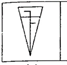
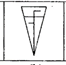
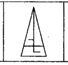
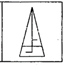
question_answer 1) Identify the false statement from the following
A)
If cohesive force = adhesive force, then angle of contact, \[\theta =90{}^\circ \]
done
clear
B)
If cohesive force < adhesive force, then angle of contact, \[\theta <90{}^\circ \]
done
clear
C)
If cohesive force > adhesive force, then angle of contact, \[\theta >90{}^\circ \]
done
clear
D)
If the radius of capillary is reduced to half, then the rise of liquid column becomes four times
done
clear
View Answer play_arrow
question_answer 2) In the case of a diatomic gas, if heat is given at constant pressure then the part of energy which is used for expansion of gas is
A)
\[\frac{2}{7}\]
done
clear
B)
\[\frac{2}{5}\]
done
clear
C)
\[\frac{3}{7}\]
done
clear
D)
\[\frac{3}{5}\]
done
clear
View Answer play_arrow
question_answer 3) Among the following which is dimensionally correct?
A)
Pressure = Force per unit volume
done
clear
B)
Pressure = Energy per unit volume
done
clear
C)
Pressure = Energy per unit area
done
clear
D)
Pressure = Force per unit volume per unit time
done
clear
View Answer play_arrow
question_answer 4) If pressure of \[C{{O}_{2}}\](real gas) in a container is given by \[p=\frac{RT}{2V-b}-\frac{a}{4{{b}^{2}}}\] Then, mass of the gas in the container is
A)
44 g
done
clear
B)
33 g
done
clear
C)
22 g
done
clear
D)
5g
done
clear
View Answer play_arrow
question_answer 5) The relative velocity of two consecutive layers is 8 \[cm{{s}^{-1}}\] and the perpendicular distance between the layers is 0.1 cm. The velocity gradient is
A)
0.8 \[{{s}^{-1}}\]
done
clear
B)
0.08 \[{{s}^{-1}}\]
done
clear
C)
8.0 \[{{s}^{-1}}\]
done
clear
D)
80 \[{{s}^{-1}}\]
done
clear
View Answer play_arrow
question_answer 6) When a projectile is fired with initial velocity u at an angle of projection 6, then its range is R. If the initial velocity is doubled at same angle of projection, then the new range will be
A)
\[R\]
done
clear
B)
\[\frac{R}{2}\]
done
clear
C)
\[2R\]
done
clear
D)
\[4R\]
done
clear
View Answer play_arrow
question_answer 7) If a resistance of R\[\Omega \] is connected in series with an inductor L and the phase angle between voltage and current is \[\frac{\pi }{4},\]then the value of inductive reactance is
A)
zero
done
clear
B)
\[R\]
done
clear
C)
\[\frac{R}{2}\]
done
clear
D)
\[\frac{R}{4}\]
done
clear
View Answer play_arrow
question_answer 8) If two waves of equal amplitude and frequency interfere each other then the ratio of intensity when the two waves arrive in phase to that when they arrive \[90{}^\circ \]out of phase is
A)
\[\sqrt{2}:1\]
done
clear
B)
\[1:1\]
done
clear
C)
\[4:1\]
done
clear
D)
\[2:1\]
done
clear
View Answer play_arrow
question_answer 9) In a transistor, forward bias is always smaller than the reverse bias because it
A)
helps to produce large voltage gain
done
clear
B)
helps to maintain a constant base current
done
clear
C)
helps in avoiding excessive heating of transistor
done
clear
D)
None of the above
done
clear
View Answer play_arrow
question_answer 10) If the total power rating of a group of electric lamps is 1000 W and they are connected across an AC voltage \[E=200\,\sin \,(310\,t+60{}^\circ )\] Then, the rms value of the circuit current is
A)
\[10\,\sqrt{2}A\]
done
clear
B)
\[20\,\sqrt{2}A\]
done
clear
C)
\[10A\]
done
clear
D)
\[20A\]
done
clear
View Answer play_arrow
question_answer 11) For a particle starting from rest, the acceleration varies with time according to relation, \[A=-a{{\omega }^{2}}\sin \omega t\] The displacement of this particle at a time t will be
A)
\[a\,\omega \,\cos \omega t\]
done
clear
B)
\[a\,\sin \omega t\]
done
clear
C)
\[a\,\omega \sin \omega t\]
done
clear
D)
\[-\frac{1}{2}(a{{\omega }^{2}}\sin \omega t)\,{{t}^{2}}\]
done
clear
View Answer play_arrow
question_answer 12) If a body is moving with uniform speed v in a circle of radius r, then tangential acceleration will be
A)
zero
done
clear
B)
\[\frac{v}{r}\]
done
clear
C)
\[\frac{{{v}^{2}}}{r}\]
done
clear
D)
\[\frac{v}{{{r}^{2}}}\]
done
clear
View Answer play_arrow
question_answer 13) By using the equation \[\tan \theta =\frac{rg}{{{v}^{2}}},\]the value of \[\theta \] (angle of banking) for a cyclist taking a curve can be calculated. But the equation is (symbols have their usual meanings)
A)
dimensionally correct only
done
clear
B)
numerically correct only
done
clear
C)
both dimensionally and numerically correct
done
clear
D)
neither dimensionally nor numerically correct
done
clear
View Answer play_arrow
question_answer 14) The point at which the gravitational potential and the gravitational field due to earth is zero is at
A)
earths surface
done
clear
B)
a height \[{{R}_{e}}\] from earths surface (\[{{R}_{e}}\] = radius of earth)
done
clear
C)
a point below the earths surface
done
clear
D)
infinity
done
clear
View Answer play_arrow
question_answer 15) If the breaking force for a wire is F, then the value of breaking force on increasing the thickness of wire 2 times will be
A)
F
done
clear
B)
2F
done
clear
C)
4F
done
clear
D)
8F
done
clear
View Answer play_arrow
question_answer 16) The Youngs modulus of a material is 3 times its modulus of rigidity, the volume elasticity is
A)
zero
done
clear
B)
infinity
done
clear
C)
\[2\times {{10}^{10}}N\text{-}{{m}^{2}}\]
done
clear
D)
\[3\times {{10}^{10}}N\text{-}{{m}^{-2}}\]
done
clear
View Answer play_arrow
question_answer 17) An arc of radius r carries charge. The linear density of charge is K and the arc subtends an angle of \[60{}^\circ \] at the centre. What is the value of electric potential at the centre?
A)
\[\frac{\lambda }{16\,{{\varepsilon }_{0}}}\]
done
clear
B)
\[\frac{\lambda }{8\,{{\varepsilon }_{0}}}\]
done
clear
C)
\[\frac{\lambda }{12\,{{\varepsilon }_{0}}}\]
done
clear
D)
\[\frac{\lambda }{4\,{{\varepsilon }_{0}}}\]
done
clear
View Answer play_arrow
question_answer 18) Newtons law of cooling is a special case of
A)
Plancks law
done
clear
B)
Kirchhoffs law
done
clear
C)
Wiens law
done
clear
D)
Stefans law
done
clear
View Answer play_arrow
question_answer 19) At a certain temperature, if the r.m.s. velocity of\[{{O}_{2}}\]is 400 m/s then at the same temperature the r.m.s. velocity of\[{{H}_{2}}\]molecules will be
A)
400 m/s
done
clear
B)
1600 m/s
done
clear
C)
3200 m/s
done
clear
D)
6400 m/s
done
clear
View Answer play_arrow
question_answer 20) The moment of inertia of a thin circular lamina of mass 1 kg and diameter 0.2 m rotating about one of its diameter is
A)
\[2.5\times {{10}^{-3}}kg\text{-}{{m}^{2}}\]
done
clear
B)
\[4\times {{10}^{-2}}kg\text{-}{{m}^{2}}\]
done
clear
C)
\[5\times {{10}^{-3}}kg\text{-}{{m}^{2}}\]
done
clear
D)
\[5.5\times {{10}^{-3}}kg\text{-}{{m}^{2}}\]
done
clear
View Answer play_arrow
question_answer 21) If the kinetic energy of a body is decreased by 36%, then its momentum is decreased by
A)
6%
done
clear
B)
8%
done
clear
C)
20%
done
clear
D)
36%
done
clear
View Answer play_arrow
question_answer 22) A man is riding on a cycle with velocity 7.2 km/h up a hill having a slope 1 in 20. The total mass of the man and cycle is 100 kg. The power of the man is
A)
98 W
done
clear
B)
125 W
done
clear
C)
175 W
done
clear
D)
200 W
done
clear
View Answer play_arrow
question_answer 23) When a ring of mass 10 kg and diameter 0.4 m is rotated about its axis it makes 2100 rev/min. The angular momentum of the ring will be
A)
\[0.4\,kg\,{{m}^{2}}{{s}^{-1}}\]
done
clear
B)
\[4.4\,kg\,{{m}^{2}}{{s}^{-1}}\]
done
clear
C)
\[44\,kg\,{{m}^{2}}{{s}^{-1}}\]
done
clear
D)
\[88\,kg\,{{m}^{2}}{{s}^{-1}}\]
done
clear
View Answer play_arrow
question_answer 24) Two electric bulbs one rated 40 W-200 V and other 100 W -200 V are connected in a house wiring circuit. Which one of the following statement about them is correct?
A)
The resistance of the filaments in both the bulbs are same
done
clear
B)
The resistance of filament in 40 W bulb is more than the resistance of 100 W bulb
done
clear
C)
The resistance of the filament in 100 W bulb is more than the resistance of 40 W bulb
done
clear
D)
None of the above
done
clear
View Answer play_arrow
question_answer 25) In the hydrogen atom, the electron is making \[6.6\times {{10}^{15}}\text{rps}\,.\]If the radius of the orbit is \[0.53\times {{10}^{-10}}m,\] then magnetic field produced at the centre of the orbit is
A)
0.14 T
done
clear
B)
1.4T
done
clear
C)
12.5 T
done
clear
D)
140 T
done
clear
View Answer play_arrow
question_answer 26) According to Boolean Algebra \[\overline{\overline{A}\cdot \overline{B}}\cdot A\]equals to
A)
\[A\]
done
clear
B)
\[A+B\]
done
clear
C)
\[\overline{A+B}\]
done
clear
D)
\[\overline{A\cdot B}\]
done
clear
View Answer play_arrow
question_answer 27) The mobility of free electrons is greater than that of free holes because
A)
they are light
done
clear
B)
they require low energy to continue their motion
done
clear
C)
they carry negative charge
done
clear
D)
they mutually collide less
done
clear
View Answer play_arrow
question_answer 28) A light ray is incident by grazing, one of the face of a prism and after refraction ray does not emerge out, what should be the angle of prism while critical angle is C?
A)
\[A>2C\]
done
clear
B)
\[A<2C\]
done
clear
C)
\[A=2C\]
done
clear
D)
None of these
done
clear
View Answer play_arrow
question_answer 29) A man can see only between 75 cm to 200 cm. The power .of lens to correct the near point will be
A)
\[-3D\]
done
clear
B)
\[-\,8/3D\]
done
clear
C)
\[+3D\]
done
clear
D)
\[+\,8/3D\]
done
clear
View Answer play_arrow
question_answer 30)
A double convex lens made up of a material of refractive index \[{{\mu }_{1}}\] is immersed inside two liquids of refractive indices \[{{\mu }_{2}}\] and \[{{\mu }_{3}}\] as shown in figure. If \[{{\mu }_{2}}>{{\mu }_{1}}<{{\mu }_{3}}\] and a wide parallel beam of light incident on the lens from the left, then the lens will give rise to
A)
a convergent beam and a divergent beam
done
clear
B)
two different divergent beams
done
clear
C)
two different convergent beams
done
clear
D)
a single convergent beam
done
clear
View Answer play_arrow
question_answer 31) A radioactive nucleus is being produced at a constant rate a per second. Its decay constant is \[\lambda \]. If \[{{N}_{0}}\]is the number of nuclei at time t = 0, then maximum number of nuclei possible are
A)
\[{{N}_{0}}\]
done
clear
B)
\[{{N}_{0}}+\frac{\alpha }{\lambda }\]
done
clear
C)
\[\frac{\alpha }{\lambda }\]
done
clear
D)
\[\frac{\lambda }{\alpha }+{{N}_{0}}\]
done
clear
View Answer play_arrow
question_answer 32) The bob of a simple pendulum of mass m and length ; is droped from the horizontal position strikes a block of the same mass elastically placed on a horizontal frictionless table. The kinetic energy of the block will be
A)
zero
done
clear
B)
\[mgl\]
done
clear
C)
\[\frac{mgl}{2}\]
done
clear
D)
\[2mgl\]
done
clear
View Answer play_arrow
question_answer 33) Metallic ropes are suspended on the carriers which take inflammable material because
A)
they keep the body of the carrier in contact with the earth
done
clear
B)
they keep the centre of gravity of the carrier nearer to the earth
done
clear
C)
they controlled the speed of the carrier
done
clear
D)
nothing should be placed under the carrier
done
clear
View Answer play_arrow
question_answer 34) The terminal potential difference of a cell is greater than its emf when it is
A)
in open circuit
done
clear
B)
being charged
done
clear
C)
being discharged
done
clear
D)
being either charged or discharged.
done
clear
View Answer play_arrow
question_answer 35) If a magnet is suspended at an angle \[30{}^\circ \] to the magnetic meridian, it makes an angle of \[45{}^\circ \] with the horizontal. The real dip is
A)
\[{{\tan }^{-1}}\left( \sqrt{\frac{3}{2}} \right)\]
done
clear
B)
\[{{\tan }^{-1}}\left( \frac{\sqrt{3}}{2} \right)\]
done
clear
C)
\[{{\tan }^{-1}}\left( \sqrt{3} \right)\]
done
clear
D)
\[{{\tan }^{-1}}\left( \frac{2}{\sqrt{3}} \right)\]
done
clear
View Answer play_arrow
question_answer 36) A Cu disc of radius 0.1 m rotates about its centre with 10 rps in a uniform magnetic field of 0.1 T. The emf induced across the radius of the disc is
A)
\[10\pi \,mV\]
done
clear
B)
\[20\pi \,mV\]
done
clear
C)
\[\frac{\pi }{10}\,V\]
done
clear
D)
\[\frac{\pi }{10}\,V\]
done
clear
View Answer play_arrow
question_answer 37) The coefficient self inductance of a solenoid is 0.18 mH. If a core of soft iron of relative permeability 900 is inserted, then the coefficient of self-inductance will becomes
A)
0.0002 mH
done
clear
B)
0.006 mH
done
clear
C)
5.4 mH
done
clear
D)
162 mH
done
clear
View Answer play_arrow
question_answer 38) If a resonant AC circuit contains a capacitor of capacitance \[{{10}^{-6}}F\] and an inductor of \[{{10}^{-4}}H,\] then the frequency of electrical oscillations will be
A)
\[10\,Hz\]
done
clear
B)
\[{{10}^{5}}\,Hz\]
done
clear
C)
\[\frac{10}{2\pi }\,Hz\]
done
clear
D)
\[\frac{{{10}^{5}}}{2\pi }\,Hz\]
done
clear
View Answer play_arrow
question_answer 39) Threshold frequency for a metal is 1015 Hz. If light of wavelength \[4000\overset{\text{o}}{\mathop{\text{A}}}\,\] falls on its surface, then which of the following statement is correct?
A)
Photo electrons come out with zero speed.
done
clear
B)
Photo electrons come out with \[{{10}^{3}}\]m/s speed.
done
clear
C)
Photo electrons come out with \[{{10}^{5}}\]m/s speed.
done
clear
D)
No photo electric emission takes place.
done
clear
View Answer play_arrow
question_answer 40) In a semiconductor the concentrations of electrons and holes are \[8\times {{10}^{18}}{{m}^{-3}}\]and \[5\times {{10}^{18}}{{m}^{-3}}\]respectively. If the mobilities of electrons and holes are \[2.3\,\,{{m}^{2}}{{V}^{-1}}{{s}^{-1}}\]and \[0.01\]\[0.01\,{{m}^{2}}{{V}^{-1}}{{s}^{-1}}\]respectivey, then semiconductor is
A)
n-type and its resistivity is 0.034 \[\Omega \text{-}m\]
done
clear
B)
n-type and its resistivity is 0.34 \[\Omega \text{-}m\]
done
clear
C)
p-type and its resistivity is 0.034\[\Omega \text{-}m\]
done
clear
D)
p-type and its resistivity is 0.34\[\Omega \text{-}m\]
done
clear
View Answer play_arrow
question_answer 41) The periodic time of a particle executing SHM is 4 s. The time taken by it to go from its mean position to half the maximum displacement (amplitude) is
A)
\[1s\]
done
clear
B)
\[2s\]
done
clear
C)
\[\frac{1}{3}s\]
done
clear
D)
\[\frac{2}{3}s\]
done
clear
View Answer play_arrow
question_answer 42)
When three masses 700g, 500g and 400g are suspended at the end of a spring as shown, they are in equilibrium. If the mass of 700g is removed, then system oscillates 500 g with a period of 3 s, if further 400g 500 g mass is also removed then the system will .oscillate with a period of
A)
1s
done
clear
B)
2s
done
clear
C)
3s
done
clear
D)
4s
done
clear
View Answer play_arrow
question_answer 43) In steady state of thermal conduction, temperature of the ends A and B of a 20 cm long rod are \[100{}^\circ C\] and \[0{}^\circ C\] respectively. What will be the temperature of the rod at a point at a distance of 6 cm from the end A of the rod?
A)
\[5{}^\circ C\]
done
clear
B)
\[70{}^\circ C\]
done
clear
C)
\[-\,30{}^\circ C\]
done
clear
D)
\[-50{}^\circ C\]
done
clear
View Answer play_arrow
question_answer 44)
If three rings each of mass m and radius r are arranged as shown in figure, then the moment of inertia of the system about the axis XX will be
A)
\[3\,m{{r}^{2}}\]
done
clear
B)
\[\frac{3}{2}\,m{{r}^{2}}\]
done
clear
C)
\[7\,m{{r}^{2}}\]
done
clear
D)
\[\frac{7}{2}\,m{{r}^{2}}\]
done
clear
View Answer play_arrow
question_answer 45) If a body of mass 10 kg is dropped to the ground from a height of 5 m, then the work done by the gravitational force is (Take g= 9.8 \[m\text{/}{{s}^{2}}\])
A)
\[+\,490\,J\]
done
clear
B)
\[-\,490\,J\]
done
clear
C)
\[-\,980\,J\]
done
clear
D)
\[+\,980\,J\]
done
clear
View Answer play_arrow
question_answer 46) A mass of 10 kg is sliding along a rough horizontal surface. If the coefficient of friction is \[\frac{1}{\sqrt{3}},\] then the least force which acts at an angle of \[30{}^\circ \] to horizontal is (Take g= 10 \[m\text{/}{{s}^{2}}\])
A)
25 N
done
clear
B)
50 N
done
clear
C)
50 n
done
clear
D)
100N
done
clear
View Answer play_arrow
question_answer 47) The velocity of a particle is given by\[(10+2{{t}^{2}})\,ms.\]The average acceleration of the particle between 2 s and 5 s will be
A)
\[2\,\,m\text{/}{{s}^{2}}\]
done
clear
B)
\[4\,\,m\text{/}{{s}^{2}}\]
done
clear
C)
\[10\,\,m\text{/}{{s}^{2}}\]
done
clear
D)
\[14\,\,m\text{/}{{s}^{2}}\]
done
clear
View Answer play_arrow
question_answer 48) At the highest point of the path of a projectile, its
A)
kinetic energy is minimum
done
clear
B)
kinetic energy is maximum
done
clear
C)
potential energy is minimum
done
clear
D)
total energy is maximum
done
clear
View Answer play_arrow
question_answer 49) If a body of mass m is projected at an angle of \[45{}^\circ \]with the horizontal, then total change in momentum when it strikes the ground will be (Take air resistance negligible)
A)
\[mv\]
done
clear
B)
\[\sqrt{2}\,mv\]
done
clear
C)
\[\frac{mv}{\sqrt{2}}\]
done
clear
D)
\[2\,mv\]
done
clear
View Answer play_arrow
question_answer 50) A body moves from rest with a constant acceleration. Which one of the following graphs represents the variation of its kinetic energy K with the distance travelled\[x?\]
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Answer play_arrow
question_answer 51) An earth satellite is moved from one stable circular orbit to a further stable orbit, then among the following which one will increase?
A)
Linear orbital speed
done
clear
B)
Centripetal acceleration
done
clear
C)
Gravitational potential energy
done
clear
D)
Gravitational force
done
clear
View Answer play_arrow
question_answer 52) The value of g at the surface of the earth is 9.8\[m\text{/}{{s}^{2}}\]. At a place 480 km above the surface of earth, the value of g will be (Take radius of earth = 6400 km)
A)
4.2 \[m\text{/}{{s}^{2}}\]
done
clear
B)
7.2 \[m\text{/}{{s}^{2}}\]
done
clear
C)
8.4 \[m\text{/}{{s}^{2}}\]
done
clear
D)
9.8 \[m\text{/}{{s}^{2}}\]
done
clear
View Answer play_arrow
question_answer 53) Two long freely hanging wires firstly joined in parallel and then in series. When the two combinations are connected with a battery, then which type of force acts between the two wires in two cases?
A)
Repulsive force in both cases
done
clear
B)
Attractive force in both cases
done
clear
C)
Repulsive force when in parallel and attractive force when in series
done
clear
D)
Attractive force when in parallel and repulsive force when in series
done
clear
View Answer play_arrow
question_answer 54) Which one of the following curves represents the curve between magnetic moment and temperature of the magnet?
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Answer play_arrow
question_answer 55) An electric motor draws a current of 10 A when operated on 60 V DC supply. If the efficiency of the motor is 50%, then the resistance of the motor windings is
A)
15\[\Omega \]
done
clear
B)
3\[\Omega \]
done
clear
C)
3\[\Omega \]
done
clear
D)
6\[\Omega \]
done
clear
View Answer play_arrow
question_answer 56) In a series L-C-R circuit, having C = 2uF, L = 1 mH and R = 10\[\Omega \] when the current in the circuit is maximum at that time the ratio of energies stored in the capacitor and the inductor will be
A)
1 : 2
done
clear
B)
2 : 1
done
clear
C)
1 : 5
done
clear
D)
5 : 1
done
clear
View Answer play_arrow
question_answer 57) In the following reaction, what is the value of x? \[{}_{12}M{{g}^{24}}+{{\,}_{2}}H{{e}^{4}}\xrightarrow[{}]{{}}{{\,}_{14}}S{{i}^{x}}+{{\,}_{0}}{{n}^{1}}\]
A)
22
done
clear
B)
26
done
clear
C)
27
done
clear
D)
28
done
clear
View Answer play_arrow
question_answer 58) If before the saturation state of a diode, for the plate voltages 400 V and 200 V the currents are \[{{i}_{1}}\] and \[{{i}_{2}}\] respectively, then the ratio of \[{{i}_{1}}/{{i}_{2}}\] is
A)
\[2/1\]
done
clear
B)
\[1/2\]
done
clear
C)
\[\sqrt{2}/4\]
done
clear
D)
\[2\sqrt{2}/1\]
done
clear
View Answer play_arrow
question_answer 59)
If the logic gates are combined as shown in figure and the logic states of inputs A, B, C are as follows A = B = C = 0 and A = B = 1, C = 0, then the logic states of output D are
A)
1, 0
done
clear
B)
1, 1
done
clear
C)
0, 1
done
clear
D)
0, 0
done
clear
View Answer play_arrow
question_answer 60) Two waves \[{{y}_{1}}=a\,\,\sin 2000\pi t\] and\[{{y}_{2}}=a\,\,\sin 2008\,\pi t\] produces beats, the number of beats heard per second will be
A)
0
done
clear
B)
1
done
clear
C)
4
done
clear
D)
5
done
clear
View Answer play_arrow
question_answer 61) 4.5 g of aluminium (at. mass 27u) is deposited at cathode from \[A{{l}^{3+}}\]solution by certain quantity of electric charge. The volume of hydrogen produced at STP from \[{{H}^{+}}\] ions in solution by the same quantity of electric charge will be
A)
22.4 L
done
clear
B)
44.8 L
done
clear
C)
5.6 L
done
clear
D)
11.2 L
done
clear
View Answer play_arrow
question_answer 62) The rate of a first order reaction is \[1.5\times {{10}^{-2}}\]\[mol\,{{L}^{-}}\,{{\min }^{-1}}\] at 0.5 M concentration of the reactant. The half-life of the reaction is
A)
0.383 min
done
clear
B)
23.1 min
done
clear
C)
8.73 min
done
clear
D)
7.53 min
done
clear
View Answer play_arrow
question_answer 63) A substance \[{{A}_{x}}{{B}_{y}}\] crystallises in a face centred cubic lattice in which A atom occupies each comer of cube and atom B occupies the centres of each face of the cube. Identify the correct composition of the substance\[{{A}_{x}}{{B}_{y}}\].
A)
\[A{{B}_{3}}\]
done
clear
B)
\[{{A}_{4}}{{B}_{3}}\]
done
clear
C)
\[{{A}_{3}}B\]
done
clear
D)
Composition cannot be specified
done
clear
View Answer play_arrow
question_answer 64) The helical structure of protein is stabilized by
A)
dipeptide bonds
done
clear
B)
hydrogen bonds
done
clear
C)
ether bonds
done
clear
D)
peptide bonds
done
clear
View Answer play_arrow
question_answer 65)
The monomer of the polymer
A)
done
clear
B)
\[{{(C{{H}_{3}})}_{2}}C=C{{(C{{H}_{3}})}_{2}}\]
done
clear
C)
\[C{{H}_{3}}CH=CH\cdot C{{H}_{3}}\]
done
clear
D)
\[C{{H}_{3}}CH=C{{H}_{2}}\]
done
clear
View Answer play_arrow
question_answer 66) The enzyme which hydrolyses triglycerides to fatty acids and glycerol is called
A)
maltase
done
clear
B)
lipase
done
clear
C)
zymase
done
clear
D)
pepsin
done
clear
View Answer play_arrow
question_answer 67) The decreasing order of bond angle is
A)
\[N{{O}_{2}}>NO_{2}^{+}>NO_{2}^{-}\]
done
clear
B)
\[NO_{2}^{-}>N{{O}_{2}}>NO_{2}^{+}\]
done
clear
C)
\[NO_{2}^{+}>N{{O}_{2}}>NO_{2}^{-}\]
done
clear
D)
\[NO_{2}^{+}>NO_{2}^{-}>N{{O}_{2}}\]
done
clear
View Answer play_arrow
question_answer 68) The strongest reducing agent among the following is
A)
\[{{F}^{-}}\]
done
clear
B)
\[C{{l}^{-}}\]
done
clear
C)
\[B{{r}^{-}}\]
done
clear
D)
\[{{I}^{-}}\]
done
clear
View Answer play_arrow
question_answer 69) Which of the following compounds is resistant to nucleophilic attack by hydroxyl ion?
A)
Methylacetate
done
clear
B)
Acetonitrile
done
clear
C)
Acetamide
done
clear
D)
Diethylether
done
clear
View Answer play_arrow
question_answer 70) Which of the following is considered to be an anticancer species?
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Answer play_arrow
question_answer 71) \[C{{N}^{-}}\] is a strong field ligand. This is due to the fact that
A)
it carries negative charge
done
clear
B)
it is a pseudohalide
done
clear
C)
it can accept electrons from metal species
done
clear
D)
it forms high spin complexes with metal species
done
clear
View Answer play_arrow
question_answer 72) The correct order of solubility of the sulphates of alkaline earth metals in water is
A)
Be > Ca > Mg > Ba> Sr
done
clear
B)
Mg >Be >Ba > Ca > Sr
done
clear
C)
Be > Mg > Ca > Sr >Ba
done
clear
D)
Mg > Ca > Ba > Be > Sr
done
clear
View Answer play_arrow
question_answer 73) In blast furnace, iron oxide is reduced to iron by
A)
carbon
done
clear
B)
limestone
done
clear
C)
CO
done
clear
D)
zinc
done
clear
View Answer play_arrow
question_answer 74) Aniline when diazotised in cold and then treated with dimethyl aniline gives a coloured product. Its structure would be
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Answer play_arrow
question_answer 75) 100 cc of 0.6 N \[{{H}_{2}}S{{O}_{4}}\] and 200 cc of 0.3 N \[HCl\] were mixed together. The normality of the solution will be
A)
0.2 N
done
clear
B)
0.4 N
done
clear
C)
0.8 N
done
clear
D)
0.6 N
done
clear
View Answer play_arrow
question_answer 76) The absolute enthalpy of neutralisation of the reaction,\[MgO(s)+2HCl(aq)\xrightarrow{{}}MgC{{l}_{2}}(aq)+{{H}_{2}}O(l)\]will be
A)
less than -57.33 kJ\[mo{{l}^{-1}}\]
done
clear
B)
-57.33 kJ \[mo{{l}^{-1}}\]
done
clear
C)
greater than -57.33 kJ \[mo{{l}^{-1}}\]
done
clear
D)
57.33 kJ \[mo{{l}^{-1}}\]
done
clear
View Answer play_arrow
question_answer 77) Which of the following solutions will have the highest boiling point?
A)
\[0.1\,M\,FeC{{l}_{3}}\]
done
clear
B)
\[0.1\,M\,BaC{{l}_{2}}\]
done
clear
C)
\[0.1\,M\,NaCl\]
done
clear
D)
\[0.1\,M\,urea\]
done
clear
View Answer play_arrow
question_answer 78) Which of the following is least reactive in a nucleophilic substitution reaction?
A)
\[{{(C{{H}_{3}})}_{3}}C-Cl\]
done
clear
B)
\[C{{H}_{2}}=CHCl\]
done
clear
C)
\[C{{H}_{3}}C{{H}_{2}}Cl\]
done
clear
D)
\[C{{H}_{2}}=CHC{{H}_{2}}Cl\]
done
clear
View Answer play_arrow
question_answer 79) Which one of the following alkenes will react faster with \[{{H}_{2}}\]under catalytic hydrogenation conditions? (R = Alkyl substituent)
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Answer play_arrow
question_answer 80)
The major organic product formed from the following reaction
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Answer play_arrow
question_answer 81)
In the compound
A)
S, S
done
clear
B)
R, S
done
clear
C)
S, R
done
clear
D)
R, R
done
clear
View Answer play_arrow
question_answer 82) Which one of the following compounds is most acidic?
A)
\[Cl-C{{H}_{2}}-C{{H}_{2}}-OH\]
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Answer play_arrow
question_answer 83) Number of atoms of oxygen present in 10.6 g of \[N{{a}_{2}}C{{O}_{3}}\]will be
A)
\[6.02\times {{10}^{23}}\]
done
clear
B)
\[12.04\times {{10}^{22}}\]
done
clear
C)
\[1.806\times {{10}^{23}}\]
done
clear
D)
\[31.80\times {{10}^{28}}\]
done
clear
View Answer play_arrow
question_answer 84) The surface tension of which of the following liquid is maximum?
A)
\[{{H}_{2}}O\]
done
clear
B)
\[{{C}_{6}}{{H}_{6}}\]
done
clear
C)
\[C{{H}_{3}}OH\]
done
clear
D)
\[{{C}_{2}}{{H}_{5}}OH\]
done
clear
View Answer play_arrow
question_answer 85) Which of the following polymer can be used for lubrication and as an insulator?
A)
PVC
done
clear
B)
PTFE
done
clear
C)
SBR
done
clear
D)
PMMA
done
clear
View Answer play_arrow
question_answer 86)
State the product available in the following reaction.
A)
cyanobenzene
done
clear
B)
benzoic acid
done
clear
C)
cyanoethane
done
clear
D)
p-aminoazobenzene
done
clear
View Answer play_arrow
question_answer 87) The correct order of acid strength is
A)
\[HClO<HCl{{O}_{2}}<HCl{{O}_{3}}<HCl{{O}_{4}}\]
done
clear
B)
\[HCl{{O}_{4}}<HClO<HCl{{O}_{2}}<HCl{{O}_{3}}\]
done
clear
C)
\[HCl{{O}_{2}}<HCl{{O}_{3}}<HCl{{O}_{4}}<HClO\]
done
clear
D)
\[HCl{{O}_{4}}<HCl{{O}_{3}}<HCl{{O}_{2}}<HClO\]
done
clear
View Answer play_arrow
question_answer 88) In the equation\[4M+8C{{N}^{-}}+2{{H}_{2}}O+{{O}_{2}}\to 4\,{{[M\,{{(CN)}_{2}}]}^{-}}+4O{{H}^{-}}\]Identify the metal M.
A)
Copper
done
clear
B)
Iron
done
clear
C)
Silver
done
clear
D)
Zinc
done
clear
View Answer play_arrow
question_answer 89) Which one of the following is expected to exhibit optical isomerism? (en = ethylenediamine)
A)
\[Cis-[Pt{{(N{{H}_{3}})}_{2}}C{{l}_{2}}]\]
done
clear
B)
\[Trans-[Co{{(en)}_{2}}C{{l}_{2}}]\]
done
clear
C)
\[Trans-[Pt{{(N{{H}_{3}})}_{2}}C{{l}_{2}}]\]
done
clear
D)
\[Cis-[Co{{(en)}_{2}}C{{l}_{2}}]\]
done
clear
View Answer play_arrow
question_answer 90) The best method for the separation of naphthalene and benzoic acid from their mixture is
A)
chromatography
done
clear
B)
crystallization
done
clear
C)
distillation
done
clear
D)
sublimation
done
clear
View Answer play_arrow
question_answer 91) The standard emf of a galvanic cell involving cell reaction with n = 2 is found to be 0.295 V at\[25{}^\circ C\]. The equilibrium constant of the reaction would be
A)
\[2.0\times {{10}^{11}}\]
done
clear
B)
\[4.0\times {{10}^{12}}\]
done
clear
C)
\[1.0\times {{10}^{2}}\]
done
clear
D)
\[1.0\times {{10}^{10}}\]
done
clear
View Answer play_arrow
question_answer 92) Milk is
A)
fat dispersed in water
done
clear
B)
fat dispersed in milk
done
clear
C)
fat dispersed in fat
done
clear
D)
water dispersed in milk
done
clear
View Answer play_arrow
question_answer 93) The magnetic moment of \[Cu{{2}^{+}}\] ion is
A)
5.63 BM
done
clear
B)
4.19 BM
done
clear
C)
3.9 BM
done
clear
D)
1.73 BM
done
clear
View Answer play_arrow
question_answer 94) The hydr6gen electrode is dipped in a solution of pH = 3 at \[25{}^\circ C\]. The potential of the cell would be (the value of 2.303 RT/F is 0.059V)
A)
0.177 V
done
clear
B)
0.087 V
done
clear
C)
-0.177 V
done
clear
D)
0.059 V
done
clear
View Answer play_arrow
question_answer 95) If the bond energies of H?H, Br?Br and H?Br are 433 192 and 364 kJ \[mo{{l}^{-1}}\]respectively, then \[\Delta H{}^\circ \] for the reaction \[{{H}_{2}}(g)+B{{r}_{2}}(g)\xrightarrow{{}}2HBr(g)\]is
A)
\[~-\,261\text{ }kJ\]
done
clear
B)
\[+103\text{ }kJ\]
done
clear
C)
\[+\,261\,kJ\]
done
clear
D)
\[-103\,kJ\]
done
clear
View Answer play_arrow
question_answer 96) Which of the following molecules has trigonal planar geometry?
A)
\[I{{F}_{3}}\]
done
clear
B)
\[PC{{l}_{3}}\]
done
clear
C)
\[N{{H}_{3}}\]
done
clear
D)
\[B{{F}_{3}}\]
done
clear
View Answer play_arrow
question_answer 97) Which of the following compounds is obtained when HCHO is reacted with ammonia?
A)
Urotropine
done
clear
B)
Formaldehyde ammonia
done
clear
C)
Methyl amine
done
clear
D)
Ethyl amine
done
clear
View Answer play_arrow
question_answer 98) Which one of the following structures represents the peptide chain?
A)
\[-\overset{\begin{smallmatrix} \text{H} \\ \text{ }\!\!|\!\!\text{ } \end{smallmatrix}}{\mathop{\text{N}}}\,-\underset{\begin{smallmatrix} \text{ }\!\!|\!\!\text{ }\!\!|\!\!\text{ } \\ \text{O} \end{smallmatrix}}{\mathop{\text{C}}}\,-\underset{\begin{smallmatrix} \text{ }\!\!|\!\!\text{ } \\ \text{H} \end{smallmatrix}}{\mathop{\text{C}}}\,-\underset{\text{ }\!\!|\!\!\text{ }}{\overset{\text{ }\!\!|\!\!\text{ }}{\mathop{\text{C}}}}\,-\text{NH}-\overset{\begin{smallmatrix} \text{O} \\ \text{ }\!\!|\!\!\text{ }\!\!|\!\!\text{ } \end{smallmatrix}}{\mathop{\text{C}}}\,-\text{NH}-\]
done
clear
B)
\[-\overset{\begin{smallmatrix} \text{H} \\ \text{ }\!\!|\!\!\text{ } \end{smallmatrix}}{\mathop{\text{N}}}\,-\underset{\begin{smallmatrix} \text{ }\!\!|\!\!\text{ }\!\!|\!\!\text{ } \\ \text{O} \end{smallmatrix}}{\mathop{\text{C}}}\,-\underset{\text{ }\!\!|\!\!\text{ }}{\overset{\text{ }\!\!|\!\!\text{ }}{\mathop{\text{C}}}}\,-\underset{\text{ }\!\!|\!\!\text{ }}{\overset{\text{ }\!\!|\!\!\text{ }}{\mathop{\text{C}}}}\,-\underset{\text{ }\!\!|\!\!\text{ }}{\overset{\text{ }\!\!|\!\!\text{ }}{\mathop{\text{C}}}}\,-\overset{\begin{smallmatrix} \text{H} \\ \text{ }\!\!|\!\!\text{ } \end{smallmatrix}}{\mathop{\text{N}}}\,-\underset{\text{ }\!\!|\!\!\text{ }}{\overset{\text{ }\!\!|\!\!\text{ }}{\mathop{\text{C}}}}\,-\underset{\text{ }\!\!|\!\!\text{ }}{\overset{\text{ }\!\!|\!\!\text{ }}{\mathop{\text{C}}}}\,-\underset{\text{ }\!\!|\!\!\text{ }}{\overset{\text{ }\!\!|\!\!\text{ }}{\mathop{\text{C}}}}\,-\]
done
clear
C)
\[-\overset{\begin{smallmatrix} \text{H} \\ \text{ }\!\!|\!\!\text{ } \end{smallmatrix}}{\mathop{\text{N}}}\,-\underset{\text{ }\!\!|\!\!\text{ }}{\overset{\text{ }\!\!|\!\!\text{ }}{\mathop{\text{C}}}}\,-\underset{\begin{smallmatrix} \text{ }\!\!|\!\!\text{ }\!\!|\!\!\text{ } \\ \text{O} \end{smallmatrix}}{\mathop{\text{C}}}\,-\overset{\begin{smallmatrix} \text{H} \\ \text{ }\!\!|\!\!\text{ } \end{smallmatrix}}{\mathop{\text{N}}}\,-\underset{\text{ }\!\!|\!\!\text{ }}{\overset{\text{ }\!\!|\!\!\text{ }}{\mathop{\text{C}}}}\,-\underset{\begin{smallmatrix} \text{ }\!\!|\!\!\text{ }\!\!|\!\!\text{ } \\ \text{O} \end{smallmatrix}}{\mathop{\text{C}}}\,\]\[-\overset{\begin{smallmatrix} \text{H} \\ \text{ }\!\!|\!\!\text{ } \end{smallmatrix}}{\mathop{\text{N}}}\,-\underset{\text{ }\!\!|\!\!\text{ }}{\overset{\text{ }\!\!|\!\!\text{ }}{\mathop{\text{C}}}}\,-\underset{\begin{smallmatrix} \text{ }\!\!|\!\!\text{ }\!\!|\!\!\text{ } \\ \text{O} \end{smallmatrix}}{\mathop{\text{C}}}\,-\overset{\begin{smallmatrix} \text{H} \\ \text{ }\!\!|\!\!\text{ } \end{smallmatrix}}{\mathop{\text{N}}}\,-\underset{\text{ }\!\!|\!\!\text{ }}{\overset{\text{ }\!\!|\!\!\text{ }}{\mathop{\text{C}}}}\,-\]
done
clear
D)
\[-\underset{\text{ }\!\!|\!\!\text{ }}{\overset{\text{ }\!\!|\!\!\text{ }}{\mathop{\text{C}}}}\,-\overset{\begin{smallmatrix} \text{H} \\ \text{ }\!\!|\!\!\text{ } \end{smallmatrix}}{\mathop{\text{N}}}\,-\underset{\text{ }\!\!|\!\!\text{ }}{\overset{\text{ }\!\!|\!\!\text{ }}{\mathop{\text{C}}}}\,-\underset{\text{ }\!\!|\!\!\text{ }}{\overset{\text{ }\!\!|\!\!\text{ }}{\mathop{\text{C}}}}\,-\overset{\begin{smallmatrix} \text{O} \\ \text{ }\!\!|\!\!\text{ } \end{smallmatrix}}{\mathop{\text{C}}}\,-\underset{\begin{smallmatrix} \text{ }\!\!|\!\!\text{ } \\ \text{H} \end{smallmatrix}}{\mathop{\text{N}}}\,-\underset{\text{ }\!\!|\!\!\text{ }}{\overset{\text{ }\!\!|\!\!\text{ }}{\mathop{\text{C}}}}\,-\underset{\text{ }\!\!|\!\!\text{ }}{\overset{\text{ }\!\!|\!\!\text{ }}{\mathop{\text{C}}}}\,\] \[-\overset{\text{ }\!\!|\!\!\text{ }}{\mathop{\text{N}}}\,-\underset{\begin{smallmatrix} \text{ }\!\!|\!\!\text{ }\!\!|\!\!\text{ } \\ \text{O} \end{smallmatrix}}{\mathop{\text{C}}}\,-\underset{\text{ }\!\!|\!\!\text{ }}{\overset{\text{ }\!\!|\!\!\text{ }}{\mathop{\text{C}}}}\,-\underset{\text{ }\!\!|\!\!\text{ }}{\overset{\text{ }\!\!|\!\!\text{ }}{\mathop{\text{C}}}}\,-\]
done
clear
View Answer play_arrow
question_answer 99) Which of the following is responsible for depletion of the ozone layer in the upper strata of the atmosphere?
A)
Polyhalogens
done
clear
B)
Ferocenes
done
clear
C)
Fullerenes
done
clear
D)
Freons
done
clear
View Answer play_arrow
question_answer 100) The radioactive isotope \[{}_{27}^{60}Co\] which is used in the treatment of cancer can be made by (n, p) reaction. For this reaction the target nucleus is
A)
\[{}_{28}^{58}Ni\]
done
clear
B)
\[{}_{27}^{59}Co\]
done
clear
C)
\[{}_{28}^{60}Ni\]
done
clear
D)
\[{}_{27}^{60}Co\]
done
clear
View Answer play_arrow
question_answer 101) Reaction of HBr with propene in the presence of peroxide gives
A)
isopropyl bromide
done
clear
B)
3-bromopropane
done
clear
C)
allylbromide
done
clear
D)
n-propylbromide
done
clear
View Answer play_arrow
question_answer 102) For a first order reaction, \[A\to B,\] the reaction rate at reactant concentration of 0.01 M is found to be\[2.0\times {{10}^{-15}}mol\,{{L}^{-1}}{{s}^{-1}}.\] The half-life period of the reaction is
A)
220 s
done
clear
B)
30 s
done
clear
C)
300 s
done
clear
D)
347 s
done
clear
View Answer play_arrow
question_answer 103) At \[90{}^\circ C,\] pure water has\[[{{H}_{3}}{{O}^{+}}]={{10}^{-6}}mol\text{/}L.\] The value of \[{{K}_{w}}\] at \[90{}^\circ C\]is
A)
10-6
done
clear
B)
10-8
done
clear
C)
10-12
done
clear
D)
10-14
done
clear
View Answer play_arrow
question_answer 104) 50 mL of 0.1 M\[HCl\] and 50 mL of 0.2 M NaOH are mixed. The pH of the resulting solution is
A)
1.30
done
clear
B)
4.2
done
clear
C)
12.70
done
clear
D)
11.70
done
clear
View Answer play_arrow
question_answer 105) Using anhydrous \[AlC{{l}_{3}}\]as catalyst, which one of the following reactions produce ethyl-benzene (PhEt)?
A)
\[{{H}_{3}}C-C{{H}_{2}}OH+{{C}_{6}}{{H}_{6}}\]
done
clear
B)
\[C{{H}_{3}}-CH=C{{H}_{2}}+{{C}_{6}}{{H}_{6}}\]
done
clear
C)
\[{{H}_{2}}C=C{{H}_{2}}+{{C}_{6}}{{H}_{6}}\]
done
clear
D)
\[{{H}_{3}}C-C{{H}_{3}}+{{C}_{6}}{{H}_{6}}\]
done
clear
View Answer play_arrow
question_answer 106) Which of the following contains cobalt?
A)
Vitamin A
done
clear
B)
Vitamin C
done
clear
C)
Vitamin \[{{B}_{12}}\]
done
clear
D)
Vitamin K
done
clear
View Answer play_arrow
question_answer 107)
The product A is
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Answer play_arrow
question_answer 108) Alizarin is a
A)
nitro dye
done
clear
B)
azo dye
done
clear
C)
anthraquinone dye
done
clear
D)
phthalein dye
done
clear
View Answer play_arrow
question_answer 109) Names of some compounds are given one is not correct in IUPAC system?
A)
\[\begin{align} & C{{H}_{3}}-\underset{OH}{\mathop{\underset{\mathbf{|}}{\mathop{CH}}\,}}\,-\underset{C{{H}_{3}}}{\mathop{\underset{\mathbf{|}}{\mathop{CH}}\,}}\,-C{{H}_{3}} \\ & \,\,\,\,\,\,\text{3-methyl-2-butanol} \\ \end{align}\]
done
clear
B)
\[\begin{align} & C{{H}_{3}}-C\equiv C-CH{{(C{{H}_{3}})}_{2}} \\ & \,\,\,\,\,\,\text{4-methyl-2-butanol} \\ \end{align}\]
done
clear
C)
\[\begin{align} & C{{H}_{3}}-\underset{C{{H}_{3}}}{\mathop{\underset{\mathbf{|}}{\mathop{C{{H}_{2}}}}\,}}\,-\underset{C{{H}_{2}}}{\mathop{\underset{||}{\mathop{C}}\,}}\,-\underset{C{{H}_{3}}}{\mathop{\underset{|}{\mathop{C{{H}_{2}}}}\,}}\, \\ & \text{2-ethyl-3-methyl-but-1-ene} \\ \end{align}\]
done
clear
D)
\[\begin{align} & C{{H}_{3}}-C{{H}_{2}}-C{{H}_{2}}-\underset{C{{H}_{2}}C{{H}_{3}}}{\mathop{\underset{|}{\mathop{CH}}\,}}\,-\overset{C{{H}_{3}}}{\mathop{\overset{|}{\mathop{CH}}\,}}\,-C{{H}_{2}}C{{H}_{3}} \\ & \,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\text{3-methyl-4-ethylheptane} \\ \end{align}\]
done
clear
View Answer play_arrow
question_answer 110) Which of the following is the electro: deficient molecule?
A)
\[{{B}_{2}}{{H}_{6}}\]
done
clear
B)
\[{{C}_{2}}{{H}_{6}}\]
done
clear
C)
\[P{{H}_{3}}\]
done
clear
D)
\[Si{{H}_{4}}\]
done
clear
View Answer play_arrow
question_answer 111) At \[25{}^\circ C\] the dissociation constant of a base BOH is\[1.0\times {{10}^{-12}}\]. The concentration of hydroxyl ions in 0.01 M aqueous solution of the base would be
A)
\[2.0\times {{10}^{-6}}mol\,{{L}^{-1}}\]
done
clear
B)
\[1.0\times {{10}^{-5}}mol\,{{L}^{-1}}\]
done
clear
C)
\[1.0\times {{10}^{-6}}mol\,{{L}^{-1}}\]
done
clear
D)
\[1.0\times {{10}^{-7}}mol\,{{L}^{-1}}\]
done
clear
View Answer play_arrow
question_answer 112) Which of the following compounds is not aromatic?
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Answer play_arrow
question_answer 113) Two nodal planes are present in
A)
\[\overset{*}{\mathop{\pi }}\,2{{p}_{x}}\]
done
clear
B)
\[\sigma 2{{p}_{z}}\]
done
clear
C)
\[\pi 2{{p}_{x}}\]
done
clear
D)
\[\pi 2{{p}_{y}}\]
done
clear
View Answer play_arrow
question_answer 114) Which of the following will be most readily dehydrated under acidic conditions?
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Answer play_arrow
question_answer 115) A solid compound \[X\] on heating gives \[C{{O}_{2}}\]gas and a residue. The residue mixed with water to give \[Y\]. On passing an excess of \[C{{O}_{2}}\]through \[Y\] in water clear solution \[Z\] is obtained. On boiling \[Z\] compound \[X\] is reformed. The compound \[X\] is
A)
\[Ca{{(HC{{O}_{3}})}_{2}}\]
done
clear
B)
\[CaC{{O}_{3}}\]
done
clear
C)
\[N{{a}_{2}}C{{O}_{3}}\]
done
clear
D)
\[{{K}_{2}}C{{O}_{3}}\]
done
clear
View Answer play_arrow
question_answer 116) Four successive members of the first row transition elements are listed below with their atomic numbers. Which one of them is expected to have the highest third ionization enthalpy?
A)
Vanadium (Z = 23)
done
clear
B)
Chromium (Z = 24)
done
clear
C)
Iron (Z = 26)
done
clear
D)
Manganese (Z = 25)
done
clear
View Answer play_arrow
question_answer 117) What is the oxidation state of phosphorus in \[C{{a}_{3}}{{P}_{2}}\]?
A)
+3
done
clear
B)
-3
done
clear
C)
+5
done
clear
D)
-5
done
clear
View Answer play_arrow
question_answer 118) The energy of second Bohr orbit of the hydrogen atom is \[-\,328\text{ }kJ\text{ }mo{{l}^{-1}};\]hence the energy of fourth Bohr orbit would be
A)
\[-\,41\text{ }kJ\text{ }mo{{l}^{-1}}\]
done
clear
B)
\[-1312\,kJ\text{ }mo{{l}^{-1}}\]
done
clear
C)
\[-164\,kJ\,mo{{l}^{-1}}\]
done
clear
D)
\[-\,82\text{ }kJ\text{ }mo{{l}^{-1}}\]
done
clear
View Answer play_arrow
question_answer 119) For d-electron, the orbital angular momentum is
A)
\[\frac{\sqrt{6}\,h}{2\pi }\]
done
clear
B)
\[\frac{\sqrt{2}\,h}{2\pi }\]
done
clear
C)
\[\frac{\,h}{2\pi }\]
done
clear
D)
\[\frac{2\,h}{\pi }\]
done
clear
View Answer play_arrow
question_answer 120) Which one of the following forms micelles in aqueous solution above certain concentration?
A)
Urea
done
clear
B)
Dodecyl trimethyl ammonium chloride
done
clear
C)
Pyridinium chloride
done
clear
D)
Glucose
done
clear
View Answer play_arrow
question_answer 121) Which of the following propagates through leaf-tip?
A)
Walking fern
done
clear
B)
Sprout-leaf plant
done
clear
C)
Marchantia
done
clear
D)
Moss
done
clear
View Answer play_arrow
question_answer 122) Common indicator organism of water pollution is
A)
Lemna pandcostata.
done
clear
B)
Eichhomia crassipes
done
clear
C)
Escherichia coli
done
clear
D)
Entamoeba histolytica
done
clear
View Answer play_arrow
question_answer 123) According to Oparin, which one of the following was not present in the primitive atmosphere of the earth?
A)
Methane
done
clear
B)
Oxygen
done
clear
C)
Hydrogen
done
clear
D)
Water vapour
done
clear
View Answer play_arrow
question_answer 124) Which one of the following precedes re-formation of the nuclear envelope during M-phase of the cell cylce?
A)
De condensation from chromosomes, and reassembly of the nuclear lamina
done
clear
B)
Transcription from chromosomes and reassembly of the nuclear lamina
done
clear
C)
Formation of the contractile ring and formation of the phragmoplast
done
clear
D)
Formation of the contractile ring and transcription from chromosomes
done
clear
View Answer play_arrow
question_answer 125) In transgenics, expression of transgene in target tissue is determined by
A)
enhancer
done
clear
B)
transgene
done
clear
C)
promoter
done
clear
D)
reporter
done
clear
View Answer play_arrow
question_answer 126) Restriction endonuclease
A)
cuts the DNA molecule randomly
done
clear
B)
cuts the DNA molecule at specific sites
done
clear
C)
restricts the synthesis of DNA inside the nucleus
done
clear
D)
synthesizes DNA
done
clear
View Answer play_arrow
question_answer 127) Earthworms are
A)
ureotelic when plenty of water is available
done
clear
B)
uricotelic when plenty of water is available
done
clear
C)
uricotelic under conditions of water scarcity
done
clear
D)
ammonotelic when plenty of water is available
done
clear
View Answer play_arrow
question_answer 128) Which one of the following has an open circulatory system?
A)
Pheretima
done
clear
B)
Periplaneta
done
clear
C)
Hirudinaria
done
clear
D)
Octopus
done
clear
View Answer play_arrow
question_answer 129) Biradial symmetry and lack of cnidoblasts are the characteristics of
A)
Starfish and sea anemone
done
clear
B)
Ctenoplana and Beroe
done
clear
C)
Aurelia and Paramecium
done
clear
D)
Hydra and starfish
done
clear
View Answer play_arrow
question_answer 130) In order to obtain virus-free plants through tissue culture, the best method is
A)
protoplast culture
done
clear
B)
embryo rescue
done
clear
C)
anther culture
done
clear
D)
meristem culture
done
clear
View Answer play_arrow
question_answer 131) Both sickle cell anaemia and Huntingtons chorea are
A)
bacteria-related diseases
done
clear
B)
congenital disorders
done
clear
C)
pollutant-induced disorders
done
clear
D)
virus-related diseases
done
clear
View Answer play_arrow
question_answer 132) Which one of the following pairs is not correctly matched?
A)
Vitamin \[{{B}_{12}}\] - Pernicious anaemia
done
clear
B)
Vitamin \[{{B}_{6}}\] - Loss of appetite
done
clear
C)
Vitamin \[{{B}_{1}}\] - Beri-beri
done
clear
D)
Vitamin \[{{B}_{2}}\] - Pellagra
done
clear
View Answer play_arrow
question_answer 133) Injury to vagus nerve in human is not likely to affect
A)
tongue movements
done
clear
B)
gastrointestinal movements
done
clear
C)
pancreatic secretion
done
clear
D)
cardiac movements
done
clear
View Answer play_arrow
question_answer 134) Environment consists of
A)
living objects of the surroundings
done
clear
B)
non-living objects of the surrounding
done
clear
C)
non-living objects, events and influences of the surroundings
done
clear
D)
living and non-living objects, events and influences of the surroundings
done
clear
View Answer play_arrow
question_answer 135) Study of pollen grains is called
A)
Spermology
done
clear
B)
Anatomy
done
clear
C)
Palynology
done
clear
D)
Aerobiology
done
clear
View Answer play_arrow
question_answer 136) The major source of pollution up to 80% of total air pollution in metropolitan cities is
A)
automobiles (traffic)
done
clear
B)
industries
done
clear
C)
pesticides
done
clear
D)
radioactivity and noise
done
clear
View Answer play_arrow
question_answer 137) Ultimate source of variations is
A)
natural selection
done
clear
B)
sexual reproduction
done
clear
C)
mutations
done
clear
D)
hormonal actions
done
clear
View Answer play_arrow
question_answer 138) Which of the following statement is correct?
A)
Xylem is made up of all living cells.
done
clear
B)
Xylem is made up of living and non-living cells.
done
clear
C)
Xylem is made up of non-living cells.
done
clear
D)
Xylem does not contain cells.
done
clear
View Answer play_arrow
question_answer 139) The boundry/transition between two or more communities sharply defined is called
A)
epilimnion
done
clear
B)
biome
done
clear
C)
anticline
done
clear
D)
ecotone
done
clear
View Answer play_arrow
question_answer 140) Unit of distance between genes in a chromosome is known as
A)
c-DNA
done
clear
B)
Morgan
done
clear
C)
Centi Morgan
done
clear
D)
Spacer
done
clear
View Answer play_arrow
question_answer 141) Age of the plant can be calculated by
A)
measuring its height
done
clear
B)
counting leaf bases
done
clear
C)
counting annual rings
done
clear
D)
All of the above
done
clear
View Answer play_arrow
question_answer 142) In detritus food chain transfer of food is
A)
detrite (dead organic matter)\[\to \]detrivores \[\to \] decomposers
done
clear
B)
detrite\[\to \]microbes\[\to \] detrivores \[\to \] decomposers
done
clear
C)
detrivores\[\to \]organic matter\[\to \]microbe\[\to \] decomposers
done
clear
D)
grass \[\to \] detrivores \[\to \] decomposers
done
clear
View Answer play_arrow
question_answer 143) Criss-cross inheritance means
A)
X-chromosome from male will pass to a male of next generation
done
clear
B)
X-chromosome from a male will pass to a female of next generation
done
clear
C)
X-chromosome from female will pass to female of next generation
done
clear
D)
None of the above
done
clear
View Answer play_arrow
question_answer 144) Cohesion force theory was proposed by
A)
JC Bose (1850)
done
clear
B)
Dixon and Jolly (1950)
done
clear
C)
Benson and Calvin (1855)
done
clear
D)
Dixon and Jolly (1894)
done
clear
View Answer play_arrow
question_answer 145) The cell cultured in vitro gives rise to complete plant. This ability of plant cell is known as
A)
growth
done
clear
B)
development
done
clear
C)
regeneration
done
clear
D)
totipotency
done
clear
View Answer play_arrow
question_answer 146) Living fossils means
A)
primitive organized organisms
done
clear
B)
extinct organisms
done
clear
C)
organisms with ancestral characters and time has not changed them
done
clear
D)
connecting link between two groups
done
clear
View Answer play_arrow
question_answer 147) Biggest source of energy on earth surface is
A)
coal
done
clear
B)
tides
done
clear
C)
atoms
done
clear
D)
solar radiation
done
clear
View Answer play_arrow
question_answer 148) The process of photosynthesis is
A)
reductive, exergonic and catabolic
done
clear
B)
reductive, endergonic and catabolic
done
clear
C)
reductive, exergonic and anabolic
done
clear
D)
reductive, endergonic and anabolic
done
clear
View Answer play_arrow
question_answer 149) Which is the correct sequence of code transfer involved in the formation of polypeptide?
A)
\[\text{DNA}-t\text{RNA}-r\text{RNA}-m\text{RNA}\]
done
clear
B)
\[t\text{RNA}-\text{DNA}-m\text{RNA}-r\text{RNA}\]
done
clear
C)
\[m\text{RNA}-t\text{RNA}-\text{DNA}-\text{amino}\,\text{acid}\]
done
clear
D)
\[\text{DNA}-m\text{RNA}-t\text{RNA}-\text{amino}\,\,\text{acid}\]
done
clear
View Answer play_arrow
question_answer 150) Cancer is the Bank of life which of the following tumour form is non-cancerous?
A)
Benign tumour
done
clear
B)
Malignant tumour
done
clear
C)
Carcinogens
done
clear
D)
All of these
done
clear
View Answer play_arrow
question_answer 151) The Cri-du-chat syndrome is caused by change in chromosome structure involving
A)
deletion
done
clear
B)
duplication
done
clear
C)
inversion
done
clear
D)
translocation
done
clear
View Answer play_arrow
question_answer 152) When synapsis is complete all along the chromosome, the cell is said to have entered a stage called
A)
zygorene
done
clear
B)
pachytene
done
clear
C)
diplotene
done
clear
D)
diakinesis
done
clear
View Answer play_arrow
question_answer 153) How does pruning help in making the hedge dense?
A)
It induces the differentiation of new shoots from the rootstock
done
clear
B)
It frees axillary buds from apical dominance
done
clear
C)
The apical shoot grows faster after pruning
done
clear
D)
It releases wound hormones
done
clear
View Answer play_arrow
question_answer 154) Which one of the following statements is correct?
A)
Neurons regulate endocrine activity, but not vice versa,
done
clear
B)
Endocrine glands regulate neural activity and nervous system regulates endocrine glands.
done
clear
C)
Neither hormones control neural activity nor the neurons control endocrine activity.
done
clear
D)
Endocrine glands regulate neural activity but not vice versa.
done
clear
View Answer play_arrow
question_answer 155) Evolutionary history of an organism is known as
A)
phylogeny
done
clear
B)
ancestry
done
clear
C)
paleontology
done
clear
D)
ontogeny
done
clear
View Answer play_arrow
question_answer 156) Sertoli cells are regulated by the pituitary hormone known as
A)
FSH
done
clear
B)
GH
done
clear
C)
prolactin
done
clear
D)
LH
done
clear
View Answer play_arrow
question_answer 157) Pulmonary artery differs from pulmonary vein in having
A)
no endothelium
done
clear
B)
strong valves
done
clear
C)
Brunners cell
done
clear
D)
thick muscular walls
done
clear
View Answer play_arrow
question_answer 158) Cerebellum portion of brain is
A)
concerned with maintainance of posture/equilibrium
done
clear
B)
responsible for olfactory functions
done
clear
C)
controls optic functions
done
clear
D)
Both (a) and (c)
done
clear
View Answer play_arrow
question_answer 159) Which of the following type of mutation involves the reverse order of genes in a chromosome?
A)
Deletion
done
clear
B)
Duplication
done
clear
C)
Inversion
done
clear
D)
Reciprocal translocation
done
clear
View Answer play_arrow
question_answer 160) Which one of the following can help in the diagnosis of a genetical disorder?
A)
ELISA
done
clear
B)
ABO blood group
done
clear
C)
PCR
done
clear
D)
NMR
done
clear
View Answer play_arrow
question_answer 161) Initiation of polypeptide chain is through
A)
methionine
done
clear
B)
glycine
done
clear
C)
lysine
done
clear
D)
leucine
done
clear
View Answer play_arrow
question_answer 162) Which one of the following is incorrect about the characteristics of protobionts (co-aservates) and microspheres as ensvisaged in the abiogenic origin of life?
A)
They were able to reproduce
done
clear
B)
They could separate combinations of molecules from the surroundings
done
clear
C)
They are partially surrounded by the surroundings
done
clear
D)
They could maintain an internal environment
done
clear
View Answer play_arrow
question_answer 163) At which stage of HIV infection does one usually shows symptoms of AIDS?
A)
When viral DNA is produced by reverse transcriptase
done
clear
B)
When HIV replicates rapidly in helper T-lymphocytes and damages large number of cells
done
clear
C)
Within these 15 days of sexual contact with an infected person
done
clear
D)
When the infecting retrovirus enters the host cells
done
clear
View Answer play_arrow
question_answer 164) When \[C{{O}_{2}}\] concentration in blood increases breathing becomes
A)
shallower and slow
done
clear
B)
there is no effect on breathing
done
clear
C)
slow and deep
done
clear
D)
faster and deeper
done
clear
View Answer play_arrow
question_answer 165) A free living nitrogen fixing cyanobacterium which can also form symbiotic association with the water fern Azolla is
A)
Tolypothrix
done
clear
B)
Chlorella
done
clear
C)
Nostoc
done
clear
D)
Anabaena
done
clear
View Answer play_arrow
question_answer 166) Which of the following hormones is not a secretion product of human placenta?
A)
Human chorionic gonadotropin
done
clear
B)
Prolactin
done
clear
C)
Oestrogen
done
clear
D)
Progesterone
done
clear
View Answer play_arrow
question_answer 167) The international unit used in biology to measure size of objects is
A)
\[\mu \,m\] (micrometer)
done
clear
B)
nm (nanometer)
done
clear
C)
\[\overset{\text{o}}{\mathop{\text{A}}}\,\] (angstrom)
done
clear
D)
All are used
done
clear
View Answer play_arrow
question_answer 168) Out of 1.7 million species of living organisms known to us, insects contributes to about
A)
0.7 million
done
clear
B)
70000
done
clear
C)
70 lakh
done
clear
D)
one lakh
done
clear
View Answer play_arrow
question_answer 169) Primitive earth had
A)
reducing atmosphere (without free oxygen)
done
clear
B)
oxidizing atmosphere (free oxygen)
done
clear
C)
Both (a) and (b)
done
clear
D)
None of the above
done
clear
View Answer play_arrow
question_answer 170) The white fibres are resistant to streaching and formed of a substance, the protein collagen, which on boiling with water yields
A)
gelatin
done
clear
B)
elastin
done
clear
C)
ossein
done
clear
D)
chondrin
done
clear
View Answer play_arrow
question_answer 171) Which one of the following is not used in production of yoghurt?
A)
Streptococcus lactis
done
clear
B)
S. thermophiles
done
clear
C)
Lactobacillus bulgaricus
done
clear
D)
Acetobacter aceti
done
clear
View Answer play_arrow
question_answer 172) Viagra is an anti-impotent drug. Its chemical salt is
A)
sildenafil citrate
done
clear
B)
nitrous oxide
done
clear
C)
nitroglycerine
done
clear
D)
digitoxyline
done
clear
View Answer play_arrow
question_answer 173) Two mating types of a variety of Paramecium are
A)
morphologically different and physio-logically similar
done
clear
B)
morphologically similar and physiologically different
done
clear
C)
physiologically similar
done
clear
D)
physiologically different
done
clear
View Answer play_arrow
question_answer 174) Correct sequence among the following is
A)
Palaeozoic \[\to \]Mesozoic \[\to \] Coenozoic
done
clear
B)
Mesozoic \[\to \] Archaeozoic \[\to \] Proterozoic
done
clear
C)
Palaeozoic \[\to \] Archaeozoic\[\to \] Coenozoic
done
clear
D)
Archaeozoic \[\to \] Palaeozoic\[\to \] Proterozoic
done
clear
View Answer play_arrow
question_answer 175) The epithelial cells lining the stomach of vertebrates is protected from damage by \[HCl\]because
A)
\[HCl\] is too dilute
done
clear
B)
the epithelial cells are resistant to the action of\[HCl\]
done
clear
C)
\[HCl\]is neutralized by alkaline gastric juice
done
clear
D)
the epithelial cells are covered with a mucous secretion
done
clear
View Answer play_arrow
question_answer 176) In situ conservation of genetic diversity is done in the form of
A)
national parks
done
clear
B)
biosphere reserves
done
clear
C)
wildlife sanctuaries
done
clear
D)
All of the above
done
clear
View Answer play_arrow
question_answer 177) Pyrenoids are centre of
A)
fat storage
done
clear
B)
starch storage
done
clear
C)
protein formation
done
clear
D)
enzyme formation
done
clear
View Answer play_arrow
question_answer 178) Damping off a seedling is due to
A)
Nematode
done
clear
B)
Albugo Candida
done
clear
C)
Fusarium oxysporium
done
clear
D)
Pythium debaryanum
done
clear
View Answer play_arrow
question_answer 179) Which one is not a vestigial organ of man?
A)
Nictitating membrane
done
clear
B)
Epiglottis or ileum
done
clear
C)
Vermiform appendix
done
clear
D)
Muscles or ear pinnae
done
clear
View Answer play_arrow
question_answer 180) Reason, will, memory, intelligence and emotions are governed by
A)
cerebellum
done
clear
B)
mid brain
done
clear
C)
medulla oblongata
done
clear
D)
cerebral hemispheres
done
clear
View Answer play_arrow
question_answer 181) Directions: Select the related word/letter/number from the given alternatives. Life : Death :: Beginning : ?
A)
Era
done
clear
B)
End
done
clear
C)
Time
done
clear
D)
Commence
done
clear
View Answer play_arrow
question_answer 182)
Arrange the following words according to English Dictionary. 1. Advertise 2. Admit 3. Addition 4. Adhesive
A)
1, 2, 3, 4
done
clear
B)
3, 4, 2, 1
done
clear
C)
3, 2, 4, 1
done
clear
D)
4, 1, 2, 3
done
clear
View Answer play_arrow
question_answer 183) Which one set of letters when sequentially placed at the gaps in the given letter series shall complete it? KLM_, KL_N, K_MN, _LMN
A)
NMLK
done
clear
B)
KLMN
done
clear
C)
LKNM
done
clear
D)
KLNM
done
clear
View Answer play_arrow
question_answer 184) Find the wrong number in the given series. 6, 14, 31, 64, 137, 280
A)
31
done
clear
B)
64
done
clear
C)
137
done
clear
D)
280
done
clear
View Answer play_arrow
question_answer 185) Pointing to a girl in a bus, a man tells his friend, She is the daughter of the only son of my fathers wife. How is the girl related to the man?
A)
Cousin
done
clear
B)
Daughter
done
clear
C)
Mother
done
clear
D)
Sister
done
clear
View Answer play_arrow
question_answer 186) There are five friends Suresh, Kaushal, Madhur, Amit and Ramesh. Suresh is shorter than Kaushal but taller than Ramesh. Madhur is the tallest. Amit is a little shorter than Kaushal but little taller than Suresh. If they stand in the order of their heights, who will be the shortest?
A)
Amit
done
clear
B)
Madhur
done
clear
C)
Ramesh
done
clear
D)
Kaushal
done
clear
View Answer play_arrow
question_answer 187) From the given alternatives, select the word which cannot be formed using the letters of the given word. HISTORICAL
A)
RICHES
done
clear
B)
CHARIOT
done
clear
C)
CHART
done
clear
D)
CHAOS
done
clear
View Answer play_arrow
question_answer 188)
Directions: Select the missing number from the given responses.
A)
64
done
clear
B)
60
done
clear
C)
52
done
clear
D)
54
done
clear
View Answer play_arrow
question_answer 189)
Directions: Select the missing number from the given responses.
A)
3
done
clear
B)
4
done
clear
C)
6
done
clear
D)
7
done
clear
View Answer play_arrow
question_answer 190) X walks Southwards and then turns right, then left and then right. In which direction is he moving now?
A)
South
done
clear
B)
North
done
clear
C)
West
done
clear
D)
South-West
done
clear
View Answer play_arrow
question_answer 191) Kalpana travelled from point B straight to C, a distance of 8 ft. She turned left and walked 5 ft away. Again, she turned left and walked 7 ft and finally turned left and walked 5 ft. How far is she from the starting point?
A)
3 ft
done
clear
B)
4 ft
done
clear
C)
1 ft
done
clear
D)
5 ft
done
clear
View Answer play_arrow
question_answer 192)
Two statements are given followed by two Conclusions I and II. You have to consider the statements to be true even if they seem to be at variance from commonly known facts. You are to decide which of the given conclusions, if any, follow from the given statements. Indicate your answer. Statements Teaching is an art. Drawing is also an art. Conclusions I. All artists are teachers. II. All artists know to draw pictures.
A)
Only Conclusion I follows
done
clear
B)
Only Conclusion II follows
done
clear
C)
Neither Conclusion I nor II follows
done
clear
D)
Both Conclusions I and II follow
done
clear
View Answer play_arrow
question_answer 193)
Which answer figure will complete the pattern in the question figure? Question Figure
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Answer play_arrow
question_answer 194)
A piece of paper is folded and cut as shown below in the question figures. From the given answer figures, indicate how it will appear when opened? Questions Figures
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Answer play_arrow
question_answer 195)
Which of the answer figure is exactly the mirror image of the given figure, when the mirror is held on the line AB? Question Figure
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Answer play_arrow
question_answer 196) Directions:- Some parts of the sentence have errors and some are correct. Find out which part of a sentence has an error corresponding to the appropriate letter (a), (b) and (c). If a sentence is free from errors, mark the answer (d).
A)
Supposing if
done
clear
B)
/ it rains
done
clear
C)
/ what shall we do?
done
clear
D)
/ No error
done
clear
View Answer play_arrow
question_answer 197) Directions:- Some parts of the sentence have errors and some are correct. Find out which part of a sentence has an error corresponding to the appropriate letter (a), (b) and (c). If a sentence is free from errors, mark the answer (d).
A)
The captain along with his team
done
clear
B)
/ are practicing very hard
done
clear
C)
/ for the forthcoming match,
done
clear
D)
/ No error
done
clear
View Answer play_arrow
question_answer 198) Directions: Out of the four alternatives, choose the one which best expresses the meaning of the given word and mark it in the answer sheet. Defer
A)
Indifferent
done
clear
B)
Defy
done
clear
C)
Differ
done
clear
D)
Postpone
done
clear
View Answer play_arrow
question_answer 199) Directions: Out of the four alternatives, choose the one which best expresses the meaning of the given word and mark it in the answer sheet. Cease
A)
Begin
done
clear
B)
Stop
done
clear
C)
Create
done
clear
D)
Dull
done
clear
View Answer play_arrow
question_answer 200) Directions: Out of the four alternatives, choose the one which best expresses the meaning of the given word and mark it in the answer sheet. Pious
A)
Religious
done
clear
B)
Sympathetic
done
clear
C)
Afraid
done
clear
D)
Faithful
done
clear
View Answer play_arrow
question_answer 201) Directions: Choose the word opposite in meaning to the given word and mark it in the answer sheet. Stingy
A)
Clean
done
clear
B)
Tight
done
clear
C)
Generous
done
clear
D)
Cheap
done
clear
View Answer play_arrow
question_answer 202) Directions: Choose the word opposite in meaning to the given word and mark it in the answer sheet. Barren
A)
Fertile
done
clear
B)
Rich
done
clear
C)
Prosperous
done
clear
D)
Positive
done
clear
View Answer play_arrow
question_answer 203) Directions: Four alternatives are given for the idiom/phrase. Choose the alternative which best expresses the meaning of the idiom/phrase and mark it in the answer sheet To look down ones nose
A)
To show anger
done
clear
B)
To retaliate
done
clear
C)
To insult in the presence of others
done
clear
D)
To regard with contempt
done
clear
View Answer play_arrow
question_answer 204) Directions: Four alternatives are given for the idiom/phrase. Choose the alternative which best expresses the meaning of the idiom/phrase and mark it in the answer sheet To shed crocodile tears
A)
To weep profusely
done
clear
B)
To pretend grief
done
clear
C)
To grieve seriously
done
clear
D)
To mock something
done
clear
View Answer play_arrow
question_answer 205) Directions: A part of the sentence is underlined which may need improvement. Alternatives are given at (a), (b) and (c) below, which may be better option. In case no improvement is needed, your answer is (d). The cheapest electronic good are manufactured in China
A)
electronical good
done
clear
B)
electrical good
done
clear
C)
electronic goods
done
clear
D)
No improvement
done
clear
View Answer play_arrow
question_answer 206) Directions: A part of the sentence is underlined which may need improvement. Alternatives are given at (a), (b) and (c) below, which may be better option. In case no improvement is needed, your answer is (d). I have been living in Delhi from 1989.
A)
eversince
done
clear
B)
since
done
clear
C)
in
done
clear
D)
No improvement
done
clear
View Answer play_arrow
question_answer 207) Directions: Out of the four alternatives, choose the one which can be substituted for the given words/sentence. Murder of a man
A)
Regicide
done
clear
B)
Fratricide
done
clear
C)
Homicide
done
clear
D)
Genocide
done
clear
View Answer play_arrow
question_answer 208) Directions: Out of the four alternatives, choose the one which can be substituted for the given words/sentence. Use of force or threats to get someone to agree to something
A)
Coercion
done
clear
B)
Conviction
done
clear
C)
Confession
done
clear
D)
Cajolement
done
clear
View Answer play_arrow
question_answer 209) Directions: In the following questions, group of four words are given. In each group, one word is correctly spelt. Find the correctly spelt word and mark it in the answer sheet.
A)
Livelihood
done
clear
B)
Livelyhood
done
clear
C)
Livlihood
done
clear
D)
Livelyhud
done
clear
View Answer play_arrow
question_answer 210) Directions: In the following questions, group of four words are given. In each group, one word is correctly spelt. Find the correctly spelt word and mark it in the answer sheet.
A)
Foyere
done
clear
B)
Foayer
done
clear
C)
Foyer
done
clear
D)
Fouyer
done
clear
View Answer play_arrow
question_answer 211) If the sum of five consecutive integers is S, then the largest of those integers in terms of S is
A)
\[\frac{S-10}{5}\]
done
clear
B)
\[\frac{S+4}{4}\]
done
clear
C)
\[\frac{S+5}{4}\]
done
clear
D)
\[\frac{S+10}{5}\]
done
clear
View Answer play_arrow
question_answer 212) The greatest among the numbers\[3\sqrt{2},3\sqrt{7},6\sqrt{5},2\sqrt{20}\] is
A)
\[3\sqrt{2}\]
done
clear
B)
\[3\sqrt{7}\]
done
clear
C)
\[6\sqrt{5}\]
done
clear
D)
\[2\sqrt{20}\]
done
clear
View Answer play_arrow
question_answer 213) The denominator of a fraction is 3 more than its numerator. If the numerator is increased by 7 and the denominator is decreased by 2, we obtain 2. The sum of numerator and denominator of the fraction is
A)
5
done
clear
B)
13
done
clear
C)
17
done
clear
D)
19
done
clear
View Answer play_arrow
question_answer 214) 47 is added to the product of 71 and an unknown number. The new number is divisible by 7, giving the quotient 98. The unknown number is a multiple of
A)
2
done
clear
B)
5
done
clear
C)
7
done
clear
D)
3
done
clear
View Answer play_arrow
question_answer 215) The least number which, when divided by 16, 18, 20 and 25 leaves 4 as remainder in each case but when divided by 7 leaves no remainder is
A)
17004
done
clear
B)
18000
done
clear
C)
18002
done
clear
D)
18004
done
clear
View Answer play_arrow
question_answer 216) A can do a work in 5 days less than the time taken by B to do it. If both of them together take 11- days, then the time taken by B alone to do the same work (in days) is
A)
15
done
clear
B)
20
done
clear
C)
25
done
clear
D)
30
done
clear
View Answer play_arrow
question_answer 217) The price of a certain television set discounted by 10% and the reduced price n then discounted by 10%. This series of successive discounts is equivalent to a single discount of
A)
20 %
done
clear
B)
19 %
done
clear
C)
18 %
done
clear
D)
11 %
done
clear
View Answer play_arrow
question_answer 218) Rahim bought a TV with 20% discount on list price. Had he bought it with 25% discount, he would have saved Rs. 500. At what price did he buy the TV?
A)
Rs. 16000
done
clear
B)
Rs. 12000
done
clear
C)
Rs. 10000
done
clear
D)
Rs. 8000
done
clear
View Answer play_arrow
question_answer 219) If \[\frac{a}{3}=\frac{b}{2},\] then value of \[\frac{2a+3b}{3a-2b}\]is
A)
\[\frac{12}{5}\]
done
clear
B)
\[\frac{5}{12}\]
done
clear
C)
\[1\]
done
clear
D)
\[\frac{12}{7}\]
done
clear
View Answer play_arrow
question_answer 220) A shopkeeper bought pens at the rate of 7 for Rs. 10 and sold them at a profit of 40%. How many pens would a customer get for Rs. 10?
A)
6
done
clear
B)
4
done
clear
C)
5
done
clear
D)
3
done
clear
View Answer play_arrow
question_answer 221) If the cost price of 10 articles is equal to the sales price of 16 articles, then the gain or loss per cent is
A)
28% profit
done
clear
B)
\[37\frac{1}{2}%\]profit
done
clear
C)
28% loss
done
clear
D)
\[37\frac{1}{2}%\]loss
done
clear
View Answer play_arrow
question_answer 222) Three persons A, B and C whose salaries together amount to Rs. 72000, spend 80, 85 and 75 per cent of their salaries respectively. If their savings are in the ratio 8 : 9 : 20, then As salary is
A)
Rs. 20000
done
clear
B)
Rs. 16000
done
clear
C)
Rs. 22000
done
clear
D)
Rs. 18000
done
clear
View Answer play_arrow
question_answer 223) Two solutions of 90% and 97% purity are mixed, resulting in 21 L of mixture of 94% purity. The quantity of the second solution in the resulting mixture, in litres, is
A)
15
done
clear
B)
12
done
clear
C)
9
done
clear
D)
6
done
clear
View Answer play_arrow
question_answer 224) A boat travels 24 km upstream in 6 h and 20 km downstream in 4 h. Then, the speed of boat in still water and the speed of water current are respectively
A)
4 km/h and 3 km/h
done
clear
B)
4.5 km/h and 0.5 km/h
done
clear
C)
4 km/h and 2 km/h
done
clear
D)
5 km/h and 2 km/h
done
clear
View Answer play_arrow
question_answer 225) If the difference between simple interest and compound interest on a certain sum of money for 3 yr at 10% per annum is Rs. 31, the sum is
A)
Rs. 500
done
clear
B)
Rs. 750
done
clear
C)
Rs. 1000
done
clear
D)
Rs. 1250
done
clear
View Answer play_arrow
question_answer 226) Which one of the following is not an objective of fiscal policy in India?
A)
Full employment
done
clear
B)
Price stability
done
clear
C)
Equitable distribution of wealth and incomes
done
clear
D)
Regulation of international trade
done
clear
View Answer play_arrow
question_answer 227) Which one of the following is not a quantitative credit control measure of a Central Bank?
A)
Bank Rate Policy
done
clear
B)
Open Market Operations
done
clear
C)
Cash Reserve Ratio
done
clear
D)
Moral Suasion
done
clear
View Answer play_arrow
question_answer 228) In which market structure is the demand curve of the market represented by the demand curve of the firm?
A)
Monopoly
done
clear
B)
Oligopoly
done
clear
C)
Duopoly
done
clear
D)
Perfect competition
done
clear
View Answer play_arrow
question_answer 229) Mixed Farming means
A)
sowing of both cash and food crops
done
clear
B)
sowing of two or more crops in the same field
done
clear
C)
sowing of two or more plants in alternate years
done
clear
D)
rearing of cattle and agriculture
done
clear
View Answer play_arrow
question_answer 230) Which of the following is deducted from NNP to arrive at NI?
A)
Indirect tax
done
clear
B)
Capital consumption allowance
done
clear
C)
Subsidy
done
clear
D)
Interest
done
clear
View Answer play_arrow
question_answer 231) Whom did Bal Gangadhar Tilak refer to as his Political Guru?
A)
Swami Vivekanand
done
clear
B)
Ram Mohan Roy
done
clear
C)
Sisir Kumar Ghosh
done
clear
D)
Dadabhai Naoroji
done
clear
View Answer play_arrow
question_answer 232) Who introduced doctrine of lapse?
A)
Lord Wellesley
done
clear
B)
Lord Curzon
done
clear
C)
Lord Dalhousie
done
clear
D)
Lord Lytton
done
clear
View Answer play_arrow
question_answer 233) Tides are caused by the gravitational pull of the
A)
Earth on the Moon
done
clear
B)
Earth on the Sun
done
clear
C)
Sun and Moon on the Earth
done
clear
D)
Moon on the Earth
done
clear
View Answer play_arrow
question_answer 234) Dry farming in India is extensively practiced in
A)
Canara Plains
done
clear
B)
Deccan Plateau
done
clear
C)
Coromandal Plains
done
clear
D)
Ganga Plains
done
clear
View Answer play_arrow
question_answer 235) The Kullu Valley is situated between
A)
Ladakh and Pirpanjal
done
clear
B)
Ranjoti and Nag Tibba
done
clear
C)
Lesser Himalayas and Siwalik
done
clear
D)
Dhauladhar and Pirpanjal
done
clear
View Answer play_arrow
question_answer 236) Terrace farming is done
A)
on the slope of hills
done
clear
B)
in dry regions
done
clear
C)
on roof tops
done
clear
D)
on mountain tops
done
clear
View Answer play_arrow
question_answer 237) The colour change in the Chameleon is due to the presence of
A)
haemoglobin
done
clear
B)
chromatophore
done
clear
C)
chlorophyll
done
clear
D)
pneumatophore
done
clear
View Answer play_arrow
question_answer 238) The deficiency of iodine leads to
A)
hyperthyroidism
done
clear
B)
goiter
done
clear
C)
midgut
done
clear
D)
diabetes
done
clear
View Answer play_arrow
question_answer 239) Two richest known sources of edible protein are
A)
meat and eggs
done
clear
B)
some algae and other microorganisms
done
clear
C)
soyabean and groundnut
done
clear
D)
milk and leafy vegetables
done
clear
View Answer play_arrow
question_answer 240) The saliva helps in the digestion of
A)
proteins
done
clear
B)
starch
done
clear
C)
fibres
done
clear
D)
fats
done
clear
View Answer play_arrow
question_answer 241) Which of the following correctly explains the phenomenon of Test Tube Baby?
A)
When every process of embryo formation is in the test tube
done
clear
B)
When the embryo develops in a test tube
done
clear
C)
When the fertilization is external and development is internal
done
clear
D)
When the fertilization is internal and development is external
done
clear
View Answer play_arrow
question_answer 242) Heart attack occurs due to
A)
bacterial attack on the heart
done
clear
B)
stopping of heart beat
done
clear
C)
lack of supply of blood to the heart itself
done
clear
D)
impairment of hearts working due to unknown reasons
done
clear
View Answer play_arrow
question_answer 243) Direct conversion of solar energy with the use of a photovoltaic cell results in the production of
A)
optical energy
done
clear
B)
electrical energy
done
clear
C)
thermal energy
done
clear
D)
mechanical energy
done
clear
View Answer play_arrow
question_answer 244) A man inside an artificial satellite feels weightlessness because the force of attraction due to earth is
A)
zero at that place
done
clear
B)
is balanced by the force of attraction due to moon
done
clear
C)
equal to the centripetal force
done
clear
D)
non-effective due to particular design of the satellite
done
clear
View Answer play_arrow
question_answer 245) When a detergent is added to pure water, its surface tension
A)
increases
done
clear
B)
decreases
done
clear
C)
remains constant
done
clear
D)
becomes infinite
done
clear
View Answer play_arrow
question_answer 246) Supernova is
A)
a comet
done
clear
B)
an asteroid
done
clear
C)
an exploding star
done
clear
D)
a black hole
done
clear
View Answer play_arrow
question_answer 247) The device that converts computer output for transmission over telephone lines is called
A)
interface
done
clear
B)
interpreter
done
clear
C)
modem
done
clear
D)
I/O port
done
clear
View Answer play_arrow
question_answer 248) A bug in a programme is a
A)
statement
done
clear
B)
error
done
clear
C)
signature
done
clear
D)
Both (b) and (c)
done
clear
View Answer play_arrow
question_answer 249) Wood spirit is
A)
methyl alcohol
done
clear
B)
ethyl alcohol
done
clear
C)
butyl alcohol
done
clear
D)
propyl alcohol
done
clear
View Answer play_arrow
question_answer 250) Which of the following processes is used for the production of Biodiesel?
A)
Transamination
done
clear
B)
Transcription
done
clear
C)
Trans-esterification
done
clear
D)
Translation
done
clear
View Answer play_arrow
question_answer 251) One property of ammonia is
A)
it is insoluble in water
done
clear
B)
it is a odourless gas
done
clear
C)
it is a yellowish gas
done
clear
D)
its aqueous solution turns red litmus to blue
done
clear
View Answer play_arrow
question_answer 252) Of the following, which one pollutes the air of a big city?
A)
Copper
done
clear
B)
Chromium
done
clear
C)
Lead
done
clear
D)
Calcium
done
clear
View Answer play_arrow
question_answer 253) Ultraviolet light of Suns radiation is prevented from reaching the Earths atmosphere by the layer of
A)
Oxygen
done
clear
B)
Hydrogen
done
clear
C)
Ozone
done
clear
D)
Helium
done
clear
View Answer play_arrow
question_answer 254) Greater population can be supported on the Earth only if we eat more
A)
mutton
done
clear
B)
eggs
done
clear
C)
plant products
done
clear
D)
beef
done
clear
View Answer play_arrow
question_answer 255) Varahamihira is
A)
an astronaut
done
clear
B)
a space shuttle
done
clear
C)
a power station
done
clear
D)
an ancient astronomer
done
clear
View Answer play_arrow
question_answer 256) Fire fighting clothes are made from
A)
mica
done
clear
B)
asbestos
done
clear
C)
talc
done
clear
D)
steatite
done
clear
View Answer play_arrow
question_answer 257) Tummalapalli in Andhra Pradesh has recently come on the world map for its largest
A)
uranium deposits
done
clear
B)
tungsten deposits
done
clear
C)
coal deposits
done
clear
D)
bauxite ore deposits
done
clear
View Answer play_arrow
question_answer 258) As per the provisional result of the 2011 Census, the density of population in India is
A)
325
done
clear
B)
352
done
clear
C)
372
done
clear
D)
382
done
clear
View Answer play_arrow
question_answer 259) Rath Yatra at Puri is celebrated in honour of
A)
Lord Rama
done
clear
B)
Lord Shiva
done
clear
C)
Lord Jagannarh
done
clear
D)
Lord Vishnu
done
clear
View Answer play_arrow
question_answer 260) Gagan Narang, whose name has been recommended for Rajiv Gandhi Khel Ratna Award is a famous
A)
motor car racer
done
clear
B)
cricketer
done
clear
C)
air rifle shooter
done
clear
D)
footballer
done
clear
View Answer play_arrow
question_answer 261) The five permanent members of the United Nations Security Council are
A)
Canada, China, France, USA, UK
done
clear
B)
China, France, Russia, USA, UK
done
clear
C)
Japan, Germany, Russia, USA, UK
done
clear
D)
Germany, China, Russia, USA, UK
done
clear
View Answer play_arrow
question_answer 262) Which city has the headquarters of two railway zones in India?
A)
Hubli
done
clear
B)
New Delhi
done
clear
C)
Mumbai
done
clear
D)
Jabalpur
done
clear
View Answer play_arrow
question_answer 263) Who acts as the Chairman of the State Planning Board?
A)
Governor
done
clear
B)
Chief Minister
done
clear
C)
Leader of the opposition
done
clear
D)
Speaker
done
clear
View Answer play_arrow
question_answer 264) Air bladder in fish acts as
A)
accessory respiratory organ
done
clear
B)
hydrostatic organ
done
clear
C)
both accessory respiratory organ and hydrostatic organ
done
clear
D)
primary respiratory organ
done
clear
View Answer play_arrow
question_answer 265) Which of the following is called Blue Planet?
A)
Saturn
done
clear
B)
Earth
done
clear
C)
Jupiter
done
clear
D)
Mars
done
clear
View Answer play_arrow
question_answer 266) In cauliflower plant, the useful part is
A)
underground stem
done
clear
B)
root
done
clear
C)
young inflorescence
done
clear
D)
leaves
done
clear
View Answer play_arrow
question_answer 267) Aurobindo Ghosh was arrested in connection with
A)
Alipore Bomb Case
done
clear
B)
Kolhapur Bomb Case
done
clear
C)
Lahore Conspiracy Case
done
clear
D)
Kakori Case
done
clear
View Answer play_arrow
question_answer 268) Manchester of South India is
A)
Coimbatore
done
clear
B)
Madurai
done
clear
C)
Bengaluru
done
clear
D)
Chennai
done
clear
View Answer play_arrow
question_answer 269) The acid stored in batteries is
A)
sulphuric acid
done
clear
B)
nitric acid
done
clear
C)
hydrochloric acid
done
clear
D)
acetic acid
done
clear
View Answer play_arrow
question_answer 270) Heavy metal pollution of water is caused by
A)
paints
done
clear
B)
wood burning
done
clear
C)
acid plants
done
clear
D)
domestic sewage
done
clear
View Answer play_arrow
question_answer 271) Which of the following Public Undertakings has not been conferred with Maharatna Status?
A)
SAIL
done
clear
B)
BHEL
done
clear
C)
ONGC
done
clear
D)
Coal India Limited
done
clear
View Answer play_arrow
question_answer 272) Who is known as the Indian Bismarck?
A)
Vallabhbhai Patel
done
clear
B)
Subhash Chandra Bose
done
clear
C)
Bhagat Singh
done
clear
D)
Bal Gangadhar Tilak
done
clear
View Answer play_arrow
question_answer 273) Which one of the four regions above the Earth has smallest height (km)?
A)
Stratosphere
done
clear
B)
Mesosphere
done
clear
C)
Thermosphere
done
clear
D)
Troposphere
done
clear
View Answer play_arrow
question_answer 274) Which State of India shows the lowest population as per Census 2011?
A)
Manipur
done
clear
B)
Tripura
done
clear
C)
Puducherry
done
clear
D)
Sikkim
done
clear
View Answer play_arrow
question_answer 275) Rail Bandhu is
A)
Website of each railway division
done
clear
B)
the onboard medical officer in every train to attend to medical emergencies
done
clear
C)
security guard in every AC coach of Rajdhani/ Shatabdi and AC Duronto Express
done
clear
D)
an onboard magazine of Indian Railways to be available in Rajdhani/Shatabdi/AC Duronto Express
done
clear
View Answer play_arrow
question_answer 276) Which one of the following ceased to be a Fundamental Right under the Constitution?
A)
Right to Education
done
clear
B)
Right to Work
done
clear
C)
Right to Property
done
clear
D)
Right to Equality before Law
done
clear
View Answer play_arrow
question_answer 277) Coffee Research Institute of India is located at
A)
Yercaud
done
clear
B)
Wayanad
done
clear
C)
Coorg
done
clear
D)
Coimbatore
done
clear
View Answer play_arrow
question_answer 278) How many major seaports are there in India?
A)
6
done
clear
B)
9
done
clear
C)
10
done
clear
D)
12
done
clear
View Answer play_arrow
question_answer 279) The members of the Rajya Sabha are elected for a term of
A)
two years
done
clear
B)
four years
done
clear
C)
five years
done
clear
D)
six years
done
clear
View Answer play_arrow
question_answer 280) When was National Emergency imposed by the President of India on the grounds of internal disorder?
A)
1962
done
clear
B)
1965
done
clear
C)
1971
done
clear
D)
1975
done
clear
View Answer play_arrow
question_answer 281) Which One of the following is an example for Monopoly?
A)
SPIC
done
clear
B)
Indian Railways
done
clear
C)
HMT
done
clear
D)
Hindustan Unilever Limited
done
clear
View Answer play_arrow
question_answer 282) A black body can absorb radiations of
A)
lower wavelengths only
done
clear
B)
intermediate wavelengths only
done
clear
C)
higher wavelengths only
done
clear
D)
all wavelengths
done
clear
View Answer play_arrow
question_answer 283) The only Hindu courtier of Akbar who accepted Deen-e-Illahi was
A)
Todermal
done
clear
B)
Birbal
done
clear
C)
Tansen
done
clear
D)
Man Singh
done
clear
View Answer play_arrow
question_answer 284) The chemical which is used in art and craft and can cause anemia and leukaemia is
A)
benzene
done
clear
B)
dioxine
done
clear
C)
phthalate
done
clear
D)
aldrin
done
clear
View Answer play_arrow
question_answer 285) Which Party was established by Subhash Chandra Bose after he came out of Indian National Congress?
A)
Indian National Army
done
clear
B)
Republican Party
done
clear
C)
Forward Block
done
clear
D)
Socialist Party
done
clear
View Answer play_arrow
question_answer 286) The Rama Krishna Mission was established by
A)
Vivekananda
done
clear
B)
Rama Krishna
done
clear
C)
MG Ranade
done
clear
D)
Keshab Chandra Sen
done
clear
View Answer play_arrow
question_answer 287) The year of accession of Kanishka to thro was
A)
AD 108
done
clear
B)
AD 78
done
clear
C)
AD 58
done
clear
D)
AD 128
done
clear
View Answer play_arrow
question_answer 288) Total number of muscles present in our bod is
A)
565
done
clear
B)
656
done
clear
C)
665
done
clear
D)
556
done
clear
View Answer play_arrow
question_answer 289) Which of the following Mughal Emperors credited with composition of Hindi songs?
A)
Babar
done
clear
B)
Akbar
done
clear
C)
Jahangir
done
clear
D)
Shahjahan
done
clear
View Answer play_arrow
question_answer 290) WTO came into existence in the year
A)
1977
done
clear
B)
1985
done
clear
C)
1995
done
clear
D)
1950
done
clear
View Answer play_arrow
question_answer 291) Hydraulic machines work under the Principle of
A)
Newtons Law
done
clear
B)
Joules Law
done
clear
C)
Pascals Law
done
clear
D)
Floatation Law
done
clear
View Answer play_arrow
question_answer 292) Who invented aeroplane?
A)
Orville Wright and Wilbur Wright
done
clear
B)
Sir Frank Whittle
done
clear
C)
Michael Faraday
done
clear
D)
Christian Huygens
done
clear
View Answer play_arrow
question_answer 293) World Bank helps countries with loans for the purpose of
A)
reconstruction and development
done
clear
B)
stimulating private investment
done
clear
C)
tailing foreign exchange crises
done
clear
D)
meeting deficits in government budget
done
clear
View Answer play_arrow
question_answer 294) Which is not a Central Service?
A)
Indian Police Service ((IPS)
done
clear
B)
Indian Foreign Service (IFS)
done
clear
C)
Indian Audit and Accounts Service (IAAS)
done
clear
D)
Indian Revenue Service (IRS)
done
clear
View Answer play_arrow
question_answer 295) The first Indian State which was created on linguistic basis following along draw agitation is
A)
Andhra Pradesh
done
clear
B)
Asom
done
clear
C)
Tamil Nadu
done
clear
D)
Karriataka
done
clear
View Answer play_arrow
question_answer 296) The Great Barrier reef is
A)
conglomeration of Corals in Australian waters
done
clear
B)
mountains range in Utah, USA
done
clear
C)
salt hills of Afghanistan
done
clear
D)
sub-oceanic mountain in South China Sea
done
clear
View Answer play_arrow
question_answer 297) The Panama Canal links
A)
Mediterranean Sea with Red Sea
done
clear
B)
Atlantic Ocean with Indian Ocean
done
clear
C)
Indian Ocean with Mediterranean
done
clear
D)
Atlantic Ocean with Pacific Ocean
done
clear
View Answer play_arrow
question_answer 298) C, BASIC, COBOL, and Java are examples of ....... language.
A)
low level
done
clear
B)
computer
done
clear
C)
system programing
done
clear
D)
high - level
done
clear
View Answer play_arrow
question_answer 299) .............. is approximately one billion bytes.
A)
Kilobyte
done
clear
B)
Bit
done
clear
C)
Gigabyte
done
clear
D)
Megabyte
done
clear
View Answer play_arrow
question_answer 300) The wildlife week is celebrated from
A)
2-8 October
done
clear
B)
1-7 June
done
clear
C)
16-22 April
done
clear
D)
14 -20 January
done
clear
View Answer play_arrow
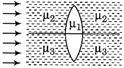
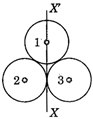
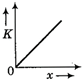
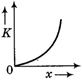
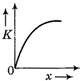
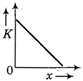
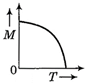
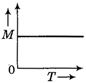
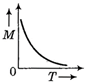
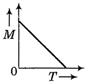
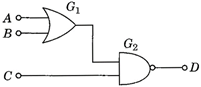
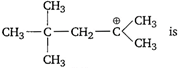
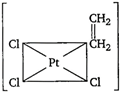
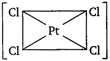
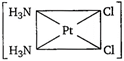
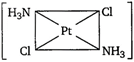
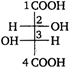 configuration at \[{{C}_{2}}\] and \[{{C}_{3}}\] atoms are
configuration at \[{{C}_{2}}\] and \[{{C}_{3}}\] atoms are
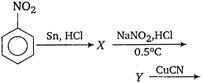

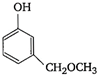
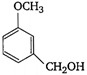

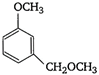
 Answer Figures
Answer Figures
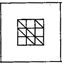
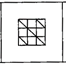
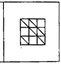
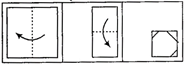 Answer Figures
Answer Figures
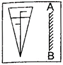 Answer Figures
Answer Figures