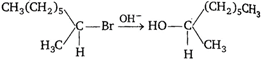question_answer 1) A body floats with one-third of its volume outside water and three-fourth of its volume outside another liquid. The density of another liquid is
A)
\[\frac{9}{4}g/c{{m}^{3}}\]
done
clear
B)
\[4.9g/c{{m}^{3}}\]
done
clear
C)
\[\frac{8}{3}g/c{{m}^{3}}\]
done
clear
D)
\[\frac{3}{8}g/c{{m}^{3}}\]
done
clear
View Answer play_arrow
question_answer 2)
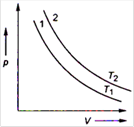
For a certain mass of gas, the isothermal curves between p and V at \[{{T}_{1}}\] and \[{{T}_{2}}\]temperatures are 1 and 2 as shown in figure. Then,
A)
\[{{T}_{1}}={{T}_{2}}\]
done
clear
B)
\[{{T}_{1}}<{{T}_{2}}\]
done
clear
C)
\[{{T}_{1}}>{{T}_{2}}\]
done
clear
D)
Nothing can be predicted
done
clear
View Answer play_arrow
question_answer 3) In simple harmonic motion, when the displacement is one-half of the amplitude, what fraction of the total energy and its kinetic energy is
A)
zero
done
clear
B)
\[\frac{1}{2}\]
done
clear
C)
\[\frac{1}{4}\]
done
clear
D)
\[\frac{3}{4}\]
done
clear
View Answer play_arrow
question_answer 4)
Two bodies M and N of equal masses are suspended from two separate massless springs of spring constants \[{{k}_{1}}\] and \[{{k}_{2}}\]respectively. If the two bodies oscillate vertically such that their maximum velocities are equal, the ratio of the amplitudes of vibrations of M to that of N is
A)
\[\frac{{{k}_{1}}}{{{k}_{2}}}\]
done
clear
B)
\[\sqrt{\frac{{{k}_{1}}}{{{k}_{2}}}}\]
done
clear
C)
\[\frac{{{k}_{2}}}{{{k}_{1}}}\]
done
clear
D)
\[\sqrt{\frac{{{k}_{2}}}{{{k}_{1}}}}\]
done
clear
View Answer play_arrow
question_answer 5)
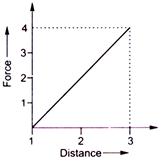
A 10 kg mass moves 3.0 m against a retarding force shown in figure. If the force is zero at the beginning, how much kinetic energy is changed?
A)
+ 6 J
done
clear
B)
- 6 J
done
clear
C)
12 J
done
clear
D)
- 12 J
done
clear
View Answer play_arrow
question_answer 6) A reference frame attached to the earth
A)
is an inertial frame by definition
done
clear
B)
cannot be an inertial frame because the earth is revolving around the sun
done
clear
C)
is an inertial frame because Newtons laws are applicable in this frame
done
clear
D)
cannot be an inertial frame because the earth is rotating about its own axis
done
clear
View Answer play_arrow
question_answer 7) An iron piece falls from a height of 1 km on the ground. If all the energy is converted into heat, the rise of temperature of the iron piece will be (specific heat of iron\[=0.1\text{ }kcal/kg{}^\circ C\])
A)
\[0.233{}^\circ C\]
done
clear
B)
\[2.33{}^\circ C\]
done
clear
C)
\[23.8{}^\circ C\]
done
clear
D)
\[233{}^\circ C\]
done
clear
View Answer play_arrow
question_answer 8) A charged particle moves along the axis of a current carrying solenoid. It experiences
A)
an accelerating force
done
clear
B)
a retarding force
done
clear
C)
a radial force
done
clear
D)
no force
done
clear
View Answer play_arrow
question_answer 9) A straight section PQ of a circuit lies along x-axis at \[x=-\frac{a}{2}\,\,\,to\,\,\,x=+\frac{a}{2}\] and carries a steady current (i), the magnetic field due to section PQ at a point x = + a will be
A)
proportional to z
done
clear
B)
proportional to a2
done
clear
C)
proportional to \[\frac{1}{a}\]
done
clear
D)
equal to zero
done
clear
View Answer play_arrow
question_answer 10) The focal length of the objective of a compound microscope is 2 cm and that of eyepeice of microscope is 5 cm. These two lenses are separated by a distance of 25 cm. When the microscope is focussed for the minimum distance of distinct vision, the magnification will be
A)
5
done
clear
B)
30
done
clear
C)
56.5
done
clear
D)
125
done
clear
View Answer play_arrow
question_answer 11) The moment of inertia of two equal masses each of mass m at separation L connected by a rod of mass M, about an axis passing through centre and perpendicular to length of rod is
A)
\[\frac{\left( M+3m \right){{L}^{2}}}{12}\]
done
clear
B)
\[\frac{\left( M+6m \right){{L}^{2}}}{12}\]
done
clear
C)
\[\frac{M{{L}^{2}}}{4}\]
done
clear
D)
\[\frac{M{{L}^{2}}}{12}\]
done
clear
View Answer play_arrow
question_answer 12) A wire of length L carries a current i. It is bent in the form of a circle. The magnetic moment of current loop (in \[A\text{-}{{m}^{2}}\]) is
A)
\[\frac{i{{L}^{2}}}{4{{\pi }^{2}}}\]
done
clear
B)
\[i\pi {{L}^{2}}\]
done
clear
C)
\[\frac{i{{L}^{2}}}{4\pi }\]
done
clear
D)
\[4\pi i{{L}^{2}}\]
done
clear
View Answer play_arrow
question_answer 13) In an atom bomb, a temperature of about 10 million degree is developed at the moment of explosion. In what region of At spectrum do the wavelength corresponding to maximum energy density lie, if the light source is in the atom bomb?
A)
Ultraviolet region
done
clear
B)
Visible region
done
clear
C)
Infrared region
done
clear
D)
X-ray region
done
clear
View Answer play_arrow
question_answer 14) The absolute temperature of a gas B increased 3 times. The root mean square velocity of the molecules will become
A)
3 times
done
clear
B)
9 times
done
clear
C)
\[\frac{1}{3}\] times
done
clear
D)
\[\sqrt{3}\]times
done
clear
View Answer play_arrow
question_answer 15) The position x of a particle varies with timer as \[x=a{{t}^{2}}-b{{t}^{3}}.\] The acceleration of the particle will be zero at time t equal to
A)
\[\frac{2a}{3b}\]
done
clear
B)
\[\frac{1}{b}\]
done
clear
C)
\[\frac{a}{3b}\]
done
clear
D)
0
done
clear
View Answer play_arrow
question_answer 16) A proton and an \[\alpha \]-particle enter a uniform magnetic field with the same velocity. The time period of rotation of \[\alpha \]- particle will be
A)
two times as that of proton
done
clear
B)
half as that of proton
done
clear
C)
four times as that of proton
done
clear
D)
same as that of proton
done
clear
View Answer play_arrow
question_answer 17) The half-life of radium is 1620 yr and its atomic weight is 226 g/mol. The number of atoms that will decay from its 1 g sample per second is
A)
\[3.61\times {{10}^{10}}\]
done
clear
B)
\[3.6\times {{10}^{12}}\]
done
clear
C)
\[3.1\times {{10}^{15}}\]
done
clear
D)
\[31.1\times {{10}^{15}}\]
done
clear
View Answer play_arrow
question_answer 18) The density of a substance at \[0{}^\circ C\] is 10 g/cc and at \[100{}^\circ C,\] its density is 9.7 g/cc. The coefficient of linear expansion of substance is
A)
\[{{10}^{-4}}\]
done
clear
B)
\[{{10}^{-2}}\]
done
clear
C)
\[{{10}^{-3}}\]
done
clear
D)
\[{{10}^{2}}\]
done
clear
View Answer play_arrow
question_answer 19) The kinetic energy of a body becomes four times its initial value. The new linear momentum will be
A)
eight times that of initial value
done
clear
B)
four times that of initial value
done
clear
C)
twice of the initial value
done
clear
D)
remain as the initial value
done
clear
View Answer play_arrow
question_answer 20) The dimensions of torque are
A)
\[\left[ M{{L}^{3}}{{T}^{-3}} \right]\]
done
clear
B)
\[\left[ M{{L}^{-1}}{{T}^{-1}} \right]\]
done
clear
C)
\[\left[ M{{L}^{2}}{{T}^{-2}} \right]\]
done
clear
D)
\[\left[ M{{T}^{-2}} \right]\]
done
clear
View Answer play_arrow
question_answer 21) At 0 K temperature, a p-type semiconductor
A)
has equal number of holes and free electrons
done
clear
B)
has few holes but no free electrons
done
clear
C)
has few holes and few free electrons
done
clear
D)
does not have any charge carrier
done
clear
View Answer play_arrow
question_answer 22) The coefficient of mutual inductance when magnetic flux changes by \[2\times {{10}^{-2}}\]Wb and current changes by 0.01 A, will be
A)
8 H
done
clear
B)
4 H
done
clear
C)
3 H
done
clear
D)
2 H
done
clear
View Answer play_arrow
question_answer 23) When a wire is stretched and its radius becomes r/2 then its resistance will be
A)
zero
done
clear
B)
2 R
done
clear
C)
8 R
done
clear
D)
16 R
done
clear
View Answer play_arrow
question_answer 24) If equation of a sound wave is y = 0.0015 sin (62.8x + 314t) then its wavelength will be
A)
2 unit
done
clear
B)
0.3 unit
done
clear
C)
0.1 unit
done
clear
D)
0.2 unit
done
clear
View Answer play_arrow
question_answer 25) The latent heat of vaporization of water is 2240 J. If the work done in the process of vaporization of 1 g is 168 J, then increase in internal energy will be
A)
1904 J
done
clear
B)
2072 J
done
clear
C)
2240 J
done
clear
D)
2408 J
done
clear
View Answer play_arrow
question_answer 26) The statement that does not belong to Bohrs model of atom, is
A)
Energy of the electrons in the orbit is quantized
done
clear
B)
The electron in the orbit nearest to the nucleus is in lowest energy state
done
clear
C)
Electrons revolve in different orbits around the nucleus
done
clear
D)
The electrons emit energy during revolution due to the presence of Coulombic forces of attraction
done
clear
View Answer play_arrow
question_answer 27) In the. reaction, \[Po\xrightarrow{-\alpha }Pb\xrightarrow{-\beta }Bi,\] if \[Bi\] belongs to group 15, to which Po belongs?
A)
13
done
clear
B)
14
done
clear
C)
15
done
clear
D)
16
done
clear
View Answer play_arrow
question_answer 28) The average kinetic energy of an ideal gas per molecule in SI units at \[25{}^\circ C\] will be
A)
\[6.17\times {{10}^{-21}}\,kJ\]
done
clear
B)
\[6.17\times {{10}^{-21}}\,J\]
done
clear
C)
\[6.17\times {{10}^{-20}}\,J\]
done
clear
D)
\[7.16\times {{10}^{-20}}\,J\]
done
clear
View Answer play_arrow
question_answer 29) For the reaction of one mole of zinc dust with one mole of sulphuric acid in a bomb calorimeter, AE and W correspond to
A)
\[\Delta E<0,\]\[W=0\]
done
clear
B)
\[\Delta E=0,\,\,W<0\]
done
clear
C)
\[\Delta E>0,\,\,W=0\]
done
clear
D)
\[\Delta E=0,\,\,W>0\]
done
clear
View Answer play_arrow
question_answer 30) The molal elevation constant of water is\[0.52{}^\circ C\]. The boiling point of 1.0 molal aqueous KCl solution (assuming complete dissociation of KCl), therefore, should be
A)
\[98.96{}^\circ C\]
done
clear
B)
\[100.52{}^\circ C\]
done
clear
C)
\[101.04{}^\circ C\]
done
clear
D)
\[107.01{}^\circ C\]
done
clear
View Answer play_arrow
question_answer 31) If the density of water is \[1\,gc{{m}^{-3}}\]then, the volume occupied by one molecule of water is approximately
A)
\[18\,c{{m}^{3}}\]
done
clear
B)
\[22400\,c{{m}^{3}}\]
done
clear
C)
\[6.02\times {{10}^{-23}}c{{m}^{3}}\]
done
clear
D)
\[3.0\times {{10}^{-23}}\,c{{m}^{3}}\]
done
clear
View Answer play_arrow
question_answer 32) In which one of the following gaseous equilibria, \[{{K}_{p}}\] is less than \[{{K}_{c}}\]?
A)
\[{{N}_{2}}{{O}_{4}}\rightleftharpoons 2N{{O}_{2}}\]
done
clear
B)
\[2S{{O}_{2}}+{{O}_{2}}\rightleftharpoons 2S{{O}_{3}}\]
done
clear
C)
\[2HI\rightleftharpoons {{H}_{2}}+{{I}_{2}}\]
done
clear
D)
\[{{N}_{2}}+{{O}_{2}}\rightleftharpoons 2NO\]
done
clear
View Answer play_arrow
question_answer 33) EDTA has coordination number
A)
3
done
clear
B)
4
done
clear
C)
5
done
clear
D)
6
done
clear
View Answer play_arrow
question_answer 34) The lanthanoids contraction relates to
A)
atomic radii
done
clear
B)
atomic as well as \[{{M}^{3+}}\]radii
done
clear
C)
valence electrons
done
clear
D)
oxidation states
done
clear
View Answer play_arrow
question_answer 35) Solder is an alloy of
A)
\[Pb+Sn\]
done
clear
B)
\[Pb+Sn+Zn\]
done
clear
C)
\[Pb+Zn\]
done
clear
D)
\[Sn+Zn\]
done
clear
View Answer play_arrow
question_answer 36) Which of the following compounds reacts with, an aqueous solution of\[Ag{{(N{{H}_{2}})}_{2}}OH\]?
A)
Ethane
done
clear
B)
Ethene
done
clear
C)
1-butyne
done
clear
D)
2-butyne
done
clear
View Answer play_arrow
question_answer 37) 2-methoxy butane is obtained by reacting diazomethane with
A)
2-butanol
done
clear
B)
1-butanol
done
clear
C)
2-butanone
done
clear
D)
butanol
done
clear
View Answer play_arrow
question_answer 38) The formation of aldehyde from alkyl cyanide is related with the name
A)
Stephen
done
clear
B)
Rosenmund
done
clear
C)
Wurtz
done
clear
D)
HVZ reaction
done
clear
View Answer play_arrow
question_answer 39) Which of the following can be used for the preparation of propane?
A)
\[C{{H}_{3}}CH=C{{H}_{2}}\xrightarrow[2.\,\,AgN{{O}_{3}}/NaOH]{1.\,\,{{B}_{2}}{{H}_{6}}}\]
done
clear
B)
\[C{{H}_{3}}C{{H}_{2}}C{{H}_{2}}Cl\xrightarrow[2.\,{{H}_{2}}{{O}_{2}}]{1.\,Mg/ether}\]
done
clear
C)
\[C{{H}_{3}}C{{H}_{2}}C{{H}_{2}}I\xrightarrow[{}]{HI/\Delta 150{}^\circ C}\]
done
clear
D)
\[C{{H}_{3}}C{{H}_{2}}C{{H}_{2}}COONa\xrightarrow[\Delta ]{NaOH(CaO)}\]
done
clear
View Answer play_arrow
question_answer 40) The reactivities of\[C{{H}_{3}}Cl,\]\[C{{H}_{3}}C{{H}_{2}}C{{H}_{2}}Cl\]and \[{{C}_{6}}{{H}_{5}}Cl\]are in the order
A)
\[C{{H}_{3}}Cl>C{{H}_{3}}C{{H}_{2}}C{{H}_{2}}Cl>{{C}_{6}}{{H}_{5}}Cl\]
done
clear
B)
\[C{{H}_{3}}C{{H}_{2}}C{{H}_{2}}Cl>C{{H}_{3}}Cl>{{C}_{6}}{{H}_{5}}Cl\]
done
clear
C)
\[{{C}_{6}}{{H}_{5}}Cl>C{{H}_{3}}C{{H}_{2}}C{{H}_{2}}Cl>C{{H}_{3}}Cl\]
done
clear
D)
\[C{{H}_{3}}Cl>{{C}_{6}}{{H}_{5}}Cl>C{{H}_{3}}C{{H}_{2}}C{{H}_{2}}Cl\]
done
clear
View Answer play_arrow
question_answer 41)
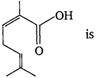
The IUPAC name of the compound
A)
2-ethenyl-3-methyl cyclohexa-1, 3-diene
done
clear
B)
2, 5-dimethyl-hepta-2, 6-dienoic acid
done
clear
C)
2, 6-dimethyl hepta-2, 5-dienoic acid
done
clear
D)
2, 3-dimethyl epoxy ethane
done
clear
View Answer play_arrow
question_answer 42) Which of the following is not isomeric with diethyl ether?
A)
Methyl n-propyl ether
done
clear
B)
Butan-1-ol
done
clear
C)
2-methyl propan-2-ol
done
clear
D)
Butan-2-one
done
clear
View Answer play_arrow
question_answer 43)
The following reaction is described as
A)
\[{{S}_{E}}2\]
done
clear
B)
\[{{S}_{N}}2\]
done
clear
C)
\[{{S}_{N}}1\]
done
clear
D)
\[{{S}_{N}}0\]
done
clear
View Answer play_arrow
question_answer 44) Which of the following species is paramagnetic in nature?
A)
Carbonium ion
done
clear
B)
Free radical
done
clear
C)
Carbene
done
clear
D)
Nitrene
done
clear
View Answer play_arrow
question_answer 45) Which of the following type of forces are present in nylon-66?
A)
van der Waals forces of attraction
done
clear
B)
Hydrogen bonding
done
clear
C)
Three dimensional network of bonds
done
clear
D)
Metallic bonding
done
clear
View Answer play_arrow
question_answer 46) Benzenediazonium chloride on reaction with phenol in weakly basic medium gives
A)
diphenyl ether
done
clear
B)
p-hydroxyazobenzene
done
clear
C)
chlorobenzene
done
clear
D)
benzene
done
clear
View Answer play_arrow
question_answer 47) Which of the following statements are correct?
A)
Aniline is a stronger base than ethyl amine
done
clear
B)
Aniline is a stronger base than p-methoxyaniline
done
clear
C)
Aniline must be acetylated before nitration with an acid derivative
done
clear
D)
Aniline is soluble in an ammonium hydroxide solution
done
clear
View Answer play_arrow
question_answer 48) The species which has the same number of electrons in the outermost as well as penultimate shell is
A)
\[{{F}^{-}}\]
done
clear
B)
\[C{{a}^{2+}}\]
done
clear
C)
\[{{O}^{2-}}\]
done
clear
D)
\[M{{g}^{2+}}\]
done
clear
View Answer play_arrow
question_answer 49) In which one of the following cases, breaking of covalent bond takes place?
A)
Boiling of \[{{H}_{2}}O\]
done
clear
B)
Melting of\[KCN\]
done
clear
C)
Boiling of\[C{{F}_{4}}\]
done
clear
D)
Melting of\[Si{{O}_{2}}\]
done
clear
View Answer play_arrow
question_answer 50) The coefficients of \[{{I}^{-}},\,IO_{3}^{-}\] and \[{{H}^{+}}\]in the redox reaction,\[{{I}^{-}}+IO_{3}^{-}+{{H}^{+}}\xrightarrow{{}}{{I}_{2}}+{{H}_{2}}O\]in the balanced form respectively are
A)
5, 1, 6
done
clear
B)
1, 5, 6
done
clear
C)
6, 1, 5
done
clear
D)
5, 6, 1
done
clear
View Answer play_arrow
question_answer 51) Which one does not matched?
A)
Thyroid - Goitre
done
clear
B)
Enzyme - Substrate
done
clear
C)
Antigen - Antibody
done
clear
D)
Pancreas - Glissons capsule
done
clear
View Answer play_arrow
question_answer 52) Ducts of Bellini are found in
A)
liver
done
clear
B)
intestine
done
clear
C)
kidney
done
clear
D)
pancreas
done
clear
View Answer play_arrow
question_answer 53) Where is protein digestion accomplished?
A)
Rectum
done
clear
B)
Duodenum
done
clear
C)
Stomach
done
clear
D)
lleum
done
clear
View Answer play_arrow
question_answer 54) The covering of the lung is called
A)
peritoneum
done
clear
B)
perichondrium
done
clear
C)
pericardium
done
clear
D)
pleura
done
clear
View Answer play_arrow
question_answer 55) Haemoglobin is found dissolved in the
A)
bird
done
clear
B)
man
done
clear
C)
frog
done
clear
D)
earthworm
done
clear
View Answer play_arrow
question_answer 56) The conversion of a protein waste, the ammonia into urea occurs in
A)
liver
done
clear
B)
kidney
done
clear
C)
gall bladder
done
clear
D)
intestine
done
clear
View Answer play_arrow
question_answer 57) Mouth part type in mosquito is
A)
sucking and piercing type
done
clear
B)
biting and chewing type
done
clear
C)
sponging type
done
clear
D)
None of the above
done
clear
View Answer play_arrow
question_answer 58) Sarcomere is the distance between
A)
A and I- bands
done
clear
B)
two I-bands
done
clear
C)
two Z- lines
done
clear
D)
Z and A bands
done
clear
View Answer play_arrow
question_answer 59) In case of blockage of functioning of islets of Langerhans, which hormone will be in short supply and what will be its effect?
A)
Thyroxine - Retardation of growth
done
clear
B)
Cortison - Tetany
done
clear
C)
Insulin - Blood glucose level rises
done
clear
D)
Adrenalin - Heart beat increases
done
clear
View Answer play_arrow
question_answer 60) Tissue culture technique can produce indefinite number of new plants from a small parental tissue. The economic importance of the technique is in raising
A)
genetically uniform population of an elite species
done
clear
B)
homozygous diploid plants
done
clear
C)
development of new species
done
clear
D)
variants through picking up somaclonal variations
done
clear
View Answer play_arrow
question_answer 61) Germ pore present on the surface of pollen grain represents
A)
specialized thickening of inline
done
clear
B)
specialized thickening of exine
done
clear
C)
area, where exine is thin or absent
done
clear
D)
area, where tectum is absent
done
clear
View Answer play_arrow
question_answer 62) 6-furfuryl amino purine, 2,4-dichloro phenoxy acetic acid and indole-3-acetic acid are
A)
kinetin, synthetic auxin and natural auxin
done
clear
B)
natural auxin, gibberellin and kinetin
done
clear
C)
synthetic auxin, kinetin and natural auxin
done
clear
D)
gibberellin, natural auxin and kinetin
done
clear
View Answer play_arrow
question_answer 63) Twin develops from
A)
different ova
done
clear
B)
different sperms
done
clear
C)
different zygote
done
clear
D)
same zygote
done
clear
View Answer play_arrow
question_answer 64) In a forest or pond ecosystem, pyramid of energy is
A)
always inverted
done
clear
B)
always upright
done
clear
C)
spindle-shaped
done
clear
D)
Both upright and inverted depending upon ecosystem
done
clear
View Answer play_arrow
question_answer 65) Gas being produced by paddy fields and involved in global warming is
A)
\[C{{O}_{2}}\]
done
clear
B)
chlorine
done
clear
C)
methane
done
clear
D)
\[{{H}_{2}}S\]
done
clear
View Answer play_arrow
question_answer 66) Which antiviral substance is produced in response to viral infection for restricting its multiplication?
A)
Virion
done
clear
B)
Antigen
done
clear
C)
Interferon
done
clear
D)
Antibody
done
clear
View Answer play_arrow
question_answer 67) A man shows outbursts of emotions, quarrelsome behaviour and conflicts with others is suffering from
A)
schizophrenia
done
clear
B)
borderline personality disorder
done
clear
C)
addictive disorder
done
clear
D)
mood disorders
done
clear
View Answer play_arrow
question_answer 68) Which one is used for preparation of bread?
A)
Aspergillus
done
clear
B)
Lactobacillus
done
clear
C)
Streptobacillus
done
clear
D)
Saccharomyces cerevisiae
done
clear
View Answer play_arrow
question_answer 69) Restriction nucleases are
A)
present in mammalian cells for degeneration of DNA of dead cells
done
clear
B)
synthesised by bacteria for their defence
done
clear
C)
used for in vitro DNA synthesis
done
clear
D)
used in genetic engineering for uniting two DNA molecules
done
clear
View Answer play_arrow
question_answer 70) A bacteriophage possesses an enzyme
A)
urease
done
clear
B)
protease
done
clear
C)
dehydrogenase
done
clear
D)
lysozyme
done
clear
View Answer play_arrow
question_answer 71) A bacterium with flagella ail around is
A)
peritrichous
done
clear
B)
lophotrichous
done
clear
C)
cephalotrichous
done
clear
D)
amphitrichous
done
clear
View Answer play_arrow
question_answer 72) Sandfly is a causative agent of
A)
typhoid
done
clear
B)
dysentery
done
clear
C)
kala- azar
done
clear
D)
sleeping sickness
done
clear
View Answer play_arrow
question_answer 73) Yeast produces an enzyme complex that is responsible for fermentation. The enzyme complex is
A)
zymase
done
clear
B)
aldolase
done
clear
C)
invertase
done
clear
D)
dehydrogenase
done
clear
View Answer play_arrow
question_answer 74) Pyrenoids are the centres for formation of
A)
starch
done
clear
B)
enzymes
done
clear
C)
fat
done
clear
D)
proteins
done
clear
View Answer play_arrow
question_answer 75) Animals devoid of respiratory, excretory and circulatory organs are
A)
sponges
done
clear
B)
threadworms
done
clear
C)
tapeworm
done
clear
D)
liver fluke
done
clear
View Answer play_arrow
question_answer 76) The train service - Thar Express between India and Pakistan, originates in India from
A)
Jaisalmer
done
clear
B)
Jodhpur
done
clear
C)
Bikaner
done
clear
D)
Banner
done
clear
View Answer play_arrow
question_answer 77) Which one of the following cricketers has been awarded the Rajiv Gandhi Khel Ratna Award for the year 2007?
A)
Sachin Tendulkar
done
clear
B)
Saurav Ganguly
done
clear
C)
MS Dhoni
done
clear
D)
Virender Sehwag
done
clear
View Answer play_arrow
question_answer 78) Garba is a dance form of
A)
Gujarat
done
clear
B)
Rajasthan
done
clear
C)
Orissa
done
clear
D)
Asom
done
clear
View Answer play_arrow
question_answer 79) Which one of the following countries has become the world leader in the carbon credit business in 2007?
A)
India
done
clear
B)
Brazil
done
clear
C)
Mexico
done
clear
D)
China
done
clear
View Answer play_arrow
question_answer 80) Who is named as the Flying Sikh of India?
A)
Mohinder Singh
done
clear
B)
Ajit Pal Singh
done
clear
C)
Joginder Singh
done
clear
D)
Milkha Singh
done
clear
View Answer play_arrow
question_answer 81) What is the tenure of the Prime Minister of India?
A)
Conterminous with the tenure of the Lok Sabha
done
clear
B)
Conterminous with the tenure of the President
done
clear
C)
As long as he enjoys the support of a majority in the Lok Sabha
done
clear
D)
Five years
done
clear
View Answer play_arrow
question_answer 82) What was the reason for Gandhijis support to decentralization of power?
A)
Decentralization ensures more participation of the people into democracy
done
clear
B)
India had decentralization of power in the past
done
clear
C)
Decentralization was essential for the economic development of the country
done
clear
D)
Decentralization can prevent communalism
done
clear
View Answer play_arrow
question_answer 83) Which of the following according to Mahatma Gandhi is the strongest force in the world?
A)
Non-violence of the brave
done
clear
B)
Non-violence of the weak
done
clear
C)
Non-violence of the coward
done
clear
D)
Non-violence of the downtrodden
done
clear
View Answer play_arrow
question_answer 84) Why was the name of Kuber Singh, a 17-year-old student of GD Goenka World School, in the news on 20th January, 2009?
A)
He was declared as the winner of the Sanjay Chopra Award
done
clear
B)
He broke the record for fastest memorizing of a 9 digit number
done
clear
C)
He witnessed the Swearing-in-Ceremony of President Barack Obama of USA, as an official invitee
done
clear
D)
He solved the crossword puzzle of the famous USA magazine
done
clear
View Answer play_arrow
question_answer 85) Who among the following were members of the Swaraj Party? 1. Motilal Nehru 2. Sardar Patel 3. Gopal Krishna Gokhale
A)
1 and 2
done
clear
B)
1 only
done
clear
C)
2 and 3
done
clear
D)
1, 2 and 3
done
clear
View Answer play_arrow
question_answer 86) Consent of the people means Consent of
A)
a few people
done
clear
B)
all people
done
clear
C)
majority of the people
done
clear
D)
leader of the people
done
clear
View Answer play_arrow
question_answer 87) For how many categories has AR Rehman, the Golden Award Winner for original sound track in the film Slum Dog Millionaire been nominated at the Oscars?
A)
1
done
clear
B)
2
done
clear
C)
3
done
clear
D)
9
done
clear
View Answer play_arrow
question_answer 88) Which of the following is not a Panchayati Raj Institution?
A)
Gram Sabha
done
clear
B)
Gram Panchayat
done
clear
C)
Gram Cooperative Society
done
clear
D)
Nyaya Panchayat
done
clear
View Answer play_arrow
question_answer 89) If the radius of blood vessels of a person decreases his/her blood pressure will
A)
increase
done
clear
B)
decrease
done
clear
C)
remain unaffected
done
clear
D)
increase for males and decrease for females
done
clear
View Answer play_arrow
question_answer 90) Cell or tissue death within a living body is called as
A)
Neutrophitia
done
clear
B)
Nephrosis
done
clear
C)
Necrosis
done
clear
D)
Neoplasia
done
clear
View Answer play_arrow
question_answer 91) Insufficient blood supply in human body is referred as
A)
Ischemia
done
clear
B)
Hyperemia
done
clear
C)
Hemostasis
done
clear
D)
Haemorrhage
done
clear
View Answer play_arrow
question_answer 92)
Match the following: Union Territory Jurisdiction (High Court) A. Pondicherry 1. Kerala B. Andaman and Nicobar Islands 2. Bombay C. Lakshadweep 3. Madras D. Daman and Diu 4. Calcutta
A)
A-3 B-4 C-1 D-2
done
clear
B)
A-1 B-3 C-4 D-2
done
clear
C)
A-1 B-2 C-3 D-4
done
clear
D)
A-1 B-4 C-3 D-2
done
clear
View Answer play_arrow
question_answer 93) Which of the following is an extra- constitutional and non - statutory body?
A)
Finance Commission
done
clear
B)
Planning Commission
done
clear
C)
Union Public Service Commission
done
clear
D)
Election Commission
done
clear
View Answer play_arrow
question_answer 94)
Which of the following is true regarding No Confidence Motion in the Parliament? 1. There is no mention of it in the Constitution. 2. A period of six months must lapse between the introduction of one No Confidence Motion and another. 3. At least 100 persons must support such a motion before it is introduced in the House. 4. It can be introduced in the Lok Sabha only.
A)
2 and 4
done
clear
B)
1, 2, 3 and 4
done
clear
C)
1, 2 and 3
done
clear
D)
1 and 4
done
clear
View Answer play_arrow
question_answer 95) Deep fried food materials are carcinogenic because they are rich in
A)
fats
done
clear
B)
hydrocarbons
done
clear
C)
cooking oil
done
clear
D)
nicotine
done
clear
View Answer play_arrow
question_answer 96) With which province of Canada has India signed a historic MoU to boost cooperation and investment in the field of geology and mineral resources?
A)
British Columbia
done
clear
B)
Ontario
done
clear
C)
Quebec
done
clear
D)
Alberta
done
clear
View Answer play_arrow
question_answer 97)
Which of these statements is/are correct? 1. Both HELINA and Namica are future antitank missiles of India. 2. Both HELINA and Namica are versions of Nag Missile.
A)
1 only
done
clear
B)
2 only
done
clear
C)
Both 1 and 2
done
clear
D)
Neither 1 nor 2
done
clear
View Answer play_arrow
question_answer 98) With which Western country has India signed a Counter Terrorism Initiative that includes steps to check financing of terror activities, joint probe in cases of bomb blasts besides cooperation in cyber and border security?
A)
France
done
clear
B)
UK
done
clear
C)
US
done
clear
D)
Germany
done
clear
View Answer play_arrow
question_answer 99) Which of these countries has its independence day on the same day as India has (August 15)?
A)
Vietnam
done
clear
B)
Sri Lanka
done
clear
C)
Ecuador
done
clear
D)
Korea
done
clear
View Answer play_arrow
question_answer 100) Which bowler recently rook his first and One Day Internationals 26th hat-trick?
A)
Muttiah Murlidharan
done
clear
B)
ZaheerKhan
done
clear
C)
Farveez Maharoof
done
clear
D)
None of the above
done
clear
View Answer play_arrow