question_answer 1) A bar magnet is oscillating in the earth's magnetic field with a period T. What happens to its period and motion if its mass is quadrupled?
A)
Motion remains SH with time period = 4T
done
clear
B)
Motion remains SH and period remains nearly constant
done
clear
C)
Motion remains SH with time period\[=\frac{T}{2}\]
done
clear
D)
Motion remains SH with time period\[=2T\]
done
clear
View Answer play_arrow
question_answer 2) A charged particle moves through a magnetic field in a direction perpendicular to it. Then the
A)
acceleration remains unchanged
done
clear
B)
velocity remains unchanged
done
clear
C)
speed of the particle remains unchanged
done
clear
D)
direction of the particle remains unchanged
done
clear
View Answer play_arrow
question_answer 3) A man weighs 80 kg. He stands on a weighing scale in a lift which is moving upwards with a uniform acceleration of\[5\text{ }m/{{s}^{2}}\]. What would be the reading on the scale?\[(g=10m/{{s}^{2}})\]
A)
800 N
done
clear
B)
1200 N
done
clear
C)
zero
done
clear
D)
400 N
done
clear
View Answer play_arrow
question_answer 4) Which of the following units denotes the dimensions\[[M{{L}^{2}}/{{Q}^{2}}],\]where Q denotes the electric charge?
A)
\[Wb/{{m}^{2}}\]
done
clear
B)
Henry (H)
done
clear
C)
\[H/{{m}^{2}}\]
done
clear
D)
Weber (Wb)
done
clear
View Answer play_arrow
question_answer 5) The vector sum of two forces is perpendicular to their vector difference. In that case, the forces
A)
are not equal to each other in magnitude
done
clear
B)
cannot be predicted
done
clear
C)
are equal to each other
done
clear
D)
are equal to each other in magnitude
done
clear
View Answer play_arrow
question_answer 6) A ball rolls without slipping. The radius of gyration of the ball about an axis passing through its centre of mass is K. If radius of the ball be R, then the fraction of total energy associated with its rotational energy will be
A)
\[\frac{{{K}^{2}}}{{{K}^{2}}+{{R}^{2}}}\]
done
clear
B)
\[\frac{{{R}^{2}}}{{{K}^{2}}+{{R}^{2}}}\]
done
clear
C)
\[\frac{{{K}^{2}}+{{R}^{2}}}{{{R}^{2}}}\]
done
clear
D)
\[\frac{{{K}^{2}}}{{{R}^{2}}}\]
done
clear
View Answer play_arrow
question_answer 7) The acceleration due to gravity on the planet A is 9 times the acceleration due to gravity on planet B. A man jumps to a height of 2 m on the surface of A. What is the height of jump by the same person on the planet B?
A)
6 m
done
clear
B)
2/3 m
done
clear
C)
2/9 m
done
clear
D)
18 m
done
clear
View Answer play_arrow
question_answer 8) When a long spring is stretched by 2 cm, its potential energy is U. If the spring is stretched by 10 cm, the potential energy in it will be
A)
10 U
done
clear
B)
25 U
done
clear
C)
U/5
done
clear
D)
5U
done
clear
View Answer play_arrow
question_answer 9) An electron is moving round the nucleus of a hydrogen atom in a circular orbit of radius r. The Coulomb force \[\overrightarrow{F}\]between the two is
A)
\[k\frac{{{e}^{2}}}{{{r}^{3}}}\overrightarrow{r}\]
done
clear
B)
\[-k\frac{{{e}^{2}}}{{{r}^{3}}}\overrightarrow{r}\]
done
clear
C)
\[-k\frac{{{e}^{2}}}{{{r}^{2}}}\hat{r}\]
done
clear
D)
\[-k\frac{{{e}^{2}}}{{{r}^{3}}}\hat{r}\] (where\[k=\frac{1}{4\pi {{\varepsilon }_{0}}}\])
done
clear
View Answer play_arrow
question_answer 10) A charge q is located at the centre of a cube. The electric flux through any face is
A)
\[\frac{\pi q}{6(4\pi {{\varepsilon }_{0}})}\]
done
clear
B)
\[\frac{q}{6(4\pi {{\varepsilon }_{0}})}\]
done
clear
C)
\[\frac{2\pi q}{6(4\pi {{\varepsilon }_{0}})}\]
done
clear
D)
\[\frac{4\pi q}{6(4\pi {{\varepsilon }_{0}})}\]
done
clear
View Answer play_arrow
question_answer 11) An observer moves towards a stationary source of sound with a speed 1/5th of the speed of sound. The wavelength and frequency of the source emitted are\[\lambda \]and\[f\] respectively. The apparent frequency and wavelength recorded by the observer are respectively
A)
\[f,1.2\lambda \]
done
clear
B)
\[0.8f,0.8\lambda \]
done
clear
C)
\[1.2f,1.2\lambda \]
done
clear
D)
\[1.2f,\lambda \]
done
clear
View Answer play_arrow
question_answer 12) Consider a compound slab consisting of two different materials having equal thickness and thermal conductivities K and 2K respectively. The equivalent thermal conductivity of the slab is
A)
3K
done
clear
B)
\[\frac{4}{3}K\]
done
clear
C)
\[\frac{2}{3}K\]
done
clear
D)
\[\sqrt{2}K\]
done
clear
View Answer play_arrow
question_answer 13) Fuse wire is a wire of
A)
low resistance and low melting point
done
clear
B)
low resistance and high melting point
done
clear
C)
high resistance and high melting point
done
clear
D)
high resistance and low melting point
done
clear
View Answer play_arrow
question_answer 14) Solar energy is mainly caused due to
A)
fusion of protons during synthesis of heavier elements
done
clear
B)
gravitational contraction
done
clear
C)
burning of hydrogen in the oxygen
done
clear
D)
fission of uranium present in the sun
done
clear
View Answer play_arrow
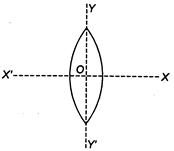
question_answer 15)
An equiconvex lens is cut into two halves along (i)\[XOX'\]and (ii)\[YOY'\]as shown in the figure. Let\[f,f',f''\]be the focal lengths of the complete lens, of each half in case (i), and of each half in case (ii), respectively.
A)
\[f'=f,f''=f\]
done
clear
B)
\[f'=2f,f''=2f\]
done
clear
C)
\[f'=f,f''=2f\]
done
clear
D)
\[f'=2f,f''=f\]
done
clear
View Answer play_arrow
question_answer 16) A diamagnetic material in a magnetic field moves
A)
perpendicular to the field
done
clear
B)
from weaker to the stronger parts of the field
done
clear
C)
from stronger to the weaker parts of the field
done
clear
D)
in none of the above directions
done
clear
View Answer play_arrow
question_answer 17) A convex lens is dipped in a liquid whose refractive index is equal to the refractive index of the lens. Then its focal length will
A)
become small, but non-zero
done
clear
B)
remain unchanged
done
clear
C)
become zero
done
clear
D)
become infinite
done
clear
View Answer play_arrow
question_answer 18) \[\overrightarrow{A}\times \overrightarrow{B}=\overrightarrow{B}\times \overrightarrow{A},\]then the angle between A and B is
A)
\[\pi \]
done
clear
B)
\[\pi /3\]
done
clear
C)
\[\pi /2\]
done
clear
D)
\[\pi /4\]
done
clear
View Answer play_arrow
question_answer 19) Two spheres of masses m and M are situated in air and the gravitational force between them is F. The space around the masses is now filled with a liquid of specific gravity 3. The gravitational force will now be
A)
\[\frac{F}{3}\]
done
clear
B)
\[\frac{F}{9}\]
done
clear
C)
\[3F\]
done
clear
D)
\[F\]
done
clear
View Answer play_arrow
question_answer 20) The potential energy of a simple harmonic oscillator when the particle is half way to its end point is
A)
\[\frac{1}{4}E\]
done
clear
B)
\[\frac{1}{2}E\]
done
clear
C)
\[\frac{2}{3}E\]
done
clear
D)
\[\frac{1}{8}E3\] (where E is the total energy)
done
clear
View Answer play_arrow
question_answer 21) In case of a forced vibration, the resonance wave becomes very sharp when the
A)
applied periodic force is small
done
clear
B)
quality factor is small
done
clear
C)
damping force is small
done
clear
D)
restoring force is small
done
clear
View Answer play_arrow
question_answer 22) A stationary particle explodes into two particles of masses\[{{m}_{1}}\]and\[{{m}_{2}}\]which move in opposite directions with velocities\[{{v}_{1}}\]and\[{{v}_{2}}\]. The ratio of their kinetic energies\[{{E}_{1}}/{{E}_{2}}\]is
A)
1
done
clear
B)
\[{{m}_{1}}{{v}_{2}}/{{m}_{2}}{{v}_{1}}\]
done
clear
C)
\[{{m}_{2}}/{{m}_{1}}\]
done
clear
D)
\[{{m}_{1}}/{{m}_{2}}\]
done
clear
View Answer play_arrow
question_answer 23) An ideal gas heat engine operates in a Carnot cycle between\[227{}^\circ C\]and\[127{}^\circ C\]. It absorbs 6 kcal at the higher temperature. The amount of heat (in kcal) converted into work is equal to
A)
1.6
done
clear
B)
1.2
done
clear
C)
4.8
done
clear
D)
3.5
done
clear
View Answer play_arrow
question_answer 24) We consider the radiation emitted by the human body. Which of the following statements is true?
A)
The radiation is emitted during the summers and absorbed during the winters
done
clear
B)
The radiation emitted lies in the ultraviolet region and hence is not visible
done
clear
C)
The radiation emitted is in the infra-red region
done
clear
D)
The radiation is emitted only during the day
done
clear
View Answer play_arrow
question_answer 25) Three capacitors each of capacity\[4\mu F\]are to be connected in such a way that the effective capacitance is\[6\mu F\]. This can be done by
A)
connecting two in series and one in parallel
done
clear
B)
connecting two in parallel and one in series
done
clear
C)
connecting all of them in series
done
clear
D)
connecting all of them in parallel
done
clear
View Answer play_arrow
question_answer 26) In a Wheatstone's bridge all the four arms have equal resistance R. If the resistance of the galvanometer arm is also R, the equivalent resistance of the combination as seen by the battery is
A)
R
done
clear
B)
2R
done
clear
C)
\[\frac{R}{4}\]
done
clear
D)
\[\frac{R}{2}\]
done
clear
View Answer play_arrow
question_answer 27) A sample of radioactive element has a mass of 10 g at an instant r = 0. The approximate mass of this element in the sample after two mean lives is
A)
3.70 g
done
clear
B)
6.30 g
done
clear
C)
1.35 g
done
clear
D)
2.50 g
done
clear
View Answer play_arrow
question_answer 28) The volume occupied by an atom is greater than the volume of the nucleus by factor of about
A)
\[{{10}^{10}}\]
done
clear
B)
\[{{10}^{15}}\]
done
clear
C)
\[{{10}^{1}}\]
done
clear
D)
\[{{10}^{5}}\]
done
clear
View Answer play_arrow
question_answer 29) In electrolysis the mass deposited on an electrode is directly proportional to
A)
current
done
clear
B)
square of current
done
clear
C)
concentration of solution
done
clear
D)
inverse of current
done
clear
View Answer play_arrow
question_answer 30) The mass of proton is 1.0073 u and that of neutron is 1.0087 u (u = atomic mass unit). The binding energy of\[2H{{e}^{4}}\]is
A)
28.4 MeV
done
clear
B)
0.061 u
done
clear
C)
0.0305 J
done
clear
D)
0.0305 erg
done
clear
View Answer play_arrow
question_answer 31) Given, mass of helium nucleus w 4.0015 u. Barrier potential of a p-n junction diode does not depend on
A)
forward bias
done
clear
B)
doping density
done
clear
C)
diode design
done
clear
D)
temperature
done
clear
View Answer play_arrow
question_answer 32) Thomson's cathode-ray tube experiment demonstrated that
A)
the e/m of electrons is greater than the e/m of protons
done
clear
B)
the e/m ratio of the cathode-ray particles changes when a different gas is placed in the discharge tube
done
clear
C)
cathode rays are streams of negatively charged ions
done
clear
D)
all the mass of an atom is essentially in the nucleus
done
clear
View Answer play_arrow
question_answer 33) Which of the following parameters does not characterize the thermodynamic state of matter?
A)
Temperature
done
clear
B)
Pressure
done
clear
C)
Work
done
clear
D)
Volume
done
clear
View Answer play_arrow
question_answer 34) In which of the following systems will the radius of the first orbit (n = 1) be minimum?
A)
Deuterium atom
done
clear
B)
Hydrogen atom
done
clear
C)
Doubly ionized lithium
done
clear
D)
Singly ionized helium
done
clear
View Answer play_arrow
question_answer 35) A long solenoid carrying a current produces a magnetic field B along its axis. If the current is doubled and the number of turns per cm is halved, the new value of the magnetic field is
A)
2B
done
clear
B)
4B
done
clear
C)
B/2
done
clear
D)
B
done
clear
View Answer play_arrow
question_answer 36)
Following diagram performs the logic function of
A)
OR gate
done
clear
B)
AND gate
done
clear
C)
XOR gate
done
clear
D)
NAND gate
done
clear
View Answer play_arrow
question_answer 37) "Parsec" is the unit of
A)
time
done
clear
B)
distance
done
clear
C)
frequency
done
clear
D)
angular acceleration
done
clear
View Answer play_arrow
question_answer 38) Which of the following is an amorphous solid?
A)
Glass
done
clear
B)
Diamond
done
clear
C)
Salt
done
clear
D)
Sugar
done
clear
View Answer play_arrow
question_answer 39) A bread gives a boy of mass 40 kg an energy of 21 k-J. If the efficiency is 28%, then the height can be climbed by him using this energy, is
A)
22.5m
done
clear
B)
15m
done
clear
C)
10m
done
clear
D)
5m
done
clear
View Answer play_arrow
question_answer 40) The kinetic energy of 1 g molecule of a gas, at normal temperature and pressure is\[(R=8.31J/mol-K)\]
A)
\[3.4\times {{10}^{3}}J\]
done
clear
B)
\[2.97\times {{10}^{3}}J\]
done
clear
C)
\[1.2\times {{10}^{2}}J\]
done
clear
D)
\[0.66\times {{10}^{4}}J\]
done
clear
View Answer play_arrow
question_answer 41) Plate current will be maximum when
A)
both the grid and anode are negative
done
clear
B)
both the grid and anode are positive
done
clear
C)
grid is positive and anode is negative
done
clear
D)
grid is negative and anode is positive
done
clear
View Answer play_arrow
question_answer 42) For driving current of 2 A for 6 min in a circuit, 1000J of work is to be done. The emf of the source in the circuit is
A)
1.38V
done
clear
B)
1.68V
done
clear
C)
2.03V
done
clear
D)
3.10V
done
clear
View Answer play_arrow
question_answer 43) An iron rod of length 2 m and cross-sectional area of\[50\text{ }m{{m}^{2}}\]is stretched by 0.5 mm, when a mass of 250 kg is hung from its lower end. Young's modulus of iron rod is
A)
\[19.6\times {{10}^{20}}N/{{m}^{2}}\]
done
clear
B)
\[19.6\times {{10}^{18}}N/{{m}^{2}}\]
done
clear
C)
\[19.6\times {{10}^{10}}N/{{m}^{2}}\]
done
clear
D)
\[19.6\times {{10}^{15}}N/{{m}^{2}}\]
done
clear
View Answer play_arrow
question_answer 44) Speed of a ball of 2 cm radius in a viscous liquid is 20 cm/s. Then the speed of 1 cm radius of ball in the same liquid is
A)
80 cm/s
done
clear
B)
40 m/s
done
clear
C)
10 cm/s
done
clear
D)
5 cm/s
done
clear
View Answer play_arrow
question_answer 45) A particle is thrown vertically upwards. Its velocity at half of the height is 10 m/s. Then the maximum height attained by it is \[(g=10\text{ }m/{{s}^{2}})\]
A)
16m
done
clear
B)
10m
done
clear
C)
8m
done
clear
D)
18m
done
clear
View Answer play_arrow
question_answer 46) The reactance of a coil when used in the AC power supply (220 V, 50 cycle/s) is\[50\,\Omega \]. The inductance of the coil is nearly
A)
0.16 H
done
clear
B)
0.22 H
done
clear
C)
2.2 H
done
clear
D)
1.6 H
done
clear
View Answer play_arrow
question_answer 47) The angle for which maximum height and horizontal range are same for a projectile is
A)
\[32{}^\circ \]
done
clear
B)
\[48{}^\circ \]
done
clear
C)
\[76{}^\circ \]
done
clear
D)
\[84{}^\circ \]
done
clear
View Answer play_arrow
question_answer 48) When a car moves on a road with uniform speed of 30 km/h, then the net resultant force on the car is
A)
the driving force, drives the car in the direction of propagation of car
done
clear
B)
the resistive force acts opposite to the direction of propagation of car
done
clear
C)
zero
done
clear
D)
none of the above
done
clear
View Answer play_arrow
question_answer 49) To draw maximum current from a combination of cells, how should the cells be grouped?
A)
Series
done
clear
B)
Parallel
done
clear
C)
Mixed
done
clear
D)
Depends upon the relative values of external and internal resistance
done
clear
View Answer play_arrow
question_answer 50) What causes change in the colours of the soap or oil films for the given beam of light?
A)
Angle of incidence
done
clear
B)
Angle of reflection
done
clear
C)
Thickness of film
done
clear
D)
None of the above
done
clear
View Answer play_arrow
question_answer 51) Carborundum is
A)
\[SiC\]
done
clear
B)
\[AlC{{l}_{3}}\]
done
clear
C)
\[A{{l}_{2}}{{(S{{O}_{4}})}_{3}}\]
done
clear
D)
\[A{{l}_{2}}{{O}_{3}}.2{{H}_{2}}O\]
done
clear
View Answer play_arrow
question_answer 52) The number of unpaired electron in\[M{{n}^{2+}}\]is
A)
4
done
clear
B)
5
done
clear
C)
6
done
clear
D)
7
done
clear
View Answer play_arrow
question_answer 53) The bond order of nitrogen molecule is
A)
2
done
clear
B)
2.5
done
clear
C)
3
done
clear
D)
none of these
done
clear
View Answer play_arrow
question_answer 54) Paracetamol is used as
A)
antipyretic
done
clear
B)
analgesics
done
clear
C)
antiseptics
done
clear
D)
antibiotics
done
clear
View Answer play_arrow
question_answer 55) The number of water molecules in Mohr?s salt is
A)
5
done
clear
B)
6
done
clear
C)
7
done
clear
D)
10
done
clear
View Answer play_arrow
question_answer 56) Which of the following is most basic?
A)
\[P{{H}_{3}}\]
done
clear
B)
\[As{{H}_{3}}\]
done
clear
C)
\[Sb{{H}_{3}}\]
done
clear
D)
\[N{{H}_{3}}\]
done
clear
View Answer play_arrow
question_answer 57) The half-life of\[_{6}{{C}^{14}}\](if its\[\lambda \]is\[2.31\times {{10}^{-4}}\]) is
A)
\[2\times {{10}^{2}}yr\]
done
clear
B)
\[3\times {{10}^{3}}yr\]
done
clear
C)
\[3.5\times {{10}^{4}}yr\]
done
clear
D)
\[4\times {{10}^{3}}yr\]
done
clear
View Answer play_arrow
question_answer 58) Oxidation number of Mn atom in\[KMn{{O}_{4}}\]is
A)
6
done
clear
B)
7
done
clear
C)
14
done
clear
D)
5
done
clear
View Answer play_arrow
question_answer 59) Benzoic acid is dissolved in benzene, van't Hoff factor will be
A)
1
done
clear
B)
0.5
done
clear
C)
1.5
done
clear
D)
2
done
clear
View Answer play_arrow
question_answer 60) The Gibbs free energy for a reversible reaction at equilibrium is
A)
positive
done
clear
B)
negative
done
clear
C)
zero
done
clear
D)
can be positive or negative
done
clear
View Answer play_arrow
question_answer 61) Which of the following electronic configuration is not possible?
A)
\[1{{s}^{2}},\text{ }2{{s}^{2}}\]
done
clear
B)
\[3{{d}^{10}},\text{ }4{{s}^{2}},4{{p}^{2}}\]
done
clear
C)
\[1{{s}^{2}},\text{ }2{{s}^{2}},\text{ }2{{p}^{6}}\]
done
clear
D)
\[1{{s}^{2}},\text{ }2{{s}^{2}},\text{ }2{{p}^{2}},\text{ }3{{s}^{1}}\]
done
clear
View Answer play_arrow
question_answer 62) Aldehydes and ketones can be distinguished by using
A)
Lucas reagent
done
clear
B)
Tollen's reagent
done
clear
C)
lodoform test
done
clear
D)
Phenyl hydrazine
done
clear
View Answer play_arrow
question_answer 63) Which radioactive elements is inert gas?
A)
\[Ra\]
done
clear
B)
\[Rn\]
done
clear
C)
\[Pu\]
done
clear
D)
\[Po\]
done
clear
View Answer play_arrow
question_answer 64) Which of the following has tetrahedral structure?
A)
\[NH_{4}^{+}\]
done
clear
B)
\[CO_{3}^{2-}\]
done
clear
C)
\[{{K}_{4}}[Fe{{(CN)}_{6}}]\]
done
clear
D)
None of these
done
clear
View Answer play_arrow
question_answer 65) Which is strongest acid?
A)
\[HClO\]
done
clear
B)
\[HCl{{O}_{2}}\]
done
clear
C)
\[HCl{{O}_{3}}\]
done
clear
D)
\[HCl{{O}_{4}}\]
done
clear
View Answer play_arrow
question_answer 66) If phosphoric acid is allowed to react with sufficient quantity of\[NaOH,\]the product obtained is
A)
\[N{{a}_{3}}P{{O}_{4}}\]
done
clear
B)
\[Na{{H}_{2}}P{{O}_{4}}\]
done
clear
C)
\[N{{a}_{2}}HP{{O}_{4}}\]
done
clear
D)
\[NaHP{{O}_{3}}\]
done
clear
View Answer play_arrow
question_answer 67) The concentration unit, independent of temperature would be
A)
molality
done
clear
B)
molarity
done
clear
C)
normality
done
clear
D)
weight per volume
done
clear
View Answer play_arrow
question_answer 68) Hexamethylenetetramine formed by the reaction of formaldehyde and
A)
ammonia
done
clear
B)
primary amine
done
clear
C)
secondary amine
done
clear
D)
tertiary amine
done
clear
View Answer play_arrow
question_answer 69) During Lassaigne's test N and S present in an organic compound changes into
A)
\[N{{a}_{2}}S\]and\[NaCN\]
done
clear
B)
\[NaSCN\]
done
clear
C)
\[N{{a}_{2}}S{{O}_{4}}\]and\[NaCN\]
done
clear
D)
\[N{{a}_{2}}S\]and\[NaCN\]
done
clear
View Answer play_arrow
question_answer 70) The quantum of light energy is
A)
photon
done
clear
B)
neutron
done
clear
C)
electron
done
clear
D)
proton
done
clear
View Answer play_arrow
question_answer 71) When ethyl alcohol reacts with\[C{{l}_{2}}\]in presence of\[NaOH,\]the product will be
A)
\[C{{H}_{3}}Cl\]
done
clear
B)
\[{{C}_{2}}{{H}_{5}}Cl\]
done
clear
C)
\[CC{{l}_{3}}.CHO\]
done
clear
D)
\[CHC{{l}_{3}}\]
done
clear
View Answer play_arrow
question_answer 72) The oxidation number of Fe in\[F{{e}_{3}}{{O}_{4}}\]is
A)
\[+8/3\]
done
clear
B)
\[+3/8\]
done
clear
C)
\[-8/3\]
done
clear
D)
\[-3/8\]
done
clear
View Answer play_arrow
question_answer 73) When lead nitrate is heated, it gives
A)
\[N{{O}_{2}}\]
done
clear
B)
\[PbO\]
done
clear
C)
both and
done
clear
D)
NO
done
clear
View Answer play_arrow
question_answer 74) Size of colloidal particle is
A)
\[{{10}^{-9}}m\text{ }to\text{ }{{10}^{-7}}m\]
done
clear
B)
\[{{10}^{-4}}cm\text{ }to\text{ }{{10}^{-7}}cm\]
done
clear
C)
\[{{10}^{-6}}m\text{ }to\text{ }{{10}^{-8}}m\]
done
clear
D)
\[{{10}^{-6}}cm\text{ }to\text{ }{{10}^{-9}}cm\]
done
clear
View Answer play_arrow
question_answer 75) Gun metal is an alloy of
A)
\[Mg,\text{ }Sn,\text{ }Zn\]
done
clear
B)
\[Cu,\text{ }Ni,\text{ }Zn\]
done
clear
C)
\[Cu,\text{ }Sn,\text{ }Zn\]
done
clear
D)
\[Cu,\text{ }Sb,\text{ }Zn,\text{ }Pb\]
done
clear
View Answer play_arrow
question_answer 76) \[{{C}_{6}}{{H}_{6}}+C{{l}_{2}}\xrightarrow{light}P\]Product 'P' is
A)
\[{{C}_{6}}{{H}_{5}}Cl\]
done
clear
B)
\[{{C}_{6}}{{H}_{4}}C{{l}_{2}}\]
done
clear
C)
\[{{C}_{6}}{{H}_{6}}C{{l}_{6}}\]
done
clear
D)
none of these
done
clear
View Answer play_arrow
question_answer 77) \[{{C}_{6}}{{H}_{6}}+C{{H}_{3}}\xrightarrow[{}]{anhy.AlC{{l}_{3}}}{{C}_{6}}{{H}_{5}}C{{H}_{3}}+HCl\] Above reaction is
A)
Kolbe?s synthesis
done
clear
B)
Wurtz reaction
done
clear
C)
Wurtz-Fittig reaction
done
clear
D)
Friedel-Craft's reaction
done
clear
View Answer play_arrow
question_answer 78) Which of the following substance is used as an ant knocking compound?
A)
Tetraethyl lead
done
clear
B)
Lead tetrachloride
done
clear
C)
Lead acetate
done
clear
D)
Ethyl acetate
done
clear
View Answer play_arrow
question_answer 79) An orbital, in which\[n=4,l=2\]is expressed as
A)
\[4p\]
done
clear
B)
\[4d\]
done
clear
C)
\[4f\]
done
clear
D)
\[3d\]
done
clear
View Answer play_arrow
question_answer 80) Which of the following gas is maximum present in atmosphere?
A)
\[Ar\]
done
clear
B)
\[C{{O}_{2}}\]
done
clear
C)
\[He\]
done
clear
D)
\[Xe\]
done
clear
View Answer play_arrow
question_answer 81) Hydrolysis of acetonitrile in acidic medium gives
A)
\[C{{H}_{3}}C{{H}_{2}}N{{H}_{2}}\]
done
clear
B)
\[C{{H}_{3}}COOH\]
done
clear
C)
\[C{{H}_{3}}CON{{H}_{2}}\]
done
clear
D)
\[C{{H}_{3}}CHO\]
done
clear
View Answer play_arrow
question_answer 82) IUPAC name\[{{C}_{2}}{{H}_{5}}CN\]is
A)
ethyl cyanide
done
clear
B)
propionitrile
done
clear
C)
propane nitrite
done
clear
D)
cyano ethane
done
clear
View Answer play_arrow
question_answer 83) Which of the following is organometallic compound?
A)
\[C{{H}_{3}}MgBr\]
done
clear
B)
\[{{(C{{H}_{3}}COO)}_{2}}Pb\]
done
clear
C)
\[C{{H}_{3}}COONa\]
done
clear
D)
All of these
done
clear
View Answer play_arrow
question_answer 84) Which of the following is good conductor of electricity?
A)
Diamond
done
clear
B)
Graphite
done
clear
C)
Solid\[NaCl\]
done
clear
D)
Wood
done
clear
View Answer play_arrow
question_answer 85) ?Equal volumes of gases under similar conditions of pressure and temperature posses equal number of molecules". This law is given by
A)
Avogadro
done
clear
B)
Berzelius
done
clear
C)
Gay Lussac
done
clear
D)
Graham
done
clear
View Answer play_arrow
question_answer 86) Most favourable condition for ionic bonding are
A)
high charge, small cadon, large anion
done
clear
B)
low charge, large cation, small anion
done
clear
C)
low charge, large cation, large anion
done
clear
D)
high charge, small cadon, small anion
done
clear
View Answer play_arrow
question_answer 87) In aluminothermite process\['Al'\]act as a/an
A)
flux
done
clear
B)
oxidising agent
done
clear
C)
reducing agent
done
clear
D)
solder
done
clear
View Answer play_arrow
question_answer 88) The electronic configuradon of nitrogen can be expressed as
A)
\[1{{s}^{2}},2{{s}^{2}},2p_{x}^{2},p_{y}^{1}p_{z}^{0}\]
done
clear
B)
\[1{{s}^{2}},2{{s}^{2}},2p_{x}^{1},p_{y}^{1},p_{z}^{1}\]
done
clear
C)
\[1{{s}^{2}},2{{s}^{2}},2p_{x}^{3},p_{y}^{0},p_{z}^{0}\]
done
clear
D)
\[1{{s}^{2}},2{{s}^{2}},2p_{x}^{1},p_{y}^{2},p_{z}^{0}\]
done
clear
View Answer play_arrow
question_answer 89) Which of the following is formed by the action of water on\[N{{a}_{2}}{{O}_{2}}\]?
A)
Ha
done
clear
B)
\[{{O}_{2}}\]
done
clear
C)
\[{{N}_{2}}\]
done
clear
D)
\[C{{O}_{2}}\]
done
clear
View Answer play_arrow
question_answer 90) Glycerol has
A)
1-primary-OH, 2-secondary-OH
done
clear
B)
2-primary-OH, 1 secondary-OH
done
clear
C)
3-primary-OH
done
clear
D)
1-primary-OH, 1 secondary OH, 1-tertiary OH
done
clear
View Answer play_arrow
question_answer 91) The long form of periodic table based on
A)
atomic number
done
clear
B)
atomic mass
done
clear
C)
electronic configuration
done
clear
D)
effective nuclear charge
done
clear
View Answer play_arrow
question_answer 92) Catalyst increases the rate of reaction
A)
by decreasing\[{{E}_{a}}\]
done
clear
B)
by increasing\[{{E}_{a}}\]
done
clear
C)
by decreasing A
done
clear
D)
by increasing entropy
done
clear
View Answer play_arrow
question_answer 93) Chlorobenzene is prepared commercially by
A)
Dew's process
done
clear
B)
Decon's process
done
clear
C)
Raschig process
done
clear
D)
Etard's process
done
clear
View Answer play_arrow
question_answer 94) In a radioactive decay, an emitted electron comes from
A)
the nucleus of atom
done
clear
B)
the orbit with principal quantum number
done
clear
C)
the inner orbital of the atom
done
clear
D)
the outer most orbit of the atom
done
clear
View Answer play_arrow
question_answer 95) Oleic, stearic, palmitic acids are
A)
nucleic acids
done
clear
B)
ammo acids
done
clear
C)
fatty acids
done
clear
D)
none of these
done
clear
View Answer play_arrow
question_answer 96) The weight of one molecule of a compound \[{{C}_{60}}{{H}_{122}}\]is
A)
\[1.3\times {{10}^{-20}}g\]
done
clear
B)
\[5.01\times {{10}^{-21}}g\]
done
clear
C)
\[3.72\times {{10}^{13}}g\]
done
clear
D)
\[1.4\times {{10}^{-21}}g\]
done
clear
View Answer play_arrow
question_answer 97) The value of 1 amu is equal to
A)
\[1.66\times {{10}^{-24}}g\]
done
clear
B)
12.00 g
done
clear
C)
\[1.9924\times {{10}^{-24}}g\]
done
clear
D)
1.0 g
done
clear
View Answer play_arrow
question_answer 98) Which of the following compound cannot reduce Fehling solution?
A)
HCOOH
done
clear
B)
\[C{{H}_{3}}COOH\]
done
clear
C)
HCHO
done
clear
D)
\[C{{H}_{3}}CHO\]
done
clear
View Answer play_arrow
question_answer 99) The metal present in vitamin\[{{B}_{12}}\]is
A)
iron
done
clear
B)
manganese
done
clear
C)
cobalt
done
clear
D)
mercury
done
clear
View Answer play_arrow
question_answer 100) The product obtained, heating ethanol with cone.\[{{H}_{2}}S{{O}_{4}}\]at\[165{}^\circ C-170{}^\circ C,\]is
A)
\[{{({{C}_{2}}{{H}_{5}})}_{2}}S{{O}_{4}}\]
done
clear
B)
\[C{{H}_{3}}COOH\]
done
clear
C)
\[C{{H}_{2}}=C{{H}_{2}}\]
done
clear
D)
\[{{C}_{2}}{{H}_{5}}HS{{O}_{4}}\]
done
clear
View Answer play_arrow
question_answer 101) Who coined the term antibiotics?
A)
Woodruff
done
clear
B)
Darwin
done
clear
C)
Selman Waksman
done
clear
D)
Flemming
done
clear
View Answer play_arrow
question_answer 102) The study of relationship between size and shape is called
A)
Allopatric
done
clear
B)
Allelopathy
done
clear
C)
Allomecry
done
clear
D)
Allogamy
done
clear
View Answer play_arrow
question_answer 103) Moulting hormone is secreted by
A)
corpora cardiacum
done
clear
B)
corpora allata
done
clear
C)
neurosecretory hormone
done
clear
D)
prothoracic gland
done
clear
View Answer play_arrow
question_answer 104) Haemodialysis helps in patient having
A)
anaemia
done
clear
B)
uremia
done
clear
C)
goitre
done
clear
D)
diabetes
done
clear
View Answer play_arrow
question_answer 105) Movements by pseudopodia of Amoeba are due to change in
A)
pressure
done
clear
B)
atmosphere
done
clear
C)
temperature
done
clear
D)
viscosity
done
clear
View Answer play_arrow
question_answer 106) A sponge harmful to oyster industry is
A)
Cliona
done
clear
B)
Euspongia
done
clear
C)
Hyolonema
done
clear
D)
Spongilla
done
clear
View Answer play_arrow
question_answer 107) During meiosis crossing over occurs at
A)
pachytene
done
clear
B)
zygotene
done
clear
C)
leptotene
done
clear
D)
diakinesis
done
clear
View Answer play_arrow
question_answer 108) Matrix of bone is composed of
A)
chondrin
done
clear
B)
ossein
done
clear
C)
osteon
done
clear
D)
auxin
done
clear
View Answer play_arrow
question_answer 109) It From outer to inside the sequence of three bones present in the middle ear of mammals is
A)
malleus, stapes and incus
done
clear
B)
stapes, malleus and incus
done
clear
C)
malleus, incus and stapes
done
clear
D)
incus, malleus and stapes
done
clear
View Answer play_arrow
question_answer 110) The reabsorprion of water in the kidney is under the control of
A)
LH
done
clear
B)
ADH
done
clear
C)
STH
done
clear
D)
ACTH
done
clear
View Answer play_arrow
question_answer 111) Glucagon is secreted by
A)
beta cells of pancreas
done
clear
B)
beta cells of islets of Langerhans
done
clear
C)
alpha cells of pancreas
done
clear
D)
adrenal cortex
done
clear
View Answer play_arrow
question_answer 112) Which pan of a sperm enters into an ovum during fertilization?
A)
Head
done
clear
B)
Tail
done
clear
C)
Whole of it
done
clear
D)
Middle piece
done
clear
View Answer play_arrow
question_answer 113) Yersninia pestis is responsible for
A)
syphilis
done
clear
B)
whooping cough
done
clear
C)
plague
done
clear
D)
leprosy
done
clear
View Answer play_arrow
question_answer 114) Male Anopheles does not transmit malarial parasite because
A)
it catches fever
done
clear
B)
it is too small to carry parasite
done
clear
C)
the parasite is killed in its stomach
done
clear
D)
it does not drink blood
done
clear
View Answer play_arrow
question_answer 115) Protein present in silk fibre is
A)
casein
done
clear
B)
keratin
done
clear
C)
elastin
done
clear
D)
fibroin
done
clear
View Answer play_arrow
question_answer 116) Ovulation takes place in a month between
A)
11-14 day
done
clear
B)
14-16 day
done
clear
C)
15-28 day
done
clear
D)
21-26 day
done
clear
View Answer play_arrow
question_answer 117) Sugar fructose is present in the secrection of
A)
Bartholin's gland
done
clear
B)
Cowper's gland
done
clear
C)
perineal glands
done
clear
D)
prostate gland
done
clear
View Answer play_arrow
question_answer 118) In sea anemone, the symmetry is
A)
radial
done
clear
B)
bilateral
done
clear
C)
spherical
done
clear
D)
absent
done
clear
View Answer play_arrow
question_answer 119) The colour of bone marrow in foetus is
A)
red
done
clear
B)
yellow
done
clear
C)
brown
done
clear
D)
none of these
done
clear
View Answer play_arrow
question_answer 120) Compound squamous epithelium is found in
A)
stomach
done
clear
B)
intestine
done
clear
C)
trachea
done
clear
D)
pharynx
done
clear
View Answer play_arrow
question_answer 121) Sacculus rotundus is a dilated part at posterior end of
A)
ileum
done
clear
B)
oesophagus
done
clear
C)
ilium
done
clear
D)
colon
done
clear
View Answer play_arrow
question_answer 122) Liver of man is
A)
bilobed
done
clear
B)
3-lobed
done
clear
C)
4-lobed
done
clear
D)
5-lobed
done
clear
View Answer play_arrow
question_answer 123) Cysricercus is the larval stage of
A)
pinworm
done
clear
B)
tapeworm
done
clear
C)
roundworm
done
clear
D)
liver fluke
done
clear
View Answer play_arrow
question_answer 124) Solenocytes are associated with
A)
respiration
done
clear
B)
digestion
done
clear
C)
excretion
done
clear
D)
nutrition
done
clear
View Answer play_arrow
question_answer 125) Which of following organ can be called a son of "blood bank" ?
A)
Heart
done
clear
B)
Spleen
done
clear
C)
Liver
done
clear
D)
Lungs
done
clear
View Answer play_arrow
question_answer 126) Colle's fracture is associated with
A)
femur
done
clear
B)
ulna
done
clear
C)
humerus
done
clear
D)
radius
done
clear
View Answer play_arrow
question_answer 127) The vital capacity of human being is about
A)
1200 mL
done
clear
B)
4800 mL
done
clear
C)
2400 mL
done
clear
D)
3600 mL
done
clear
View Answer play_arrow
question_answer 128) The percentage of \[{{O}_{2}}\]in inhaled air is about
A)
21%
done
clear
B)
16%
done
clear
C)
79%
done
clear
D)
4%
done
clear
View Answer play_arrow
question_answer 129) The gland often referred in relation to AIDS is
A)
adrenal
done
clear
B)
thyroid
done
clear
C)
thymus
done
clear
D)
pituitary
done
clear
View Answer play_arrow
question_answer 130) The hormone which stimulates constriction of gall bladder
A)
gastrin
done
clear
B)
secretin
done
clear
C)
glucagon
done
clear
D)
cholecystokinin
done
clear
View Answer play_arrow
question_answer 131) Broca's area is connected with
A)
learning and reasoning
done
clear
B)
speech function
done
clear
C)
receiving the impulses from eyes
done
clear
D)
sensation of smell
done
clear
View Answer play_arrow
question_answer 132) The cavity of diencephalon is known as
A)
\[\text{I}\] ventricle
done
clear
B)
\[\text{II}\] ventricle
done
clear
C)
\[\text{III}\] ventricle
done
clear
D)
iter
done
clear
View Answer play_arrow
question_answer 133) Normal level of urea in blood plasma is
A)
80-100 mg/100mL blood
done
clear
B)
18-38 mg/100 mL blood
done
clear
C)
30-40 mg/100 mL blood
done
clear
D)
1-10mg/100 mL blood
done
clear
View Answer play_arrow
question_answer 134) Sporogony of malarial parasite occurs in
A)
liver of man
done
clear
B)
RBCs of man
done
clear
C)
salivary glands of mosquito
done
clear
D)
stomach wall of mosquito
done
clear
View Answer play_arrow
question_answer 135) Which vitamin should not be stored ?
A)
Calciferol
done
clear
B)
Retinol
done
clear
C)
Niacin
done
clear
D)
Ascorbic acid
done
clear
View Answer play_arrow
question_answer 136) Which one of the following has an open circulatory system ?
A)
Pheretima
done
clear
B)
Periplaneta
done
clear
C)
Hirudinaria
done
clear
D)
Octopus
done
clear
View Answer play_arrow
question_answer 137) The cranial capacity of Peking man was about
A)
900 cc
done
clear
B)
1660 cc
done
clear
C)
1075 cc
done
clear
D)
1450 cc
done
clear
View Answer play_arrow
question_answer 138) Connecting link between annelids and mollusks is
A)
Neopilina
done
clear
B)
Peripatus
done
clear
C)
Periplaneta
done
clear
D)
Limulus
done
clear
View Answer play_arrow
question_answer 139) Labium majora of a female mammal is homologous to
A)
penis
done
clear
B)
prostate gland
done
clear
C)
epididymis
done
clear
D)
scrotal sac
done
clear
View Answer play_arrow
question_answer 140) Eustachian canal connects
A)
middle ear with external ear
done
clear
B)
middle ear with internal ear
done
clear
C)
external ear with internal ear
done
clear
D)
pharynx with middle ear
done
clear
View Answer play_arrow
question_answer 141) Shell of molluscs is derived from
A)
foot
done
clear
B)
mantle
done
clear
C)
ctenidia
done
clear
D)
placoid
done
clear
View Answer play_arrow
question_answer 142) Which is common between earthworm, leech and centipede ?
A)
They have Malpighian tubules
done
clear
B)
They are hermaphrodite
done
clear
C)
They have ventral nerve cord
done
clear
D)
They have no legs
done
clear
View Answer play_arrow
question_answer 143) Pneumatic bones are expected to be found in
A)
house lizard
done
clear
B)
flying fish
done
clear
C)
pigeon
done
clear
D)
tadpole of frog
done
clear
View Answer play_arrow
question_answer 144) The velocity of conduction of nerve impulse in frog is
A)
300 m/s
done
clear
B)
same as of electricity
done
clear
C)
faster than sound
done
clear
D)
30 m/s
done
clear
View Answer play_arrow
question_answer 145) Which compound has very important role in prebiotic evolution ?
A)
\[S{{O}_{2}}\]
done
clear
B)
NO
done
clear
C)
\[C{{H}_{4}}\]
done
clear
D)
\[S{{O}_{3}}\]
done
clear
View Answer play_arrow
question_answer 146) How many sperm cells are present in an average (3 cc) ejaculation?
A)
200 million
done
clear
B)
300 million
done
clear
C)
400 million
done
clear
D)
500 million
done
clear
View Answer play_arrow
question_answer 147) A man of 'A' blood group marries a woman of AB' blood group, which type of progeny would indicate that man is heterozygous ?
A)
O
done
clear
B)
B
done
clear
C)
A
done
clear
D)
AB
done
clear
View Answer play_arrow
question_answer 148) Which one of the following is not a vestigial structure in Homo sapiens ?
A)
Third molar
done
clear
B)
Epiglottis
done
clear
C)
Plica semilunaris
done
clear
D)
Pyramidalis muscle
done
clear
View Answer play_arrow
question_answer 149) Balbiani rings are the structural features of
A)
allosomes
done
clear
B)
polytene chromosomes
done
clear
C)
autosomes
done
clear
D)
Lampbrush chromosomes
done
clear
View Answer play_arrow
question_answer 150) The permanent decrease in population number occurs due to
A)
migration
done
clear
B)
natality
done
clear
C)
emigration
done
clear
D)
mortality
done
clear
View Answer play_arrow
question_answer 151) Benthic organisms are found in
A)
surface of marine water
done
clear
B)
middle of water in sea
done
clear
C)
bottom of sea
done
clear
D)
on ground
done
clear
View Answer play_arrow
question_answer 152) In Whittaker's system of classification, prokaryotes belong to the kingdom
A)
Monera
done
clear
B)
Protista
done
clear
C)
Animalia
done
clear
D)
Fungi
done
clear
View Answer play_arrow
question_answer 153) The process which cannot cake place in the absence of viruses is
A)
transformation
done
clear
B)
conjugation
done
clear
C)
translocation
done
clear
D)
transduction
done
clear
View Answer play_arrow
question_answer 154) Water bloom is generally caused by
A)
green algae
done
clear
B)
blue-green algae
done
clear
C)
bacteria
done
clear
D)
Hydrilla
done
clear
View Answer play_arrow
question_answer 155) Clinging roots are found in
A)
Orchids
done
clear
B)
Trapa
done
clear
C)
Podostemon
done
clear
D)
Screwpine
done
clear
View Answer play_arrow
question_answer 156) Winged petioles are characteristic of
A)
Podostemon
done
clear
B)
Citrus
done
clear
C)
neem
done
clear
D)
banana
done
clear
View Answer play_arrow
question_answer 157) Seed may be defined as
A)
ripened ovule
done
clear
B)
fertilized ovary
done
clear
C)
the gynoecium
done
clear
D)
embryo
done
clear
View Answer play_arrow
question_answer 158) Which one of die following is a saprophytic Biyophyte ?
A)
Ricda fluitans
done
clear
B)
Buxbaumia aphylla
done
clear
C)
Funaria hygrometrica
done
clear
D)
Sphagnum
done
clear
View Answer play_arrow
question_answer 159) A genophore is made up of
A)
a single double stranded DNA
done
clear
B)
a single stranded DNA
done
clear
C)
UNA and histories
done
clear
D)
histones and non-histones
done
clear
View Answer play_arrow
question_answer 160) Who was awarded Nobel Prize for the synthesis of RNA?
A)
A Kornberg
done
clear
B)
H Khorana
done
clear
C)
Nirenberg
done
clear
D)
S Ochoa
done
clear
View Answer play_arrow
question_answer 161) Potato leaf roll or leaf curl of papaya are caused by
A)
fungi
done
clear
B)
viruses
done
clear
C)
bacteria
done
clear
D)
nematodes
done
clear
View Answer play_arrow
question_answer 162) Iodine is obtained from
A)
Laminaria
done
clear
B)
Chlorella
done
clear
C)
Potysiphonia
done
clear
D)
Porphyra
done
clear
View Answer play_arrow
question_answer 163) Covered smut of barley is caused by
A)
Ustilago hordei
done
clear
B)
Tilltetia caries
done
clear
C)
Ustilago nuda
done
clear
D)
Colletotrichum falcatum
done
clear
View Answer play_arrow
question_answer 164) Retort cells occur in
A)
Funaria
done
clear
B)
Pogonatum
done
clear
C)
Porella
done
clear
D)
Sphagnum
done
clear
View Answer play_arrow
question_answer 165) The jute fibres anatomically are
A)
bast fibres
done
clear
B)
cortical fibres
done
clear
C)
xylem fibres
done
clear
D)
pith fibres
done
clear
View Answer play_arrow
question_answer 166) Phloem parenchyma is absent in
A)
monocot stem
done
clear
B)
dicotstem
done
clear
C)
dicot root
done
clear
D)
dicot leaf
done
clear
View Answer play_arrow
question_answer 167) Parachute type dispersal occurs in
A)
tomato
done
clear
B)
mustard
done
clear
C)
pea
done
clear
D)
cotton
done
clear
View Answer play_arrow
question_answer 168) Root hair absorbs water from soil through
A)
turgor pressure
done
clear
B)
ion exchange
done
clear
C)
osmosis
done
clear
D)
DPD
done
clear
View Answer play_arrow
question_answer 169) Plants need one of the following for ATP formation
A)
N, P
done
clear
B)
N, Cu
done
clear
C)
N.Ca
done
clear
D)
K
done
clear
View Answer play_arrow
question_answer 170) In CAM plants, \[C{{O}_{2}}\]acceptor is
A)
RuBP
done
clear
B)
PEP
done
clear
C)
OAA
done
clear
D)
PGA
done
clear
View Answer play_arrow
question_answer 171) The energy currency of cell is
A)
NAD
done
clear
B)
GDP
done
clear
C)
RNA
done
clear
D)
ATP
done
clear
View Answer play_arrow
question_answer 172) Which one of the following is a natural growth inhibitor?
A)
NAA
done
clear
B)
ABA
done
clear
C)
IAA
done
clear
D)
GA
done
clear
View Answer play_arrow
question_answer 173) Which one is a short day plant?
A)
Brossica compestris
done
clear
B)
Rdphanus sativus
done
clear
C)
Glycine max
done
clear
D)
Papaversomniferum
done
clear
View Answer play_arrow
question_answer 174) Florigen is produced in the region of
A)
leaves
done
clear
B)
fruit
done
clear
C)
root
done
clear
D)
trunk
done
clear
View Answer play_arrow
question_answer 175) "Sanjeevani boori" is
A)
Selaginella kraussiana
done
clear
B)
Selaginella chrysocaculos
done
clear
C)
Selaginella bryopteris
done
clear
D)
None of the above
done
clear
View Answer play_arrow
question_answer 176) The process of decay of dead organic matter is known as
A)
denitrification
done
clear
B)
nitrification
done
clear
C)
nitrogen fixation
done
clear
D)
ammonification
done
clear
View Answer play_arrow
question_answer 177) Apical dominance w caused by
A)
auxin
done
clear
B)
gibberellin
done
clear
C)
kinetin
done
clear
D)
ABA
done
clear
View Answer play_arrow
question_answer 178) Cystolith is made up of
A)
silica
done
clear
B)
calcium carbonate
done
clear
C)
calcium oxalate
done
clear
D)
inulin
done
clear
View Answer play_arrow
question_answer 179) Growth of pollen tube towards embryo sac is
A)
geotropism
done
clear
B)
thigmotaxis
done
clear
C)
chemotaxis
done
clear
D)
all of these
done
clear
View Answer play_arrow
question_answer 180) Ozone depletion in stratosphere would result in
A)
forest fires
done
clear
B)
global warming
done
clear
C)
skin cancer incidence
done
clear
D)
none of these
done
clear
View Answer play_arrow
question_answer 181) Molybdenum causes
A)
mottling
done
clear
B)
wilting
done
clear
C)
reclamation
done
clear
D)
chloros is
done
clear
View Answer play_arrow
question_answer 182) Which of the following is a test cross?
A)
\[Tt\times tt\]
done
clear
B)
\[TT\times tt\]
done
clear
C)
\[Tt\times Tt\]
done
clear
D)
\[tt\times tt\]
done
clear
View Answer play_arrow
question_answer 183) Tetradynamous stamens are found in family
A)
Malvaceae
done
clear
B)
Solanaceae
done
clear
C)
Crudferae
done
clear
D)
Liliaceae
done
clear
View Answer play_arrow
question_answer 184) The witches broom of legume is caused by
A)
virus
done
clear
B)
bacteria
done
clear
C)
fungus
done
clear
D)
mycoplasma
done
clear
View Answer play_arrow
question_answer 185) Opium is obtained from
A)
Oryza sativa
done
clear
B)
Coffea Arabica
done
clear
C)
Thea sinensis
done
clear
D)
Papaver somniferum
done
clear
View Answer play_arrow
question_answer 186) A pigment which absorbs red and far red light is
A)
carotene
done
clear
B)
xanthophyll
done
clear
C)
phytochrome
done
clear
D)
cytochrome
done
clear
View Answer play_arrow
question_answer 187) 1:2:1 phenotypic and genotypic ratio is found in
A)
complementary genes
done
clear
B)
blending inheritance
done
clear
C)
multiple alleles
done
clear
D)
pseudo alleles
done
clear
View Answer play_arrow
question_answer 188) From which of the following plant is a medicine for respiratory disorders obtained?
A)
Ephedra
done
clear
B)
Eucalyptus
done
clear
C)
Cannabis
done
clear
D)
Saccharum
done
clear
View Answer play_arrow
question_answer 189) Osmotic pressure of a solution is
A)
greater than pure solvent
done
clear
B)
less than pure solvent
done
clear
C)
equal to pure solvent
done
clear
D)
less than or greater than pure solvent
done
clear
View Answer play_arrow
question_answer 190) Decarboxyladon is involved in
A)
electron transport system
done
clear
B)
glycolysis
done
clear
C)
Krebs' cycle
done
clear
D)
lactic acid fermentation
done
clear
View Answer play_arrow
question_answer 191) Ginger multiplies vegetatively by
A)
tuber
done
clear
B)
corm
done
clear
C)
sucker
done
clear
D)
rhizome
done
clear
View Answer play_arrow
question_answer 192) Damaged sieve tubes are sealed by deposition of
A)
pectin
done
clear
B)
callus
done
clear
C)
suberin
done
clear
D)
lignin
done
clear
View Answer play_arrow
question_answer 193) During meiosis crossing over occurs between which part of homologous chromosome?
A)
Sister chromatids
done
clear
B)
Non-sister chromatids
done
clear
C)
Genes
done
clear
D)
Alleles
done
clear
View Answer play_arrow
question_answer 194) Phytotron is
A)
a controlled condition chamber for tissue culture
done
clear
B)
leaf culture process
done
clear
C)
special culture of plants
done
clear
D)
root culture process
done
clear
View Answer play_arrow
question_answer 195) The backbone of RNA consists of which of the following sugar?
A)
Deoxyribose
done
clear
B)
Ribose
done
clear
C)
Sucrose
done
clear
D)
Maltose
done
clear
View Answer play_arrow
question_answer 196) Pigment present in cyanobacteria is
A)
r-phycocyanin
done
clear
B)
r-phycoerythrin
done
clear
C)
c-phycocyanin
done
clear
D)
anthocyanin
done
clear
View Answer play_arrow
question_answer 197) Stop codons are
A)
\[\text{AUG,}\,\text{GUG}\]
done
clear
B)
\[\text{UAA,}\,\text{UGA,}\,\text{UAG}\]
done
clear
C)
\[\text{UAC,}\,\text{UGG}\]
done
clear
D)
\[\text{AGU,}\,\text{AGA,}\,\text{UAC}\]
done
clear
View Answer play_arrow
question_answer 198) A lake with nutrients is called
A)
trophic
done
clear
B)
euphoric
done
clear
C)
oligotrophic
done
clear
D)
eutrophic
done
clear
View Answer play_arrow
question_answer 199) Expanded green stem of Opuntia is called
A)
phylloclade
done
clear
B)
tendril
done
clear
C)
bulbs
done
clear
D)
cladode
done
clear
View Answer play_arrow
question_answer 200) "Foolish seedling disease" of rice in Japan was caused by
A)
the deficiency of nitrogen
done
clear
B)
a bacterium
done
clear
C)
a fungus
done
clear
D)
a virus
done
clear
View Answer play_arrow
 Choose the correct statement from the following.
Choose the correct statement from the following.
