question_answer 1) The length of a simple pendulum is about 100 cm known to have an accuracy of 1 mm. Its period of oscillation is 2 s determined by measuring the time for 100 oscillations using a clock of 0.1s resolution. What is the accuracy in the determined value of g?
A)
0.2%
done
clear
B)
0.5%
done
clear
C)
0.1%
done
clear
D)
2%
done
clear
View Answer play_arrow
question_answer 2) Young's modulus of the material of a wire is Y. On pulling the wire by a force F, the increase in its length is x. The potential energy of the stretched wire is:
A)
\[\frac{1}{2}Fx\]
done
clear
B)
\[\frac{1}{2}Yx\]
done
clear
C)
\[\frac{1}{2}F{{x}^{2}}\]
done
clear
D)
none of these
done
clear
View Answer play_arrow
question_answer 3) A charge situated at a certain distance along the axis of an electric dipole experience a force F. If the distance of the charge from the dipole is doubled, the force acting on it will become:
A)
2F
done
clear
B)
\[\frac{F}{2}\]
done
clear
C)
\[\frac{F}{4}\]
done
clear
D)
\[\frac{F}{8}\]
done
clear
View Answer play_arrow
question_answer 4) A coil in the shape of an equilateral triangle of side I is suspended between the pole pieces of a permanent magnet such that 8 is in plane of the coil. If due to a current i in the triangle a torque t acts on it, the side I of the triangle is:
A)
\[\frac{2}{\sqrt{3}}{{\left( \frac{\tau }{Bi} \right)}^{\frac{1}{2}}}\]
done
clear
B)
\[\frac{2}{3}\left( \frac{\tau }{Bi} \right)\]
done
clear
C)
\[2{{\left( \frac{\tau }{\sqrt{3}Bi} \right)}^{\frac{1}{2}}}\]
done
clear
D)
\[\frac{1}{\sqrt{3}}\frac{\tau }{Bi}\]
done
clear
View Answer play_arrow
question_answer 5) A certain electrical conductor has a square cross-section, 2.0 mm on a side and is 12 m long. The resistance between its ends is\[0.072\,\Omega \]. The resistivity of its material is equal to:
A)
\[2.4\times {{10}^{-6}}\Omega m\]
done
clear
B)
\[1.2\times {{10}^{-6}}\Omega m\]
done
clear
C)
\[1.2\times {{10}^{-8}}\Omega m\]
done
clear
D)
\[2.4\times {{10}^{-8}}\Omega m\]
done
clear
View Answer play_arrow
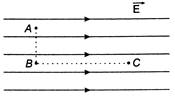
question_answer 6)
Figure shows three points A, B and C in a region of uniform electric field\[\overrightarrow{E}\].The line AB is perpendicular and BC is parallel to the field lines. Then which of the following holds good?
A)
\[{{V}_{A}}={{V}_{B}}={{V}_{C}}\]
done
clear
B)
\[{{V}_{A}}={{V}_{B}}>{{V}_{C}}\]
done
clear
C)
\[{{V}_{A}}={{V}_{B}}<{{V}_{C}}\]
done
clear
D)
\[{{V}_{A}}>{{V}_{B}}={{V}_{C}}\] Where\[{{V}_{A}},{{V}_{B}}\]and\[{{V}_{C}}\]represent the electric potentials at the points A, B and C respectively.
done
clear
View Answer play_arrow
question_answer 7) The\[(x,\text{ }y,\text{ }z)\]co-ordinates of two points A and B are given respectively as (0,3, -1) and (-2, 6, 4). The displacement vector form A to B may be given by:
A)
\[-2\hat{i}+6\hat{j}+4\hat{k}\]
done
clear
B)
\[-2\hat{i}+3\hat{j}+3\hat{k}\]
done
clear
C)
\[-2\hat{i}+3\hat{j}+5\hat{k}\]
done
clear
D)
\[2\hat{i}-3\hat{j}-5\hat{k}\]
done
clear
View Answer play_arrow
question_answer 8) In the first second of its flight, rocket ejects 1/60 of its mass with a velocity of\[2400\text{ }m{{s}^{-1}}\]. The acceleration of the rocket is:
A)
\[19.6m{{s}^{-2}}\]
done
clear
B)
\[30.2m{{s}^{-2}}\]
done
clear
C)
\[40\,m{{s}^{-2}}\]
done
clear
D)
\[49.8m{{s}^{-2}}\]
done
clear
View Answer play_arrow
question_answer 9) An ice cube is kept on an inclined plane of angle\[30{}^\circ \]. Coefficient of kinetic friction between block and incline plane is\[\frac{1}{\sqrt{3}}.\]. What is acceleration of block?
A)
Zero
done
clear
B)
\[2\text{ }m/{{s}^{2}}\]
done
clear
C)
\[1.5\text{ }m/{{s}^{2}}\]
done
clear
D)
5 m/s
done
clear
View Answer play_arrow
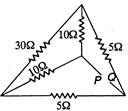
question_answer 10)
The equivalent resistance between the points P and Q in the network shown in the figure is given by:
A)
\[2.5\,\Omega \]
done
clear
B)
\[7.5\,\Omega \]
done
clear
C)
\[10\,\Omega \]
done
clear
D)
\[12.5\,\,\Omega \]
done
clear
View Answer play_arrow
question_answer 11) The magnetic field amplitude of an electromagnetic wave is\[2\times {{10}^{-7}}T\]. Ifs electric field amplitude if the wave is travelling in free space is:
A)
\[6\,V{{m}^{-1}}\]
done
clear
B)
\[60\text{ }V{{m}^{-1}}\]
done
clear
C)
\[10/6V{{m}^{-1}}\]
done
clear
D)
none of these
done
clear
View Answer play_arrow
question_answer 12) A cart is moving horizontally along a straight line with constant speed 30 m/s. A projectile is to be fired form the moving cart in such a way that it will return to the cart after the cart has moved 80 m. At what speed (relative to the cart) must the projectile be fired? (Take\[g=10\text{ }m/{{s}^{2}}\])
A)
10 m/s
done
clear
B)
\[10\,m/s\]
done
clear
C)
\[\frac{40}{3}m/s\]
done
clear
D)
None of these
done
clear
View Answer play_arrow
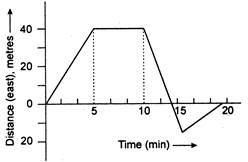
question_answer 13)
A boy begins to walk eastward along a street in front of his house and the graph of his displacement from home is shown in the following figure. His average for the whole time interval is equal to:
A)
8m/mm
done
clear
B)
6m/min
done
clear
C)
\[\frac{8}{3}m/\min \]
done
clear
D)
2 m/min
done
clear
View Answer play_arrow
question_answer 14)
What is the potential drop between points A and C in the following circuit?
A)
\[1.75\,V\]
done
clear
B)
\[2.25\,V\]
done
clear
C)
\[\frac{5}{4}\,V\]
done
clear
D)
\[\frac{4}{5}\,V\]
done
clear
View Answer play_arrow
question_answer 15) The escape velocity of a projectile on the earth's surface in\[11.2\text{ }km{{s}^{-1}}\]. A body is projected out with thrice this speed. The speed of the body far away form the earth will be:
A)
\[22.4\text{ }km{{s}^{-1}}\]
done
clear
B)
\[31.7\text{ }km\text{ }{{s}^{-1}}\]
done
clear
C)
\[33.6km{{s}^{-1}}\]
done
clear
D)
none of these
done
clear
View Answer play_arrow
question_answer 16) A body moves along a circular path of radius 10 m and the coefficient of friction is 0.5. What should be its angular speed in rad/s if it is not to slip from the surface? (\[g=9.8\text{ }m{{s}^{-2}}\])
A)
5
done
clear
B)
10
done
clear
C)
0.1
done
clear
D)
0.7
done
clear
View Answer play_arrow
question_answer 17) One end of a string of length (is connected to a particle of mass m and the other to a small peg on a smooth horizontal table. If the particle moves in a circle with speed v, the net force on the particle (directed towards the centre) is:
A)
\[T\]
done
clear
B)
\[T-\frac{m{{v}^{2}}}{l}\]
done
clear
C)
\[T+\frac{m{{v}^{2}}}{l}\]
done
clear
D)
zero T is the tension in the string
done
clear
View Answer play_arrow
question_answer 18) A body is initially at rest. It undergoes one-dimensional motion with constant acceleration. The power delivered to it at time t is proportional to:
A)
\[{{t}^{1/2}}\]
done
clear
B)
t
done
clear
C)
\[{{t}^{3/2}}\]
done
clear
D)
\[{{t}^{2}}\]
done
clear
View Answer play_arrow
question_answer 19) The displacement\[x\]of opal-tide varies with time t as\[x=a{{e}^{-\alpha t}}+b{{e}^{\beta t}}\]where a, b,\[\alpha \]and\[\beta \] are positive constants. The velocity of the particle will:
A)
go on decreasing with time
done
clear
B)
be independent of\[\alpha \]and\[\beta \]
done
clear
C)
drop to zero when\[\alpha =\beta \]
done
clear
D)
go on increasing with time
done
clear
View Answer play_arrow
question_answer 20) Two circular, similar, coaxial loops carry equal currents in the same direction. If the loops are brought nearer, what will happen?
A)
Current will increase in each loop
done
clear
B)
Current will decrease in each loop
done
clear
C)
Current will remain same in each loop
done
clear
D)
Current will increase in one and decrease in the other
done
clear
View Answer play_arrow
question_answer 21)
The figure below shows the plot of\[\frac{pV}{nT}\]versus P for oxygen gas at two different temperatures.
A)
(i) only
done
clear
B)
(i) and (ii) only
done
clear
C)
All of these
done
clear
D)
None of these
done
clear
View Answer play_arrow
question_answer 22) A Cannot engine has efficiency 1/5. Efficiency becomes 1/3 when temperature of sink is decreased by 50 K. What is the temperature of sink?
A)
325 K
done
clear
B)
375 K
done
clear
C)
300 K
done
clear
D)
350 K
done
clear
View Answer play_arrow
question_answer 23) A bar magnet has a coercively\[4\times {{10}^{3}}A{{m}^{-1}}\]. It is desired to demagnetize it by inserting it inside a solenoid 12 cm long and having 60 turns. The current carried by the solenoid should be:
A)
8 A
done
clear
B)
6 A
done
clear
C)
4.5 A
done
clear
D)
2 A
done
clear
View Answer play_arrow
question_answer 24) In a series LCR circuit the frequency of a 10 V, AC voltage source is adjusted in such a fashion that the reactance of the inductor measures \[15\,\Omega \]and that of the capacitor\[11\,\Omega \]. If\[R=3\,\Omega \]the potential difference across the series combination of Z, and C will be:
A)
8V
done
clear
B)
10 V
done
clear
C)
22V
done
clear
D)
52V
done
clear
View Answer play_arrow
question_answer 25) A circuit draws 330 W from a 110 V, 60 Hz AC line. The power factor is 0.6 and the current lags the voltage. The capacitance of a series capacitor that will result in a power factor of unity is equal to:
A)
\[31\text{ }\mu F\]
done
clear
B)
\[\text{54 }\mu F\]
done
clear
C)
\[\text{151 }\mu F\]
done
clear
D)
\[\text{201 }\mu F\]
done
clear
View Answer play_arrow
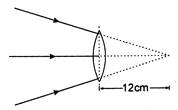
question_answer 26)
If the focal length of the lens is 20 cm, what is the distance of the image from the lens in the following figure?
A)
5.5cm
done
clear
B)
7.5cm
done
clear
C)
12.0 cm
done
clear
D)
20.0 cm
done
clear
View Answer play_arrow
question_answer 27) An open\[U-\]tube contains mercury. When 11.2 cm of water is poured into one of the arms of the tube, how high does the mercury rise in the other arm form its initial level?
A)
0.56 cm
done
clear
B)
1.35 cm
done
clear
C)
0.41 cm
done
clear
D)
2.32 cm
done
clear
View Answer play_arrow
question_answer 28) The change in the entropy of a 1 mole of an ideal gas which went through an isothermal process from an initial stat\[({{P}_{1}},{{V}_{1}},T)\]to the final state\[({{P}_{2}},{{V}_{2}},T)\]is equal to:
A)
zero
done
clear
B)
\[R\,In\,T\]
done
clear
C)
\[R\,In\,\frac{{{V}_{1}}}{{{V}_{2}}}\]
done
clear
D)
\[R\,In\,\frac{{{V}_{2}}}{{{V}_{1}}}\]
done
clear
View Answer play_arrow
question_answer 29) An un polarized beam of light is incident on a glass surface at an angle of incidence equal to the polarizing angle of the glass. Read the following statements: (i) The reflected beam is completely polarized. (ii) The refracted beam is partially polarized. (iii) The angle between the reflected and the refracted beams is\[90{}^\circ \]. Which of the above statements is/are true?
A)
(i) only
done
clear
B)
(ii)only
done
clear
C)
(i) and (iii)
done
clear
D)
All statements are correct
done
clear
View Answer play_arrow
question_answer 30) The threshold frequency for certain metal is\[3.3\times {{10}^{14}}Hz\]. If light of frequency\[8.2\times {{10}^{14}}\]Hz is incident on the metal, the cut-off voltage of the photoelectric current will be:
A)
4.9V
done
clear
B)
3.0V
done
clear
C)
2.0V
done
clear
D)
1.0V
done
clear
View Answer play_arrow
question_answer 31) Frequencies higher than 10 MHz were found not being reflected by the ionosphere on a particular day at a place. The maximum electron density of the ionosphere on the day was near to:
A)
\[1.5\times {{10}^{10}}{{m}^{-3}}\]
done
clear
B)
\[1.24\times {{10}^{12}}{{m}^{-3}}\]
done
clear
C)
\[3\times {{10}^{12}}{{m}^{-3}}\]
done
clear
D)
none of these
done
clear
View Answer play_arrow
question_answer 32) The de-Broglie wavelength of an electron, \[\alpha -\]particle and a proton all having the same kinetic energy is respectively given as\[{{\lambda }_{e}},{{\lambda }_{\alpha }}\] and\[{{\lambda }_{p}}\]. Then which of the following is not true?
A)
\[{{\lambda }_{e}}<{{\lambda }_{p}}\]
done
clear
B)
\[{{\lambda }_{p}}<{{\lambda }_{\alpha }}\]
done
clear
C)
\[{{\lambda }_{e}}<{{\lambda }_{\alpha }}\]
done
clear
D)
\[{{\lambda }_{\alpha }}<{{\lambda }_{p}}>{{\lambda }_{e}}\]
done
clear
View Answer play_arrow
question_answer 33) What is the disintegration constant of radon, if the number of its atoms diminishes by 18% in 24 h?
A)
\[2.1\times {{10}^{-3}}{{s}^{-1}}\]
done
clear
B)
\[2.1\times {{10}^{-4}}{{s}^{-1}}\]
done
clear
C)
\[2.1\times {{10}^{-5}}{{s}^{-1}}\]
done
clear
D)
\[2.1\times {{10}^{-6}}{{s}^{-1}}\]
done
clear
View Answer play_arrow
question_answer 34) Which of the following statements is true for an n-type semiconductor?
A)
The donor level lies closely below the bottom of the conduction band
done
clear
B)
The donor level lies closely above the top of the valence band
done
clear
C)
The donor level lies at the halfway mark of the forbidden energy gap
done
clear
D)
None of the above
done
clear
View Answer play_arrow
question_answer 35) Carbon, silicon and germanium have four valence electrons each. These are characterized valence and conduction bands separated by energy band gap respectively equal to\[{{(Eg)}_{C}},{{(Eg)}_{Si}}\]and\[{{(Eg)}_{Ge}}\]. Which of the following statements is true?
A)
\[{{(Eg)}_{C}}={{(Eg)}_{Si}}={{(Eg)}_{Ge}}\]
done
clear
B)
\[{{(Eg)}_{C}}>{{(Eg)}_{Si}}>{{(Eg)}_{Ge}}\]
done
clear
C)
\[{{(Eg)}_{C}}<{{(Eg)}_{Ge}}>{{(Eg)}_{Si}}\]
done
clear
D)
\[{{(Eg)}_{Si}}<{{(Eg)}_{Ge}}>{{(Eg)}_{C}}\]
done
clear
View Answer play_arrow
question_answer 36) A particle executes SHM of amplitude 25 cm and time period 3 s. What is the minimum time required for the particle to move between two points 12.5 cm on either side of the mean position?
A)
0.5s
done
clear
B)
1.0s
done
clear
C)
1.5s
done
clear
D)
2.0s
done
clear
View Answer play_arrow
question_answer 37) The speed of a wave on a string is 150 m/s when the tension is 120 N. The percentage increase in the tension in order to raise the wave speed by 20% is:
A)
44%
done
clear
B)
40%
done
clear
C)
20%
done
clear
D)
10%
done
clear
View Answer play_arrow
question_answer 38) A straight rod length L has one of its ends at the origin and the other at\[x=L\]. If the mass per unit length of the rod is given by Ay where A is constant, where is its mass centre?
A)
\[L/3\]
done
clear
B)
\[L/2\]
done
clear
C)
\[2L/3\]
done
clear
D)
\[3L/4\]
done
clear
View Answer play_arrow
question_answer 39) The image of a small electric bulb fixed on the wall of a room is to be obtained on the opposite wall 4 m away by means of a large convex lens. The maximum possible focal length of the lens required for this purpose will be:
A)
0.5m
done
clear
B)
1.0m
done
clear
C)
1.5 m
done
clear
D)
2.0 m
done
clear
View Answer play_arrow
question_answer 40) The total energy of a satellite moving with an orbital velocity v around the earth is:
A)
\[\frac{1}{2}m{{v}^{2}}\]
done
clear
B)
\[\frac{-1}{2}m{{v}^{2}}\]
done
clear
C)
\[m{{v}^{2}}\]
done
clear
D)
\[\frac{3}{2}m{{v}^{2}}\]
done
clear
View Answer play_arrow
question_answer 41) In hydrogen atom, the electron is moving round the nucleus with velocity\[2.18\times {{10}^{6}}\] m/s in an orbit of radius \[0.528\overset{\text{o}}{\mathop{\text{A}}}\,\]. The acceleration of the electron is:
A)
\[9\times {{10}^{18}}m/{{s}^{2}}\]
done
clear
B)
\[9\times {{10}^{22}}m/{{s}^{2}}\]
done
clear
C)
\[9\times {{10}^{-22}}m/{{s}^{2}}\]
done
clear
D)
\[9\times {{10}^{-12}}m/{{s}^{2}}\]
done
clear
View Answer play_arrow
question_answer 42) When a spring is stretched by a distance\[x,\]it exerts a force given by \[F=(-5x-16{{x}^{3}})N\] The work done, when the spring is stretched from 0.1 m to 0.2 m is:
A)
\[8.7\times {{10}^{-2}}J\]
done
clear
B)
\[12.2\times {{10}^{-2}}J\]
done
clear
C)
\[8.1\times {{10}^{-1}}J\]
done
clear
D)
\[12.2\times {{10}^{-1}}J\]
done
clear
View Answer play_arrow
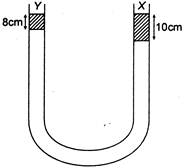
question_answer 43)
A liquid\[X\]of density\[3.36g/c{{m}^{2}}\]is poured in a \[U-\]tube, which contains Hg. Another liquid Y is poured in left arm with height 8 cm, upper levels of\[X\]and Y are same. What is density of Y?
A)
0.8g/cc
done
clear
B)
1.2g/cc
done
clear
C)
1.4 g/cc
done
clear
D)
1.6 g/cc
done
clear
View Answer play_arrow
question_answer 44) If boiling point of water is\[95{}^\circ F,\]what will be reduction at Celsius scale?
A)
\[7{}^\circ C\]
done
clear
B)
\[65{}^\circ C\]
done
clear
C)
\[63{}^\circ \text{ }C\]
done
clear
D)
\[35{}^\circ C\]
done
clear
View Answer play_arrow
question_answer 45) The motion of a particle varies with time according to the relation\[y=a(\sin \omega t+\cos \omega t)\]
A)
the motion is oscillatory but not SHM
done
clear
B)
the motion is SHM with amplitude a
done
clear
C)
the motion is SHM with amplitude\[a\sqrt{2}\]
done
clear
D)
the motion is SHM with amplitude 2a
done
clear
View Answer play_arrow
question_answer 46) Two closed organ pipes A and B, have the same length. A is wider than B. They resonate in the fundamental mode at frequencies n^ and rig respectively, then:
A)
\[{{n}_{A}}={{n}_{B}}\]
done
clear
B)
\[{{n}_{A}}>{{n}_{B}}\]
done
clear
C)
\[{{n}_{A}}<{{n}_{B}}\]
done
clear
D)
either or depending on the ratio of their diameters
done
clear
View Answer play_arrow
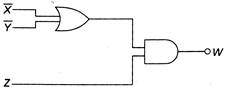
question_answer 47)
Output W is given by:
A)
\[(X+Y)Z\]
done
clear
B)
\[(\overline{X}+\overline{Y})Z\]
done
clear
C)
\[\overline{X}.\overline{Y}+\overline{Z}\]
done
clear
D)
\[(\overline{X}.\overline{Y})Z\]
done
clear
View Answer play_arrow
question_answer 48) In double slit experiment, the distance between two slits is 0.6 mm and these are illumunated with light of wavelength \[4800\overset{\text{o}}{\mathop{\text{A}}}\,\]. The angular width of first dark fringe on the screen distant 120 cm from slits will be:
A)
\[8\times {{10}^{-4}}rad\]
done
clear
B)
\[6\times {{10}^{-4}}rad\]
done
clear
C)
\[4\times {{10}^{-4}}rad\]
done
clear
D)
\[16\times {{10}^{-4}}rad\]
done
clear
View Answer play_arrow
question_answer 49) If there were no atmosphere, the average temperature on the surface of the earth would be:
A)
lower
done
clear
B)
higher
done
clear
C)
same as now
done
clear
D)
\[0{}^\circ C\]
done
clear
View Answer play_arrow
question_answer 50) The ionization energy of 10 times ionized sodium atom is:
A)
\[\frac{13.6}{11}eV\]
done
clear
B)
\[\frac{13.6}{112}eV\]
done
clear
C)
\[13.6\times {{(11)}^{2}}\,eV\]
done
clear
D)
\[13.6\text{ }eV\]
done
clear
View Answer play_arrow
question_answer 51) Freon used as refrigerant is:
A)
\[C{{F}_{2}}=C{{F}_{2}}\]
done
clear
B)
\[C{{H}_{2}}{{F}_{2}}\]
done
clear
C)
\[CC{{l}_{2}}{{F}_{2}}\]
done
clear
D)
\[C{{F}_{4}}\]
done
clear
View Answer play_arrow
question_answer 52) Formic acid can be distinguished from acetic acid by:
A)
\[NaHC{{O}_{3}}\]
done
clear
B)
Tollen's reagent
done
clear
C)
\[NaOH\]
done
clear
D)
none of these
done
clear
View Answer play_arrow
question_answer 53) Amongst the trihalides of nitrogen which one is the least basic?
A)
\[N{{F}_{3}}\]
done
clear
B)
\[NC{{l}_{3}}\]
done
clear
C)
\[NB{{r}_{3}}\]
done
clear
D)
\[N{{I}_{3}}\]
done
clear
View Answer play_arrow
question_answer 54) Aldol condensation is not given by:
A)
\[C{{H}_{3}}CHO\]
done
clear
B)
\[HCHO\]
done
clear
C)
\[C{{H}_{3}}C{{H}_{2}}CHO\]
done
clear
D)
\[C{{H}_{3}}COC{{H}_{3}}\]
done
clear
View Answer play_arrow
question_answer 55) Which pair is of isoelectronic species?
A)
\[{{K}^{+}},C{{l}^{-}},N{{a}^{+}}\]
done
clear
B)
\[{{K}^{+}},C{{l}^{-}},C{{a}^{2+}}\]
done
clear
C)
\[{{F}^{-}},A{{l}^{3+}},{{K}^{+}}\]
done
clear
D)
\[F{{e}^{2+}},C{{u}^{2+}},{{V}^{3+}}\]
done
clear
View Answer play_arrow
question_answer 56) Glycerol on heating with\[KHS{{O}_{4}}\]gives:
A)
acrolein
done
clear
B)
formic acid
done
clear
C)
oxalic acid
done
clear
D)
allyl alcohol
done
clear
View Answer play_arrow
question_answer 57) Acetaldehyde reacts with chlorine to give:
A)
\[CC{{l}_{4}}\]
done
clear
B)
\[CHC{{l}_{3}}\]
done
clear
C)
\[CC{{l}_{3}}.COC{{H}_{3}}\]
done
clear
D)
\[CC{{l}_{3}}.CHO\]
done
clear
View Answer play_arrow
question_answer 58) Hydrolysis of cyanogen gives:
A)
oxalic acid +\[N{{H}_{3}}\]
done
clear
B)
oxalic acid
done
clear
C)
\[N{{H}_{3}}\]
done
clear
D)
none of these
done
clear
View Answer play_arrow
question_answer 59) Which of the following compound shows optical isomerism?
A)
\[C{{H}_{3}}-\underset{\begin{smallmatrix} | \\ Cl \end{smallmatrix}}{\overset{\begin{smallmatrix} C{{H}_{3}} \\ | \end{smallmatrix}}{\mathop{C}}}\,-COOH\]
done
clear
B)
\[C{{H}_{3}}-\underset{\begin{smallmatrix} | \\ H \end{smallmatrix}}{\overset{\begin{smallmatrix} H \\ | \end{smallmatrix}}{\mathop{C}}}\,-COOH\]
done
clear
C)
\[C{{H}_{3}}-\underset{\begin{smallmatrix} | \\ OH \end{smallmatrix}}{\overset{\begin{smallmatrix} H \\ | \end{smallmatrix}}{\mathop{C}}}\,-COOH\]
done
clear
D)
\[C{{H}_{3}}C{{H}_{2}}C{{H}_{2}}C{{H}_{2}}OH\]
done
clear
View Answer play_arrow
question_answer 60) Number of primary alcohols of the formula. \[{{C}_{5}}{{H}_{12}}O\]are:
A)
1
done
clear
B)
4
done
clear
C)
5
done
clear
D)
3
done
clear
View Answer play_arrow
question_answer 61) Which enzyme converts sucrose into ethanol?
A)
Diastase
done
clear
B)
Invertase
done
clear
C)
Zymase
done
clear
D)
Both and
done
clear
View Answer play_arrow
question_answer 62) Which acts as auto catalyst during titration of \[KMn{{O}_{4}}\]and oxalic acid in presence\[{{H}_{2}}S{{O}_{4}}\]?
A)
\[{{H}_{2}}S{{O}_{4}}\]
done
clear
B)
\[KMnS{{O}_{4}}\]
done
clear
C)
Oxalic acid
done
clear
D)
\[MnS{{O}_{4}}\]
done
clear
View Answer play_arrow
question_answer 63) \[C{{H}_{3}}\underset{\begin{smallmatrix} | \\ C{{H}_{3}} \end{smallmatrix}}{\overset{\begin{smallmatrix} C{{H}_{3}} \\ | \end{smallmatrix}}{\mathop{C}}}\,CHO\]shows Cannizaro's reaction due to:
A)
carbon is bounded by 3 methyl groups
done
clear
B)
absence of a-hydrogen atom
done
clear
C)
due to steric effect
done
clear
D)
none of the above
done
clear
View Answer play_arrow
question_answer 64) When primary amines react with nitrous acid, the gas evolved is:
A)
\[{{N}_{2}}\]
done
clear
B)
\[N{{H}_{3}}\]
done
clear
C)
\[C{{O}_{2}}\]
done
clear
D)
\[CO\]
done
clear
View Answer play_arrow
question_answer 65) Which is a natural polymer?
A)
Protein
done
clear
B)
PVC
done
clear
C)
PMMA
done
clear
D)
SBR
done
clear
View Answer play_arrow
question_answer 66) "The greater the charge on an ion, the greater its coagulating power" is a statement of:
A)
Tyndall?s effect
done
clear
B)
Faraday?s law
done
clear
C)
Mosley?s law
done
clear
D)
Hardy-SchuIze law
done
clear
View Answer play_arrow
question_answer 67) When ice melts into water, entropy:
A)
becomes zero
done
clear
B)
decreases
done
clear
C)
increases
done
clear
D)
remain same
done
clear
View Answer play_arrow
question_answer 68) If the solubility of\[Pb{{I}_{2}}\]in its saturated solutions at\[25{}^\circ C\]be\[2\times {{10}^{-3}}\] mol/L. Its solubility product is:
A)
\[4\times {{10}^{-9}}\]
done
clear
B)
\[8\times {{10}^{-12}}\]
done
clear
C)
\[6\times {{10}^{-9}}\]
done
clear
D)
\[32\times {{10}^{-9}}\]
done
clear
View Answer play_arrow
question_answer 69) The ionisation potential of nitrogen is greater than oxygen due to:
A)
half-filled 2p-subshell
done
clear
B)
more electropositive
done
clear
C)
less electropositive
done
clear
D)
none of the above
done
clear
View Answer play_arrow
question_answer 70) A radioactive substance decays to,\[\frac{3}{4}\]th of its original value in 30 min. The half-life of the substance is:
A)
60 min
done
clear
B)
5 min
done
clear
C)
7.5mm
done
clear
D)
15 min
done
clear
View Answer play_arrow
question_answer 71) \[I-131\]is used for the treatment of:
A)
thyroid disorders
done
clear
B)
skin disorders
done
clear
C)
brain tumour
done
clear
D)
kidney stones
done
clear
View Answer play_arrow
question_answer 72) Cinnabar is:
A)
\[HgS\]
done
clear
B)
\[PbS\]
done
clear
C)
\[Sn{{O}_{2}}\]
done
clear
D)
\[PbC{{O}_{3}}\]
done
clear
View Answer play_arrow
question_answer 73) Which of the following has least bond angle?
A)
\[{{H}_{2}}O\]
done
clear
B)
\[{{H}_{2}}S\]
done
clear
C)
\[{{H}_{2}}Se\]
done
clear
D)
\[{{H}_{2}}Te\]
done
clear
View Answer play_arrow
question_answer 74) Which molecule is linear in shape?
A)
\[N{{H}_{3}}\]
done
clear
B)
\[COa\]
done
clear
C)
\[B{{F}_{3}}\]
done
clear
D)
\[{{H}_{2}}O\]
done
clear
View Answer play_arrow
question_answer 75) \[CaC{{N}_{2}}+C\]is called as:
A)
urea
done
clear
B)
thomas slag
done
clear
C)
nitrolim
done
clear
D)
triple superphosphate
done
clear
View Answer play_arrow
question_answer 76) Alcoholic solution of KOH is used for:
A)
dehydration
done
clear
B)
dehydrogenation
done
clear
C)
dehydrohalogenation
done
clear
D)
dehalogenation
done
clear
View Answer play_arrow
question_answer 77) \[{{[EDTA]}^{4-}}\]is a:
A)
monodentate ligand
done
clear
B)
bidentate ligand
done
clear
C)
quadridentate ligand
done
clear
D)
hexadentate ligand
done
clear
View Answer play_arrow
question_answer 78) 2.76g of silver carbonate on being strongly heated yield a residue weighing:
A)
2.16 g
done
clear
B)
2.48 g
done
clear
C)
2.64 g
done
clear
D)
2.32 g
done
clear
View Answer play_arrow
question_answer 79) A cylinder of gas supplied by Bharat petroleum is assumed to contain 14 kg of butane. It is normal family requires 20,000 kJ of energy per day for cooking, butane gas in the cylinder last for......... days. (\[\Delta H,\]of \[{{C}_{4}}{{H}_{10}}=-2658\text{ }kJ\]per mol)
A)
15 days
done
clear
B)
20 days
done
clear
C)
32 days
done
clear
D)
40 days
done
clear
View Answer play_arrow
question_answer 80) After losing a number of\[\alpha \]and \[\beta \]-particles \[_{92}{{U}^{238}}\]is changed to 32 Pb206. The total number of\[\alpha \]-particles lost in this process is:
A)
10
done
clear
B)
5
done
clear
C)
8
done
clear
D)
32
done
clear
View Answer play_arrow
question_answer 81) Which type of bond is present in\[{{H}_{2}}S\]molecule?
A)
Ionic bond
done
clear
B)
Covalent bond
done
clear
C)
Co-ordinate bond
done
clear
D)
All of the above
done
clear
View Answer play_arrow
question_answer 82) Anon-ideal solution was prepared by mixing 30 mL chloroform and 50 mL acetone. The volume of mixture will be:
A)
> 80 mL
done
clear
B)
< 80 mL
done
clear
C)
= 80 mL
done
clear
D)
> 80 mL
done
clear
View Answer play_arrow
question_answer 83) For the reaction\[A+B\xrightarrow[{}]{{}}C,\]it found that doubling the concentration of A increases the rate by 4 times, and doubling the concentration of B doubles the reaction rate. What is the overall order of the reaction?
A)
4
done
clear
B)
3/2
done
clear
C)
3
done
clear
D)
1
done
clear
View Answer play_arrow
question_answer 84) Na and Mg crystallize in bcc and fee type crystals respectively, then the number of atoms of Na and Mg present in the unit cell of their respective crystal is:
A)
4 and 2
done
clear
B)
9 and 14
done
clear
C)
14 and 9
done
clear
D)
2 and 4
done
clear
View Answer play_arrow
question_answer 85) The pH of 0.1M solution of the following salts increases in the order:
A)
\[NaCl<N{{H}_{4}}Cl<NaCN<HCl\]
done
clear
B)
\[HCl<N{{H}_{4}}Cl<NaCl<NaCN\]
done
clear
C)
\[NaCN<N{{H}_{4}}Cl<NaCl<HCl\]
done
clear
D)
\[HCl<NaCl<NaCN<N{{H}_{4}}Cl\]
done
clear
View Answer play_arrow
question_answer 86) The compound in which cation is isoelectronic with anion is:
A)
\[NaCl\]
done
clear
B)
\[CsF\]
done
clear
C)
\[NaI\]
done
clear
D)
\[{{K}_{2}}S\]
done
clear
View Answer play_arrow
question_answer 87) Which process of purification is represented by the following scheme? \[\underset{impure}{\mathop{Ti}}\,+2{{I}_{2}}\xrightarrow[{}]{{{250}^{o}}C}Ti{{I}_{4}}\xrightarrow[{}]{{{1400}^{o}}C}\underset{pure}{\mathop{Ti}}\,+{{I}_{2}}\]
A)
Cupellation
done
clear
B)
Poling
done
clear
C)
Electrolytic refining
done
clear
D)
Van-Arkel process
done
clear
View Answer play_arrow
question_answer 88) Bredig arc method cannot be used to prepare colloidal solution of which of the following?
A)
Pt
done
clear
B)
Fe
done
clear
C)
Ag
done
clear
D)
Au
done
clear
View Answer play_arrow
question_answer 89) In alkaline medium\[Cl{{O}_{2}}\]oxidize\[{{H}_{2}}{{O}_{2}}\]in\[{{O}_{2}}\]and reduced itself in\[C{{l}^{-}}\]then how many moles of\[{{H}_{2}}{{O}_{2}}\]will oxidize by one mole of\[Cl{{O}_{2}}\]?
A)
1.0
done
clear
B)
1.5
done
clear
C)
2.5
done
clear
D)
3.5
done
clear
View Answer play_arrow
question_answer 90) For the electrochemical cell,\[M|{{M}^{+}}||{{X}^{-}}|X,{{E}^{o}}({{M}^{+}}/M)=0.44\,V\]and \[E{}^\circ (X/{{X}^{-}})=0.33\text{ }V\]. From this data one can deduce that:
A)
\[M+X\to {{M}^{+}}+{{X}^{-}}\]is the spontaneous reaction
done
clear
B)
\[{{M}^{+}}+{{X}^{-}}\to M+X\]is the spontaneous reaction
done
clear
C)
\[{{E}_{cell}}=0.77V\]
done
clear
D)
\[{{E}_{cell}}=-0.77V\]
done
clear
View Answer play_arrow
question_answer 91) Potassium is kept in:
A)
alcohol
done
clear
B)
water
done
clear
C)
kerosene
done
clear
D)
liquid ammonia
done
clear
View Answer play_arrow
question_answer 92) Which of the following is not obtained by direct reaction of constituent elements?
A)
\[Xe{{F}_{2}}\]
done
clear
B)
\[Xe{{F}_{4}}\]
done
clear
C)
\[Xe{{O}_{3}}\]
done
clear
D)
\[XeFe\]
done
clear
View Answer play_arrow
question_answer 93) The reagent which forms crystalline osazone derivative when reacted with glucose is:
A)
Fehling solution
done
clear
B)
phenyl hydrazine
done
clear
C)
Benedict solution
done
clear
D)
hydroxylamine
done
clear
View Answer play_arrow
question_answer 94) The volume of a gas decreases from 500 cc to 300 cc when a sample gas is compressed by an average pressure of 0.6 atm. During this process 10 J of heat is liberated. The change in internal energy is:
A)
\[-2.16J\]
done
clear
B)
\[12.156\text{ }J\]
done
clear
C)
\[2.16J\]
done
clear
D)
\[101.3\text{ }J\]
done
clear
View Answer play_arrow
question_answer 95) Benzaldehyde condenses with N, N-dimethyla- niline in presence of anhydrous \[ZnC{{l}_{2}}\]to give:
A)
Michler?s ketone
done
clear
B)
azo-dye
done
clear
C)
malachite green
done
clear
D)
buffer yellow
done
clear
View Answer play_arrow
question_answer 96) Which of the following cannot be used in Friedel-Craft?s reaction?
A)
\[FeC{{l}_{3}}\]
done
clear
B)
\[FeB{{r}_{2}}\]
done
clear
C)
\[AlC{{l}_{3}}\]
done
clear
D)
\[NaCl\]
done
clear
View Answer play_arrow
question_answer 97) A gaseous hypothetical chemical equation\[2A4B+C\]is carried out in a closed vessel. The concentration of B is found to increase by\[5\times {{10}^{-3}}mol\text{ }{{L}^{-1}}\]in 10 s. The rate of appearance of B is:
A)
\[5\times {{10}^{-4}}mol\text{ }{{L}^{-1}}{{s}^{-1}}\]
done
clear
B)
\[5\times {{10}^{-5}}mol\,{{L}^{-1}}{{s}^{-1}}\]
done
clear
C)
\[6\times {{10}^{-5}}mol\,{{L}^{-1}}{{s}^{-1}}\]
done
clear
D)
\[4\times {{10}^{-4}}mol\,{{L}^{-1}}{{s}^{-1}}\]
done
clear
View Answer play_arrow
question_answer 98) Which of the following colligative properties can provide molar mass of proteins (or polymers or colloids) with greater precision?
A)
Relative lowering of vapour pressure
done
clear
B)
Elevation in boiling point
done
clear
C)
Depression in freezing point
done
clear
D)
Osmotic pressure
done
clear
View Answer play_arrow
question_answer 99) In compound X, all the bond angles are exactly\[{{109}^{o}}28,X\]is:
A)
chloromethane
done
clear
B)
iodoform
done
clear
C)
carbon tetrachloride
done
clear
D)
chloroform
done
clear
View Answer play_arrow
question_answer 100) Glass is:
A)
super cooled liquid
done
clear
B)
crystalline solid
done
clear
C)
amorphous solid
done
clear
D)
liquid crystal
done
clear
View Answer play_arrow
question_answer 101) In malaria, the product released by mosquito into blood that causes chill and fever, is called :
A)
heamatin
done
clear
B)
Schuffner's dots
done
clear
C)
haemozoin
done
clear
D)
haemotoxin
done
clear
View Answer play_arrow
question_answer 102) In Amoeba, the contractile vacuole is present:
A)
near the trailing end
done
clear
B)
near the advancing end
done
clear
C)
at the middle of the body
done
clear
D)
any where inside the body
done
clear
View Answer play_arrow
question_answer 103) The trisomy for 21st chromosome is called :
A)
Down's syndrome
done
clear
B)
Turner's syndrome
done
clear
C)
Sickle cell anaemia
done
clear
D)
Klinefelter's syndrome
done
clear
View Answer play_arrow
question_answer 104) 'Mammalian thymus' is mainly concerned with :
A)
regulation of body temperature
done
clear
B)
regulation of body growth
done
clear
C)
immunological functions
done
clear
D)
secretion of thyrotropin
done
clear
View Answer play_arrow
question_answer 105) Which of the following is the largest gland in an adult man?
A)
Thymus
done
clear
B)
Liver
done
clear
C)
Thyroid
done
clear
D)
Pancreas
done
clear
View Answer play_arrow
question_answer 106) In Pheretima, septa are absent between which segments?
A)
3/4 and 9/10
done
clear
B)
4/5 and 8/9
done
clear
C)
5/6 and 7/8
done
clear
D)
7/8 and 6/7
done
clear
View Answer play_arrow
question_answer 107) During emergency which of the following hormone is secreted?
A)
Aldosterone
done
clear
B)
Thyroxine
done
clear
C)
Adrenaline
done
clear
D)
Calcitonin
done
clear
View Answer play_arrow
question_answer 108) In which of the following animals, respiration occurs without any respiratory organ ?
A)
frog
done
clear
B)
fish
done
clear
C)
cockroach
done
clear
D)
earthworm
done
clear
View Answer play_arrow
question_answer 109) In cockroach, larval and nymphal characters are maintained by :
A)
ecdysone
done
clear
B)
salivary glands
done
clear
C)
parotid gland
done
clear
D)
juvenile hormone
done
clear
View Answer play_arrow
question_answer 110) Which of the following is a transparent tissue ?
A)
Tendon
done
clear
B)
Fibrous cartilage
done
clear
C)
Hyaline cartilage
done
clear
D)
All of these
done
clear
View Answer play_arrow
question_answer 111) Rh factor is present in :
A)
all vertebrates
done
clear
B)
all mammals
done
clear
C)
all reptiles
done
clear
D)
man and rhesus monkey only
done
clear
View Answer play_arrow
question_answer 112) In rabbit, end of a long bone is connected to another by:
A)
tendon
done
clear
B)
ligaments
done
clear
C)
muscle
done
clear
D)
cartilage
done
clear
View Answer play_arrow
question_answer 113) Which of the following cell type is capable of giving rise to other cell types in sponges ?
A)
thesocytes
done
clear
B)
pinacocytes
done
clear
C)
cnidocytes
done
clear
D)
archaeocytes
done
clear
View Answer play_arrow
question_answer 114) Thigmotaxis is not shown by:
A)
Poramecium
done
clear
B)
Amoeba
done
clear
C)
Ascaris
done
clear
D)
Hydra
done
clear
View Answer play_arrow
question_answer 115) Which is correctly matched ?
A)
Apiculture?honey bee
done
clear
B)
Pisciculture?silk moth
done
clear
C)
Sericulture?fish
done
clear
D)
Aquaculture?mosquito
done
clear
View Answer play_arrow
question_answer 116) Changes that allow the conversion of larva into adult, is called :
A)
metagenesis
done
clear
B)
alternation
done
clear
C)
metamorphosis
done
clear
D)
metastasis
done
clear
View Answer play_arrow
question_answer 117) Which of the following is secreted by mast cell ?
A)
Histamine
done
clear
B)
Heparin
done
clear
C)
Serotonin
done
clear
D)
All of these
done
clear
View Answer play_arrow
question_answer 118) 5th cranial nerve of frog is called :
A)
optic nerve
done
clear
B)
vagus
done
clear
C)
trigeminal
done
clear
D)
ophthalmic
done
clear
View Answer play_arrow
question_answer 119) Animals having a built in thermostat to maintain constant body temperature are known as :
A)
biothermic
done
clear
B)
poikilothermic
done
clear
C)
oligothennic
done
clear
D)
homeothermic
done
clear
View Answer play_arrow
question_answer 120) In earthworm, neurons are:
A)
motor
done
clear
B)
associated
done
clear
C)
sensory
done
clear
D)
all of these
done
clear
View Answer play_arrow
question_answer 121) In sex linkage, the speciality is :
A)
atavism
done
clear
B)
criss-cross inheritance
done
clear
C)
reversion
done
clear
D)
gene flow
done
clear
View Answer play_arrow
question_answer 122) Vitamin-D is synthesized in skin by the action of sunlight on :
A)
cholesterol
done
clear
B)
7-hydroxy cholesterol
done
clear
C)
cephalin cholesterol
done
clear
D)
all of the above
done
clear
View Answer play_arrow
question_answer 123) The intermediate host of Schistosoma to :
A)
snail
done
clear
B)
mosquito
done
clear
C)
housefly
done
clear
D)
sheep
done
clear
View Answer play_arrow
question_answer 124) The islets of Langerhans are found in :
A)
pancreas
done
clear
B)
stomach
done
clear
C)
liver
done
clear
D)
alimentary canal
done
clear
View Answer play_arrow
question_answer 125) In Klinefelter's syndrome, the sex chromosome compliment is :
A)
22 A + XXY
done
clear
B)
22 A + XO
done
clear
C)
22A + XY
done
clear
D)
22 A + XX
done
clear
View Answer play_arrow
question_answer 126) Lymphoid tissue is found in :
A)
thymus
done
clear
B)
tonsils
done
clear
C)
lymph nodes
done
clear
D)
all of these
done
clear
View Answer play_arrow
question_answer 127) Most widely used bioweapon is :
A)
Bacillus subtilis
done
clear
B)
Pseudomonos putida
done
clear
C)
Bacillus anthracis
done
clear
D)
none of the above
done
clear
View Answer play_arrow
question_answer 128) The vitamin which is essential for blood dotting is:
A)
vitamin-A
done
clear
B)
vitamin-B
done
clear
C)
vitamin-C
done
clear
D)
vitamin-K
done
clear
View Answer play_arrow
question_answer 129) The female genital pore of Phererima posthuma located upon the segment ?
A)
14th
done
clear
B)
16th
done
clear
C)
18th
done
clear
D)
15th
done
clear
View Answer play_arrow
question_answer 130) Polyp phase is absent in :
A)
Hydra
done
clear
B)
Aurelia
done
clear
C)
Physalia
done
clear
D)
Obelia
done
clear
View Answer play_arrow
question_answer 131) In frog heart, there are cardiac muscles which consist of fibres called :
A)
Purkinje fibres
done
clear
B)
my onemes
done
clear
C)
telodendria
done
clear
D)
columnae camae
done
clear
View Answer play_arrow
question_answer 132) Malpighian tubules are :
A)
excretory organs of insects
done
clear
B)
excretory organs of frog
done
clear
C)
respiratory organs of insects
done
clear
D)
endocrine glands of insects
done
clear
View Answer play_arrow
question_answer 133) Pacemaker is:
A)
AV node
done
clear
B)
SA node
done
clear
C)
bundle of His
done
clear
D)
Purkinje fibres
done
clear
View Answer play_arrow
question_answer 134) LH and FSH are collectively called :
A)
oxytocin
done
clear
B)
somatotrophins
done
clear
C)
luteotrophic
done
clear
D)
gonadotrophins
done
clear
View Answer play_arrow
question_answer 135) Who is known as "father of Endocrinology" ?
A)
R.H. Whittaker
done
clear
B)
Pasteur
done
clear
C)
Einthoven
done
clear
D)
Thomas Addison
done
clear
View Answer play_arrow
question_answer 136) Which of the following provides most evident proof of evolution ?
A)
Fossils
done
clear
B)
Morphology
done
clear
C)
Embryo
done
clear
D)
Vestigial organs
done
clear
View Answer play_arrow
question_answer 137) In Mollusca, eye is present over a stalk, called :
A)
ostractum
done
clear
B)
operculum
done
clear
C)
ommatophores
done
clear
D)
osphradium
done
clear
View Answer play_arrow
question_answer 138) Among the following, colonial insects are :
A)
locusts
done
clear
B)
mosquitoes
done
clear
C)
white ants
done
clear
D)
bed bug
done
clear
View Answer play_arrow
question_answer 139) In Ascaris, the coelom is :
A)
schizocoelom
done
clear
B)
pseudocoelom
done
clear
C)
true coelom
done
clear
D)
haemocoelom
done
clear
View Answer play_arrow
question_answer 140) Turbellarians' are free living :
A)
nematodes
done
clear
B)
cestodes
done
clear
C)
flat worms
done
clear
D)
trematodes
done
clear
View Answer play_arrow
question_answer 141) The characteristic larva of phylum Coelemerata' is :
A)
planula
done
clear
B)
cysricercus
done
clear
C)
rhabdiform
done
clear
D)
wriggler
done
clear
View Answer play_arrow
question_answer 142) Podocytes are the cells, present in :
A)
cortex of nephron
done
clear
B)
inner wall of Bowman's capsule
done
clear
C)
outer wall of Bowman's capsule
done
clear
D)
wall of glomerular capillaries
done
clear
View Answer play_arrow
question_answer 143) In rabbit, head of epididymis present at the head of the testis is called :
A)
vas deferens
done
clear
B)
cauda epididymis
done
clear
C)
gubemaculum
done
clear
D)
caput epididymis
done
clear
View Answer play_arrow
question_answer 144) Tendons and ligaments are specialized types of:
A)
nervous tissue
done
clear
B)
muscular tissue
done
clear
C)
epithelial tissue
done
clear
D)
fibrous connective tissue
done
clear
View Answer play_arrow
question_answer 145) In blood, \[C{{O}_{2}}\]is transported majorly as :
A)
sodium carbonate
done
clear
B)
carboxy haemoglobin
done
clear
C)
bicarbonate
done
clear
D)
\[C{{O}_{2}}\] as such
done
clear
View Answer play_arrow
question_answer 146) Animals undergoes inactive stage during winter, is known as :
A)
aestivarion
done
clear
B)
hibernation
done
clear
C)
adaptation
done
clear
D)
acclimirization
done
clear
View Answer play_arrow
question_answer 147) Kupffer cells are present in :
A)
liver
done
clear
B)
small intestine
done
clear
C)
pancreas
done
clear
D)
thyroid gland
done
clear
View Answer play_arrow
question_answer 148) The embryo at 16 celled stage is known as ;
A)
morula
done
clear
B)
gastrula
done
clear
C)
blastula
done
clear
D)
blastomere
done
clear
View Answer play_arrow
question_answer 149) Contractile vacuole in protozoan Amoeba is meant for :
A)
respiration
done
clear
B)
excretion
done
clear
C)
locomotion
done
clear
D)
osmoregulation
done
clear
View Answer play_arrow
question_answer 150) In which of the following, putrefying bacteria is present ?
A)
intestine
done
clear
B)
colon
done
clear
C)
stomach
done
clear
D)
liver
done
clear
View Answer play_arrow
question_answer 151) Jumping genes in maize were discovered by :
A)
Hugo de Vries
done
clear
B)
Barbara Me Clintock
done
clear
C)
T.H. Morgan
done
clear
D)
Mendel
done
clear
View Answer play_arrow
question_answer 152) Streptomycin is obtained from :
A)
S. treptomyces griseus
done
clear
B)
S. aureofaciens
done
clear
C)
S. venezuelae
done
clear
D)
S. ramosus
done
clear
View Answer play_arrow
question_answer 153) Binomial system of nomenclature was given by:
A)
Julian Huxley
done
clear
B)
Bentham and Hooker
done
clear
C)
Linnaeus
done
clear
D)
Casper Bauhin
done
clear
View Answer play_arrow
question_answer 154) Morphine obtained from Opium is :
A)
latex
done
clear
B)
pome
done
clear
C)
alkaloid
done
clear
D)
tannin
done
clear
View Answer play_arrow
question_answer 155) Indusium is found in :
A)
algae
done
clear
B)
ferns
done
clear
C)
moss
done
clear
D)
Cycas
done
clear
View Answer play_arrow
question_answer 156) The vacuole is lined by a membrane called :
A)
tonoplast
done
clear
B)
jacket
done
clear
C)
cell membrane
done
clear
D)
tonoplasm
done
clear
View Answer play_arrow
question_answer 157) Agar-agar is obtained from :
A)
Chlorella
done
clear
B)
Spirogyra
done
clear
C)
Ulothrix
done
clear
D)
Gelidium
done
clear
View Answer play_arrow
question_answer 158) Effect of pollution is observed first on :
A)
micro-organisms
done
clear
B)
food crop
done
clear
C)
green vegetation
done
clear
D)
herbivores
done
clear
View Answer play_arrow
question_answer 159) DNA element with ability 10 change its position, is called:
A)
caisson
done
clear
B)
transposon
done
clear
C)
intron
done
clear
D)
recon
done
clear
View Answer play_arrow
question_answer 160) Initiation codon is :
A)
\[\text{UUU}\]
done
clear
B)
\[\text{UGA}\]
done
clear
C)
\[\text{AUG}\]
done
clear
D)
\[\text{UAG}\]
done
clear
View Answer play_arrow
question_answer 161) DNA multiplication is called :
A)
translation
done
clear
B)
replication
done
clear
C)
transduction
done
clear
D)
transcription
done
clear
View Answer play_arrow
question_answer 162) Duramen is present in :
A)
inner region of secondary wood
done
clear
B)
pan of sap wood
done
clear
C)
outer region of secondary wood
done
clear
D)
region of pericycle
done
clear
View Answer play_arrow
question_answer 163) In plants, water supply is due to :
A)
osmosis
done
clear
B)
imbibition
done
clear
C)
guttation
done
clear
D)
adhesion force
done
clear
View Answer play_arrow
question_answer 164) Programmed cell death is scientifically termed as:
A)
autotomy
done
clear
B)
cell lysis
done
clear
C)
apoptosis
done
clear
D)
none of these
done
clear
View Answer play_arrow
question_answer 165) Paraffin wax is :
A)
ester
done
clear
B)
acid
done
clear
C)
monohydric alcohol
done
clear
D)
cholesterol
done
clear
View Answer play_arrow
question_answer 166) Medicine for curing respiratory problems is obtained from :
A)
Ephedra
done
clear
B)
Calotrop is
done
clear
C)
Eucalyptus
done
clear
D)
Can aft is
done
clear
View Answer play_arrow
question_answer 167) In Funaria, stomata are present on the :
A)
stem
done
clear
B)
leaves
done
clear
C)
capsule
done
clear
D)
apophysis
done
clear
View Answer play_arrow
question_answer 168) The process after glycolysis in anaerobic respiration is known as :
A)
fermentation
done
clear
B)
respiration
done
clear
C)
Krebs cycle
done
clear
D)
decomposition
done
clear
View Answer play_arrow
question_answer 169) Which is always present in photochemical smog?
A)
\[{{O}_{3}}\]
done
clear
B)
\[C{{O}_{2}}\]
done
clear
C)
\[S{{O}_{2}}\]
done
clear
D)
\[C{{H}_{4}}\]
done
clear
View Answer play_arrow
question_answer 170) Which crop utilises solar energy most efficiently?
A)
Potato
done
clear
B)
Sugarcane
done
clear
C)
Wheat
done
clear
D)
Rice
done
clear
View Answer play_arrow
question_answer 171) Viral genome in coporated host DNA is called :
A)
prophase
done
clear
B)
prophage
done
clear
C)
bacteriophage
done
clear
D)
none of these
done
clear
View Answer play_arrow
question_answer 172) In cell cycle, during which phase, chromosomes are arranged in equatorial plate?
A)
Metaphase
done
clear
B)
Anaphase
done
clear
C)
Telophase
done
clear
D)
Prophase
done
clear
View Answer play_arrow
question_answer 173) The soil which is transported by wind is known as:
A)
colluvial
done
clear
B)
eolian
done
clear
C)
alluvial
done
clear
D)
glacial soil
done
clear
View Answer play_arrow
question_answer 174) Spindle fibre is made up of:
A)
tubulin
done
clear
B)
humulin
done
clear
C)
intermediate filament
done
clear
D)
flagellin
done
clear
View Answer play_arrow
question_answer 175) The yielding of rice is increased by :
A)
Azolla
done
clear
B)
Nostoc
done
clear
C)
Anabaena
done
clear
D)
Clostridium
done
clear
View Answer play_arrow
question_answer 176) Lichen is the pioneer vegetation on which succession?
A)
hydrosere
done
clear
B)
lithosere
done
clear
C)
psammosere
done
clear
D)
xerosere
done
clear
View Answer play_arrow
question_answer 177) Law of limiting factor was given by:
A)
Leibig
done
clear
B)
Blackman
done
clear
C)
Calvin
done
clear
D)
Amon
done
clear
View Answer play_arrow
question_answer 178) In Pinus, male cone bears a large number of:
A)
ligules
done
clear
B)
anthers
done
clear
C)
micro-sporophylls
done
clear
D)
mega-sporophylls
done
clear
View Answer play_arrow
question_answer 179) Insectivorous plants grow in soil:
A)
rich in nitrates
done
clear
B)
rich in phosphates
done
clear
C)
deficient in nitrates
done
clear
D)
rich in calcium
done
clear
View Answer play_arrow
question_answer 180) Induction of flowering by low temperature treatment is :
A)
vernalisation
done
clear
B)
cryobiology
done
clear
C)
photoperiodism
done
clear
D)
pruning
done
clear
View Answer play_arrow
question_answer 181) Decomposers are :
A)
autotrophs
done
clear
B)
autoheterotrophs
done
clear
C)
organotrophs
done
clear
D)
heterotrophs
done
clear
View Answer play_arrow
question_answer 182) 'Club moss' belongs to :
A)
Algae
done
clear
B)
Pteridophyta
done
clear
C)
Fungi
done
clear
D)
Bryophyta
done
clear
View Answer play_arrow
question_answer 183) Cleavage polyembryony occurs in :
A)
Pinus
done
clear
B)
Mini Cyan
done
clear
C)
Cycas
done
clear
D)
Ephedra
done
clear
View Answer play_arrow
question_answer 184) Edible part of mushroom is :
A)
basidiocarp
done
clear
B)
secondary mycelium
done
clear
C)
primary mycelium
done
clear
D)
ternary mycelium
done
clear
View Answer play_arrow
question_answer 185) Which is the major crop in Asia?
A)
Rice
done
clear
B)
Sugarcane
done
clear
C)
Jowar
done
clear
D)
Millet
done
clear
View Answer play_arrow
question_answer 186) Most nutritious among the following is :
A)
wheat
done
clear
B)
maize
done
clear
C)
bajra
done
clear
D)
rice
done
clear
View Answer play_arrow
question_answer 187) Which of the following plant product is the hardest?
A)
Lignin
done
clear
B)
Cutin
done
clear
C)
Suberin
done
clear
D)
Sporopollenin
done
clear
View Answer play_arrow
question_answer 188) 'Ruminate endosperm' is found in :
A)
Cruciferae
done
clear
B)
Asteraceae
done
clear
C)
Euphorbiaceae
done
clear
D)
Annonaceae
done
clear
View Answer play_arrow
question_answer 189) Guttarion is due to :
A)
transpiration
done
clear
B)
root pressure
done
clear
C)
osmosis
done
clear
D)
diffusion
done
clear
View Answer play_arrow
question_answer 190) The following is used to get desired breed of plants.
A)
tissue culture
done
clear
B)
genetic engineering
done
clear
C)
inbreeding
done
clear
D)
ikebana
done
clear
View Answer play_arrow
question_answer 191) For production of haploids, we culture :
A)
shoot tip
done
clear
B)
anther
done
clear
C)
root tip
done
clear
D)
none of these
done
clear
View Answer play_arrow
question_answer 192) Calyptra is derived from :
A)
archegonia
done
clear
B)
capsule
done
clear
C)
antheridia
done
clear
D)
columella
done
clear
View Answer play_arrow
question_answer 193) Late blight of potato is caused by :
A)
Ustilago
done
clear
B)
Pseudomonas
done
clear
C)
Phytophchora infestans
done
clear
D)
Colletotricum falcatum
done
clear
View Answer play_arrow
question_answer 194) 'Clamp connection' are observed in :
A)
Basidiomycetes
done
clear
B)
Zygomycetes
done
clear
C)
Ascomycetes
done
clear
D)
Oomyceles
done
clear
View Answer play_arrow
question_answer 195) Leaf abscission is caused by :
A)
ABA
done
clear
B)
cytokinin
done
clear
C)
auxin
done
clear
D)
gibberellins
done
clear
View Answer play_arrow
question_answer 196) What is the main cause for the extinction of some species in tropical forest?
A)
Deforestation
done
clear
B)
Afforestation
done
clear
C)
Pollution
done
clear
D)
Soil erosion
done
clear
View Answer play_arrow
question_answer 197) Wilting of plant is due to :
A)
blockage of phloem
done
clear
B)
blockage of xylem
done
clear
C)
when some root and phloem are removed
done
clear
D)
tracheid is blocked
done
clear
View Answer play_arrow
question_answer 198) Most accepted theory for ascent of sap is :
A)
capillarity theory
done
clear
B)
root pressure theory
done
clear
C)
pulsation theory
done
clear
D)
transpiration pull
done
clear
View Answer play_arrow
question_answer 199) Which enzyme converts glucose into alcohol?
A)
Zymase
done
clear
B)
Diastase
done
clear
C)
Invertase
done
clear
D)
Lipase
done
clear
View Answer play_arrow
question_answer 200) Crop rotation helps in :
A)
increase soil fertility
done
clear
B)
decrease soil erosion
done
clear
C)
decrease soil fertility
done
clear
D)
decrease water loss
done
clear
View Answer play_arrow
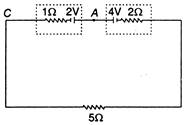 Resistances\[1\,\Omega \]and\[2\,\Omega \]represent the internal resistances of the respective cells.
Resistances\[1\,\Omega \]and\[2\,\Omega \]represent the internal resistances of the respective cells.
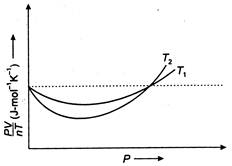 Read the following statements concerning the above curves: (i) The dotted line corresponds to the 'ideal' gas behaviour. (ii)\[{{T}_{1}}>{{T}_{2}}\] (iii) The value of\[\frac{pV}{nT}\]at the point where the curves meet on the y-axis is the same for all gases. Which of the above statement is true?
Read the following statements concerning the above curves: (i) The dotted line corresponds to the 'ideal' gas behaviour. (ii)\[{{T}_{1}}>{{T}_{2}}\] (iii) The value of\[\frac{pV}{nT}\]at the point where the curves meet on the y-axis is the same for all gases. Which of the above statement is true?