_answer 72) Which is obtained on treating phenol with cone. \[HN{{O}_{3}}\]?
A)
done
clear
B)
done
clear
C)
done
clear
D)
None of the
done
clear
View Answer play_arrow
question_answer 73) Solder is an alloy of lead with
A)
copper
done
clear
B)
zinc
done
clear
C)
nickel
done
clear
D)
tin
done
clear
View Answer play_arrow
question_answer 74) Arrange \[NH_{4}^{+}{{H}_{2}}O,\,{{H}_{3}}{{O}^{+}}\], HF and \[O{{H}^{-}}\] in increasing order of acidic nature
A)
\[{{H}_{3}}{{O}^{+}}<NH_{4}^{+}<HF<O{{H}^{-}}<{{H}_{2}}O\]
done
clear
B)
\[NH_{4}^{+}<HF<{{H}_{3}}{{O}^{+}}<{{H}_{2}}O<O{{H}^{-}}\]
done
clear
C)
\[O{{H}^{-}}<{{H}_{2}}O<NH_{4}^{+}<HF<{{H}_{3}}{{O}^{+}}\]
done
clear
D)
\[{{H}_{3}}{{O}^{+}}>HF>{{H}_{2}}O>NH_{4}^{+}>O{{H}^{-}}\]
done
clear
View Answer play_arrow
question_answer 75) Which of the following radicals, gives the apple green flame during flame test?
A)
\[B{{a}^{2+}}\]
done
clear
B)
\[S{{r}^{2+}}\]
done
clear
C)
\[C{{a}^{2+}}\]
done
clear
D)
\[N{{a}^{+}}\]
done
clear
View Answer play_arrow
question_answer 76) When chlorine is passed through concentrated solution of KOH, the compound formed is
A)
\[KlC{{O}_{4}}\]
done
clear
B)
\[KlC{{O}_{3}}\]
done
clear
C)
\[KlC{{O}_{2}}\]
done
clear
D)
\[KlCO\]
done
clear
View Answer play_arrow
question_answer 77) The equilibrium constant Kp for the reaction, \[{{H}_{2}}(g)+{{I}_{2}}(g)2HI(g)\] is
A)
more than one
done
clear
B)
less than one
done
clear
C)
equal to \[{{K}_{c}}\]
done
clear
D)
zero
done
clear
View Answer play_arrow
question_answer 78) What is the weight of oxygen that is required for the complete combustion of 2.8 kg of ethylene?
A)
9.6kg
done
clear
B)
96.0kg
done
clear
C)
6.4kg
done
clear
D)
2.8kg
done
clear
View Answer play_arrow
question_answer 79) The metal that does not displace hydrogen from an acid is
A)
Ca
done
clear
B)
Al
done
clear
C)
Zn
done
clear
D)
Hg
done
clear
View Answer play_arrow
question_answer 80) The decomposition of \[{{N}_{2}}{{O}_{5}}\] occurs as, \[2{{N}_{2}}{{O}_{5}}\xrightarrow{{}}4N{{O}_{2}}+{{O}_{2}}\], and follows first order kinetics, hence
A)
the reaction is unimolecular
done
clear
B)
the reaction is bimolecular
done
clear
C)
\[{{t}_{1/2}}\propto {{a}^{0}}\]
done
clear
D)
None of the above
done
clear
View Answer play_arrow
question_answer 81) Atomic radii of F and Ne, in \[\overset{o}{\mathop{A}}\,\], are given by
A)
0.72, 0.71
done
clear
B)
0.72, 1.6
done
clear
C)
1.6, 1.58
done
clear
D)
0.71, 0.72
done
clear
View Answer play_arrow
question_answer 82) Which pair has both members from the same period of periodic table?
A)
Cl, Br
done
clear
B)
Ca, Cl
done
clear
C)
Na, Ca
done
clear
D)
Na, Cl
done
clear
View Answer play_arrow
question_answer 83) When dilute aqueous solution of \[AgN{{O}_{3}}\](excess) is added to KI solution, positively charged sol of \[AgI\] is formed due to adsorption of
A)
\[NO_{3}^{-}\]
done
clear
B)
\[O_{2}^{-}\]
done
clear
C)
\[A{{g}^{+}}\]
done
clear
D)
\[{{K}^{+}}\]
done
clear
View Answer play_arrow
question_answer 84) Which of the following has largest ionic radius?
A)
\[C{{s}^{+}}\]
done
clear
B)
\[L{{i}^{+}}\]
done
clear
C)
\[N{{a}^{+}}\]
done
clear
D)
\[{{K}^{+}}\]
done
clear
View Answer play_arrow
question_answer 85) For \[CaC{{O}_{3}}(s)CaO(s)+C{{O}_{2}}(g)\] at\[{{927}^{o}}C\], \[\Delta H=176\,kJ;\] then \[\Delta E\] is
A)
180 kJ
done
clear
B)
186.4 kJ
done
clear
C)
166.0 kJ
done
clear
D)
160 kJ
done
clear
View Answer play_arrow
question_answer 86) The charge required to liberate one gram equivalent of an element is
A)
96500 F
done
clear
B)
1 F
done
clear
C)
1C
done
clear
D)
None of these
done
clear
View Answer play_arrow
question_answer 87) The shape of sulphate ion is
A)
square planar
done
clear
B)
trigonal
done
clear
C)
trigonal planar
done
clear
D)
tetrahedral
done
clear
View Answer play_arrow
question_answer 88) The \[H-H\] bond energy is 430 kJ \[mo{{l}^{-1}}\] and \[Cl-Cl\] bond energy is 240 kJ \[mo{{l}^{-1}}\]. \[\Delta H\] for HCl is -90 k J. The H?Cl bond energy is about
A)
180 kJ \[mo{{l}^{-1}}\]
done
clear
B)
360 kJ \[mo{{l}^{-1}}\]
done
clear
C)
213 kJ \[mo{{l}^{-1}}\]
done
clear
D)
425 kJ \[mo{{l}^{-1}}\]
done
clear
View Answer play_arrow
question_answer 89) In the equation\[{{H}_{2}}S+2HN{{O}_{3}}\xrightarrow{{}}2{{H}_{2}}O+2N{{O}_{2}}+S\] The equivalent weight of hydrogen sulphide is
A)
18
done
clear
B)
68
done
clear
C)
34
done
clear
D)
17
done
clear
View Answer play_arrow
question_answer 90) The energy released in an atom bomb explosion is mainly due to
A)
release of neutrons
done
clear
B)
release of electrons
done
clear
C)
greater mass of products than initial material
done
clear
D)
lesser mass of products than initial material
done
clear
View Answer play_arrow
question_answer 91) Highest entropy is in
A)
hydrogen
done
clear
B)
water
done
clear
C)
graphite
done
clear
D)
mercury
done
clear
View Answer play_arrow
question_answer 92) Which one will liberate Br2 from KBr?
A)
\[{{I}_{2}}\]
done
clear
B)
\[S{{O}_{2}}\]
done
clear
C)
\[Hl\]
done
clear
D)
\[C{{l}_{2}}\]
done
clear
View Answer play_arrow
question_answer 93) Nuclides
A)
have specific atomic numbers
done
clear
B)
have same number of protons
done
clear
C)
have specific atomic number and mass numbers
done
clear
D)
are isotopes
done
clear
View Answer play_arrow
question_answer 94) Arrhenius equation, is
A)
\[\Delta H=\Delta E+\Delta {{n}_{g}}RT\]
done
clear
B)
\[\Delta G=\Delta H-T\,.\,\,\Delta S\]
done
clear
C)
\[K=A\,.\,{{e}^{-{{E}_{a}}/RT}}\]
done
clear
D)
None of the above
done
clear
View Answer play_arrow
question_answer 95) In which of the following compounds, the oxidation number of iodine is fractional?
A)
\[I{{F}_{3}}\]
done
clear
B)
\[I{{F}_{5}}\]
done
clear
C)
\[I_{3}^{-}\]
done
clear
D)
\[I{{F}_{7}}\]
done
clear
View Answer play_arrow
question_answer 96) Non-directional orbital is
A)
4 p
done
clear
B)
4 d
done
clear
C)
\[4f\]
done
clear
D)
3s
done
clear
View Answer play_arrow
question_answer 97) A monoprotic acid in 1.00 M solution is 0.01% ionised. The dissociation constant of this acid is
A)
\[1\times {{10}^{-8}}\]
done
clear
B)
\[1\times {{10}^{-4}}\]
done
clear
C)
\[1\times {{10}^{-6}}\]
done
clear
D)
\[1\times {{10}^{-5}}\]
done
clear
View Answer play_arrow
question_answer 98) If both oxygen and helium gases are at the same temperature, the rate of diffusion of \[{{O}_{2}}\] is very close to
A)
4 times that of He
done
clear
B)
2 times that of He
done
clear
C)
0.35 times that of He
done
clear
D)
8 times that of helium
done
clear
View Answer play_arrow
question_answer 99) A white substance having alkaline nature in solution is
A)
\[NaN{{O}_{3}}\]
done
clear
B)
\[N{{H}_{4}}Cl\]
done
clear
C)
\[N{{a}_{2}}C{{O}_{3}}\]
done
clear
D)
\[FeC{{l}_{3}}\]
done
clear
View Answer play_arrow
question_answer 100) A solution of \[FeC{{l}_{3}}\] in water acts as acidic solution due to
A)
hydrolysis of \[F{{e}^{3+}}\]
done
clear
B)
acidic impurities
done
clear
C)
dissociation
done
clear
D)
ionization
done
clear
View Answer play_arrow
question_answer 101) Which of the following is known as pond silk?
A)
Spirogyra
done
clear
B)
Ulothrix
done
clear
C)
Nostoc
done
clear
D)
Anabaena
done
clear
View Answer play_arrow
question_answer 102) Which of the following is incorrectly matched?
A)
Rabbit- Microlacithal, isolecithal
done
clear
B)
Frog- Mesolecithal, centrolecithal
done
clear
C)
Human- Mesolecithal, telolecithal
done
clear
D)
Birds- Macrolecithal, telolecithal
done
clear
View Answer play_arrow
question_answer 103) In a copulating pair of Pheretima posthuma which of the two processes take place?
A)
Internal fertilization and reciprocal fertilization
done
clear
B)
Cross fertilization and reciprocalfertilization
done
clear
C)
Reciprocal fertilization and internal fertilization
done
clear
D)
External fertilization and internal fertilization
done
clear
View Answer play_arrow
question_answer 104) A student sets up an experiment on photosynthesis as follows: He takes soda water in a glass tumbler and add chlorophyll extracts into the contents and keeps the tumbler exposed to sunlight hoping that he has provided necessary ingredients for photosynthesis to proceed ( \[viz,C{{O}_{2}},{{H}_{2}}O,\] chlorophyll and light). What do you think what will happen after, say, a few hours of exposure of light?
A)
Photosynthesis will take place and glucose will be produced
done
clear
B)
Photosynthesis will take place and starch will be produced, which will turn the mixture turbid
done
clear
C)
Photosynthesis will not take place because \[C{{O}_{2}}\] dissolved in soda water escapes into the atmosphere
done
clear
D)
Photosynthesis will not take place because intact chloroplasts are needed for the process
done
clear
View Answer play_arrow
question_answer 105) The pesticide most persistent in the soil is
A)
DDT
done
clear
B)
BHC
done
clear
C)
dieldrin
done
clear
D)
baygon
done
clear
View Answer play_arrow
question_answer 106) An antibody is a
A)
white corpuscle which attacks invading bacteria
done
clear
B)
molecule that specifically inactivates an antigen
done
clear
C)
component of the blood
done
clear
D)
secretion of mammalian erythrocyte
done
clear
View Answer play_arrow
question_answer 107) ECG is a measure of
A)
rate of heart beat
done
clear
B)
difference in electric potential
done
clear
C)
volume of blood pumped
done
clear
D)
ventricular contraction
done
clear
View Answer play_arrow
question_answer 108) Vitreous humour is
A)
colloid
done
clear
B)
watery fluid
done
clear
C)
mucoid connective tissue
done
clear
D)
All of the above
done
clear
View Answer play_arrow
question_answer 109) In respiration, largest amount of energy is produced in
A)
anaerobic respiration
done
clear
B)
Krebs cycle
done
clear
C)
glycolysis
done
clear
D)
None of the above
done
clear
View Answer play_arrow
question_answer 110) Torpedo is commonly known as
A)
sucker fish
done
clear
B)
electric ray
done
clear
C)
globe fish
done
clear
D)
sea horse
done
clear
View Answer play_arrow
question_answer 111) Foramen of Panizzae is found in the heart of
A)
rabbit
done
clear
B)
pigeon
done
clear
C)
crocodile
done
clear
D)
frog
done
clear
View Answer play_arrow
question_answer 112) Which one of the following pesticides is banned now-a-days?
A)
DDT
done
clear
B)
Eldrin
done
clear
C)
Aldrin
done
clear
D)
Toxaphene
done
clear
View Answer play_arrow
question_answer 113) Camassial teeth are modified for
A)
crushing
done
clear
B)
tearing
done
clear
C)
grinding
done
clear
D)
cutting
done
clear
View Answer play_arrow
question_answer 114) In the anther wall
A)
tapetum lies between endothecium an middle layers
done
clear
B)
tapetum lies just inner to endothecium
done
clear
C)
middle layer is between endothecium and tapetum
done
clear
D)
endothecium lies to middle layer
done
clear
View Answer play_arrow
question_answer 115) Insectivorous plants grow in a soil
A)
deficient in iron
done
clear
B)
rich in nitrogen
done
clear
C)
deficient in nitrogen
done
clear
D)
deficient in calcium
done
clear
View Answer play_arrow
question_answer 116) Aleurone grains are rich in
A)
fat
done
clear
B)
protein
done
clear
C)
carbohydrates
done
clear
D)
auxins
done
clear
View Answer play_arrow
question_answer 117) Meiosis can be observed in
A)
tapetal cells
done
clear
B)
megaspores
done
clear
C)
microspores
done
clear
D)
spore mother cells
done
clear
View Answer play_arrow
question_answer 118) Which of the following is not an intermediate in Krebs cycle?
A)
Acetic acid
done
clear
B)
Succinyl Coenzyme - A
done
clear
C)
Malic acid
done
clear
D)
Citric acid
done
clear
View Answer play_arrow
question_answer 119) The possible beneficial aspect of grazing animals is the
A)
removal of wild animals and pests
done
clear
B)
eradication of weeds
done
clear
C)
removal of wild plants
done
clear
D)
addition of their excreta into the soil
done
clear
View Answer play_arrow
question_answer 120) Carrier proteins are involved in
A)
transport of enzymes
done
clear
B)
water transport
done
clear
C)
active transport of ions
done
clear
D)
passive transport of ions
done
clear
View Answer play_arrow
question_answer 121) McClintock is related to
A)
plasmids
done
clear
B)
retroposons
done
clear
C)
transposons
done
clear
D)
none of these
done
clear
View Answer play_arrow
question_answer 122) Chloroplasts of Spirogyra have
A)
spiral margin
done
clear
B)
smooth or waxy margin
done
clear
C)
smooth margin
done
clear
D)
None of the above
done
clear
View Answer play_arrow
question_answer 123) Vascular bundles, in which phloem is found on both sides of xylem are called
A)
collateral
done
clear
B)
bicollateral
done
clear
C)
radial
done
clear
D)
amphicribral
done
clear
View Answer play_arrow
question_answer 124) Aerenchyma is helpful in plants by
A)
providing buoyancy in hydrophytes
done
clear
B)
absorption in stilt roots
done
clear
C)
giving mechanical strength to plant
done
clear
D)
giving flexibility to plants
done
clear
View Answer play_arrow
question_answer 125) Who proposed the "Cohesion Theory" of ascent sap?
A)
Strasburger
done
clear
B)
Godlewski
done
clear
C)
Western
done
clear
D)
Dixon and Jolley
done
clear
View Answer play_arrow
question_answer 126) A child has single kidney since birth. This variation is
A)
hybridization
done
clear
B)
negative meristic
done
clear
C)
blastogenic
done
clear
D)
substantive
done
clear
View Answer play_arrow
question_answer 127) Why is CO poisonous for man?
A)
CO affects the nerves of the lungs
done
clear
B)
CO affects the diaphragm and intercoastal muscles
done
clear
C)
CO reacts with oxygen reducing percentage of \[{{O}_{2}}\]in air
done
clear
D)
Haemoglobin combines with CO instead and the product cannot dissociate
done
clear
View Answer play_arrow
question_answer 128) Duct of Bellini opens on
A)
ureter
done
clear
B)
renal papilla
done
clear
C)
duodenum
done
clear
D)
pelvis
done
clear
View Answer play_arrow
question_answer 129) Humus is essential for plant growth because
A)
it is rich in nutrients and increases the water holding capacity of soil
done
clear
B)
it increases aeration of soil
done
clear
C)
it increases porocity of soil
done
clear
D)
All of the above
done
clear
View Answer play_arrow
question_answer 130) Which of the following is viviparous?
A)
Running birds
done
clear
B)
Whales
done
clear
C)
Bats
done
clear
D)
Both (a) and (b)
done
clear
View Answer play_arrow
question_answer 131) In short homed cattle, genes for red (R) and white (r) coat colour occur. Cross between red (R R) and white (rr) produced (Rr) roan. This is an example of
A)
incomplete dominance
done
clear
B)
codominance
done
clear
C)
complementary genes
done
clear
D)
epistasis
done
clear
View Answer play_arrow
question_answer 132) Complex tissue includes
A)
collenchymas
done
clear
B)
apical meristems
done
clear
C)
conducting tissue
done
clear
D)
idioblast
done
clear
View Answer play_arrow
question_answer 133) Turpentine oil is obtained from
A)
Pinus longifolia
done
clear
B)
Melia azadirachta
done
clear
C)
Eucalyptus
done
clear
D)
All of the above
done
clear
View Answer play_arrow
question_answer 134) The sclerenchyma of the hypodermis in the Pinus needle helps in
A)
increasing the absorptive surface of the cell
done
clear
B)
checking transpiration
done
clear
C)
mechanical support
done
clear
D)
photosynthesis
done
clear
View Answer play_arrow
question_answer 135) Sponges are
A)
pelagic
done
clear
B)
free - swimming
done
clear
C)
planktonic
done
clear
D)
sessile
done
clear
View Answer play_arrow
question_answer 136) Schistosoma is a parasite found in
A)
testes of frog
done
clear
B)
liver
done
clear
C)
intestine
done
clear
D)
blood
done
clear
View Answer play_arrow
question_answer 137) The recent model for plasma membrane proposed by Singer and Nicolson is
A)
molecular - lipid model
done
clear
B)
lamellar model
done
clear
C)
unit membrane model
done
clear
D)
fluid mosaic model
done
clear
View Answer play_arrow
question_answer 138) Cells obtained from cancerous tumours are known as
A)
hybridomas
done
clear
B)
myelomas
done
clear
C)
polyclonal cells
done
clear
D)
monoclonal cells
done
clear
View Answer play_arrow
question_answer 139) Significance of meiosis lies in
A)
reduction of chromosome number to one half
done
clear
B)
maintaining constancy of chromosome number during sexual reproduction
done
clear
C)
production of genetic variability
done
clear
D)
All of the above
done
clear
View Answer play_arrow
question_answer 140) Which is the connecting link between glycolysis and Krebs cycle?
A)
Acetyl Co - A
done
clear
B)
Pyruvic acid
done
clear
C)
Both (a) and (b)
done
clear
D)
None of the above
done
clear
View Answer play_arrow
question_answer 141) Mark A correct sequence
A)
Anthesis\[\xrightarrow[{}]{{}}\]Meiosis\[\xrightarrow[{}]{{}}\]Pollination \[\xrightarrow[{}]{{}}\] Syngamy
done
clear
B)
Pollination\[\xrightarrow[{}]{{}}\]Meiosis\[\xrightarrow[{}]{{}}\]Anthesi \[\xrightarrow[{}]{{}}\]Syngamy
done
clear
C)
Anthesis\[\xrightarrow[{}]{{}}\]Pollination\[\xrightarrow[{}]{{}}\]Meiosis \[\xrightarrow[{}]{{}}\] Syngamy
done
clear
D)
Anthesis\[\xrightarrow[{}]{{}}\]Meiosis\[\xrightarrow[{}]{{}}\]Syngamy \[\xrightarrow[{}]{{}}\]Pollination
done
clear
View Answer play_arrow
question_answer 142) The relationship between the alga Microcystis and the surrounding fauna corresponds to
A)
ammensalism
done
clear
B)
parasitism
done
clear
C)
predation
done
clear
D)
exploitation
done
clear
View Answer play_arrow
question_answer 143) Lipofusin granules are found in
A)
nerve cell
done
clear
B)
cardiac muscle
done
clear
C)
red muscle
done
clear
D)
cartilage
done
clear
View Answer play_arrow
question_answer 144) Camel in its hump stores
A)
water for emergency
done
clear
B)
fat for emergency
done
clear
C)
both fat and water for emergency
done
clear
D)
fat and proteins as reserve food for emergency
done
clear
View Answer play_arrow
question_answer 145) Arrangement of three successive bases in the genetic code signifies
A)
protein
done
clear
B)
nucleic acid
done
clear
C)
plasmids
done
clear
D)
amino acids
done
clear
View Answer play_arrow
question_answer 146) Potato is a native of
A)
Brazil
done
clear
B)
Peru
done
clear
C)
Panama
done
clear
D)
Mexico
done
clear
View Answer play_arrow
question_answer 147) The anthers in Solanaceae are
A)
monothecous, introse
done
clear
B)
dithecous, extrose
done
clear
C)
dithecous, introse
done
clear
D)
monothecous, extrose
done
clear
View Answer play_arrow
question_answer 148) In India there is decline in female population as compared to males, this is because of
A)
less female reach reproductive age
done
clear
B)
number of female children are born less as there is more chance of prenatal death in case of female child
done
clear
C)
female infanticide
done
clear
D)
All of the above Female infanticide is the route cause of decline in female population in India.
done
clear
View Answer play_arrow
question_answer 149) During embryonic development endoskeleton and muscle develop from which germinal layer?
A)
Ectoderm
done
clear
B)
Endoderm
done
clear
C)
Mesoderm
done
clear
D)
Blastopore
done
clear
View Answer play_arrow
question_answer 150) The semen fluid consists of sperm cells and secretion from
A)
seminal vesical, prostate gland, Cowpers gland
done
clear
B)
seminal vesicle, testes, uterus
done
clear
C)
seminal vesicle, prostate gland, testes
done
clear
D)
seminal vesicle, Cowpers gland and testes
done
clear
View Answer play_arrow
question_answer 151) The peculiar feature of Marchantia palmata is
A)
absence of gemma cup
done
clear
B)
presence of androgynous receptacles
done
clear
C)
absence of elaters
done
clear
D)
All of the above
done
clear
View Answer play_arrow
question_answer 152) Pneumatophores are positively
A)
geotropic
done
clear
B)
phototropic
done
clear
C)
aerotropic
done
clear
D)
rheotropic
done
clear
View Answer play_arrow
question_answer 153) Entry of pollen grain through micropyle is called
A)
porogamy
done
clear
B)
chalazogamy
done
clear
C)
allogamy
done
clear
D)
geitnogamy
done
clear
View Answer play_arrow
question_answer 154) A change in the relative abundance of an allele (the allelic frequency) within a population, over a succession of generations is called
A)
micro evolution
done
clear
B)
macro evolution
done
clear
C)
co - evolution
done
clear
D)
phylogenetic evolution
done
clear
View Answer play_arrow
question_answer 155) The early man whose skeleton is almost indistinguishable from that of modem man is
A)
Neanderthal man
done
clear
B)
Peking man
done
clear
C)
Homo erectus
done
clear
D)
Cro - magnon man
done
clear
View Answer play_arrow
question_answer 156) Which of these is based on magnetic resonance?
A)
EEG
done
clear
B)
CT scanning
done
clear
C)
NMR scanning
done
clear
D)
PET scanning
done
clear
View Answer play_arrow
question_answer 157) Broad spectrum antibiotic
A)
acts on all bacteria and virus
done
clear
B)
is effective in very small amount
done
clear
C)
acts on both pathogen and host
done
clear
D)
acts on a variety of pathogenic micro organisms
done
clear
View Answer play_arrow
question_answer 158) Tunnel vision is associated with
A)
alcoholism
done
clear
B)
smoking
done
clear
C)
drug addiction
done
clear
D)
epilepsy
done
clear
View Answer play_arrow
question_answer 159) In his bacteriophage experiments, Hershey and Chase demonstrated that DNA is genetic material in
A)
TMV
done
clear
B)
Escherichia coli
done
clear
C)
\[{{T}_{2}}\]bacteriophage
done
clear
D)
Diplococcus pneumoniae
done
clear
View Answer play_arrow
question_answer 160) Man whose fossils were found in Shivalik hills
A)
Ramapithecus
done
clear
B)
Pithecanthropus
done
clear
C)
Australopithecus
done
clear
D)
Sinanthropus
done
clear
View Answer play_arrow
question_answer 161) Which of the following is autotrophic?
A)
Virus
done
clear
B)
Mycoplasma
done
clear
C)
Nosroc
done
clear
D)
All of these
done
clear
View Answer play_arrow
question_answer 162) Germination of pollen grain on the stigma is
A)
in situ germination
done
clear
B)
in vitro germination
done
clear
C)
in vivo germination
done
clear
D)
autogamy
done
clear
View Answer play_arrow
question_answer 163) The function of tracheal hair is to pass
A)
mucus out
done
clear
B)
mucus in
done
clear
C)
air out
done
clear
D)
air in
done
clear
View Answer play_arrow
question_answer 164) In Echinodermata, tube feet are related with
A)
locomotion
done
clear
B)
excretory system
done
clear
C)
respiratory system
done
clear
D)
reproductive system
done
clear
View Answer play_arrow
question_answer 165) Autoradiography technique was used to
A)
trace the path of carbon in photosynthesis
done
clear
B)
establish that the oxygen evolved during photosynthesis is by photolysis of water molecule
done
clear
C)
study photorespiration in certain plants
done
clear
D)
find out the absorption maximum of chlorophyll pigments
done
clear
View Answer play_arrow
question_answer 166) Where did an epidemic bone softening disease itai- itai occurred first?
A)
South Korea
done
clear
B)
Japan
done
clear
C)
China
done
clear
D)
Burma
done
clear
View Answer play_arrow
question_answer 167) The trapping centre of light energy I photosystem- I is
A)
\[{{P}_{660}}\]
done
clear
B)
\[{{P}_{700}}\]
done
clear
C)
\[{{P}_{680}}\]
done
clear
D)
\[{{P}_{630}}\]
done
clear
View Answer play_arrow
question_answer 168) The membrane which allows passage of certain substances more readily than others is termed as
A)
permeable
done
clear
B)
selectively permeable
done
clear
C)
semipermeable
done
clear
D)
impermeable
done
clear
View Answer play_arrow
question_answer 169) Which combination of tissues acts together to provide the support to the hypocotyl of a seedling?
A)
Epidermis and collenchyma
done
clear
B)
Xylem and parenchyma
done
clear
C)
Epidermis and parenchyma
done
clear
D)
Xylem and phloem fibres
done
clear
View Answer play_arrow
question_answer 170) The UV radiation from sun cause reaction that produces
A)
carbon monoxide
done
clear
B)
sulphur dioxide
done
clear
C)
ozone
done
clear
D)
fluorides
done
clear
View Answer play_arrow
question_answer 171) Slow muscle fibres are found in
A)
eye
done
clear
B)
leg
done
clear
C)
stomach
done
clear
D)
heart
done
clear
View Answer play_arrow
question_answer 172) Muscle pump is
A)
beating of heart
done
clear
B)
squeezing effect of muscles upon veins running through them
done
clear
C)
peristaltic wave that travel along the alimentary canal
done
clear
D)
None of the above
done
clear
View Answer play_arrow
question_answer 173) The magnitude of root pressure ranges between
A)
2-5 atm
done
clear
B)
1-5 atm
done
clear
C)
0.1 - 0.2 atm
done
clear
D)
4-6 atm
done
clear
View Answer play_arrow
question_answer 174) Guttation is a consequence of high
A)
root pressure
done
clear
B)
transpiration pull
done
clear
C)
photosynthesis
done
clear
D)
respiration
done
clear
View Answer play_arrow
question_answer 175) Which of the following part in cows stomach is specialised for microbial digestion of plant material?
A)
Rumen
done
clear
B)
Reticulum
done
clear
C)
Abomassum
done
clear
D)
Both (a) and (b)
done
clear
View Answer play_arrow
question_answer 176) Which of the following is/are not an essential micro nutrient?
A)
Boron
done
clear
B)
Nickel and cadmium
done
clear
C)
Molybdenum
done
clear
D)
Zinc
done
clear
View Answer play_arrow
question_answer 177) Khaira disease of paddy is caused by
A)
Viral attack
done
clear
B)
Mn deficiency
done
clear
C)
MLO attack
done
clear
D)
Zn deficiency
done
clear
View Answer play_arrow
question_answer 178) The gliding joints are important for gliding movements. One example of such a joint is between the
A)
zygapophyses of adjacent vertebrae
done
clear
B)
humerus and glenoid cavity
done
clear
C)
occipital condyle and odontoid process
done
clear
D)
femur and tibio fibula
done
clear
View Answer play_arrow
question_answer 179) The term hormone was given by
A)
Starling for insulin
done
clear
B)
Starling for secretin
done
clear
C)
Byliss for insulin
done
clear
D)
Byliss for secretin
done
clear
View Answer play_arrow
question_answer 180) Cushings syndrome and myxoedema are associated with these glands respectively
A)
adrenal, thyroid
done
clear
B)
thyroid, adrenal
done
clear
C)
adrenal, pituitary
done
clear
D)
parathyroid, thyroid
done
clear
View Answer play_arrow
question_answer 181) Opening and closing of flowers represent a kind of
A)
nastic movement
done
clear
B)
tropic movement
done
clear
C)
mutation
done
clear
D)
autonomic movement
done
clear
View Answer play_arrow
question_answer 182) 2,4-D is a/an
A)
insecticide
done
clear
B)
weedicide
done
clear
C)
nematicide
done
clear
D)
rodenticide
done
clear
View Answer play_arrow
question_answer 183) What is the correct order of nitrogen assimilation?
A)
\[{{N}_{2~}}\to N{{O}_{2}}\to ~N{{O}_{3}}\to N{{H}_{2}}OH~\to N{{H}_{3}}\]
done
clear
B)
\[{{N}_{2~}}\to N{{O}_{3}}\to ~N{{O}_{2}}\to N{{H}_{2}}OH~\to N{{H}_{3}}\]
done
clear
C)
\[{{N}_{2~}}\to N{{O}_{2}}\to ~N{{O}_{3}}\to N{{H}_{3}}~\to N{{H}_{2}}OH\]
done
clear
D)
\[{{N}_{2~}}\to N{{O}_{3}}\to ~N{{O}_{2}}\to N{{H}_{3}}~\to N{{H}_{2}}OH\]
done
clear
View Answer play_arrow
question_answer 184) Hormone responsible for bolting is
A)
auxin
done
clear
B)
gibberellins
done
clear
C)
ethylene
done
clear
D)
cytokinin
done
clear
View Answer play_arrow
question_answer 185) Functional kidney of frog tadpole is
A)
archinephros
done
clear
B)
pronephros
done
clear
C)
mesonephros
done
clear
D)
metanephros
done
clear
View Answer play_arrow
question_answer 186) Loop of Henie is meant for absorption of
A)
potassium
done
clear
B)
glucose
done
clear
C)
water
done
clear
D)
\[C{{O}_{2}}\]
done
clear
View Answer play_arrow
question_answer 187) In dark adaptation
A)
only cones are involved
done
clear
B)
only rods are involved
done
clear
C)
both rods and cones are involved
done
clear
D)
neither rods nor cones are involved
done
clear
View Answer play_arrow
question_answer 188) Part of ear where sound is transduced, is
A)
tympanic membrane
done
clear
B)
malleus, incus and stapes
done
clear
C)
semicircular canal
done
clear
D)
cochlea
done
clear
View Answer play_arrow
question_answer 189) A microbial mutant requiring growth factor in addition to minimal medium is
A)
auxotroph
done
clear
B)
heterotroph
done
clear
C)
autotroph
done
clear
D)
syntroph
done
clear
View Answer play_arrow
question_answer 190) When a plant has different types of leaves, the condition is known as
A)
heterophilly
done
clear
B)
anisophilly
done
clear
C)
myremecophily
done
clear
D)
None of the above
done
clear
View Answer play_arrow
question_answer 191) In plants, glycolate metabolism takes place in
A)
low concentration of \[C{{O}_{2}}\]
done
clear
B)
high concentration of \[C{{O}_{2}}\]
done
clear
C)
low concentration of oxygen
done
clear
D)
absence of oxygen
done
clear
View Answer play_arrow
question_answer 192) Aristotles lantern is a characteristic of the following class of Echinodermata
A)
Echinoidea
done
clear
B)
Ophiuroidea
done
clear
C)
Holothuroidea
done
clear
D)
Asteroidea
done
clear
View Answer play_arrow
question_answer 193) The largest corpuscles in mammalian blood are
A)
basophils
done
clear
B)
erythrocytes
done
clear
C)
monocytes
done
clear
D)
lymphocytes
done
clear
View Answer play_arrow
question_answer 194) A fatty acid not synthesized in man is
A)
oleic
done
clear
B)
linoleic
done
clear
C)
palmitoleic
done
clear
D)
stearic
done
clear
View Answer play_arrow
question_answer 195) Which of the following cell organelles remake enveloped by a single unit membrane?
A)
Mitochondria
done
clear
B)
Lysosomes
done
clear
C)
Nucleus
done
clear
D)
Chloroplast
done
clear
View Answer play_arrow
question_answer 196) The promoter region in a lac operon is distinguishable into the following number -x segments
A)
three
done
clear
B)
four
done
clear
C)
two
done
clear
D)
five
done
clear
View Answer play_arrow
question_answer 197) Old stem of Combretum has
A)
inter and intra xylary phloem
done
clear
B)
inter and extra xylary phloem
done
clear
C)
intra and extra xylary phloem
done
clear
D)
All of the above
done
clear
View Answer play_arrow
question_answer 198) Prions are
A)
infectious nucleic add
done
clear
B)
infectious lipids
done
clear
C)
infectious proteins
done
clear
D)
infectious nucleoproteins
done
clear
View Answer play_arrow
question_answer 199) When a sea diver goes very deep, he has to breath on compressed air at high pressure After sometime, he loses his strength to work and feel drowsy. This is because of
A)
compressed air
done
clear
B)
more \[C{{O}_{2}}\]diffusing into molecules
done
clear
C)
more \[{{N}_{2}}\]diffusing in blood and body fats
done
clear
D)
nervous system does not work properly
done
clear
View Answer play_arrow
question_answer 200) Salivation in man is under the control of
A)
medulla oblongata
done
clear
B)
mesencephalon
done
clear
C)
hypothalamus
done
clear
D)
cerebellum
done
clear
View Answer play_arrow