question_answer 1) Itencrierence may be seen using two independent
A)
sodium lamps
done
clear
B)
fluorescent tubes
done
clear
C)
lasers
done
clear
D)
mercury vapour lamps
done
clear
View Answer play_arrow
question_answer 2) The focal lengths of the objective aiidthe eye piece of telescope are 100 cm and 10 cm respectively. The magnification of the telescope when final image is formed at
A)
\[0.1\]
done
clear
B)
\[10\]
done
clear
C)
W 100
done
clear
D)
\[\infty \]
done
clear
View Answer play_arrow
question_answer 3) An electromagnetic wave has
A)
electric vector only
done
clear
B)
magnetic vector only
done
clear
C)
eiectric and magnetic vectors perpendicular to each other
done
clear
D)
neither the electric vector nor the magnetic vector
done
clear
View Answer play_arrow
question_answer 4) 160W-60V lamp is connected at 60 V DC Supply. The number of electrons passing trough the lamp in 1 min is
A)
\[{{10}^{19}}\]
done
clear
B)
\[{{10}^{21}}\]
done
clear
C)
\[1.6\times {{10}^{19}}\]
done
clear
D)
\[1.4\times {{10}^{20}}\] (The charge of electron \[1.6\times {{10}^{-19}}C)\]
done
clear
View Answer play_arrow
question_answer 5) If a current is allowed to pass through a circuit coasisung of two dissimilar metals, thers is eaher evolution or absorption of heat at the junction, depending upon the direction of the current. The effect is known as
A)
See back effect
done
clear
B)
Joule effect
done
clear
C)
Peltier effect
done
clear
D)
Thomson effect
done
clear
View Answer play_arrow
question_answer 6) The energy of an X-ray photon of wavelength\[1\,\overset{\text{o}}{\mathop{\text{A}}}\,\]is \[(h=6.6\times {{10}^{-34}}\text{J-s})\]
A)
24.6 keV
done
clear
B)
6.1 keV
done
clear
C)
12.3 keV
done
clear
D)
1.2 keV
done
clear
View Answer play_arrow
question_answer 7) Gravitational field is
A)
conservative
done
clear
B)
non-conservative
done
clear
C)
electromagnetic
done
clear
D)
magnetic
done
clear
View Answer play_arrow
question_answer 8) According to Maxwell's law of distribution of velocities of molecules, the most probable velocity is
A)
greater than the mean velocity
done
clear
B)
equal to the mean velocity
done
clear
C)
equal to the root mean square velocity
done
clear
D)
less than the root mean square velocity
done
clear
View Answer play_arrow
question_answer 9) The photons in a radio wave of wavelength \[3\times {{10}^{4}}cm\]have energy
A)
\[6.62\times {{10}^{-10}}\text{J}\]
done
clear
B)
\[19.86\times {{10}^{-24}}\text{J}\]
done
clear
C)
\[6.62\times {{10}^{-28}}\text{J}\]
done
clear
D)
\[2.2\times {{10}^{-35}}\text{J}\]
done
clear
View Answer play_arrow
question_answer 10) According to Bohr's theory of hydrogen atom, for the electron in the \[{{\text{n}}^{\text{th}}}\] allowed orbit the linear momentum is proportional to 1/n (ii) radius is proportional to n (iii) kinetic energy is proportional to 1/n2 (iv) angular momentum is proportional to n Choose the correct option from the codes given below:
A)
(i), (iii), (iv)
done
clear
B)
(i)
done
clear
C)
(i), (ii)
done
clear
D)
(iii)
done
clear
View Answer play_arrow
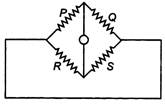
question_answer 11)
In the circuit shown the equivalent resistance between A and B is
A)
\[27\,\Omega \]
done
clear
B)
\[18\,\Omega \]
done
clear
C)
\[9\,\Omega \]
done
clear
D)
\[3\,\Omega \]
done
clear
View Answer play_arrow
question_answer 12) When two sinusoidal waves moving at right angle to each other superimpose, they produce
A)
beats
done
clear
B)
interference
done
clear
C)
stationary waves
done
clear
D)
Lissajous figure
done
clear
View Answer play_arrow
question_answer 13)
Each of the properties of sound in List-\[\text{I}\] primarily depends on one of the quantities in List-\[\text{II}\text{.}\] Select the correct answer (matching List-\[\text{I}\] with List-\[\text{II}\text{.}\]) as per code given below the lists \[\text{List}\,\text{-I}\] \[\text{List}\,\text{-II}\] (A) Loudness (1) Waveform (B) Pitch (2) Frequency (C) Quality (3) Intensity
Codes:
A)
A-1 B-2 C-3
done
clear
B)
A-3 B-2 C-1
done
clear
C)
A-2 B-3 C-1
done
clear
D)
A-2 B-1 C-3
done
clear
View Answer play_arrow
question_answer 14) One of the causes of the breadth of the spectral lines is
A)
doppler's effect
done
clear
B)
dispersion
done
clear
C)
reflection
done
clear
D)
refraction
done
clear
View Answer play_arrow
question_answer 15) In a stationary wave represented by \[y=2a\,\cos kx\,\sin \,\omega t\]the intensity at a certain point is maximum when
A)
cos toe is maximum
done
clear
B)
cos toe is minimum
done
clear
C)
sin cot is maximum
done
clear
D)
sin cot is minimum
done
clear
View Answer play_arrow
question_answer 16) If a big drop of water is broken into smaller drops the surface energy
A)
increases
done
clear
B)
decreases
done
clear
C)
remains unchanged
done
clear
D)
can increase as well as decrease
done
clear
View Answer play_arrow
A)
A,C
done
clear
B)
A,B,C
done
clear
C)
B, D
done
clear
D)
A, B, C, D
done
clear
View Answer play_arrow
question_answer 18) If the differential equation given by \[\frac{{{d}^{2}}y}{d{{t}^{2}}}+2k\frac{dy}{dt}+{{\omega }^{2}}y={{F}_{0}}\,\sin \,pt\]describes the oscillatory motion of body in a dissipative medium under the influence of a periodic force, then the state of maximum amplitude of the oscillation is a measure of
A)
free vibration
done
clear
B)
damped vibration
done
clear
C)
forced vibration
done
clear
D)
resonance
done
clear
View Answer play_arrow
question_answer 19) A grating which would be most suitable for constructing a spectrometer for the visible and ultraviolet region, should have
A)
100 lines/cm
done
clear
B)
1000 lines/cm
done
clear
C)
10000 lines/cm
done
clear
D)
1000000 lines/cm
done
clear
View Answer play_arrow
question_answer 20) A grating which would be most suitable for constructing a spectrometer for the visible and ultraviolet region should have \[1000000\text{ }lines\text{ }c{{m}^{-1}}.\] Directions : The following 5 (.five) items consists of two statements, one labelled the 'Assertion 'and the other labelled the 'Reason R'. You are to examine these two statements carefully and decide if the Assertion A and the Reason R are individually true and of so, whether the reason is correct explanation of the Assertion. Select your answer to these items using the codes given below and mark your answer sheet accordingly. Codes: Both A and R are true and R is the correct explanation of A. Both A and R are true and R is NOT a correct explanation of A. A is true but R is false. A is false but R is true. Assertion : The angular momentum under a central force is constant. Reason (R): Inverse square law of force is conservative.
A)
Both A and R are true and R is the correct explanation of A.
done
clear
B)
Both A and R are true and R is NOT a correct explanation of A.
done
clear
C)
A is true but R is false.
done
clear
D)
A is false but R is true.
done
clear
View Answer play_arrow
question_answer 21) Assertion The upper surface of the Wings of an aeroplane is made convex and the Lower surface is made concave. Reason (R) : The air currents at the top have A smaller and thus less pressure at the Bottom than at the top.
A)
Both A and R are true and R is the correct explanation of A.
done
clear
B)
Both A and R are true and R is NOT a correct explanation of A.
done
clear
C)
A is true but R is false.
done
clear
D)
A is false but R is true.
done
clear
View Answer play_arrow
question_answer 22) Assertion : All the rain drops hit the Surface of theearth with the same constant velocity. Reason (R) : An object falling through a viscous medium eventually attains a terminal velocity.
A)
Both A and R are true and R is the correct explanation of A.
done
clear
B)
Both A and R are true and R is NOT a correct explanation of A.
done
clear
C)
A is true but R is false.
done
clear
D)
A is false but R is true.
done
clear
View Answer play_arrow
question_answer 23) Assertion : Photoelectric effect can take Place only with an electron bound in the atom. Reason(R) : Electron is a fermion where as proton is a boson.
A)
Both A and R are true and R is the correct explanation of A.
done
clear
B)
Both A and R are true and R is NOT a correct explanation of A.
done
clear
C)
A is true but R is false.
done
clear
D)
A is false but R is true.
done
clear
View Answer play_arrow
question_answer 24) Assertion : Fission of \[_{\text{92}}^{\text{235}}\text{U}\]is brought about by a thermal neutron, whereas that of \[_{\text{92}}^{\text{238}}\text{U}\]is brought about by a fast neutron. Reason (R): \[_{\text{92}}^{\text{235}}\text{U}\]is an even-odd nucleus, whereas \[_{\text{92}}^{\text{238}}\text{U}\]is an even-even nucleus.
A)
Both A and R are true and R is the correct explanation of A.
done
clear
B)
Both A and R are true and R is NOT a correct explanation of A.
done
clear
C)
A is true but R is false.
done
clear
D)
A is false but R is true.
done
clear
View Answer play_arrow
question_answer 25) 25. Damping on sharpness of resonance has
A)
no effect
done
clear
B)
more damping, sharper resonance
done
clear
C)
less damping, sharper resonance
done
clear
D)
less damping, less sharp resonance
done
clear
View Answer play_arrow
question_answer 26) 26. A superconducting material is
A)
ferromagnetic
done
clear
B)
ferroelectric
done
clear
C)
diamagnetic
done
clear
D)
paramagnetic
done
clear
View Answer play_arrow
question_answer 27) 27. If the free electron density be n and relaxation time be \[\tau ,\]the electrical conductivity of a conductor may be expressed as
A)
\[\frac{ne\tau }{{{m}_{e}}}\]
done
clear
B)
\[\frac{n{{e}^{2}}\tau }{{{m}_{e}}}\]
done
clear
C)
\[\frac{n{{e}^{2}}}{\tau {{m}_{e}}}\]
done
clear
D)
\[\frac{{{m}_{e}}{{e}^{2}}\tau }{n}\]
done
clear
View Answer play_arrow
question_answer 28) The spacing between the principal planes of a crystal is \[4\,\overset{\text{o}}{\mathop{\text{A}}}\,\]. It is found that first order bragg reflection of a beam of monochromatic X-rays occurs at an angle of \[30{}^\circ \]. The wavelength of X-rays is
A)
\[4\,\overset{\text{o}}{\mathop{\text{A}}}\,\]
done
clear
B)
\[2\,\overset{\text{o}}{\mathop{\text{A}}}\,\]
done
clear
C)
\[1\,\overset{\text{o}}{\mathop{\text{A}}}\,\]
done
clear
D)
\[1.5\,\overset{\text{o}}{\mathop{\text{A}}}\,\]
done
clear
View Answer play_arrow
question_answer 29) An ideal gas is allowed to expand freely against a vacuum in a rigid insulated container. The gas undergoes
A)
an increase in its internal energy
done
clear
B)
a decrease in its internal energy
done
clear
C)
neither an increase nor a decrease in its temperature or internal energy
done
clear
D)
a decrease in temperature
done
clear
View Answer play_arrow
question_answer 30) The relation between electric field vector\[\mathbf{\vec{E}}\] the displacement vector \[\mathbf{\vec{D}}\] and the polarization vector \[\mathbf{\vec{P}}\] for a dielectric placed in electric field \[\mathbf{\vec{E}}\] is given by
A)
\[\mathbf{\vec{P}}={{\varepsilon }_{0}}\mathbf{\vec{E}}+\mathbf{\vec{D}}\]
done
clear
B)
\[\mathbf{\vec{P}}=\mathbf{\vec{D}}+\mathbf{\vec{E}}\]
done
clear
C)
\[\mathbf{\vec{D}}={{\varepsilon }_{0}}\mathbf{\vec{E}}+\mathbf{\vec{P}}\]
done
clear
D)
\[\mathbf{\vec{E}}=\mathbf{\vec{D}}+\mathbf{\vec{P}}\]
done
clear
View Answer play_arrow
question_answer 31) If Young's modulus of elasticity Y for a material is one and half times its rigidity coefficient \[\eta \] the Poisson ratio \[\sigma \] will be
A)
\[+\frac{2}{3}\]
done
clear
B)
\[-\frac{1}{4}\]
done
clear
C)
\[+\frac{1}{4}\]
done
clear
D)
\[-\frac{2}{3}\]
done
clear
View Answer play_arrow
question_answer 32) Which one of the following is not a fundamental \[\text{SI}\] unit ?
A)
Ampere
done
clear
B)
Candela
done
clear
C)
Newton
done
clear
D)
Kelvin
done
clear
View Answer play_arrow
question_answer 33)
The formula for horizontal range of a projectile is \[S=\frac{{{v}^{2}}\sin {{\,}^{2}}\text{ }\!\!\theta\!\!\text{ }}{2g}\] where v is initial speed, \[\text{ }\!\!\theta\!\!\text{ }\] angle of inclination and g is acceleration due to gravity. The Wheatstone bridge shown in figure can be used to determine the range if the following arrangement is made
A)
Q proportional to v2 P proportional to g R proportional to \[{{\sin }^{2}}\text{ }\!\!\theta\!\!\text{ }\]
done
clear
B)
Q proportional to g P proportional to v2 R proportional to \[{{\sin }^{2}}\text{ }\!\!\theta\!\!\text{ }\]
done
clear
C)
Q proportional to g P proportional to \[{{\sin }^{2}}\text{ }\!\!\theta\!\!\text{ }\] R proportional to v2
done
clear
D)
Q proportional to \[{{\sin }^{2}}\text{ }\!\!\theta\!\!\text{ }\] P proportional to v2 R proportional to g
done
clear
View Answer play_arrow
question_answer 34) One Becquerel is defined as
A)
1 disintegration per sec
done
clear
B)
106 disintegration per sec
done
clear
C)
\[3.7\times {{10}^{10}}\] disintegration per sec
done
clear
D)
103 disintegration per sec
done
clear
View Answer play_arrow
question_answer 35) The shortest wavelength which can be obtained in hydrogen spectrum is\[(R={{10}^{7}}{{\text{m}}^{\text{-1}}})\]
A)
\[1000\,\overset{\text{o}}{\mathop{\text{A}}}\,\]
done
clear
B)
\[800\,\overset{\text{o}}{\mathop{\text{A}}}\,\]
done
clear
C)
\[1300\,\overset{\text{o}}{\mathop{\text{A}}}\,\]
done
clear
D)
\[2100\,\overset{\text{o}}{\mathop{\text{A}}}\,\]
done
clear
View Answer play_arrow
question_answer 36) In an ideal choke, ratio of its inductance L to its DC resistance R is
A)
infinity
done
clear
B)
zero
done
clear
C)
unity
done
clear
D)
hundred
done
clear
View Answer play_arrow
question_answer 37) A transformer is often filled with oil. The oil used should have
A)
low viscosity
done
clear
B)
high dielectric strength
done
clear
C)
low boiling point
done
clear
D)
high thermal conducting
done
clear
View Answer play_arrow
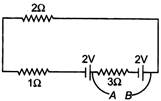
question_answer 38)
For the circuit shown in figure the potential difference between A and B will be (in volt)
A)
2
done
clear
B)
1.5
done
clear
C)
1.0
done
clear
D)
zero
done
clear
View Answer play_arrow
question_answer 39) An electron microscope is superior to an optical microscope in
A)
having better resolving power
done
clear
B)
being easy to handle
done
clear
C)
low cost
done
clear
D)
quickness of observation
done
clear
View Answer play_arrow
question_answer 40) A zener diode is used for
A)
rectification
done
clear
B)
modulation
done
clear
C)
detection
done
clear
D)
voltage regulation
done
clear
View Answer play_arrow
question_answer 41) A black body at a temperature of \[227{}^\circ C\] radiates heat at the rate of 5 \[c{{m}^{-2}}{{s}^{-1}}.\] temperature of \[727{}^\circ C\] the rate of heat radiated per unit area in cal \[c{{m}^{-2}}{{s}^{-1}}\] is
A)
400
done
clear
B)
80
done
clear
C)
40
done
clear
D)
15
done
clear
View Answer play_arrow
question_answer 42) With the rise of temperature the resistivity of a semiconductor
A)
remains unchanged
done
clear
B)
increases
done
clear
C)
decreases
done
clear
D)
first increases and then decreases
done
clear
View Answer play_arrow
question_answer 43) The terminal velocity of spherical ball of radius a falling through a viscous liquid a proportional to
A)
\[a\]
done
clear
B)
\[{{a}^{2}}\]
done
clear
C)
\[{{a}^{3}}\]
done
clear
D)
\[{{a}^{-1}}\]
done
clear
View Answer play_arrow
question_answer 44) The physical quantity which is not a unit of energy is
A)
volt-coulomb
done
clear
B)
Me V-sec
done
clear
C)
henry (ampere)2
done
clear
D)
farad-(volt)2
done
clear
View Answer play_arrow
question_answer 45)
45.
A)
a charged hollow metallic sphere of radius OP with centre at O
done
clear
B)
a charged solid metallic sphere of radius OP with centre at O
done
clear
C)
a uniformly charged non-conducting sphere of radius OP with centre at O
done
clear
D)
a uniformly charged non-conducting hollow sphere of radius OP with centre at O
done
clear
View Answer play_arrow
question_answer 46) 46. ??Good emitters are good absorbers?? is a statement concluded from
A)
Newton?s law of cooling
done
clear
B)
Stefan?s law of radiation
done
clear
C)
Provost?s theory
done
clear
D)
Kirchhoff?s law
done
clear
View Answer play_arrow
question_answer 47)
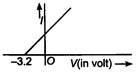
47. In a photoelectric experiment the relation between applied potential difference between cathode and anode V and the photoelectric current \[I\] was found to be shown in graph below. If planck?s constant \[h=6.6\times {{10}^{-34}}\]J-s, the frequency of incident radiation would be nearly (in s-1)
A)
\[0.436\times {{10}^{18}}\]
done
clear
B)
\[0.436\times {{10}^{17}}\]
done
clear
C)
\[0.775\times {{10}^{15}}\]
done
clear
D)
\[0.775\times {{10}^{16}}\]
done
clear
View Answer play_arrow
question_answer 48) For hydrogen atom electron in \[{{\text{n}}^{\text{th}}}\] Bohr orbit, the ratio of radius of orbit to its de-Broglie wavelength is
A)
\[\frac{n}{2\pi }\]
done
clear
B)
\[\frac{{{n}^{2}}}{2\pi }\]
done
clear
C)
\[\frac{1}{2\pi n}\]
done
clear
D)
\[\frac{1}{2\pi {{n}^{2}}}\]
done
clear
View Answer play_arrow
question_answer 49)
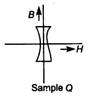
If the B-H curves of two samples of P and Q of iron are as shown below, then which one of the following statements is correct?
A)
Both P and Q are suitable for making permanent magnet
done
clear
B)
P is suitable for making permanent magnet and Q for making electromagnet
done
clear
C)
P is suitable for making electromagnet and Q is suitable for permanent magnet
done
clear
D)
Both P and Q are suitable for making electromagnets
done
clear
View Answer play_arrow
question_answer 50) A current passing through a coil of self inductance of 2 mH changes at the rate of 20 mA s-1. The emf induced in the coil is
A)
\[10\mu V\]
done
clear
B)
\[40\mu V\]
done
clear
C)
10 mV
done
clear
D)
40 mV
done
clear
View Answer play_arrow
question_answer 51) In the electrolytic cell electron flows from
A)
cathode to anode in solution
done
clear
B)
cathode to anode through external circuit
done
clear
C)
cathode to anode through internal circuit
done
clear
D)
cathode to cathode through external circuit
done
clear
View Answer play_arrow
question_answer 52) In first order reaction, the concentration of reactant decreases from \[800\text{ }mol\text{ }c{{m}^{-3}}\]to \[50\text{ }mol\text{ }c{{m}^{-3}}\]in \[2\times {{10}^{4}}s.\]. The rate constant is
A)
\[2\times {{10}^{4}}{{s}^{-1}}\]
done
clear
B)
\[3.45\times {{10}^{-5}}{{s}^{-1}}\]
done
clear
C)
\[1.386\times {{10}^{-4}}{{s}^{-1}}\]
done
clear
D)
\[2\times {{10}^{-4}}{{s}^{-1}}\]
done
clear
View Answer play_arrow
question_answer 53) \[{{H}_{3}}B{{O}_{3}}\]is
A)
mono basic add and weak Lewis add
done
clear
B)
mono basic and weak Bronsted add
done
clear
C)
mono basic and strong Lewis add
done
clear
D)
tri basic and weak Bronsted acid
done
clear
View Answer play_arrow
question_answer 54) During depression of freezing point in a solution, the following are in equilibrium
A)
liquid solvent-solid solvent
done
clear
B)
liquid solute-solid solute
done
clear
C)
liquid solute-solid solvent
done
clear
D)
liquid solvent-solid solute
done
clear
View Answer play_arrow
question_answer 55) Benzamide on reaction with \[POC{{l}_{3}}\] gives
A)
aniline
done
clear
B)
chlorobenzene
done
clear
C)
phenyl amine
done
clear
D)
phenyl nitrile
done
clear
View Answer play_arrow
question_answer 56) The acid having \[O-O\] bond is
A)
\[{{H}_{2}}{{S}_{2}}{{O}_{3}}\]
done
clear
B)
\[{{H}_{2}}{{S}_{2}}{{O}_{6}}\]
done
clear
C)
\[{{H}_{2}}{{S}_{2}}{{O}_{8}}\]
done
clear
D)
\[{{H}_{2}}{{S}_{4}}{{O}_{6}}\]
done
clear
View Answer play_arrow
question_answer 57) A sodium salt on treatment with \[MgC{{l}_{2}}\] gives white propitiate on heating. The anion of sodium salt is
A)
\[HCO_{3}^{-}\]
done
clear
B)
\[CO_{3}^{2-}\]
done
clear
C)
\[NO_{3}^{-}\]
done
clear
D)
\[SO_{4}^{2-}\]
done
clear
View Answer play_arrow
question_answer 58) According to molecular orbital theory which is correct statement regarding \[{{O}_{2}}\]?
A)
Paramagnetic and bond order \[<O_{2}^{-}\]
done
clear
B)
Paramagnetic and bond order \[>O_{2}^{-}\]
done
clear
C)
Diamagnetic and bond order \[<O_{2}^{-}\]
done
clear
D)
Diamagnetic and bond order \[>O_{2}^{-}\]
done
clear
View Answer play_arrow
question_answer 59) Adsorption of gases on solid surface is exothermic since
A)
enthalpy is positive
done
clear
B)
entropy is negative
done
clear
C)
entropy increases
done
clear
D)
free energy increases
done
clear
View Answer play_arrow
question_answer 60) Which of the following is not oxidised by ozone?
A)
\[KI\]
done
clear
B)
\[FeS{{O}_{4}}\]
done
clear
C)
\[KMn{{O}_{4}}\]
done
clear
D)
\[{{K}_{2}}Mn{{O}_{4}}\]
done
clear
View Answer play_arrow
question_answer 61) Which is necessary for conversion of butane-2-one into propanoic acid?
A)
Tollen reagent
done
clear
B)
Fehling solution
done
clear
C)
\[NaOH/{{I}_{2}}/{{H}^{+}}\]
done
clear
D)
\[NaOH/NaI/{{H}^{+}}\]
done
clear
View Answer play_arrow
question_answer 62) When phenyl magnesium bromide is reacted with ethyl alcohol, product is
A)
phenol
done
clear
B)
t-butyl benzene
done
clear
C)
benzene
done
clear
D)
t-butyl phenyl ether
done
clear
View Answer play_arrow
question_answer 63) The number of radial nodes of 3s and 2p orbital are
A)
\[2,0\]
done
clear
B)
\[0,2\]
done
clear
C)
\[1,2\]
done
clear
D)
\[2,1\]
done
clear
View Answer play_arrow
question_answer 64) The ratio of rates of diffusion of helium and methane under same conditions of temperature and pressure will be
A)
\[4:1\]
done
clear
B)
\[2:1\]
done
clear
C)
\[1:1\]
done
clear
D)
\[1:2\]
done
clear
View Answer play_arrow
question_answer 65) A metal nitrate reacts with \[KI\] to give black precipitate which turns to orange on addition of \[KI\]. Metal is
A)
\[H{{g}^{2+}}\]
done
clear
B)
\[B{{i}^{3+}}\]
done
clear
C)
\[P{{b}^{2+}}\]
done
clear
D)
\[C{{u}^{+}}\]
done
clear
View Answer play_arrow
question_answer 66) 2-acetoxy benzoic add is
A)
antiseptic
done
clear
B)
antibiotic
done
clear
C)
dye
done
clear
D)
aspirin
done
clear
View Answer play_arrow
question_answer 67) Silica is soluble in
A)
\[HCl\]
done
clear
B)
\[HN{{O}_{3}}\]
done
clear
C)
\[{{H}_{2}}S{{O}_{4}}\]
done
clear
D)
\[HF\]
done
clear
View Answer play_arrow
question_answer 68) The use of alums in purifying muddy water , involves
A)
absorption
done
clear
B)
adsorption
done
clear
C)
dialysis
done
clear
D)
coagulation
done
clear
View Answer play_arrow
question_answer 69) Which type of metal form effective catalyst?
A)
Alkali metal
done
clear
B)
Transition metal
done
clear
C)
Alkaline earth metal
done
clear
D)
Radioactive metal
done
clear
View Answer play_arrow
question_answer 70) The rusting of iron is catalysed by
A)
\[Fe\]
done
clear
B)
\[{{O}_{2}}\]
done
clear
C)
\[{{H}^{+}}\]
done
clear
D)
\[Zn\]
done
clear
View Answer play_arrow
question_answer 71) IUPAC name of the following compound is \[\underset{CN}{\mathop{\underset{|}{\mathop{C}}\,}}\,{{H}_{2}}-\underset{CN}{\mathop{\underset{|}{\mathop{C}}\,}}\,H-\underset{CN}{\mathop{\underset{|}{\mathop{C}}\,}}\,{{H}_{2}}\]
A)
\[1,2,3,-tricyanopropane\]
done
clear
B)
\[1-2,3-cyanopropane\]
done
clear
C)
\[1-2,\text{ }3-propanetrinitrile\]
done
clear
D)
\[3-cyano-1,\text{ }5-dinitrile\]
done
clear
View Answer play_arrow
question_answer 72) Standard free energy change for an equilibrium is zero, the value of \[{{K}_{p}}\] is
A)
zero
done
clear
B)
\[1\]
done
clear
C)
\[2\]
done
clear
D)
\[100\]
done
clear
View Answer play_arrow
question_answer 73) Hydrolysis of \[PC{{l}_{5}}\] gives
A)
\[{{H}_{3}}P{{O}_{3}}\]
done
clear
B)
\[HP{{O}_{3}}\]
done
clear
C)
\[{{H}_{3}}P{{O}_{2}}\]
done
clear
D)
\[{{H}_{3}}P{{O}_{4}}\]
done
clear
View Answer play_arrow
question_answer 74) Which is responsible for 'green house effect'?
A)
Methane
done
clear
B)
\[C{{O}_{2}}\]
done
clear
C)
\[CO\]
done
clear
D)
\[{{N}_{2}}\]
done
clear
View Answer play_arrow
question_answer 75) In solid \[CuS{{O}_{4}}\].\[5{{H}_{2}}O\] copper is coordinated to number of water molecules
A)
\[5\]
done
clear
B)
\[4\]
done
clear
C)
\[1\]
done
clear
D)
\[2\]
done
clear
View Answer play_arrow
question_answer 76) Most stable carbonium ion is
A)
\[p-N{{O}_{2}}-{{C}_{6}}{{H}_{4}}-CH_{2}^{+}\]
done
clear
B)
\[{{C}_{6}}{{H}_{5}}CH_{2}^{+}\]
done
clear
C)
\[p-Cl-{{C}_{6}}{{H}_{4}}-CH_{2}^{+}\]
done
clear
D)
\[p-C{{H}_{3}}O-{{C}_{6}}{{H}_{4}}-CH_{2}^{+}\]
done
clear
View Answer play_arrow
question_answer 77) \[0.2\] molal acid HX ionises 20%. \[{{k}_{f}}\] for acid is\[1-86\text{ }molalit{{y}^{-1}}\]. Freezing point is
A)
\[-0.45\]
done
clear
B)
\[-0.50\]
done
clear
C)
\[-0.31\]
done
clear
D)
\[-0.53\]
done
clear
View Answer play_arrow
question_answer 78) Natural rubber is polymer of
A)
trans-isoprene
done
clear
B)
cis-isoprene
done
clear
C)
mixture of cis and trans-isoprene
done
clear
D)
None of the above
done
clear
View Answer play_arrow
question_answer 79) The most stable conformation of n-butane is
A)
skew
done
clear
B)
eclipsed
done
clear
C)
staggered
done
clear
D)
None of these
done
clear
View Answer play_arrow
question_answer 80) Antidote for carbon monoxide is
A)
carborundum
done
clear
B)
carbogen
done
clear
C)
carbonic acid
done
clear
D)
pure oxygen
done
clear
View Answer play_arrow
question_answer 81) Among the following, the molecule with highest dipole moment is
A)
\[C{{H}_{3}}Cl\]
done
clear
B)
\[C{{H}_{2}}C{{l}_{2}}\]
done
clear
C)
\[CHC{{l}_{3}}\]
done
clear
D)
\[CC{{l}_{4}}\]
done
clear
View Answer play_arrow
question_answer 82) Which are isoelectronic and isostructural?
A)
\[NO_{3}^{-},CO_{3}^{2-}\]
done
clear
B)
\[S{{O}_{3}},NO_{3}^{-}\]
done
clear
C)
\[ClO_{3}^{-},CO_{3}^{2-}\]
done
clear
D)
\[CO_{3}^{2-},S{{O}_{3}}\]
done
clear
View Answer play_arrow
question_answer 83) The species having tetrahedral shape is
A)
\[{{[PdC{{l}_{4}}]}^{2-}}\]
done
clear
B)
\[[Ni(CN)]_{4}^{2-}\]
done
clear
C)
\[{{[Pd{{(CN)}_{4}}]}^{2-}}\]
done
clear
D)
\[{{[NiC{{l}_{4}}]}^{2-}}\]
done
clear
View Answer play_arrow
question_answer 84) Total number of lone pairs of electrons on \[Xe\]in \[XeO{{F}_{4}}\] is
A)
\[0\]
done
clear
B)
\[1\]
done
clear
C)
\[2\]
done
clear
D)
\[3\]
done
clear
View Answer play_arrow
question_answer 85) The number of nodal planes in \[{{p}_{x}}\] orbital is
A)
\[1\]
done
clear
B)
\[2\]
done
clear
C)
\[3\]
done
clear
D)
\[0\]
done
clear
View Answer play_arrow
question_answer 86) Which has the highest nucleophilicity?
A)
\[{{F}^{-}}\]
done
clear
B)
\[O{{H}^{-}}\]
done
clear
C)
\[CH_{3}^{-}\]
done
clear
D)
\[NH_{2}^{-}\]
done
clear
View Answer play_arrow
question_answer 87) Which will exhibit geometrical isomerism?
A)
\[3-phenyl-1-butene\]
done
clear
B)
\[1-phenyl-2-butene\]
done
clear
C)
\[2-phenyl-1-butene\]
done
clear
D)
\[1,\text{ }1-diphenyl-1-propene\]
done
clear
View Answer play_arrow
question_answer 88) Which is the most thermodynamically stable allotropic form of phosphorus?
A)
Red
done
clear
B)
White
done
clear
C)
Black
done
clear
D)
Yellow
done
clear
View Answer play_arrow
question_answer 89) When \[Pb{{O}_{2}}\] reacts with cone \[HN{{O}_{3}},\] the gas evolved is
A)
\[N{{O}_{2}}\]
done
clear
B)
\[{{O}_{2}}\]
done
clear
C)
\[{{N}_{2}}\]
done
clear
D)
\[{{N}_{2}}O\]
done
clear
View Answer play_arrow
question_answer 90) In which crystal alternate tetrahedral voids are occupied?
A)
\[NaCl\]
done
clear
B)
\[Ca{{F}_{2}}\]
done
clear
C)
\[ZnS\]
done
clear
D)
\[N{{a}_{2}}O\]
done
clear
View Answer play_arrow
question_answer 91) In thermodynamics, a process is called reversible when
A)
surrounding and system change into each other
done
clear
B)
there is no boundary between system and surrounding
done
clear
C)
the surrounding are always in equilibrium with the system
done
clear
D)
the system changes into surrounding spontaneously
done
clear
View Answer play_arrow
question_answer 92) In the standard is ation of \[N{{a}_{2}}{{S}_{2}}{{O}_{3}}\] using \[{{K}_{2}}C{{r}_{2}}{{O}_{7}}\] by iodometry, the equivalent weight of \[{{K}_{2}}C{{r}_{2}}{{O}_{7}}\] is
A)
\[\frac{molecular\text{ }weight}{2}\]
done
clear
B)
\[\frac{molecular\text{ }weight}{6}\]
done
clear
C)
\[\frac{molecular\text{ }weight}{3}\]
done
clear
D)
same as molecular weight
done
clear
View Answer play_arrow
question_answer 93) The correct order of first ionisation potential is
A)
\[K>Na>Li\]
done
clear
B)
\[Be>Mg>Ca\]
done
clear
C)
\[B>C>N\]
done
clear
D)
\[Ge>Si>C\]
done
clear
View Answer play_arrow
question_answer 94) The slag formed in extraction of copper is
A)
\[CuFe{{S}_{2}}\]
done
clear
B)
\[C{{u}_{2}}O+FeS\]
done
clear
C)
\[C{{u}_{2}}S+FeO\]
done
clear
D)
\[FeSi{{O}_{3}}\]
done
clear
View Answer play_arrow
question_answer 95) The root mean square velocity of ideal gas at constant pressure varies with density as
A)
\[{{d}^{2}}\]
done
clear
B)
\[d\]
done
clear
C)
\[\sqrt{d}\]
done
clear
D)
\[\frac{1}{\sqrt{d}}\]
done
clear
View Answer play_arrow
question_answer 96) The correct order of acidic strength is
A)
\[C{{l}_{2}}{{O}_{7}}>S{{O}_{2}}>{{P}_{4}}{{O}_{10}}\]
done
clear
B)
\[C{{O}_{2}}>{{N}_{2}}{{O}_{5}}>S{{O}_{3}}\]
done
clear
C)
\[N{{a}_{2}}O>MgO>A{{l}_{2}}{{O}_{3}}\]
done
clear
D)
\[{{K}_{2}}O>CaO>MgO\]
done
clear
View Answer play_arrow
question_answer 97) Standard solution of \[KN{{O}_{3}}\]is used to make salt bridge because
A)
velocity of \[{{K}^{+}}\] is greater than \[NO_{3}^{-}\]
done
clear
B)
velocity of \[NO_{3}^{-}\] is greater than \[{{K}^{+}}\]
done
clear
C)
velocity of \[{{K}^{+}}\] and \[NO_{3}^{-}\] are same
done
clear
D)
\[KN{{O}_{3}}\]is highly soluble in water
done
clear
View Answer play_arrow
question_answer 98) Anhydrous ferric chloride is prepared by
A)
heating \[FeC{{l}_{3}}\]at high temperature h stream of air
done
clear
B)
heating iron in stream of dry chlorine
done
clear
C)
heating ferric oxide with hydrochloric acid
done
clear
D)
reacting iron with hydrochloric acid
done
clear
View Answer play_arrow
question_answer 99) Which represent mode of hybridization \[s{{p}^{2}}-s{{p}^{2}}-sp-sp\] from left to right?
A)
\[{{H}_{2}}C=CH-C\equiv CH\]
done
clear
B)
\[HC\equiv C-C\equiv CH\]
done
clear
C)
\[{{H}_{2}}C=C=C=C{{H}_{2}}\]
done
clear
D)
done
clear
View Answer play_arrow
question_answer 100) \[N{{a}^{23}}\] is more stable isotope of\[Na\]. The process by which \[_{11}N{{a}^{24}}\] can undergo radioactive decay is
A)
\[\beta \]-rays
done
clear
B)
\[\alpha \]-rays
done
clear
C)
\[{{\beta }^{+}}\]-rays
done
clear
D)
\[\gamma \]-rays
done
clear
View Answer play_arrow
question_answer 101) Contractile vacuoles in Protozoa serve the purpose of
A)
water circulation
done
clear
B)
digestion
done
clear
C)
osmoregularion
done
clear
D)
excretion
done
clear
View Answer play_arrow
question_answer 102) A ceil organelle that is exceptionally rich in hydrolytic enzymes is
A)
ribosome
done
clear
B)
endoplasmic reticulum
done
clear
C)
lysosome
done
clear
D)
mitochondria
done
clear
View Answer play_arrow
question_answer 103) Pearls are produced by the animals of phylum
A)
Annelida
done
clear
B)
Arthropoda
done
clear
C)
Mollusca
done
clear
D)
Echinodermata
done
clear
View Answer play_arrow
question_answer 104) Restriction enzymes are
A)
enzymes that restrict uncontrolled growth of cell, which results in cancer
done
clear
B)
enzymes that restrict DNA synthesis to the minimum necessary level and prevent polyploidy
done
clear
C)
enzymes that cleave phosphodi ester linkages in DNA only at a specific sequence yielding fragments of reproducible sizes
done
clear
D)
enzymes that cleave polycistronic m-RNA to yield individual cisirons
done
clear
View Answer play_arrow
question_answer 105) An analysis of a DNA (double stranded) sample yielded 18% cytosine. What would be the percentage of other bases in this sample?
A)
\[T-32%,\text{ }A-32%,\text{ }G-18%\]
done
clear
B)
\[T-32%,A-18%,G-32%\]
done
clear
C)
\[T-18%,\text{ }A-32%,\text{ }G-32%\]
done
clear
D)
\[1-40%,\text{ }A-22%,\text{ }G-20%\]
done
clear
View Answer play_arrow
question_answer 106) Which of the following is a mismatch?
A)
Amphetamine-a stimulant
done
clear
B)
Morphine - an opiate narcotic
done
clear
C)
LSD - a hallucinogen
done
clear
D)
Cocaine - a sedative/tranquillizer
done
clear
View Answer play_arrow
question_answer 107) Which of the following is not related to sex chromosome X or Y?
A)
Turner's syndrome
done
clear
B)
Ktinefelter's syndrome
done
clear
C)
Down's syndrome
done
clear
D)
Haemophilia and colour blindness
done
clear
View Answer play_arrow
question_answer 108) Which of the following is a part of pectoral girdle?
A)
Ilium
done
clear
B)
Ischium
done
clear
C)
Acetabulum
done
clear
D)
Glenoid cavity
done
clear
View Answer play_arrow
question_answer 109) Gir sanctuary is mainly for
A)
rhino
done
clear
B)
tiger
done
clear
C)
lion
done
clear
D)
elephant
done
clear
View Answer play_arrow
question_answer 110) Which of the following organs is called the graveyard of RBCs?
A)
Thymus
done
clear
B)
Liver
done
clear
C)
Spleen
done
clear
D)
Kidney
done
clear
View Answer play_arrow
question_answer 111) Cyanides like KCN, NaCN are lethal to humans because they
A)
interfere with protein synthesis
done
clear
B)
stop the electron transport chain in the last step by combining with cytochrome oxidase
done
clear
C)
stop DNA-replication
done
clear
D)
induce accumulation of waste materials inside the cell
done
clear
View Answer play_arrow
question_answer 112) Which one is an iron storage protein?
A)
Myosin
done
clear
B)
Glutelin
done
clear
C)
Ferritine
done
clear
D)
Immunoglobulin
done
clear
View Answer play_arrow
question_answer 113) Which of the following is a mismatch?
A)
\[\text{ }\!\!\beta\!\!\text{ -}\]cells of islets of Langerhans--insulin
done
clear
B)
\[\text{ }\!\!\alpha\!\!\text{ -}\]celis of islets ofLangerhans--insulin
done
clear
C)
Leydig cells -- testosterone
done
clear
D)
Corpus luteum -- progesterone
done
clear
View Answer play_arrow
question_answer 114) The genetic material of AIDS virus in
A)
double stranded DNA
done
clear
B)
double stranded RNA
done
clear
C)
single stranded RNA
done
clear
D)
single stranded DNA
done
clear
View Answer play_arrow
question_answer 115) Lac is produced by lac insect as
A)
secretion from the body
done
clear
B)
excretion from the body
done
clear
C)
faecal matter
done
clear
D)
excess food oozing out of body
done
clear
View Answer play_arrow
question_answer 116) Water vascular system is characteristic of
A)
Protozoa
done
clear
B)
Porifera
done
clear
C)
Annelida
done
clear
D)
Echinodermata
done
clear
View Answer play_arrow
question_answer 117) Science, which deals with the study of ageing is known as
A)
Teratology
done
clear
B)
Gerontology
done
clear
C)
Limnology
done
clear
D)
Palaeontology
done
clear
View Answer play_arrow
question_answer 118) Taxonomically, which of the following set is matched correctly?
A)
Cattle fish, jelly fish, silver fish
done
clear
B)
Bat, pigeon, kite
done
clear
C)
Lobsters, spider, shrimps
done
clear
D)
Oyster, otter, Octopus
done
clear
View Answer play_arrow
question_answer 119) Which of the following is not a restriction endonuclease?
A)
\[\text{EcoRI}\]
done
clear
B)
\[\text{Hind-III}\]
done
clear
C)
Pst\[\text{-I}\]
done
clear
D)
DNAse\[\text{-I}\]
done
clear
View Answer play_arrow
question_answer 120) Who proposed theory of imprinting?
A)
Niko Tinbergen
done
clear
B)
Charles Darwin
done
clear
C)
Konrad Lorenz
done
clear
D)
Karl Van Frish
done
clear
View Answer play_arrow
question_answer 121) Which of the following has a triple helix structure?
A)
Haemoglobin
done
clear
B)
Keratin
done
clear
C)
Lysozyme
done
clear
D)
Collagen
done
clear
View Answer play_arrow
question_answer 122) Which of the following statement is true?
A)
Saltatory conduction is seen in non-myelinated nerve fibres
done
clear
B)
Nissl's granules are found in muscle fibres
done
clear
C)
Non-myelinated nerve fibres do not possess nodes of Ranvier
done
clear
D)
Non-myelinated nerve fibres are completely enclosed by myelin sheath
done
clear
View Answer play_arrow
question_answer 123) Which combination is mismatched?
A)
Vitamin-D-Rickets
done
clear
B)
Thiamine-Beri-beri
done
clear
C)
Vitamin-K-Sterility
done
clear
D)
Niacin-Pellagra
done
clear
View Answer play_arrow
question_answer 124) Which of the following gases does not cause acid rain?
A)
Sulphur dioxide
done
clear
B)
Methane
done
clear
C)
Nitrous oxide
done
clear
D)
Carbon monoxide
done
clear
View Answer play_arrow
question_answer 125) Five-kingdom scheme to classify living beings has been proposed by
A)
J B S Haldane
done
clear
B)
G G Simpson
done
clear
C)
E Mayr
done
clear
D)
R H Whittaker
done
clear
View Answer play_arrow
question_answer 126) A normal woman is married with a man having hypertrichosis condition. They got one daughter and one son. What is the possibility of this daughter to have hypertrichosis condition?
A)
0%
done
clear
B)
25%
done
clear
C)
50%
done
clear
D)
100%
done
clear
View Answer play_arrow
question_answer 127) Sea horse is
A)
a bird
done
clear
B)
a mammal
done
clear
C)
an amphibian
done
clear
D)
a fish
done
clear
View Answer play_arrow
question_answer 128) The centrum of eighth vertebra of frog is
A)
procoelous
done
clear
B)
heterocoelous
done
clear
C)
amphicoelous
done
clear
D)
opisthocoelous
done
clear
View Answer play_arrow
question_answer 129) The terminal end of chromosomes is called
A)
centromere
done
clear
B)
telomere
done
clear
C)
chromomere
done
clear
D)
metamere
done
clear
View Answer play_arrow
question_answer 130) Which one of the following animals lay eggs yet the female secretes milk?
A)
Bat
done
clear
B)
Kangaroo
done
clear
C)
Platypus
done
clear
D)
Ostrich
done
clear
View Answer play_arrow
question_answer 131) Which one of the following amino acids is not found in proteins?
A)
Arginine
done
clear
B)
Ornithine
done
clear
C)
Aspartic acid
done
clear
D)
Tyrosine
done
clear
View Answer play_arrow
question_answer 132) In human eyes, colour perception is done by
A)
rod cells only
done
clear
B)
cone cells only
done
clear
C)
Both [a] and [b]
done
clear
D)
choroid layer cells
done
clear
View Answer play_arrow
question_answer 133) ADH regulates the permeability of
A)
proximal convoluted tubule
done
clear
B)
collecting tubule and distal convoluted tubule
done
clear
C)
ascending limb of loop of Henie
done
clear
D)
descending limb of loop of Henie
done
clear
View Answer play_arrow
question_answer 134) Which of the following embryonic membranes structure is excretory in function?
A)
Amnion
done
clear
B)
Allantois
done
clear
C)
Yolk sac
done
clear
D)
Vitelline chorion
done
clear
View Answer play_arrow
question_answer 135) Which of the following is a mismatch?
A)
Giraffe - Lamarck
done
clear
B)
Drosophila - Morgan
done
clear
C)
Galapagos island - Darwin's finches
done
clear
D)
Origin of species - Mendel
done
clear
View Answer play_arrow
question_answer 136) Most of the carbon dioxide is transported in blood is
A)
a gas
done
clear
B)
carbaminohaemoglobin
done
clear
C)
serum carbaminoproteins
done
clear
D)
\[HCO_{3}^{-}\]
done
clear
View Answer play_arrow
question_answer 137) Which of the following type of cartilage is found in tervertebral disc of mammal?
A)
Hyaline cartilage
done
clear
B)
Fibrous cartilage
done
clear
C)
Calcified cartilage
done
clear
D)
Elastic cartilage
done
clear
View Answer play_arrow
question_answer 138) Archaeopteryx is
A)
a living fossil
done
clear
B)
a mammal
done
clear
C)
a connecting link between Annelida and Arthropoda
done
clear
D)
a connecting link between reptiles and birds
done
clear
View Answer play_arrow
question_answer 139) Ink glands are found in
A)
sponge
done
clear
B)
star fish
done
clear
C)
sepia
done
clear
D)
jellyfish
done
clear
View Answer play_arrow
question_answer 140) Which of the following is a harmful social insect?
A)
Termite
done
clear
B)
Bombyxmori
done
clear
C)
Tachardia lacca
done
clear
D)
Apis indica
done
clear
View Answer play_arrow
question_answer 141) Which of the following set is a mis-match?
A)
Phosphoenol pyruvate carboxykinase - Gluconeogenesis
done
clear
B)
Phospho dehydrogenase- Glycolysis
done
clear
C)
Succinate dehydrogenase - Kerbs cyclye
done
clear
D)
Urease
done
clear
View Answer play_arrow
question_answer 142) The largest endangered bird in Indiais
A)
vulture
done
clear
B)
flamingo
done
clear
C)
great Indian bustard
done
clear
D)
great Indian hornbill
done
clear
View Answer play_arrow
question_answer 143) 93. A woman has haemophilic son and three normal children. Her genotype and that of her husband with respect to this gene would be
A)
\[\text{XX}\,\text{and}\,{{\text{X}}^{\text{h}}}\text{Y}\]
done
clear
B)
\[{{\text{X}}^{h}}{{\text{X}}^{h}}\,\text{and}\,{{\text{X}}^{\text{h}}}\text{Y}\]
done
clear
C)
\[{{\text{X}}^{h}}{{\text{X}}^{h}}\,\text{and}\,\text{XY}\]
done
clear
D)
\[{{\text{X}}^{h}}\text{X}\,\text{and}\,\text{XY}\]
done
clear
View Answer play_arrow
question_answer 144) The causative agent of filaria is
A)
Wuchereria bancrofti
done
clear
B)
Leishmania donovani
done
clear
C)
Plasmodium vivax
done
clear
D)
Trypanosoma gambiens
done
clear
View Answer play_arrow
question_answer 145) The copying of mRNA to DNA is known as
A)
replication
done
clear
B)
transcription
done
clear
C)
translation
done
clear
D)
reverse transcription
done
clear
View Answer play_arrow
question_answer 146) Which of the following does not make a nest of its own?
A)
Crow
done
clear
B)
Parrot
done
clear
C)
Cuckoo
done
clear
D)
Sparrow
done
clear
View Answer play_arrow
question_answer 147) Which of the following is not a defence evolved by a prey to avoid predators?
A)
Ejection of noxious chemicals
done
clear
B)
Possession of toxic hairs
done
clear
C)
Mimicry of inedible objects
done
clear
D)
Secretion of pheromones
done
clear
View Answer play_arrow
question_answer 148) The sea snakes have
A)
cylindrical tail
done
clear
B)
dry homey scale at tail end
done
clear
C)
laterally compressed tail
done
clear
D)
dorsoventrally flattened tail
done
clear
View Answer play_arrow
question_answer 149) Pernicious anaemia is caused due to
A)
absence of vitamin-K
done
clear
B)
lack of vitamin\[-{{B}_{12}}\]
done
clear
C)
lack of vitamin-C
done
clear
D)
presence of intrinsic factor
done
clear
View Answer play_arrow
question_answer 150) How many molecules of oxygen can bind to a molecule of haemoglobin?
A)
One
done
clear
B)
Two
done
clear
C)
Three
done
clear
D)
Four
done
clear
View Answer play_arrow
question_answer 151) Insectivorous plant grow generally in soil that are deficient in
A)
water
done
clear
B)
nitrogen
done
clear
C)
potassium
done
clear
D)
calcium
done
clear
View Answer play_arrow
question_answer 152) While entering in the neck of a fern archegonium, sperm shows
A)
phototaxy
done
clear
B)
chemotaxy
done
clear
C)
thermotaxy
done
clear
D)
cyclosis
done
clear
View Answer play_arrow
question_answer 153) Plants which flower only once in their life are termed as
A)
monochlamydous
done
clear
B)
monocious
done
clear
C)
monocarpic
done
clear
D)
monosomic
done
clear
View Answer play_arrow
question_answer 154) Triticum vulgare has been found to be presently evolved as
A)
diploid
done
clear
B)
tetraploid
done
clear
C)
pentaploid
done
clear
D)
hexaploid
done
clear
View Answer play_arrow
question_answer 155) Which of the following clogs the cavity of the xylem vessels?
A)
Tyiosis
done
clear
B)
Cystolith
done
clear
C)
Hydathode
done
clear
D)
Raphide
done
clear
View Answer play_arrow
question_answer 156) Which of the following has multiflagellate sperms?
A)
Equisetum
done
clear
B)
Riccia
done
clear
C)
Lycopodium
done
clear
D)
Anthoceros
done
clear
View Answer play_arrow
question_answer 157) Which of the following is known as 'bag moss' ?
A)
Polytrichum
done
clear
B)
Funaria
done
clear
C)
Sphagnum
done
clear
D)
Porella
done
clear
View Answer play_arrow
question_answer 158) Which of the following is known as 'living fossil'?
A)
Pinus
done
clear
B)
Rhynia
done
clear
C)
Gnetum
done
clear
D)
Ginkgo
done
clear
View Answer play_arrow
question_answer 159) Which of following is connecting link between glycolysis and Krebs cycle?
A)
Puruvic acid
done
clear
B)
Isocitric acid
done
clear
C)
Acetyl Co-A
done
clear
D)
phoglyceric acid
done
clear
View Answer play_arrow
question_answer 160) The respiratory process of yeast is
A)
rarely anaerobic
done
clear
B)
anaerobic
done
clear
C)
purely aerobic
done
clear
D)
Both [a] and [b]
done
clear
View Answer play_arrow
question_answer 161) Which of the following is important source of edible protein ?
A)
Spirogyra
done
clear
B)
Porphyra
done
clear
C)
Spirulina
done
clear
D)
Cephaleuros
done
clear
View Answer play_arrow
question_answer 162) The male gametophyte in angiosperm is reduced to
A)
one cell
done
clear
B)
two cells
done
clear
C)
three cells
done
clear
D)
four cells
done
clear
View Answer play_arrow
question_answer 163) Recently discovered anti-cancer drug is obtained from
A)
Taxus
done
clear
B)
Tagetes
done
clear
C)
Tamarix
done
clear
D)
Thea
done
clear
View Answer play_arrow
question_answer 164) Pollen embryo sacs are
A)
Pollen like embryo sacs
done
clear
B)
pollen developed in embryo sacs
done
clear
C)
embryo sacs developed from pollen
done
clear
D)
fused pollen and embryo sac
done
clear
View Answer play_arrow
question_answer 165) The plant hormone produced by Rhizobium nodulation is
A)
IBA
done
clear
B)
NAA
done
clear
C)
2, 4-D
done
clear
D)
IAA
done
clear
View Answer play_arrow
question_answer 166) The tunica-corpus theory was proposed by
A)
Hofmeister
done
clear
B)
Nageli
done
clear
C)
Strabusrger
done
clear
D)
Schmidt
done
clear
View Answer play_arrow
question_answer 167) The part of castor seed that yields oil is
A)
cotyledon
done
clear
B)
caruncle
done
clear
C)
endosperm
done
clear
D)
nucellus
done
clear
View Answer play_arrow
question_answer 168) The character found only in halophytes is
A)
sunken stomata
done
clear
B)
vivipary
done
clear
C)
velamen tissue
done
clear
D)
heterophylly
done
clear
View Answer play_arrow
question_answer 169) A hyaline bisexual and self-fertilized flower that does not open at all is
A)
chasmogamaus
done
clear
B)
apogamous
done
clear
C)
cleistogamous
done
clear
D)
polygamous
done
clear
View Answer play_arrow
question_answer 170) Modification of petiole into leaf like structure is called
A)
cladode
done
clear
B)
phylloclade
done
clear
C)
phyllode
done
clear
D)
pistillode
done
clear
View Answer play_arrow
question_answer 171) Which one of the following is a pseudocarp ?
A)
Apple
done
clear
B)
Guava
done
clear
C)
Tomato
done
clear
D)
Banana
done
clear
View Answer play_arrow
question_answer 172) The part of the flower of Crocus that yields saffron is
A)
calyx
done
clear
B)
corolla
done
clear
C)
perianth
done
clear
D)
style and stigma
done
clear
View Answer play_arrow
question_answer 173) Largest moss is
A)
Pogonatum
done
clear
B)
Funaria
done
clear
C)
Dawsonia
done
clear
D)
Polytrichum
done
clear
View Answer play_arrow
question_answer 174) Oil is reserve food in
A)
Chlamydomonas
done
clear
B)
Oedogonium
done
clear
C)
Vaucheria
done
clear
D)
Chara
done
clear
View Answer play_arrow
question_answer 175) Which of the following statement is correct?
A)
Viruses are obligate parasites
done
clear
B)
All fungi are pathogenic
done
clear
C)
All algae are eukaryotes
done
clear
D)
Bacteria are always harmful to mankind
done
clear
View Answer play_arrow
question_answer 176) Late blight of potato is caused by
A)
Cystopus
done
clear
B)
Phytophthora
done
clear
C)
Altemaria
done
clear
D)
Ustilago
done
clear
View Answer play_arrow
question_answer 177) Viruses contain
A)
only RNA
done
clear
B)
only DNA
done
clear
C)
either DNA or RNA
done
clear
D)
neither DNA nor RNA
done
clear
View Answer play_arrow
question_answer 178) The fungus without mycelium is
A)
Puccmia
done
clear
B)
Phytophthora
done
clear
C)
Rhizopus
done
clear
D)
Saccharomyces
done
clear
View Answer play_arrow
question_answer 179) A T-series bacteriophage' can be recognized by its
A)
tadpole shape
done
clear
B)
rounded shape
done
clear
C)
irregular shape
done
clear
D)
rhomboidal shape
done
clear
View Answer play_arrow
question_answer 180) In gymnosperms, the seeds are naked because they lack
A)
integument
done
clear
B)
nucellus
done
clear
C)
pericarp
done
clear
D)
perianth
done
clear
View Answer play_arrow
question_answer 181) The smallest free living organism is
A)
virus
done
clear
B)
mycoplasma
done
clear
C)
diatom
done
clear
D)
cyano bacterium
done
clear
View Answer play_arrow
question_answer 182) Name the fungus that is edible
A)
Penicillium
done
clear
B)
Mucor
done
clear
C)
Rhizopus
done
clear
D)
Morchell
done
clear
View Answer play_arrow
question_answer 183) Characters of both conifers and cycads are found in
A)
Ginkgo
done
clear
B)
Ephedra
done
clear
C)
Cupressus
done
clear
D)
Tsuga
done
clear
View Answer play_arrow
question_answer 184) Cork is obtained from
A)
Quercus suber
done
clear
B)
Pinus roxbiirghii
done
clear
C)
Cedrus deodara
done
clear
D)
Mangifera indica
done
clear
View Answer play_arrow
question_answer 185) The number of RNA molecules in 60S sub-particles of 80S ribosomes are
A)
five
done
clear
B)
four
done
clear
C)
three
done
clear
D)
two
done
clear
View Answer play_arrow
question_answer 186) The number of ATP molecules required to initiate p-oxidation is
A)
1
done
clear
B)
2
done
clear
C)
3
done
clear
D)
4
done
clear
View Answer play_arrow
question_answer 187) Sprouting of potato under storage condition can be prevented by
A)
auxin
done
clear
B)
gibberellin
done
clear
C)
ethylene
done
clear
D)
cylokinin
done
clear
View Answer play_arrow
question_answer 188) The components of PS-l are located on the
A)
stroma
done
clear
B)
stroma thylakoid
done
clear
C)
granum thyiakoid
done
clear
D)
outer surface of stromal and granal thylakoid
done
clear
View Answer play_arrow
question_answer 189) The final electron acceptor in pseudocyclic electron transport in chloroplast is
A)
\[NAD{{P}^{+}}\]
done
clear
B)
ferredoxin
done
clear
C)
oxygen
done
clear
D)
\[NA{{D}^{+}}\]
done
clear
View Answer play_arrow
question_answer 190) Photolysis of water during photosynthesis occurs with the help of
A)
PS-II
done
clear
B)
PS-I
done
clear
C)
ferredoxin
done
clear
D)
cytochrome
done
clear
View Answer play_arrow
question_answer 191) The' lock and key' theory of enzyme structure and function was proposed by
A)
Morgan
done
clear
B)
Robertson
done
clear
C)
Brown
done
clear
D)
Fischer
done
clear
View Answer play_arrow
question_answer 192) Photosynthetic evolution of oxygen requires
A)
Coenzyme
done
clear
B)
Fe-enzyme
done
clear
C)
Mn-enzyme
done
clear
D)
Zn-enzyme
done
clear
View Answer play_arrow
question_answer 193) For good growth of pollen tube, necessary element is
A)
Ca
done
clear
B)
B
done
clear
C)
Mg
done
clear
D)
Mo
done
clear
View Answer play_arrow
question_answer 194) The organelle associated with photorespiration is
A)
lysosome
done
clear
B)
peroxisome
done
clear
C)
glyoxysome
done
clear
D)
spherosome
done
clear
View Answer play_arrow
question_answer 195) The mutagenic agent among following is
A)
ethyl methane
done
clear
B)
ethylene
done
clear
C)
2, 4-D
done
clear
D)
IAA
done
clear
View Answer play_arrow
question_answer 196) The number of bases per helical turn in ZDNA is
A)
10
done
clear
B)
11
done
clear
C)
12
done
clear
D)
13
done
clear
View Answer play_arrow
question_answer 197) Ribosomal RNA (rRNA) is synthesized in
A)
nucleolus
done
clear
B)
nucleosome
done
clear
C)
cytoplasm
done
clear
D)
ribosome
done
clear
View Answer play_arrow
question_answer 198) Green-house effect with respect to global climate refers to
A)
cooling and moist condition
done
clear
B)
warming effect
done
clear
C)
increase rainfall and greenery
done
clear
D)
desertification
done
clear
View Answer play_arrow
question_answer 199) In India, tropical rain forest occur in
A)
Jamrnu and Kashinir
done
clear
B)
Andaman
done
clear
C)
UttarPradesh
done
clear
D)
Himachal Pradesh
done
clear
View Answer play_arrow
question_answer 200) Plant length is increased by
A)
apical meristem
done
clear
B)
lateral meristem
done
clear
C)
dermatogens
done
clear
D)
periblem
done
clear
View Answer play_arrow
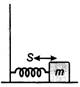
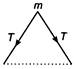 The vibrations are simple harmonic in
The vibrations are simple harmonic in
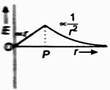 The above figure shows electric field E at a distance r in any direction from the origin O. The electric field E is due to
The above figure shows electric field E at a distance r in any direction from the origin O. The electric field E is due to