question_answer 1) Kirchhoffs first law, \[i.,e.,\Sigma i=0\] at a junction, deals with the conservation of:
A)
angular momentum
done
clear
B)
linear momentum
done
clear
C)
energy
done
clear
D)
charge
done
clear
View Answer play_arrow
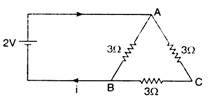
question_answer 2)
The current in the following circuit is :
A)
1 A
done
clear
B)
\[\frac{2}{3}A\]
done
clear
C)
\[\frac{2}{9}A\]
done
clear
D)
\[\frac{1}{8}A\]
done
clear
View Answer play_arrow
question_answer 3) There are three copper wires of length and cross-sectional area (L, A), (2L, (1/2) A), ((1/2) L, 2A). In which case is the resistance minimum?
A)
It is the same in all three cases
done
clear
B)
Wire of cross-sectional area 2 A
done
clear
C)
Wire of cross-sectional area A
done
clear
D)
Wire of cross-sectional area \[\frac{1}{2}A\]
done
clear
View Answer play_arrow
question_answer 4) A 100 W-200 V bulb is connected to a 160 V power supply. The power consumption would be :
A)
125 W
done
clear
B)
100 W
done
clear
C)
80 W
done
clear
D)
64 W
done
clear
View Answer play_arrow
question_answer 5) Two parallel wires in free space arc 10 cm apart, and each carries a current of 10A, in the same direction. The force, one wire exerts on the other, per metre of length, is :
A)
\[2\times {{10}^{-7}}N,\]repulsive
done
clear
B)
\[2\times {{10}^{-7}}N,\]attractive
done
clear
C)
\[2\times {{10}^{-4}}N,\]repulsive
done
clear
D)
\[2\times {{10}^{-4}}N,\]attractive
done
clear
View Answer play_arrow
question_answer 6) A straight wire of diameter 0.5 mm carrying a current of 1A is replaced by another wire of 1 mm diameter carrying the same current. The strength of magnetic field far away is :
A)
unchanged
done
clear
B)
quarter of its earlier value
done
clear
C)
half of the earlier value
done
clear
D)
twice the earlier value
done
clear
View Answer play_arrow
question_answer 7) A positively charged particle moving due east enters a region of uniform magnetic field directed vertically upwards. The particle will :
A)
continue to move due east
done
clear
B)
move in a circular orbit with its speed unchanged
done
clear
C)
move in a circular orbit with its speed increased
done
clear
D)
gets deflected vertically upwards
done
clear
View Answer play_arrow
question_answer 8) In an AC circuit with voltage V and current I the power dissipated is :
A)
depends on the phase between V and I
done
clear
B)
\[\frac{1}{\sqrt{2}}VI\]
done
clear
C)
\[\frac{1}{2}VI\]
done
clear
D)
VI
done
clear
View Answer play_arrow
question_answer 9) The primary winding of transformer has 500 turns whereas its secondary has 5000 turns. The primary is connected to an AC supply of 20 V-50 Hz. The secondary will have an output of:
A)
2 V, 5 Hz
done
clear
B)
200 V, 500 Hz
done
clear
C)
2V, 50 Hz
done
clear
D)
200 V, 50 Hz
done
clear
View Answer play_arrow
question_answer 10) Due to the ends magnetic field, charged cosmic ray particles :
A)
can never reach the poles
done
clear
B)
can never reach the equator
done
clear
C)
require less kinetic energy to reach the equator than the poles
done
clear
D)
require greater kinetic energy to reach the equator, than the poles
done
clear
View Answer play_arrow
question_answer 11) If \[{{\varepsilon }_{0}}\]and \[{{\mu }_{0}}\] are respectively the electric permittivity and magnetic permeability of free space, \[\varepsilon \]and\[\mu \] are the corresponding quantities in a medium, the index of refraction of the medium is :
A)
\[\sqrt{\frac{{{\varepsilon }_{0}}{{\mu }_{0}}}{\varepsilon \mu }}\]
done
clear
B)
\[\sqrt{\frac{\varepsilon \mu }{{{\varepsilon }_{0}}{{\mu }_{0}}}}\]
done
clear
C)
\[\sqrt{\frac{{{\varepsilon }_{0}}\mu }{\varepsilon {{\mu }_{0}}}}\]
done
clear
D)
\[\sqrt{\frac{\varepsilon }{{{\varepsilon }_{0}}}}\]
done
clear
View Answer play_arrow
question_answer 12) The kinetic energy of an accelerated through a potential of 100 V is :
A)
\[1.60\times {{10}^{-17}}J\]
done
clear
B)
\[1.16\times {{10}^{4}}k\]
done
clear
C)
418.6 cal
done
clear
D)
\[6.626\times {{10}^{-34}}Ws\]
done
clear
View Answer play_arrow
question_answer 13) The most penetrating radiation out of the following is :
A)
gamma-rays
done
clear
B)
alpha particles
done
clear
C)
beta-rays
done
clear
D)
X-rays
done
clear
View Answer play_arrow
question_answer 14) When hydrogen atom is in its first excited level, its radius is :
A)
four times, its ground state radius
done
clear
B)
twice, its ground state radius
done
clear
C)
same as its ground state radius
done
clear
D)
half of its ground state radius
done
clear
View Answer play_arrow
question_answer 15) The energy of ground electronic state of hydrogen atom is - 13.6 eV. The energy of the first excited state will be :
A)
- 54.4 eV
done
clear
B)
-27.2eV
done
clear
C)
- 6.8 eV
done
clear
D)
- 3.4 eV
done
clear
View Answer play_arrow
question_answer 16) Which of the following is true?
A)
The stopping potential increases with increasing intensity of incident light
done
clear
B)
The photo current increases with increasing intensity of light
done
clear
C)
The current in photocell increases with increasing frequency of light
done
clear
D)
The photo current is proportional to applied voltage
done
clear
View Answer play_arrow
question_answer 17) The activity of a radioactive sample is measured as 9750 counts per minute at t = 0 and as 975 counts per minute at T = 5 minutes. The decay constant is approximately:
A)
0.922 per minute
done
clear
B)
0.691 per minute
done
clear
C)
0.461 per minute
done
clear
D)
0.230 per minute
done
clear
View Answer play_arrow
question_answer 18) Which of the following is used as a moderator in nuclear reactors?
A)
Plutonium
done
clear
B)
Cadmium
done
clear
C)
Heavy water
done
clear
D)
Uranium
done
clear
View Answer play_arrow
question_answer 19) The stable nucleus that has a radius half that of\[F{{e}^{56}}\] is :
A)
\[L{{i}^{7}}\]
done
clear
B)
\[N{{a}^{21}}\]
done
clear
C)
\[{{S}^{16}}\]
done
clear
D)
\[C{{a}^{40}}\]
done
clear
View Answer play_arrow
question_answer 20) A free neutron decays into a proton, an electron and :
A)
a beta particle
done
clear
B)
an alphaparticle
done
clear
C)
an antineutrino
done
clear
D)
a neutrino
done
clear
View Answer play_arrow
question_answer 21) In a fission reaction \[_{92}^{236}U{{\to }^{117}}X{{+}^{117}}Y+n+n\]the binding energy per nucleon of X and Y is 8.5 MeV whereas of 236U is 7.6 MeV. The total energy liberated will be about:
A)
2000 MeV
done
clear
B)
200 MeV
done
clear
C)
2 MeV
done
clear
D)
200 MeV
done
clear
View Answer play_arrow
question_answer 22) To obtain a p-type germanium semiconductor, it must be doped with :
A)
phosphorus
done
clear
B)
indium
done
clear
C)
antimony
done
clear
D)
arsenic
done
clear
View Answer play_arrow
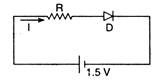
question_answer 23)
The diode used in the circuit shown in the figure has a constant voltage drop of 0.5 V at all currents and a maximum power rating of 100 mill watt. What should be the value of the resistor R, connected in series with the diode, for obtaining maximum current?
A)
200\[\Omega \]
done
clear
B)
6.67 \[\Omega \]
done
clear
C)
5\[\Omega \]
done
clear
D)
1.5\[\Omega \]
done
clear
View Answer play_arrow
question_answer 24) The correct relationship between the two current gains a and p in a transistor is :
A)
\[\beta =\frac{1+\alpha }{\beta }\]
done
clear
B)
\[\alpha =\frac{\beta }{1+\beta }\]
done
clear
C)
\[\alpha =\frac{\beta }{1-\beta }\]
done
clear
D)
\[\beta =\frac{\alpha }{1+\alpha }\]
done
clear
View Answer play_arrow
question_answer 25)
The following truth table belongs to which of the following four gates?
A
B
C
1
1
0
1
0
0
0
1
0
0
0
1
A)
NOR
done
clear
B)
XOR
done
clear
C)
NAND
done
clear
D)
OR
done
clear
View Answer play_arrow
question_answer 26) The force F on a sphere of radius r moving in a medium with velocity v given by\[F=6\pi \,\eta rv\]. The dimensions of r) are :
A)
\[\left[ M{{L}^{-3}} \right]\]
done
clear
B)
\[\left[ ML{{T}^{2}} \right]\]
done
clear
C)
\[\left[ M{{T}^{-1}} \right]\]
done
clear
D)
\[\left[ M{{L}^{-1}}{{T}^{-1}} \right]\]
done
clear
View Answer play_arrow
question_answer 27) Find the torque of a force \[\mathbf{\vec{F}}=-3\mathbf{\hat{i}}+\mathbf{\hat{j}}+5\mathbf{\hat{k}}\]acting at the point \[\vec{r}=-7\mathbf{\hat{i}}+3\mathbf{\hat{j}}+16\mathbf{\hat{k}}\]:
A)
\[-21\mathbf{\hat{i}}+3\mathbf{\hat{j}}+5\mathbf{\hat{k}}\]
done
clear
B)
\[-14\mathbf{\hat{i}}+3\mathbf{\hat{j}}+16\mathbf{\hat{k}}\]
done
clear
C)
\[4\mathbf{\hat{i}}+4\mathbf{\hat{j}}+6\mathbf{\hat{k}}\]
done
clear
D)
\[14\mathbf{\hat{i}}+38\mathbf{\hat{j}}+16\mathbf{\hat{k}}\]
done
clear
View Answer play_arrow
question_answer 28) The position x of a particle varies with time t, as\[x=a{{t}^{2}}-d{{t}^{3}}\]. The acceleration of the particle will be zero at time t equal to :
A)
zero
done
clear
B)
\[\frac{a}{3b}\]
done
clear
C)
\[\frac{2a}{3b}\]
done
clear
D)
\[\frac{a}{b}\]
done
clear
View Answer play_arrow
question_answer 29) If a car at rest accelerates uniformly to a speed of 144 km/h in 20 s, it covers a distance of :
A)
2880 m
done
clear
B)
1440 m
done
clear
C)
400 m
done
clear
D)
20 m
done
clear
View Answer play_arrow
question_answer 30) A ball, whose kinetic energy is E, is thrown at an angle of \[45{}^\circ \] with the horizontal, its kinetic energy at the highest point of its flight will be :
A)
zero
done
clear
B)
E/2
done
clear
C)
\[E/\sqrt{2}\]
done
clear
D)
E
done
clear
View Answer play_arrow
question_answer 31) Two bodies of masses m and 4m are moving with equal kinetic energy. The ratio of their linear momenta is :
A)
1 : 4
done
clear
B)
1 : 2
done
clear
C)
1 : 1
done
clear
D)
4 : 1
done
clear
View Answer play_arrow
question_answer 32) In a carbon monoxide molecule, the carbon and the oxygen atoms are separated by a distance \[1.12\times {{10}^{-10}}m\]. The distance of the centre of mass, from the carbon atom is :
A)
\[o.64\times {{10}^{-10}}m\]
done
clear
B)
\[o.56\times {{10}^{-10}}m\]
done
clear
C)
\[o.51\times {{10}^{-10}}m\]
done
clear
D)
\[o.48\times {{10}^{-10}}m\]
done
clear
View Answer play_arrow
question_answer 33) A cart of mass M is tied to one end of a massless rope of length 10 m. The other end of the rope is in the hands of a man of mass M. The entire system is on a smooth horizontal surface. The man is at x = 0 and the cart at x = 10 m. If the man pulls the cart by the rope, the man and the cart will meet at the point :
A)
they will never meet
done
clear
B)
x = 10 m
done
clear
C)
x= 5m
done
clear
D)
x = 0
done
clear
View Answer play_arrow
question_answer 34) A metal ball of mass 2 kg moving with a velocity of 36 km/h has a head on collision with a stationary ball of mass 3 kg. If after the collision, the two balls move together, the loss in kinetic energy due to collision is :
A)
140 J
done
clear
B)
100 J
done
clear
C)
60 J
done
clear
D)
40 J
done
clear
View Answer play_arrow
question_answer 35) A couple produces :
A)
no motion
done
clear
B)
linear and rotational motion
done
clear
C)
purely rotational motion
done
clear
D)
purely linear motion
done
clear
View Answer play_arrow
question_answer 36) The escape velocity of a body on the surface of the earth is 11.2 km/s. If the earths mass increases to twice its present value and the radius of the earth becomes half, the escape velocity would become :
A)
44.8 km/s
done
clear
B)
22.4 km/s
done
clear
C)
11.2 km/s (remain unchanged)
done
clear
D)
5.6 km/s
done
clear
View Answer play_arrow
question_answer 37) The period of revolution of planet A round the sun is 8 times that of B. The distance of A from the sun is how many times greater than that of B from the sun?
A)
5
done
clear
B)
4
done
clear
C)
3
done
clear
D)
2
done
clear
View Answer play_arrow
question_answer 38) A hollow sphere is filled with water. It is hung by a long thread. As the water flows out of a hole at the bottom, the period of oscillation will:
A)
first increase and then decrease
done
clear
B)
first decrease and then increase
done
clear
C)
go on increasing
done
clear
D)
go on decreasing
done
clear
View Answer play_arrow
question_answer 39) Two simple harmonic motions with the same frequency act on a particle at right angles i.e., along x and y axis. If the two amplitudes are equal and the phase difference is \[\pi \]/2, the resultant motion will be :
A)
a circle
done
clear
B)
an ellipse with the major axis along y-axis
done
clear
C)
an ellipse with the major axis along x-axis
done
clear
D)
a straight line inclined at 45° to the x-axis
done
clear
View Answer play_arrow
question_answer 40) The equation of a travelling wave is : y = 60 cos (18001 - 6x)where y is in microns, t in second and x in metres. The ratio of maximum particle velocity to velocity of wave propagation is :
A)
3.6
done
clear
B)
\[3.6\times {{10}^{-4}}\]
done
clear
C)
\[3.6\times {{10}^{-6}}\]
done
clear
D)
\[3.6\times {{10}^{11}}\]
done
clear
View Answer play_arrow
question_answer 41) Standing waves are produced in a 10 m long stretched string. If the string vibrates in 5 segments and the wave velocity is 20 m/s, the frequency is :
A)
10 Hz
done
clear
B)
5 Hz
done
clear
C)
4 Hz
done
clear
D)
2 Hz
done
clear
View Answer play_arrow
question_answer 42) A pulse of a wave train travels along a stretched string and reaches the fixed end of the string. It will be reflected back with :
A)
a phase change of 180° with velocity reversed
done
clear
B)
the same phase as the incident pulse with no reversal of velocity
done
clear
C)
a phase change of 180° with no reversal of velocity
done
clear
D)
the same phase as the incident pulse but with velocity reversed
done
clear
View Answer play_arrow
question_answer 43) A cylindrical resonance tube open at both ends, has a fundamental frequency\[f\], in air. If half of the length is dipped vertically in water, the fundamental frequency of the air column will be:
A)
\[2f\]
done
clear
B)
\[3f/2\]
done
clear
C)
\[f\]
done
clear
D)
\[f\]/2
done
clear
View Answer play_arrow
question_answer 44) A sample of gas expands from volume \[{{\text{V}}_{\text{1}}}\]to \[{{\text{V}}_{\text{2}}}\text{.}\]The amount of work done by the gas is greatest when the expansion is :
A)
adiabatic
done
clear
B)
isobaric
done
clear
C)
isothermal
done
clear
D)
equal in all above cases
done
clear
View Answer play_arrow
question_answer 45) A black body is at temperature of 500 K. It emits energy at rate which is proportional to :
A)
\[{{(500)}^{4}}\]
done
clear
B)
\[{{(500)}^{3}}\]
done
clear
C)
\[{{(500)}^{2}}\]
done
clear
D)
500
done
clear
View Answer play_arrow
question_answer 46) The efficiency of a Carnot engine operating between temperatures of 100°C and - 23°C will be :
A)
\[\frac{100-23}{273}\]
done
clear
B)
\[\frac{100+23}{373}\]
done
clear
C)
\[\frac{100+23}{100}\]
done
clear
D)
\[\frac{100-23}{100}\]
done
clear
View Answer play_arrow
question_answer 47) The focal lengths of a converging lens measured for violet, green and red colours are \[{{f}_{v\,}},{{f}_{g,}}{{f}_{r}}\]respectively. We will find :
A)
\[{{f}_{g}}>{{f}_{r}}\]
done
clear
B)
\[{{f}_{v}}<{{f}_{r}}\]
done
clear
C)
\[{{f}_{v}}>{{f}_{r}}\]
done
clear
D)
\[{{f}_{v}}={{f}_{r}}\]
done
clear
View Answer play_arrow
question_answer 48) An astronomical telescope of ten-fold angular magnification has a length of 44 cm. The focal length of the objective is :
A)
440 cm
done
clear
B)
44 cm
done
clear
C)
40 cm
done
clear
D)
4 cm
done
clear
View Answer play_arrow
question_answer 49) Electromagnetic radiation of frequency n, velocity v and wavelength \[\lambda \], in air, enters a glass slab of refractive index \[\mu \]. The frequency, wavelength and velocity of light in the glass slab will be, respectively:
A)
\[\frac{n}{\mu },\frac{\lambda }{\mu },v\]
done
clear
B)
\[n,\lambda \frac{v}{\mu }\]
done
clear
C)
\[n,\frac{\lambda }{\mu },\frac{v}{\mu }\]
done
clear
D)
\[\frac{n}{\mu },\frac{\lambda }{\mu },\frac{v}{\mu }\]
done
clear
View Answer play_arrow
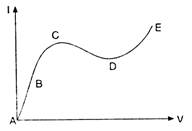
question_answer 50)
From the graph between current I and voltage V shown in figure, identify the portion corresponding to negative resistance:
A)
DE
done
clear
B)
CD
done
clear
C)
BC
done
clear
D)
AB
done
clear
View Answer play_arrow
question_answer 51) Which sequence correctly describes a relative bond strength of oxygen molecule, super-oxide ion and peroxide ion:
A)
\[{{O}_{2}}>O_{2}^{-}>O_{2}^{2-}\]
done
clear
B)
\[{{O}_{2}}>O_{2}^{-}<O_{2}^{2-}\]
done
clear
C)
\[{{O}_{2}}<O_{2}^{-}>O_{2}^{2-}\]
done
clear
D)
\[{{O}_{2}}<O_{2}^{-}<O_{2}^{2-}\]
done
clear
View Answer play_arrow
question_answer 52) 1 mole of \[C{{H}_{4}}\] contains:
A)
3.0g of carbon
done
clear
B)
\[1.81\times {{10}^{23}}\] molecules of \[C{{H}_{4}}\]
done
clear
C)
4 g atoms of hydrogen
done
clear
D)
\[6.02\times {{10}^{23}}\] atoms of H
done
clear
View Answer play_arrow
question_answer 53) In two H-atoms A and B the electrons move around the nucleus in-circular orbit of radius Y and 4r respectively. The ratio of the times taken by them to complete one revolution is:
A)
\[1 : 2\]
done
clear
B)
\[2 : 1\]
done
clear
C)
\[1 : 4\]
done
clear
D)
\[1 : 8\]
done
clear
View Answer play_arrow
question_answer 54)
The table given below lists the bond dissociation energy \[({{E}_{diss}})\]for single co-valent bonds formed between C and atoms A, B, D, E Bond \[({{E}_{diss}})\,(kcal\,-\,mo{{l}^{-1}})\] C-A 240 C-B 382 C-D 276 C-E 486
Which of the atoms has smallest size?
A)
E
done
clear
B)
B
done
clear
C)
D
done
clear
D)
A
done
clear
View Answer play_arrow
question_answer 55) The ionisation energy of gaseous Na atom is \[495.8\text{ }kJ\text{ }mo{{l}^{-1}}\]. The lowest possible frequency of light that can ionise a Na atom is:
A)
\[1.24\times {{10}^{12}}{{s}^{-1}}\]
done
clear
B)
\[3.15\times {{10}^{14}}{{s}^{-1}}\]
done
clear
C)
\[4.76\times {{10}^{14}}{{s}^{-1}}\]
done
clear
D)
\[1.24\times {{10}^{15}}{{s}^{-1}}\]
done
clear
View Answer play_arrow
question_answer 56) A sudden large jump between the values of second and third ionization energy of an element would be associated with the electronic configuration:
A)
\[1{{s}^{2}},2{{s}^{2}}2{{p}^{6}},3{{s}^{2}},3{{p}^{3}}\]
done
clear
B)
\[1{{s}^{2}},2{{s}^{2}}2{{p}^{6}},3{{s}^{2}}\]
done
clear
C)
\[1{{s}^{2}},2{{s}^{2}}2{{p}^{6}},3{{s}^{1}}\]
done
clear
D)
\[1{{s}^{2}},2{{s}^{2}}2{{p}^{6}},3{{s}^{2}}3{{p}^{1}}\]
done
clear
View Answer play_arrow
question_answer 57) A \[\sigma \] bonded molecule \[M{{X}_{3}}\] is T-shaped. The number of non-bonding pair of electrons is:
A)
1
done
clear
B)
3
done
clear
C)
2
done
clear
D)
can be predicted only if atomic number of M is known
done
clear
View Answer play_arrow
question_answer 58) The value of \[\Delta {{H}_{O-H}}\] n is\[109\text{ }kcal\text{ }mo{{l}^{-1}}\]. Then formation of one mole of water in gaseous state from H(g) ang O(g) is accompanied by:
A)
absorption of 218 kcal of energy
done
clear
B)
release of 218 kcal of energy
done
clear
C)
release of 109 kcal of energy
done
clear
D)
unpredictable
done
clear
View Answer play_arrow
question_answer 59) One of the following is Bronsted acid but not a Bronsted base:
A)
\[{{H}_{2}}O\]
done
clear
B)
\[N{{H}_{3}}\]
done
clear
C)
\[{{H}_{2}}S\]
done
clear
D)
\[HCO_{3}^{-}\]
done
clear
View Answer play_arrow
question_answer 60) Calculate the heat of following reaction \[Na(s)+0.5C{{l}_{2}}(g)\xrightarrow{{}}NaCl(s);\Delta {{H}^{o}}?\] Given \[{{H}_{2}}(g)+C{{l}_{2}}(g)\xrightarrow{{}}2HCl,\]\[\Delta {{H}^{o}}=152kcal\] \[2Na(s)+2HCl(g)\xrightarrow{{}}2NaCl(s)+{{H}_{2}}(g),\]\[\Delta {{H}^{o}}=152kcal\]
A)
\[54\text{ }kcal\]
done
clear
B)
\[\text{98 }kcal\]
done
clear
C)
\[\text{196 }kcal\]
done
clear
D)
\[\text{108 }kcal\]
done
clear
View Answer play_arrow
question_answer 61) The equilibrium constant for the reaction \[N{{H}_{4}}N{{O}_{2}}(s){{N}_{2}}(g)+2{{H}_{2}}O(g)\] is given by:
A)
\[\frac{[{{N}_{2}}]{{[{{H}_{2}}O]}^{2}}}{[N{{H}_{4}}N{{O}_{2}}]}\]
done
clear
B)
\[\frac{[{{N}_{2}}]{{[2{{H}_{2}}O]}^{2}}}{{{[N{{H}_{4}}N{{O}_{2}}]}^{2}}}\]
done
clear
C)
\[\frac{[N{{H}_{4}}N{{O}_{2}}]}{[{{N}_{2}}]{{[{{H}_{2}}O]}^{2}}}\]
done
clear
D)
\[[{{N}_{2}}]{{[{{H}_{2}}O]}^{2}}\]
done
clear
View Answer play_arrow
question_answer 62) In a closed vessel of volume V, a moles of nitrogen and b moles of oxygen are made to react to give nitric oxide, according to reactio\[{{N}_{2}}+{{O}_{2}}2NO\] If at equilibrium, 2x of moles of NO are obtained then:
A)
\[{{K}_{c}}=\frac{4{{x}^{2}}}{(a-x)\,(b-x)}.\frac{1}{V}\]
done
clear
B)
\[{{K}_{c}}=\frac{4{{x}^{2}}}{(a-x)\,(b-x)}.V\]
done
clear
C)
\[{{K}_{c}}=\frac{{{x}^{2}}}{(a-x)\,(b-x)}.V\]
done
clear
D)
\[{{K}_{c}}=\frac{4{{x}^{2}}}{(a-x)\,(b-x)}\]
done
clear
View Answer play_arrow
question_answer 63) The molality of 1 L solution of \[93%{{H}_{2}}S{{O}_{4}}\](w/v) having density \[1.84\text{ }g/mL\] is:
A)
\[1.043\text{ }m\]
done
clear
B)
\[0.143m\]
done
clear
C)
10.43 m
done
clear
D)
none of these
done
clear
View Answer play_arrow
question_answer 64) Reaction that takes place at graphite anode in dry cell is:
A)
\[M{{n}^{+}}+2{{e}^{-}}\xrightarrow{{}}Mn(s)\]
done
clear
B)
\[Mn(s)+\xrightarrow{{}}M{{n}^{+}}+{{e}^{-}}+1.5V\]
done
clear
C)
\[Z{{n}^{2+}}(s)+2{{e}^{-}}\xrightarrow{{}}Zn(s)\]
done
clear
D)
\[Zn(s)\xrightarrow{{}}Z{{n}^{2+}}+2{{e}^{-}}\]
done
clear
View Answer play_arrow
question_answer 65) The radius of \[N{{a}^{+}}\] is 95 pm and that of \[C{{l}^{-}}\] ion is 181 pm. Predict the co-ordination number of \[N{{a}^{+}}\].
A)
8
done
clear
B)
6
done
clear
C)
4
done
clear
D)
unpredictable
done
clear
View Answer play_arrow
question_answer 66) A reaction \[2A\to B\] follows a second order kinetics. Doubling the concentration of A will increase the rate of formation of B by a factor of:
A)
\[1/4\]
done
clear
B)
4
done
clear
C)
\[1/2\]
done
clear
D)
2
done
clear
View Answer play_arrow
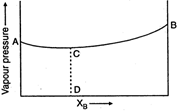
question_answer 67)
The diagram given above is a vapour pressure composition diagram for a binary solution of A and B. In the solution A......B interactions are:
A)
smaller than A....... A and B.......B interactions
done
clear
B)
similar to A........ A and B........B interactions
done
clear
C)
greater than A ........ A and B........ B interactions
done
clear
D)
unpredictable
done
clear
View Answer play_arrow
question_answer 68) Calculate the volume occupied by 7.0 g of nitrogen gas at \[{{27}^{o}}C\] and 750 mm Hg pressure.
A)
\[62.32\text{ }L\]
done
clear
B)
\[6.232\text{ }L\]
done
clear
C)
\[623.2\text{ }L~\]
done
clear
D)
\[0.623\text{ }L\]
done
clear
View Answer play_arrow
question_answer 69) If \[_{92}{{U}^{235}}\] is assumed to decay only by emitting two a and one \[\beta \]- particles. The possible product of decay is;
A)
\[_{89}A{{c}^{277}}\]
done
clear
B)
\[_{89}A{{c}^{235}}\]
done
clear
C)
\[_{89}A{{c}^{211}}\]
done
clear
D)
\[_{89}A{{c}^{237}}\]
done
clear
View Answer play_arrow
question_answer 70) The colour of \[{{K}_{2}}C{{r}_{2}}{{O}_{7}}\] changes from red orange to lemon yellow on treatment with aqueous \[KOH\] because of:
A)
oxidation of potassium hydroxide to potassium peroxide
done
clear
B)
conversion of dichromate ion to chromate
done
clear
C)
reduction of \[Cr\] (VI) to \[Cr\] (III)
done
clear
D)
formation of chromium hydroxide
done
clear
View Answer play_arrow
question_answer 71) Which one of the following metals cannot be extracted by using Al as a reducing agent?
A)
\[W\] from \[W{{O}_{3}}\]
done
clear
B)
\[Mn\] from \[M{{n}_{3}}{{O}_{4}}\]
done
clear
C)
\[Cr\] from \[C{{r}_{2}}{{O}_{3}}\]
done
clear
D)
\[Na\] from \[N{{a}_{2}}O\]
done
clear
View Answer play_arrow
question_answer 72) The numbers of a and P particles emitted in the transformation \[_{92}{{U}^{238}}{{\xrightarrow{{}}}_{92}}{{U}^{234}}\]
A)
\[2,1\]
done
clear
B)
\[1,2\]
done
clear
C)
\[1,1\]
done
clear
D)
\[1,0\]
done
clear
View Answer play_arrow
question_answer 73) The maximum bond angle in hydrides of group 16-elements is in:
A)
\[{{H}_{2}}Te\]
done
clear
B)
\[{{H}_{2}}Se\]
done
clear
C)
\[{{H}_{2}}S\]
done
clear
D)
\[{{H}_{2}}O\]
done
clear
View Answer play_arrow
question_answer 74) \[N{{O}_{2}}\] is obtained by heating :
A)
\[NaN{{O}_{3}}\]
done
clear
B)
\[AgN{{O}_{3}}\]
done
clear
C)
\[CsN{{O}_{3}}\]
done
clear
D)
\[KN{{O}_{3}}\]
done
clear
View Answer play_arrow
question_answer 75) An ingredient of baking powder is:
A)
sodium bicarbonate
done
clear
B)
sodium carbonate
done
clear
C)
borax
done
clear
D)
sodium chloride
done
clear
View Answer play_arrow
question_answer 76) Stable compounds in \[+1\] oxidation state are formed by:
A)
\[B\]
done
clear
B)
\[Al\]
done
clear
C)
\[Ga\]
done
clear
D)
\[Tl\]
done
clear
View Answer play_arrow
question_answer 77) Which one of the following configurations is correct for alkaline earth elements?
A)
\[1{{s}^{2}}2{{s}^{2}}2{{p}^{3}}\]
done
clear
B)
\[1{{s}^{2}}2{{s}^{2}}2{{p}^{6}}3{{s}^{2}}\]
done
clear
C)
\[1{{s}^{2}}2{{s}^{2}}2{{p}^{6}}3{{s}^{2}}3{{p}^{2}}\]
done
clear
D)
\[1{{s}^{2}}2{{s}^{2}}2{{p}^{6}}\]
done
clear
View Answer play_arrow
question_answer 78) Perches process is used to prepare:
A)
\[BaC{{O}_{3}}\]
done
clear
B)
\[L{{i}_{2}}C{{O}_{3}}\]
done
clear
C)
\[{{K}_{2}}C{{O}_{3}}\]
done
clear
D)
\[N{{a}_{2}}C{{O}_{3}}\]
done
clear
View Answer play_arrow
question_answer 79) The gold numbers of gelatin, albumin and strach are 0.005, 0.15 and 2.5 respectively. Which is the best protective colloid?
A)
Gelatin
done
clear
B)
Albumin
done
clear
C)
Starch
done
clear
D)
None of these
done
clear
View Answer play_arrow
question_answer 80) \[K{{O}_{2}}+C{{O}_{2}}\xrightarrow{{}}(gas)\], Which gas is this?
A)
\[{{H}_{2}}\]
done
clear
B)
\[{{N}_{2}}\]
done
clear
C)
\[{{O}_{2}}\]
done
clear
D)
\[CO\]
done
clear
View Answer play_arrow
question_answer 81) Which of the following interhalogen does not exist?
A)
\[Cl{{F}_{2}}\]
done
clear
B)
\[Cl{{F}_{5}}\]
done
clear
C)
\[Cl{{F}_{3}}\]
done
clear
D)
\[ClF\]
done
clear
View Answer play_arrow
question_answer 82) If a person is injured by the shot of gun and all the bullets could not be removed. It may cause poisoning by:
A)
\[Tl\]
done
clear
B)
\[Pb\]
done
clear
C)
\[As\]
done
clear
D)
carbon
done
clear
View Answer play_arrow
question_answer 83) Silicon is the main constituent of:
A)
plants
done
clear
B)
rocks
done
clear
C)
alloys
done
clear
D)
animals
done
clear
View Answer play_arrow
question_answer 84) Which of the following is known as aqua-regia?
A)
\[3HCl+HN{{O}_{3}}\]
done
clear
B)
\[3HN{{O}_{3}}+HCl\]
done
clear
C)
\[{{H}_{3}}P{{O}_{4}}+{{H}_{2}}S{{O}_{4}}\]
done
clear
D)
\[HCl+C{{H}_{3}}COOH\]
done
clear
View Answer play_arrow
question_answer 85) Silver bromide is used in photography because it is:
A)
soluble in \[N{{H}_{4}}OH\]
done
clear
B)
insoluble in acids
done
clear
C)
photosensitive
done
clear
D)
soluble in hypo-solution
done
clear
View Answer play_arrow
question_answer 86) The matte obtained by smelting copper pyrites with coke and sand contains mainly:
A)
\[FeS+ZnS\]
done
clear
B)
\[C{{u}_{2}}S+FeS\]
done
clear
C)
\[CuS+Fe{{S}_{2}}\]
done
clear
D)
\[ZnS+CuS\]
done
clear
View Answer play_arrow
question_answer 87) In the metal carbonyls of general formula \[M{{(CO)}_{x}}\] where M = metal, \[x=4\]the metal is bonded to:
A)
oxygen
done
clear
B)
carbon
done
clear
C)
carbon and oxygen
done
clear
D)
\[C\equiv O\] triple bond
done
clear
View Answer play_arrow
question_answer 88) Which one of the following is common example of fibres?
A)
Nylon-66
done
clear
B)
Nylon-6
done
clear
C)
Buna-S
done
clear
D)
Bakelite
done
clear
View Answer play_arrow
question_answer 89) Which one of the following species does not exist?
A)
\[{{[SnC{{l}_{6}}]}^{3-}}\]
done
clear
B)
\[{{[GeC{{l}_{6}}]}^{2-}}\]
done
clear
C)
\[{{[SiC{{l}_{6}}]}^{2-}}\]
done
clear
D)
\[{{[CC{{l}_{6}}]}^{2-}}\]
done
clear
View Answer play_arrow
question_answer 90) \[(CuS{{O}_{4}}+N{{H}_{4}}OH)\] gives a deep blue complex of:
A)
cupra ammonium sulphate
done
clear
B)
cupra ammonium hydroxide
done
clear
C)
sodium hexametaphosphate
done
clear
D)
ammonal
done
clear
View Answer play_arrow
question_answer 91) Which one of the following is not an ore of magnesium?
A)
Dolomite
done
clear
B)
Carnallite
done
clear
C)
Gypsum
done
clear
D)
Magnesite
done
clear
View Answer play_arrow
question_answer 92) Which one of the following will be able to show geometrical isomerism?
A)
\[M{{A}_{4}}\]
done
clear
B)
\[M{{A}_{3}}B\]
done
clear
C)
\[M{{(AA)}_{2}}\]
done
clear
D)
\[MABCD\]
done
clear
View Answer play_arrow
question_answer 93) Which of the following imparts violet colouration to the Bunsen burner non luminous flam?
A)
\[KCl\]
done
clear
B)
\[CaC{{l}_{2}}\]
done
clear
C)
\[BaC{{l}_{2}}\]
done
clear
D)
\[NaCl\]
done
clear
View Answer play_arrow
question_answer 94) Sodium tripolyphosphate used in industrial detergent and softening of water is obtained by the hydrolysis of:
A)
orthophosphate
done
clear
B)
trimetaphosphate
done
clear
C)
sodium dihydrogenphosphate
done
clear
D)
none of the above
done
clear
View Answer play_arrow
question_answer 95) \[RCH=C{{H}_{2}}\xrightarrow[{{C}_{2}}{{H}_{5}}OH]{Na/N{{H}_{3}}_{(l)}}RC{{H}_{2}}C{{H}_{3}}\], This reaction is called as:
A)
Fischer-Siper reaction
done
clear
B)
Clemmensen reaction
done
clear
C)
Birch reduction
done
clear
D)
Amdt-Eistert synthesis
done
clear
View Answer play_arrow
question_answer 96) The compound A in the following sequence of reaction is a silver salt of:
A)
butyric acid
done
clear
B)
isobutyric acid
done
clear
C)
propanoic acid
done
clear
D)
none of these
done
clear
View Answer play_arrow
question_answer 97) In order to get propane gas which of the following should be subjected to soda lime decarboxylation ?
A)
Sodium formate
done
clear
B)
Mixture of sodium acetate and sodium ethanoate
done
clear
C)
Sodium butyrate
done
clear
D)
Sodium propionate
done
clear
View Answer play_arrow
question_answer 98) Which of the following species is less reactive than benzene towards electrophilic substituation reactions?
A)
Aniline
done
clear
B)
Nitrobenzene
done
clear
C)
Bromobenzene
done
clear
D)
Phenol
done
clear
View Answer play_arrow
question_answer 99) A compound has simplest formula \[C{{H}_{2}}\]. To which hydrocarbon series does it belong ?
A)
Alkynes
done
clear
B)
Cycloalkanes
done
clear
C)
Alkanes
done
clear
D)
None of these
done
clear
View Answer play_arrow
question_answer 100) The change in the optical rotation (with time) of freshly prepared solution of sugar is known as:
A)
mutarotation
done
clear
B)
rotatory motion
done
clear
C)
specific rotation
done
clear
D)
inversion
done
clear
View Answer play_arrow
question_answer 101) The long bones which are solid may be eaten up by special cells and make them hollow. These cells are:
A)
chondroblast
done
clear
B)
chondroclasts
done
clear
C)
octeoblasts
done
clear
D)
osteoclasts
done
clear
View Answer play_arrow
question_answer 102) A significant effect of ultraviolet radiation is the:
A)
production of base tautomeres
done
clear
B)
breakage of phosphodiester bonds
done
clear
C)
formation of pyremidine dimers
done
clear
D)
induction of photosynthesis
done
clear
View Answer play_arrow
question_answer 103) Tracer elements are defined as:
A)
micronutrient elements
done
clear
B)
radioisotope elements
done
clear
C)
macronutrient elements
done
clear
D)
vitamins
done
clear
View Answer play_arrow
question_answer 104) The place where RNA polymerase attaches with the DNA is called:
A)
promoter site
done
clear
B)
operator site
done
clear
C)
activator site
done
clear
D)
repressor site
done
clear
View Answer play_arrow
question_answer 105) Which one of the following is a secretory tissue?
A)
Sclerenchyma
done
clear
B)
Companion cells
done
clear
C)
Hydathode
done
clear
D)
Sclereids
done
clear
View Answer play_arrow
question_answer 106) A pigment concerned with both floral induction and seed germination is:
A)
anthocyanin
done
clear
B)
phytochrome
done
clear
C)
phycocyanin
done
clear
D)
xanthophylls
done
clear
View Answer play_arrow
question_answer 107) Translocation of carbohydrates in flowering plants occur in the form of:
A)
glucose
done
clear
B)
maltose
done
clear
C)
starch
done
clear
D)
sucrose
done
clear
View Answer play_arrow
question_answer 108) Which one of the following elements play important role in denitrogen fixation?
A)
Mn
done
clear
B)
MO
done
clear
C)
2n
done
clear
D)
Cu
done
clear
View Answer play_arrow
question_answer 109) At constant temperature, the rate of transpiration will be higher at:
A)
sea level
done
clear
B)
1 km below sea level
done
clear
C)
1 km above sea level
done
clear
D)
1.5 km above sea level
done
clear
View Answer play_arrow
question_answer 110) Complete oxidation of a molecule of glucose yield :
A)
15 ATP molecules
done
clear
B)
36 ATP molecules
done
clear
C)
2 ATP molecules
done
clear
D)
8 ATP molecules
done
clear
View Answer play_arrow
question_answer 111) Water can be absorbed from a hypertonic external solution by :
A)
adding more water in the external solution
done
clear
B)
auxin treated cells
done
clear
C)
adding a buffer in the external solution
done
clear
D)
cytokinin treated cells
done
clear
View Answer play_arrow
question_answer 112) For process of photosynthesis, all except one of the following items are essential. Point out the exception :
A)
water, minerals
done
clear
B)
light, chlorophyll
done
clear
C)
\[C{{O}_{2}}\]and optimum temperature
done
clear
D)
\[{{O}_{2}}\] and sucrose
done
clear
View Answer play_arrow
question_answer 113) Germinating wheat seeds are put in a Ganongs respirometer filled with mercury or saturated sodium chloride solution. What will happen to it after 24 hours when apparatus is set?
A)
Level will rise
done
clear
B)
Level will fall
done
clear
C)
Level remains as such
done
clear
D)
Seeds will die
done
clear
View Answer play_arrow
question_answer 114) In chiasmata meiosis, the disial segments of the daughter chromalid segregate from eacli other, ar:
A)
anaphase
done
clear
B)
anaphase
done
clear
C)
both (a) and (b)
done
clear
D)
diakinesis
done
clear
View Answer play_arrow
question_answer 115) A structure called synaptomeal complex, which is considered to be a physical structure which is associated with synapsis of homologous chromosomes was discovered by:
A)
King
done
clear
B)
Moses
done
clear
C)
Griffith
done
clear
D)
Balbiam
done
clear
View Answer play_arrow
question_answer 116) If a cross is made between two plants of Aa Bb cc Dd and Aa bb cc DD genotype then what will be the expected frequency of obtaining a progeny with AA Bb Cc Dd ?
A)
1/128
done
clear
B)
1/64
done
clear
C)
1/32
done
clear
D)
1/16
done
clear
View Answer play_arrow
question_answer 117) The garden pea (Piswn sativiim) has 14 chromosomes in each of its body cells. The number of linkage group expected in the species is :
A)
more than 7
done
clear
B)
less than 7
done
clear
C)
7
done
clear
D)
14
done
clear
View Answer play_arrow
question_answer 118) Given are the statements regarding linkages of genes: (i) The strength of the linkage is determined by the distance between the 2 genes in question. (ii) The strength of the linkage is directly proportional to the distance between the two genes. The two genes are said to be linked when they fail to show independent assortment. Out of these statements:
A)
all are correct
done
clear
B)
(i) and (ii) are correct
done
clear
C)
(i) and (iii) are correct
done
clear
D)
(ii) and (vi) are correct
done
clear
View Answer play_arrow
question_answer 119) Independent gene that copy other gene. so as to produce a similar effect are known as :
A)
complementary gene
done
clear
B)
epistatic gene
done
clear
C)
lethal gene
done
clear
D)
duplicate gene
done
clear
View Answer play_arrow
question_answer 120) Out of the given four combination which one possess the integrated protein synthesizing machinery:
A)
m-RNA. r-RNA and amino acid
done
clear
B)
t-RNA, ribosome. nucleus and m-RNA
done
clear
C)
ribosomes. r-RNA and amino acid
done
clear
D)
ribosomes. r-RNA. m-RNA and amino acid
done
clear
View Answer play_arrow
question_answer 121) In pseudo-allelisms, genes are :
A)
functionally allelic and structurally non-allelic
done
clear
B)
structurally as well as functionally allelic
done
clear
C)
structurally allelic and functionally non-allelic
done
clear
D)
structurally as well as functionally non-allelic
done
clear
View Answer play_arrow
question_answer 122) Recessive epistasis is defined as :
A)
a situation in which a gene pair in homozygous recessive condition masks the effect of one or both the member of another gene pair
done
clear
B)
a situation in which the dominant allele of the first gene pair masks the effect of both the members of the second gene pair and the dominant allele of the second gene pair masks the effect of both the members of the first gene pair
done
clear
C)
a situation in which one allele does not allow its alternative form to express
done
clear
D)
a situation in which the dominant allele of one gene pair masks the effect of both members of another gene pair
done
clear
View Answer play_arrow
question_answer 123) Clove oil is obtained from :
A)
wood of Santalum album
done
clear
B)
leaf of Syzygium aromaticum
done
clear
C)
flower bud of Syzygium aromadcum
done
clear
D)
rhizome of Veiiveria ziganoides
done
clear
View Answer play_arrow
question_answer 124) Chewing gum is the latex, obtained from the bark of:
A)
PIumeria rubra
done
clear
B)
Ficus hispida
done
clear
C)
Achras sapota
done
clear
D)
Streblus asper
done
clear
View Answer play_arrow
question_answer 125) The alkaloid known as reserpine is obtained from the root of which plant?
A)
Papaver somnifenim
done
clear
B)
Zingiber officinal
done
clear
C)
Hemidesmus indieus
done
clear
D)
Rauwolffia serpentine
done
clear
View Answer play_arrow
question_answer 126) Natural rubber is obtained from the latex of
A)
Calotropis procera
done
clear
B)
Ricinus communis
done
clear
C)
Nerium indicum
done
clear
D)
Hevea brasiliensis
done
clear
View Answer play_arrow
question_answer 127) Out of the given following disease, which disease results due to disorder in lysosomes ?
A)
Arthritic joints
done
clear
B)
Hurles syndrome
done
clear
C)
Tay Sachs disease
done
clear
D)
All of these
done
clear
View Answer play_arrow
question_answer 128) Besides C-AMP, which one of the following molecules acts as a second messenger in biological system ?
A)
C-DNA
done
clear
B)
C-GTP
done
clear
C)
\[C{{a}^{++}}\]
done
clear
D)
ATP
done
clear
View Answer play_arrow
question_answer 129) In plants lysosomes are not present, then the function of lysosomes is taken by which of the following orgenelles ?
A)
sphaerosomes
done
clear
B)
vacuoles
done
clear
C)
aleurone layer
done
clear
D)
all of these
done
clear
View Answer play_arrow
question_answer 130) Out of the given following cell structure which show polarity is :
A)
microtubules
done
clear
B)
mitochondria
done
clear
C)
endoplasmic reticulum
done
clear
D)
microbodies
done
clear
View Answer play_arrow
question_answer 131) Which one of the following is catalysed by the presence of enzyme peptidyl transferase?
A)
Peptide bond formation
done
clear
B)
Transfer of a ammo acid
done
clear
C)
Transfer of ribosomes units
done
clear
D)
Transfer of peptide bonds
done
clear
View Answer play_arrow
question_answer 132) The anticodon region is an important part of the structure of:
A)
r-RNA
done
clear
B)
m-RNA
done
clear
C)
r-RNA
done
clear
D)
in-RNA
done
clear
View Answer play_arrow
question_answer 133) Which of the following cell organelles is not a micro body?
A)
Peroxisome
done
clear
B)
Lomasome
done
clear
C)
Ribosome
done
clear
D)
Sphaerosome
done
clear
View Answer play_arrow
question_answer 134) Microfilaments are long, thin, contractile rods (solid) of 6-10 nm diameter. These help in :
A)
movement of cell membrane during mobility and endocytosis
done
clear
B)
contraction of muscles, functioning of microvilli and undulation of cell membrane
done
clear
C)
cytoplasmic streming, cleavage of animal celt formation of pseudopodia
done
clear
D)
all of the above
done
clear
View Answer play_arrow
question_answer 135) To no fibrils of desmosomes are made up of :
A)
chitin and keratin
done
clear
B)
keratin and actin
done
clear
C)
actin and desmin
done
clear
D)
keratin and desmin
done
clear
View Answer play_arrow
question_answer 136) In lower plants motile cells bears two flagella. One is called whiplash or acronernatic. Which is smooth and the second one is called tinsel or rough or pleuronematic or pantonematic and bears lateral hair like appendages called :
A)
flimmers
done
clear
B)
mastigonemes
done
clear
C)
both (a) and (b)
done
clear
D)
none of these
done
clear
View Answer play_arrow
question_answer 137) Plasmids are ideal vectors for gene cloning as :
A)
they replicate freely outside the bacterial cells
done
clear
B)
they are self-replicating within the bacterial cell
done
clear
C)
they can be multiplied by culruring
done
clear
D)
they can be multiplied in a laboratory using enzyme
done
clear
View Answer play_arrow
question_answer 138) DNA finger priming has proved useful in Forensic science. It involve use of :
A)
minisatellite
done
clear
B)
ribosomal DNA
done
clear
C)
cDNA
done
clear
D)
bacterial DNA
done
clear
View Answer play_arrow
question_answer 139) The synonym of family Labiatae, Umbelliferae, Compositae and Gramineae are :
A)
Lcguminosae, Acanthaceae, Asteraceae and Lamiaceae
done
clear
B)
Solanaceae. Aricaceae, Apiaceae, Poaceae
done
clear
C)
Lamiaceae, Apiaceae, Asteraceae, Poaceae
done
clear
D)
Lilliaceae, Cucurbitaceae, Poaceae, Asleraceae
done
clear
View Answer play_arrow
question_answer 140) The following features outline a system of plant classification : (i) unisexual flower are the most primitive within the angiosperm (ii) polyphyletic origin of angiosperm (iii) monocotyledons have been considered more primitive than dicotyledons Which one of the following system of classification represent above feature ?
A)
Linnaeus
done
clear
B)
Engter and Pranti
done
clear
C)
Rendle
done
clear
D)
Hutchinson
done
clear
View Answer play_arrow
question_answer 141) The following statements outlines the major feature of a system of classification : (i) Monophylctic origin of angiosperm (ii) Dicotyledons are the primitive over monocotyledons (iii) Division ofdicotyledonae into Ugnosae and Herbaceae Which of the following system of classification represents above features ?
A)
Linnaeus
done
clear
B)
Bentham and Hooker
done
clear
C)
Engler and Pranti
done
clear
D)
Hutchinson
done
clear
View Answer play_arrow
question_answer 142) To how many families to the following vegetable belong ? Pea, soyabcan, radish, cauliflower, cabbage, potato, tomato.
A)
two
done
clear
B)
three
done
clear
C)
four
done
clear
D)
five
done
clear
View Answer play_arrow
question_answer 143) Industrial production of ethanol from starch is brought about by a certain species of :
A)
Penicillin
done
clear
B)
Azotobacter
done
clear
C)
Saccharomyces
done
clear
D)
Lactobaciflus
done
clear
View Answer play_arrow
question_answer 144) In 1928, a scientist discovered an antibiotic which was found to be very effective. The name of scientist and the antibiotic respectively are :
A)
Woodruff and actinomycin
done
clear
B)
Waksman and streptomycin
done
clear
C)
Cesar Milstein and rifampicin
done
clear
D)
Alexander l-lemming and penicillin
done
clear
View Answer play_arrow
question_answer 145) Advancement in genetic engineering has been possible due to :
A)
oncogenes
done
clear
B)
exonucleases
done
clear
C)
transposons
done
clear
D)
endonucleases
done
clear
View Answer play_arrow
question_answer 146) Apart from DNA in the bacterial nucleoid, there is a circular extra chromosomal DNA, called :
A)
plasmid
done
clear
B)
meso some
done
clear
C)
chromosome
done
clear
D)
none of these
done
clear
View Answer play_arrow
question_answer 147) Seed habit was first originated in members of:
A)
ferns
done
clear
B)
cycadofilicales
done
clear
C)
gymnosperm
done
clear
D)
angiosperm
done
clear
View Answer play_arrow
question_answer 148) If Gnetum is not a flowering plant, it may be mistaken for a:
A)
tree fem
done
clear
B)
monocot plant
done
clear
C)
dicot plant
done
clear
D)
none of these
done
clear
View Answer play_arrow
question_answer 149) Which of the following is not functionally analogous with other in the group ?
A)
Antheridium
done
clear
B)
Archegonium
done
clear
C)
Oogonium
done
clear
D)
Ovule
done
clear
View Answer play_arrow
question_answer 150) Which one of the following gymnosperms shows the complete absence of archegonia in this ovules?
A)
Cycas
done
clear
B)
Ephedra
done
clear
C)
Gnetum
done
clear
D)
Ginfego
done
clear
View Answer play_arrow
question_answer 151) The occurrence of more than one embryo in a seed in know as :
A)
polar nuclei
done
clear
B)
polyembryony
done
clear
C)
porogamy
done
clear
D)
protandry
done
clear
View Answer play_arrow
question_answer 152) The deteriorative processes that naturally. Terminate their functional life are collectively called :
A)
sensescence
done
clear
B)
abscission
done
clear
C)
wilting
done
clear
D)
plasmolysis
done
clear
View Answer play_arrow
question_answer 153) Which of the following elements are considered essential for photolysis of water ?
A)
Ca and Cl
done
clear
B)
Mn and Cl
done
clear
C)
Zn and I
done
clear
D)
None of these
done
clear
View Answer play_arrow
question_answer 154) The development of eye in vertebrate embryology is studied under :
A)
neurogenesis
done
clear
B)
organogenesis
done
clear
C)
nologenesis
done
clear
D)
all of these
done
clear
View Answer play_arrow
question_answer 155) The Noble Prize for the discovery of TCA cycle and ATP biosynthesis was awarded to :
A)
Hans Krebs
done
clear
B)
Lipman
done
clear
C)
Krebs and Lipman jointly
done
clear
D)
Vishniac and Ochoa
done
clear
View Answer play_arrow
question_answer 156) Which of the following is not a feature of mangrove plant ?
A)
Production of respiration roots called pneumatophores
done
clear
B)
lack of slit like prop root
done
clear
C)
viviparous method of germination of seeds
done
clear
D)
all of the above
done
clear
View Answer play_arrow
question_answer 157) A and B genes are linked. What should be the genotype of progeny in a cross between AB/ab and ab/ab ?
A)
AA bb and aa bb
done
clear
B)
Aa Bb and aa bb
done
clear
C)
AA BB and aa bb
done
clear
D)
None of these
done
clear
View Answer play_arrow
question_answer 158) The part of m-RNA which removed after transcription by RNA spliting is :
A)
exons
done
clear
B)
liexane
done
clear
C)
introns
done
clear
D)
none of these
done
clear
View Answer play_arrow
question_answer 159) A spindle is organised during a process of meiosis, sometime formation of spindle is arrested by the use of:
A)
nitrous acid
done
clear
B)
cellulose
done
clear
C)
colchicine
done
clear
D)
phosphate
done
clear
View Answer play_arrow
question_answer 160) A phenomenon which include all those heritable changes, which after the phenotype of an individual is called ?
A)
Mutation
done
clear
B)
Lethality
done
clear
C)
Both (a) and (b)
done
clear
D)
None of these
done
clear
View Answer play_arrow
question_answer 161) The main difference between active and passive transport across cell membrane is that:
A)
active transport is more rapid than passive transport
done
clear
B)
passive transport is non-selective
done
clear
C)
passive transport is due to the concentration gradient across the cell membrane where as active transport is due to metabolic energy
done
clear
D)
passive transport is confined to anions, while acrive transport is confined to cations only
done
clear
View Answer play_arrow
question_answer 162) Which of the following does not obtain its nitrogen from captured animal i.e., is not an insectivorous plant ?
A)
Orobanche
done
clear
B)
Utriciilaria
done
clear
C)
Drosera
done
clear
D)
All of these
done
clear
View Answer play_arrow
question_answer 163)
List-I List-II A. Diameter of the helix 1. \[34\,\overset{\text{o}}{\mathop{\text{A}}}\,\] B. Distance between two adjacent 2. \[20\,\overset{\text{o}}{\mathop{\text{A}}}\,\] C. Distance between 2 complete turns 3. \[3\,\overset{\text{o}}{\mathop{\text{A}}}\,\] D. Length of the hydrogen bonds 4. \[4.4\,\overset{\text{o}}{\mathop{\text{A}}}\,\]
Codes:
A)
A-1 B-3 C-2 D-4
done
clear
B)
A-2 B-4 C-1 D-3
done
clear
C)
A-4 B-2 C-3 D-1
done
clear
D)
A-1 B-4 C-2 D-3
done
clear
View Answer play_arrow
question_answer 164) Isochromosis are formed as a result of :
A)
longitudinal splitting of centromere
done
clear
B)
pairing of identical chromosomes
done
clear
C)
disjunction of bivalents
done
clear
D)
transverse division of centromere
done
clear
View Answer play_arrow
question_answer 165) The most widespread but distinctive cytogenerics process involved in speciation of higher plant is :
A)
autosomal mutation
done
clear
B)
polyploidy
done
clear
C)
apomixes
done
clear
D)
interspecific hybridization
done
clear
View Answer play_arrow
question_answer 166) The genetic code is said to be degenerate and universal which means that: (i) codon are common for higher and lower organisms (ii) amino acids may have more than one codon (iii) all amino acids have more than one codon Out of the above statement, correct statement is :
A)
(ii) and (iii) are correct
done
clear
B)
only the (iii) statement is correct
done
clear
C)
(i) and (ii) are correct
done
clear
D)
all are correct
done
clear
View Answer play_arrow
question_answer 167) Zinc is considered essential for the formation of plant hormone :
A)
abscisic acid
done
clear
B)
in dole acetic acid
done
clear
C)
gibberellic acid
done
clear
D)
none of these
done
clear
View Answer play_arrow
question_answer 168) The low temperature requirement for flowering was first noticed by :
A)
Nawaschin
done
clear
B)
Klippast
done
clear
C)
MacLeod
done
clear
D)
Lysenko
done
clear
View Answer play_arrow
question_answer 169) Which of the following codon is related to UAA and UAG functions ?
A)
UUU
done
clear
B)
UGA
done
clear
C)
AUG
done
clear
D)
GUG
done
clear
View Answer play_arrow
question_answer 170) When carbohydrates are the respiratory substrates the Respiratory Quotient (R.Q.) becomes:
A)
zero
done
clear
B)
more than one
done
clear
C)
less than one
done
clear
D)
one
done
clear
View Answer play_arrow
question_answer 171) The plants which can refix \[C{{O}_{2}}\] released during respiration and use it during photosynthesis are known as ;
A)
succulents
done
clear
B)
mesophytes
done
clear
C)
hydrophytes
done
clear
D)
non-succlents
done
clear
View Answer play_arrow
question_answer 172) Out of the following which one is not a millet:
A)
Triticum
done
clear
B)
Penicillium
done
clear
C)
Pennisetum
done
clear
D)
Eleiisine.
done
clear
View Answer play_arrow
question_answer 173) Litmus is a natural dye which is obtained from:
A)
algae
done
clear
B)
fungi
done
clear
C)
lichen
done
clear
D)
mosses
done
clear
View Answer play_arrow
question_answer 174) Commercially cork is obtained from llie bark of:
A)
Mangifera indica
done
clear
B)
Avadirachra indica
done
clear
C)
Qiterciis suber
done
clear
D)
Ficiis bengalensis
done
clear
View Answer play_arrow
question_answer 175) The part of Papaver sainniferwn from which opium is obtained is:
A)
seed
done
clear
B)
mature fruit
done
clear
C)
unripe fruit
done
clear
D)
stem and leaf
done
clear
View Answer play_arrow
question_answer 176) Calmodulein is a calcium protein complex in microtubules and microfilaments. It is involved in :
A)
cell-differenciation
done
clear
B)
synthesis of ATP
done
clear
C)
breakdown of ATP
done
clear
D)
cell motility
done
clear
View Answer play_arrow
question_answer 177) Who among the following discovered microtubules ?
A)
de Robertis and Franchi
done
clear
B)
Watson and Crick
done
clear
C)
Ziner and Lederberg
done
clear
D)
Boveri and Van Beneden
done
clear
View Answer play_arrow
question_answer 178) In idioblasts. crystals of calcium oxalate exist in different shapes needle like crystals of calcium oxalates are called :
A)
raphids
done
clear
B)
cystoliths
done
clear
C)
otoliths
done
clear
D)
sphaerophids
done
clear
View Answer play_arrow
question_answer 179) Nitrogenous excretory (waste) products in plants cells are :
A)
alkaloids like quinine and morphine
done
clear
B)
tannins and resins
done
clear
C)
latex and essential oils
done
clear
D)
all of the above
done
clear
View Answer play_arrow
question_answer 180) Raphids, sphaeraphides, druses are minerals crystals in plants cells of Nerium. Lefnna, Eichhornia, onion. These are chemically:
A)
\[CaC{{O}_{3}}\]
done
clear
B)
calcium oxalate
done
clear
C)
silica
done
clear
D)
\[CaS{{O}_{4}}\]
done
clear
View Answer play_arrow
question_answer 181) Insulin is a fan shaped carbohydrats crystals and is found in cell sap of:
A)
tubers of dahlia
done
clear
B)
tubers of Jeruselum (artichoke)
done
clear
C)
dandelion
done
clear
D)
all of the above
done
clear
View Answer play_arrow
question_answer 182) Most common mineral crystals are of calcium oxalate \[[Ca({{C}_{2}}{{p}_{4}}),2{{H}_{2}}O].\] When these are satellite in shape and found in clusters, are called:
A)
sphaeraphides
done
clear
B)
druse
done
clear
C)
both (a) and (b)
done
clear
D)
cistolith
done
clear
View Answer play_arrow
question_answer 183) Smallest chromosomes (size \[0.025\mu \]) are found in algae. Largest eukaryotic normal chrom- osome is \[30\mu \] in size and is found in :
A)
Trilliuin
done
clear
B)
Ophioglossum
done
clear
C)
Radiolarians
done
clear
D)
Mays
done
clear
View Answer play_arrow
question_answer 184) Which protein of nucleoplasm activates DNA to replicate the transcribe?
A)
Histone
done
clear
B)
Non-histone
done
clear
C)
Phospho protein
done
clear
D)
Nucleoproteins
done
clear
View Answer play_arrow
question_answer 185) Experiment to demonstrate importance of nucleus in the controlling growth and heredity were performed on :
A)
Acetabularia
done
clear
B)
Neurospora
done
clear
C)
eucocytes
done
clear
D)
star fish
done
clear
View Answer play_arrow
question_answer 186) Which one of the following phyla is characterized by the absence of a true coelom characterised by the absence of a true coelom?
A)
Echinodermata
done
clear
B)
Annelida
done
clear
C)
Mollusca
done
clear
D)
Nematoda
done
clear
View Answer play_arrow
question_answer 187) Which of the following is the correct pairing of the classification group and common example?
A)
Porifera-sea fan
done
clear
B)
Crustacea-cattle fish
done
clear
C)
Plathylielminthes-Neries
done
clear
D)
Mastigophora-Volvox
done
clear
View Answer play_arrow
question_answer 188) Flowers of which one of the following plants bears 2 minute scales called lodicules ?
A)
Gossypidin herbarium
done
clear
B)
Helianthns annuus
done
clear
C)
Triticum vulgare
done
clear
D)
Citrus medico.
done
clear
View Answer play_arrow
question_answer 189) Gynan drophore is found in the members of family :
A)
Brassica ceae
done
clear
B)
Capparidaceae
done
clear
C)
Malvaceae
done
clear
D)
Ranunculaceae
done
clear
View Answer play_arrow
question_answer 190) The type specimen used by tlie author in the original publication is known as :
A)
lectotype
done
clear
B)
isotype
done
clear
C)
halotype
done
clear
D)
syntype
done
clear
View Answer play_arrow
question_answer 191) Syngensious stamens occur in members of family :
A)
Malvaceae
done
clear
B)
Orchidaceae
done
clear
C)
Solanaceae
done
clear
D)
Asteraceae
done
clear
View Answer play_arrow
question_answer 192) Biofertilizers are used to increase the availability of nutrient like :
A)
nitrogen
done
clear
B)
phosphorus
done
clear
C)
potassium
done
clear
D)
both (a) and (b)
done
clear
View Answer play_arrow
question_answer 193) Which bacterium was the first biopesticide to be used on a commercial scale in the world?
A)
Bacillus thiiringiensis
done
clear
B)
Bac.Uli.is polymyxa
done
clear
C)
Bacillus brevis
done
clear
D)
Bacillits lichenifonnis
done
clear
View Answer play_arrow
question_answer 194) Out of the following which is not concerned with biotechnology ?
A)
Biogas production
done
clear
B)
Sewage treatment
done
clear
C)
Bio fertilizers
done
clear
D)
Wood seasoning
done
clear
View Answer play_arrow
question_answer 195) Pentadiplandra brazzeana produces a protein called brazzein which is about 2,000 times as sweet as sugar. This plant belongs to :
A)
Australia
done
clear
B)
South America
done
clear
C)
Africa
done
clear
D)
Canada
done
clear
View Answer play_arrow
question_answer 196) Classification of algae into II classes was made by Fritsh on the basis of flagellation, pigmentation and type of reserve food. out of this. the main criteria used in algal classification/grouping of algae is :
A)
chemical composition of cell wall
done
clear
B)
type of pigmentation
done
clear
C)
unicellular sex organ
done
clear
D)
aquatic liabitat
done
clear
View Answer play_arrow
question_answer 197) The minimum number of chromosome1 recorded in an algae are n = 2. The algae is :
A)
Laininaria
done
clear
B)
Porphyra
done
clear
C)
Netrium
done
clear
D)
Chlamydomonas
done
clear
View Answer play_arrow
question_answer 198) A members of RJiodophyceae (red algae) which is widely used as weed is called irish moss. Its alternative name is :
A)
Ficus
done
clear
B)
Laminaria
done
clear
C)
Ulothrix
done
clear
D)
Chondrus
done
clear
View Answer play_arrow
question_answer 199) From a cell wall of Chondrus a sulphated polysaccharides is obtained which is used clearingagentinliquas (beer).The substance is:
A)
funor
done
clear
B)
agar
done
clear
C)
carrageenm
done
clear
D)
none of these
done
clear
View Answer play_arrow
question_answer 200) In angiosperm. the egg is :
A)
completely surrounded by a wall
done
clear
B)
surrounded by a wall only at the micro pylal end
done
clear
C)
surrounded by a wall only at chalazal end
done
clear
D)
devoid of wall
done
clear
View Answer play_arrow