question_answer 1) A simple pendulum is made of a body which a hollow sphere is containing mercury suspended by means of a wire. If a little mercury is drained off, the period of pendulum will
A)
remain unchanged
done
clear
B)
increase
done
clear
C)
decrease
done
clear
D)
become erratic
done
clear
View Answer play_arrow
question_answer 2) Two balloons are filled, one with pure He gas and the other by air, respectively. If the pressure and temperature of these balloons are same then the number of molecules per unit volume is
A)
more in the He filled balloon
done
clear
B)
same in both balloons
done
clear
C)
more in air filled balloon
done
clear
D)
in the ratio of 1 : 4
done
clear
View Answer play_arrow
question_answer 3) Which of the following logic gates is an universal gate?
A)
OR
done
clear
B)
NOT
done
clear
C)
AND
done
clear
D)
NAND
done
clear
View Answer play_arrow
question_answer 4) We consider a thermodynamic system. If \[\text{ }\!\!\Delta\!\!\text{ U}\]represents the increase in its internal energy and W the work done by the system, which of the following statements is true?
A)
\[\Delta U=-W\] in an adiabatic process
done
clear
B)
\[\Delta U=W\] in an isothermal process
done
clear
C)
\[\Delta U=-W\]in an isothermal process
done
clear
D)
\[\Delta U=W\] in an adiabatic process
done
clear
View Answer play_arrow
question_answer 5) Aerofils are so designed that the speed of air
A)
on top side is more than on lower side
done
clear
B)
on fop side is less than on lower side,
done
clear
C)
is same on both sides
done
clear
D)
is turbulent
done
clear
View Answer play_arrow
question_answer 6) According to Bernoullis equation \[\frac{P}{\rho g}+h+\frac{1}{2}\frac{{{v}^{2}}}{g}=\text{constant}\] The terms A, B and C are generally called respectively:
A)
gravitational head, pressure head and velocity head
done
clear
B)
gravity, gravitational head and velocity head
done
clear
C)
pressure head, gravitational head and velocity head
done
clear
D)
gravity, pressure and velocity head
done
clear
View Answer play_arrow
question_answer 7) The time of reverberation of a room A is one second. What will be the time (in seconds) of reverberation of a room, having all the dimensions double of those of room A?
A)
2
done
clear
B)
4
done
clear
C)
\[\frac{1}{2}\]
done
clear
D)
1
done
clear
View Answer play_arrow
question_answer 8) For sky wave propagation of 10 MHz signal, what should be the minimum electron density in ionosphere?
A)
\[\tilde{\ }1.2\times {{10}^{12}}{{m}^{-3}}\]
done
clear
B)
\[\tilde{\ }{{10}^{6}}\,{{m}^{-3}}\]
done
clear
C)
\[\tilde{\ }{{10}^{14}}\,{{m}^{-3}}\]
done
clear
D)
\[\tilde{\ }{{10}^{22}}\,{{m}^{-3}}\]
done
clear
View Answer play_arrow
question_answer 9) If the mass of moon is \[\frac{\text{M}}{\text{81}}\text{,}\]where M is the mass of earth, find the distance of the point from the moon, where gravitational field due to earth and moon cancel each other. Given that distance between earth and moon is 60R, where R is the radius of earth.
A)
6R
done
clear
B)
BR
done
clear
C)
2R
done
clear
D)
4R
done
clear
View Answer play_arrow
question_answer 10) 27 small drops each having charge q and radius r coalesce to from big drop. How many times charge and capacitance will become?
A)
3, 27
done
clear
B)
27, 3
done
clear
C)
27, 27
done
clear
D)
3, 3
done
clear
View Answer play_arrow
question_answer 11) The magnetic moment has dimensions of
A)
\[[LA]\]
done
clear
B)
\[[{{L}^{2}}A]\]
done
clear
C)
\[[L{{T}^{-1}}A]\]
done
clear
D)
\[[{{L}^{2}}{{T}^{-1}}A]\]
done
clear
View Answer play_arrow
question_answer 12) Which of the following circular rods, (given radius r and length\[l\]) each made of the same material and whose ends are maintained at the same temperature will conduct most heat?
A)
\[r=2{{r}_{0}};l=2{{l}_{0}}\]
done
clear
B)
\[r=2{{r}_{0}};l={{l}_{0}}\]
done
clear
C)
\[r={{r}_{0}};l={{l}_{0}}\]
done
clear
D)
\[r={{r}_{0}};l=2{{l}_{0}}\]
done
clear
View Answer play_arrow
question_answer 13) For a satellite moving in an orbit around the earth, the ratio of kinetic energy to potential energy is
A)
2
done
clear
B)
\[\frac{1}{2}\]
done
clear
C)
\[\frac{1}{\sqrt{2}}\]
done
clear
D)
\[\sqrt{2}\]
done
clear
View Answer play_arrow
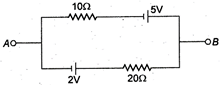
question_answer 14)
The current in the given circuit is
A)
0.3 A
done
clear
B)
0.1 A
done
clear
C)
0.4 A
done
clear
D)
0.2 A
done
clear
View Answer play_arrow
question_answer 15) Interference was observed in an interference chamber when air was present. Now, the chamber is evacuated and if the same light is used, a careful observation will show
A)
no interference
done
clear
B)
interference with bright band
done
clear
C)
interference with dark bands
done
clear
D)
interference in which breadth of the fringe will be slightly increased
done
clear
View Answer play_arrow
question_answer 16) When a charged particle moving with velocity \[\text{\vec{v}}\]is subjected to a magnetic field of induction \[\text{\vec{B},}\] the force on it is non-zero. This implies that
A)
angle between \[\text{\vec{v}}\]and \[\text{\vec{B},}\] is necessarily \[90{}^\circ \]
done
clear
B)
angle between\[\text{\vec{v}}\]and\[\text{\vec{B},}\]can have any value other than \[\text{9}{{\text{0}}^{o}}\]
done
clear
C)
angle between\[\text{\vec{v}}\]and \[\text{\vec{B},}\]can have any value other than zero and \[{{180}^{o}}\]
done
clear
D)
angle between \[\text{\vec{v}}\]and \[\text{\vec{B},}\]is either zero or \[{{180}^{o}}\]
done
clear
View Answer play_arrow
question_answer 17) A article moves along a straight line OX. At a time t (in seconds) the distance \[x\](in metres) of the particle from O is given by \[x=40+12t-{{t}^{3}}\] How long would the particle travel before coming to rest?
A)
24 m
done
clear
B)
40 m
done
clear
C)
56 m
done
clear
D)
16 m
done
clear
View Answer play_arrow
question_answer 18) Which one of the following statements is true?
A)
Both light and sound waves in air are transverse
done
clear
B)
The sound waves in air are longitudinal while the light waves are transverse
done
clear
C)
Both light and sound waves in air are longitudinal
done
clear
D)
Both light and sound waves can travel in vacuum
done
clear
View Answer play_arrow
question_answer 19) When power is drawn from the secondary coil of the transformer, the dynamic resistance
A)
increases
done
clear
B)
decreases
done
clear
C)
remains unchanged
done
clear
D)
changes erratically
done
clear
View Answer play_arrow
question_answer 20) A reference frame attached to the earth
A)
is an inertial frame by definition
done
clear
B)
cannot be an inertial frame because earth is revolving round the sun
done
clear
C)
is an inertial frame because Newtons laws are applicable
done
clear
D)
is an inertial frame because the earth is rotating about its own axis
done
clear
View Answer play_arrow
question_answer 21) Pressure of an ideal gas is increased by keeping temperature constant. What is the effect on kinetic energy of molecules?
A)
Increase
done
clear
B)
Decrease
done
clear
C)
No change
done
clear
D)
Cant be determined
done
clear
View Answer play_arrow
question_answer 22) What maximum frequency can be reflected from ionosphere?
A)
5 MHz
done
clear
B)
6 GHz
done
clear
C)
5 kHz
done
clear
D)
500 MHz
done
clear
View Answer play_arrow
question_answer 23) A telescope has focal length of objective and eyepiece as 200 cm and 5 cm respectively. What is magnification of telescope?
A)
40
done
clear
B)
80
done
clear
C)
50
done
clear
D)
0.01
done
clear
View Answer play_arrow
question_answer 24) A small disc of .radius 2 cm is cut from a disc of radius 6 cm. If the distance between their centres is 3.2 cm, what is the shift in the centre of mass of the disc?
A)
0.4 cm
done
clear
B)
2.4 cm
done
clear
C)
1.8 cm
done
clear
D)
1.2 cm
done
clear
View Answer play_arrow
question_answer 25) Which of the following is not a process involved in fabrication of 1C?
A)
Polymerization
done
clear
B)
Diffusion
done
clear
C)
Photolithography
done
clear
D)
Metallisation
done
clear
View Answer play_arrow
question_answer 26) Permanent magnet has properties retentivity and coercivity respectively
A)
high-high
done
clear
B)
low-low
done
clear
C)
. low-high
done
clear
D)
high-low
done
clear
View Answer play_arrow
question_answer 27) Hydrogen bomb is based upon
A)
fission
done
clear
B)
fusion
done
clear
C)
chemical reaction
done
clear
D)
transmutation
done
clear
View Answer play_arrow
question_answer 28) What is not true for equipotential surface for uniform electric field?
A)
Equipotential surface is flat
done
clear
B)
Equipotential surface is spherical
done
clear
C)
Electric lines are perpendicular to equipotential surface
done
clear
D)
Work done is zero
done
clear
View Answer play_arrow
question_answer 29) A closed organ pipe of length 20 cm is sounded with tuning fork in resonance. What is the frequency of tuning fork? \[(v=332\,m/s)\]
A)
300 Hz
done
clear
B)
350 Hz
done
clear
C)
375 Hz
done
clear
D)
415 Hz
done
clear
View Answer play_arrow
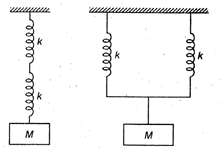
question_answer 30)
Two identical springs of spring constant k are connected in series and parallel as shown in figure. A mass M is suspended from them. The ratio of their frequencies of vertical oscillation will be
A)
2:1
done
clear
B)
1.5
done
clear
C)
2.1
done
clear
D)
2.5
done
clear
View Answer play_arrow
question_answer 31) The area of the acceleration-displacement curve of a body gives
A)
impulse
done
clear
B)
change in momentum per unit mass
done
clear
C)
change in KE per unit mass
done
clear
D)
total change in energy
done
clear
View Answer play_arrow
question_answer 32) The speed of earths rotation about its axis is \[\omega .\]Its speed is increased to\[x\]times to make the effective acceleration due to gravity equal to zero at the equator. Then \[x\] is
A)
1
done
clear
B)
8.5
done
clear
C)
17
done
clear
D)
34
done
clear
View Answer play_arrow
question_answer 33) Hard X-rays for the study of fractures in bones should have a minimum wavelength of \[{{10}^{-11}}m.\] The accelerating voltage for electrons in X-ray machine should be
A)
\[<\,124\,kV\]
done
clear
B)
\[>\,124\,kV\]
done
clear
C)
between 60 kV and 70 kV
done
clear
D)
= 100 kV
done
clear
View Answer play_arrow
question_answer 34) In photoelectric effect, the electrons are ejected from metals if the incident light has a certain minimum
A)
wavelength
done
clear
B)
frequency
done
clear
C)
amplitude
done
clear
D)
angle of incidence
done
clear
View Answer play_arrow
question_answer 35)
A)
12
done
clear
B)
6
done
clear
C)
8/3
done
clear
D)
3
done
clear
View Answer play_arrow
question_answer 36) A leaf which contains only green pigments, is illuminated by a laser light of wavelength \[\text{0}\text{.6328 }\!\!\mu\!\!\text{ m}\text{.}\]It would appear to be
A)
brown
done
clear
B)
black
done
clear
C)
red
done
clear
D)
green
done
clear
View Answer play_arrow
question_answer 37) The operation of a nuclear reactor is said to be critical, if the multiplication factor \[(k)\] has a value
A)
1
done
clear
B)
1.5
done
clear
C)
2.1
done
clear
D)
2.5
done
clear
View Answer play_arrow
question_answer 38) A galvanometer having a resistance of \[8\,\Omega \]is shunted by a wire of resistance \[2\,\Omega .\] If the total current is 1 A, the part of it passing through the shunt will be
A)
0.25 A
done
clear
B)
0.8 A
done
clear
C)
0.2 A
done
clear
D)
0.5 A
done
clear
View Answer play_arrow
question_answer 39) Flash light equipped with a new set of batteries, produces bright white light. As the batteries wear out
A)
the light intensity gets reduced with no change in its colour
done
clear
B)
light colour changes first to yellow andthen red with no change in intensity
done
clear
C)
it stops working suddenly while giving white light
done
clear
D)
colour changes to red and also intensity gets reduced
done
clear
View Answer play_arrow
question_answer 40) If alpha, beta and gamma rays carry same momentum, which has the longest wavelength?
A)
Alpha rays
done
clear
B)
Beta rays
done
clear
C)
Gamma rays
done
clear
D)
None, all have same wavelength
done
clear
View Answer play_arrow
question_answer 41) An amplifier has a voltage gain \[{{A}_{v}}=1000.\]The voltage gain in dB is
A)
30 dB
done
clear
B)
60 dB
done
clear
C)
3 dB
done
clear
D)
20 dB
done
clear
View Answer play_arrow
question_answer 42) When you make ice cubes, the entropy of water
A)
does not change
done
clear
B)
increases
done
clear
C)
decreases
done
clear
D)
may either increase or decrease depending on the process used
done
clear
View Answer play_arrow
question_answer 43) Half-lives of two radioactive substances A and B are respectively 20 min and 40 min. Initially the samples of A and B have equal number of nuclei. After 80 min the ratio of remaining number of A and B nuclei is
A)
1 : 16
done
clear
B)
4 : 1
done
clear
C)
1:4
done
clear
D)
1 : 1
done
clear
View Answer play_arrow
question_answer 44) If equation of a sound wave is \[y=0.0015\sin (62.8x+314t)\] then its wavelength will be
A)
0.1 unit
done
clear
B)
2 unit
done
clear
C)
0.3 unit
done
clear
D)
0.2 unit
done
clear
View Answer play_arrow
question_answer 45) A projectile is thrown in the upward direction making an angle of \[{{60}^{0}}\] with the horizontal direction with a velocity of \[147\,m{{s}^{-1}}.\] Then the time after which its inclination with the horizontal is \[{{45}^{o}},\] is
A)
15s
done
clear
B)
10.98 s
done
clear
C)
5.49 s
done
clear
D)
2.745 s
done
clear
View Answer play_arrow
question_answer 46) A motor cycle is going on an overbridge of radius R. The driver maintains a constant speed. As the motor cycle is ascending on the overbridge, the normal force on it
A)
increases
done
clear
B)
decreases
done
clear
C)
remains the same
done
clear
D)
fluctuates erratically
done
clear
View Answer play_arrow
question_answer 47) If we throw a body upwards with velocity of \[4\text{ }m/s,\] at what height does its kinetic energy reduce to half of the initial value? (Take\[g=10\,m{{s}^{-2}}\])
A)
4 m
done
clear
B)
2 m
done
clear
C)
1 m
done
clear
D)
0.4 m
done
clear
View Answer play_arrow
question_answer 48) Two glass plates are separated by water. If surface tension of water is 75 dynes/cm and area of each plate wetted by water is \[\text{8}\,\text{c}{{\text{m}}^{\text{2}}}\]and the distance between the plates is 0.12 mm, then the force applied to separate the two plates is
A)
\[{{10}^{2}}\]dyne
done
clear
B)
\[{{10}^{4}}\] dyne
done
clear
C)
\[{{10}^{5}}\]dyne
done
clear
D)
\[{{10}^{6}}\]dyne
done
clear
View Answer play_arrow
question_answer 49)
Which of the following is true for rays coming from infinity?
A)
Two images are formed
done
clear
B)
Continuous image is formed between focal points of upper and lower lens
done
clear
C)
One image is formed
done
clear
D)
None of the above
done
clear
View Answer play_arrow
question_answer 50) The north pole of a long horizontal bar magnet is being brought closer to a vertical conducting plane along the perpendicular direction. The direction of the induced current in the conducting plane will be
A)
horizontal
done
clear
B)
vertical
done
clear
C)
clockwise
done
clear
D)
anticlockwise
done
clear
View Answer play_arrow
question_answer 51) Calcium carbide reacts with \[{{\text{H}}_{\text{2}}}\text{O}\]to produce
A)
ethane
done
clear
B)
methane
done
clear
C)
ethylene
done
clear
D)
acetylene
done
clear
View Answer play_arrow
question_answer 52) Propyne and propene can be distinguished by
A)
\[\text{dil}\text{.}\,\text{KMn}{{\text{O}}_{4}}\]
done
clear
B)
\[\text{conc}\text{.}\,{{\text{H}}_{\text{2}}}\text{S}{{\text{O}}_{\text{4}}}\]
done
clear
C)
\[\text{AgN}{{\text{O}}_{\text{3}}}\]in ammonia
done
clear
D)
\[\text{B}{{\text{r}}_{\text{2}}}\]in \[\text{CC}{{\text{l}}_{\text{4}}}\]
done
clear
View Answer play_arrow
question_answer 53) Oxidation number of Fe in\[\text{F}{{\text{e}}_{\text{2}}}{{\text{O}}_{\text{3}}}\] is
A)
\[\frac{1}{2}\]
done
clear
B)
\[\frac{2}{6}\]
done
clear
C)
\[\frac{8}{3}\]
done
clear
D)
\[\frac{3}{2}\]
done
clear
View Answer play_arrow
question_answer 54) Bauxite is an ore of
A)
Al
done
clear
B)
Ca
done
clear
C)
Cu
done
clear
D)
Ni
done
clear
View Answer play_arrow
question_answer 55) The best source of vitamin A is
A)
beans
done
clear
B)
pulses
done
clear
C)
orange
done
clear
D)
carrot
done
clear
View Answer play_arrow
question_answer 56) Plaster of Paris is
A)
\[CaS{{O}_{4}}.\frac{1}{2}{{H}_{2}}O\]
done
clear
B)
\[CaS{{O}_{4}}.2{{H}_{2}}O\]
done
clear
C)
\[CaS{{O}_{4}}.{{H}_{2}}O\]
done
clear
D)
\[CaS{{O}_{4}}.4{{H}_{2}}O\]
done
clear
View Answer play_arrow
question_answer 57) Penicillin is
A)
analgesic
done
clear
B)
antipyretic
done
clear
C)
antimalarials
done
clear
D)
antibiotic
done
clear
View Answer play_arrow
question_answer 58) The most stable compound is
A)
\[\text{LiF}\]
done
clear
B)
\[\text{ }\!\!~\!\!\text{ LiCl}\]
done
clear
C)
\[\text{ }\!\!~\!\!\text{ LiBr}\]
done
clear
D)
\[\text{LiI}\]
done
clear
View Answer play_arrow
question_answer 59) Heavy water is
A)
\[CaS{{O}_{4}}\]
done
clear
B)
water contain \[CaS{{O}_{4}},MgS{{O}_{4}}\]
done
clear
C)
\[{{D}_{2}}O\]
done
clear
D)
water contain\[CaC{{O}_{3}}\]
done
clear
View Answer play_arrow
question_answer 60) In the extraction of copper from its sulphide ore, the metal is formed by reduction of \[\text{C}{{\text{u}}_{\text{2}}}\text{O}\] with
A)
\[FeS\]
done
clear
B)
\[CO\]
done
clear
C)
\[C{{u}_{2}}S\]
done
clear
D)
\[S{{O}_{2}}\]
done
clear
View Answer play_arrow
question_answer 61) \[\text{Ba}{{\text{O}}_{\text{2}}}\]and ozone reacts to produce
A)
\[\text{Ba}\]
done
clear
B)
\[B{{a}_{2}}{{O}_{3}}\]
done
clear
C)
\[BaO\]
done
clear
D)
\[Ba{{(OH)}_{3}}\]
done
clear
View Answer play_arrow
question_answer 62) Heisenberg uncertainty principle can be explained as
A)
\[\Delta x\ge \frac{\Delta p\times h}{4\pi }\]
done
clear
B)
\[\Delta x\,\times \,\Delta p\ge \frac{h}{4\pi }\]
done
clear
C)
\[\Delta x\,\times \,\Delta p\ge \frac{h}{\pi }\]
done
clear
D)
\[\,\Delta p\ge \frac{\pi h}{\,\Delta x}\]
done
clear
View Answer play_arrow
question_answer 63) For decolourisation of 1 mole of \[\text{KMn}{{\text{O}}_{\text{4}}}\text{,}\]the moles of \[{{\text{H}}_{\text{2}}}{{\text{O}}_{\text{2}}}\]required is
A)
\[\frac{1}{2}\]
done
clear
B)
\[\frac{3}{2}\]
done
clear
C)
\[\frac{5}{2}\]
done
clear
D)
\[\frac{7}{2}\]
done
clear
View Answer play_arrow
question_answer 64) Bleaching powder is obtained by treating \[\text{C}{{\text{l}}_{\text{2}}}\] with
A)
\[Ca{{(OH)}_{2}}\]
done
clear
B)
\[CaO\]
done
clear
C)
\[CaC{{O}_{3}}\]
done
clear
D)
\[CaC{{l}_{2}}\]
done
clear
View Answer play_arrow
question_answer 65) Magnalium contains
A)
\[\text{Mg}\,\text{+}\,\text{Al}\]
done
clear
B)
\[\text{ }\!\!~\!\!\text{ Mg}\,\text{+}\,\text{Cu}\]
done
clear
C)
\[\text{Mg}\,\text{+}\,\text{Fe}\]
done
clear
D)
\[\text{ }\!\!~\!\!\text{ Mg}\,\text{+ Mn}\]
done
clear
View Answer play_arrow
question_answer 66) The de-Broglie wavelenght of a particle with mass 1 kg and velocity 100 m/s is
A)
\[6.6\,\,\times \,\,{{10}^{-33}}\,m\]
done
clear
B)
\[6.6\,\,\times \,\,{{10}^{-36}}\,m\]
done
clear
C)
\[6.6\,\,\times \,\,{{10}^{33}}\,m\]
done
clear
D)
\[3.3\,\,\times \,\,{{10}^{-36}}\,m\]
done
clear
View Answer play_arrow
question_answer 67) The volume of a gas measured at \[\text{27}{{\,}^{\text{o}}}\text{C}\]and 1 atm pressure is 10 L. To reduce the volume to 2L at 1 atm. pressure, the temperature required is
A)
60 K
done
clear
B)
75 K
done
clear
C)
150 K
done
clear
D)
225 K
done
clear
View Answer play_arrow
question_answer 68) Isotonic solutions have
A)
same vapour pressure
done
clear
B)
same osmotic pressure
done
clear
C)
same boiling point
done
clear
D)
same temperature
done
clear
View Answer play_arrow
question_answer 69) Epsom salt is
A)
\[BaS{{O}_{4}}.2{{H}_{2}}O\]
done
clear
B)
\[CaS{{O}_{4}}.{{H}_{2}}O\]
done
clear
C)
\[MgS{{O}_{4}}.2{{H}_{2}}O\]
done
clear
D)
\[MgS{{O}_{4}}.7{{H}_{2}}O\]
done
clear
View Answer play_arrow
question_answer 70) Paracetamol is an
A)
analgesic
done
clear
B)
antipyretic
done
clear
C)
both [a] and [b]
done
clear
D)
none of the above
done
clear
View Answer play_arrow
question_answer 71) The number of moles of oxygen obtained by the electrolytic decomposition of 108 g water is
A)
2.5
done
clear
B)
3
done
clear
C)
5
done
clear
D)
7.5
done
clear
View Answer play_arrow
question_answer 72) The change in entropy for the fusion of 1 mole of ice is [\[\text{mp}\,\text{of}\,\text{ice}\,\,\text{=}\,\,\text{273}\,\text{K,}\], molar enthalpy of fusion for ice\[=\,\,6.0\,\text{kJ}\,\text{mo}{{\text{l}}^{-1}}\]]
A)
\[11.73\,J{{K}^{-1}}\,mo{{l}^{-1}}\]
done
clear
B)
\[18.84\,J{{K}^{-1}}\,mo{{l}^{-1}}\]
done
clear
C)
\[21.97\,J{{K}^{-1}}\,mo{{l}^{-1}}\]
done
clear
D)
\[24.47\,J{{K}^{-1}}\,mo{{l}^{-1}}\]
done
clear
View Answer play_arrow
question_answer 73) Which does not give a precipitate with \[\text{AgN}{{\text{O}}_{\text{3}}}\]solution?
A)
\[[Co{{(N{{H}_{3}})}_{6}}]C{{l}_{3}}\]
done
clear
B)
\[[Co{{(N{{H}_{3}})}_{5}}Cl]C{{l}_{2}}\]
done
clear
C)
\[[Co{{(N{{H}_{3}})}_{4}}C{{l}_{2}}]Cl\]
done
clear
D)
\[[Co{{(N{{H}_{3}})}_{3}}C{{l}_{3}}]\]
done
clear
View Answer play_arrow
question_answer 74) Total volume of atoms present in a face centred cubic unit cell of a metal is (r is atomic radius)
A)
\[\frac{16}{3}\pi {{r}^{3}}\]
done
clear
B)
\[\frac{20}{3}\pi {{r}^{3}}\]
done
clear
C)
\[\frac{24}{3}\pi {{r}^{3}}\]
done
clear
D)
\[\frac{12}{3}\pi {{r}^{3}}\]
done
clear
View Answer play_arrow
question_answer 75) Pure silicon doped with phosphorus is a
A)
metallic conductor
done
clear
B)
insulator
done
clear
C)
n-type semiconductor
done
clear
D)
p-type semiconductor
done
clear
View Answer play_arrow
question_answer 76) Which of the following is a biodegradable polymer?
A)
Cellulose
done
clear
B)
Polythene
done
clear
C)
Polyvinyl chloride
done
clear
D)
Nylon 6
done
clear
View Answer play_arrow
question_answer 77) What is X in the following nuclear reaction? \[{{\,}_{7}}N{{\,}^{14}}+{{\,}_{1}}N{{\,}^{1}}\xrightarrow[{}]{{}}{{\,}_{8}}{{O}^{15}}+X\]
A)
\[{{\,}_{0}}{{n}^{1}}\]
done
clear
B)
\[{{\,}_{-1}}{{e}^{0}}\]
done
clear
C)
\[{{\,}_{+\,1}}{{e}^{0}}\]
done
clear
D)
\[\gamma \]
done
clear
View Answer play_arrow
question_answer 78) Solubility product of \[\text{PbC}{{\text{l}}_{\text{2}}}\]at \[298\,K\]is \[1\times {{10}^{-6}}.\] At this temperature solubility of \[\text{PbC}{{\text{l}}_{\text{2}}}\]in mol/L is
A)
\[{{(1\,\times \,{{10}^{-6}})}^{1/2}}\]
done
clear
B)
\[{{(1\,\times \,{{10}^{-6}})}^{1/3}}\]
done
clear
C)
\[{{(0.25\,\times \,{{10}^{-6}})}^{1/3}}\]
done
clear
D)
\[{{(0.25\,\times \,{{10}^{-6}})}^{1/2}}\]
done
clear
View Answer play_arrow
question_answer 79) The pH of a 0.001 M solution of \[\text{HCl}\]is
A)
0
done
clear
B)
3
done
clear
C)
5
done
clear
D)
10
done
clear
View Answer play_arrow
question_answer 80) Gold number is associated with
A)
amount of gold
done
clear
B)
protective colloids
done
clear
C)
purple of cassius
done
clear
D)
electrophoresis
done
clear
View Answer play_arrow
question_answer 81) Noble gases are used in discharge tubes to give different colours. Reddish-orange glow is due to
A)
Ar
done
clear
B)
Ne
done
clear
C)
Xe
done
clear
D)
Kr
done
clear
View Answer play_arrow
question_answer 82) The set representing the correct order for first ionisation potential is
A)
\[\text{ }\!\!~\!\!\text{ K}\,\text{}\,\text{Na}\,\text{}\,\text{Li}\]
done
clear
B)
\[\text{ }\!\!~\!\!\text{ Be Mg Ca}\]
done
clear
C)
\[\text{ }\!\!~\!\!\text{ B C N}\]
done
clear
D)
\[Ge>Si>C\]
done
clear
View Answer play_arrow
question_answer 83) Dry ice is
A)
solid\[\text{C}{{\text{O}}_{\text{2}}}\]
done
clear
B)
solid camphor
done
clear
C)
solid\[\text{S}{{\text{O}}_{\text{2}}}\]
done
clear
D)
solid\[\text{N}{{\text{O}}_{\text{2}}}\]
done
clear
View Answer play_arrow
question_answer 84) Methanol and ethanol are miscible in water due to
A)
covalent character
done
clear
B)
hydrogen bonding character
done
clear
C)
oxygen bonding character
done
clear
D)
none of the above
done
clear
View Answer play_arrow
question_answer 85) Which of the following does not have optical isomer?
A)
\[[Co{{(N{{H}_{3}})}_{3}}C{{l}_{3}}]\]
done
clear
B)
\[[Co{{(e{{n}_{3}})}_{3}}]C{{l}_{2}}\]
done
clear
C)
\[[Co(e{{n}_{2}})C{{l}_{2}}]Cl\]
done
clear
D)
\[[Co(en){{(N{{H}_{3}})}_{2}}C{{l}_{2}}]Cl\]
done
clear
View Answer play_arrow
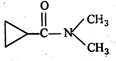
question_answer 86)
IUPAC name of the following compound
A)
\[\text{N,}\,\text{N}-\] dimethyl cyclopropaneear box amide
done
clear
B)
N-methyl cyclopropan amide
done
clear
C)
cyclopropion amide
done
clear
D)
none of the above
done
clear
View Answer play_arrow
question_answer 87) The product of following reaction is \[2C{{H}_{3}}MgBr+C{{H}_{2}}O\xrightarrow{{}}\]
A)
\[C{{H}_{3}}OH\]
done
clear
B)
\[{{C}_{2}}{{H}_{5}}OH\]
done
clear
C)
\[C{{H}_{4}}\]
done
clear
D)
\[{{C}_{2}}{{H}_{6}}\]
done
clear
View Answer play_arrow
question_answer 88) Freon used as refrigerant is
A)
\[C{{F}_{2}}=C{{F}_{2}}\]
done
clear
B)
\[C{{H}_{2}}{{F}_{2}}\]
done
clear
C)
\[CC{{l}_{2}}{{F}_{2}}\]
done
clear
D)
\[C{{F}_{4}}\]
done
clear
View Answer play_arrow
question_answer 89) Lucas reagent is
A)
anhy. \[\text{ZnC}{{\text{l}}_{\text{2}}}\]and \[\text{N}{{\text{H}}_{\text{3}}}\]
done
clear
B)
anhy. \[\text{ZnC}{{\text{l}}_{\text{2}}}\]and \[\text{CaC}{{\text{l}}_{\text{2}}}\]
done
clear
C)
anhy. \[\text{ZnC}{{\text{l}}_{\text{2}}}\]and cone. \[\text{HCl}\]
done
clear
D)
anhy. \[\text{ZnC}{{\text{l}}_{\text{2}}}\]and \[\text{HCl}\] gas
done
clear
View Answer play_arrow
question_answer 90) The enzyme which can catalyse the conversion of glucose to ethanol is
A)
zymase
done
clear
B)
invertase
done
clear
C)
maltase
done
clear
D)
diastase
done
clear
View Answer play_arrow
question_answer 91) When dihydroxy acetone reacts with HI04, the product is/are
A)
HCHO
done
clear
B)
HCOOH
done
clear
C)
HCHO and HCOOH
done
clear
D)
HCHO and \[\text{C}{{\text{O}}_{2}}\]
done
clear
View Answer play_arrow
question_answer 92) Which of the following does not reduce Fehlings solution?
A)
Benzaldehyde
done
clear
B)
Formic acid
done
clear
C)
Glucose
done
clear
D)
Fructose
done
clear
View Answer play_arrow
question_answer 93) Sodium formate on heating gives
A)
oxalic acid and \[{{\text{H}}_{\text{2}}}\]
done
clear
B)
sodium oxalate and \[{{\text{H}}_{\text{2}}}\]
done
clear
C)
sodium oxalate
done
clear
D)
\[\text{C}{{\text{O}}_{\text{2}}}\]and caustic soda
done
clear
View Answer play_arrow
question_answer 94) Reaction of ethyl formate with excess of \[\text{C}{{\text{H}}_{\text{3}}}\text{Mgl}\] followed by hydrolysis gives
A)
\[n-\]propyl alcohol
done
clear
B)
isopropyl alcohol
done
clear
C)
acetaldehyde
done
clear
D)
acetone
done
clear
View Answer play_arrow
question_answer 95) Hydrolysis ofphenyl isocyanide forms
A)
benzoicacid
done
clear
B)
formic acid
done
clear
C)
acetic acid
done
clear
D)
none of these
done
clear
View Answer play_arrow
question_answer 96) Styrene can be purified by
A)
simple distillation
done
clear
B)
fractional distillation
done
clear
C)
steam distillation
done
clear
D)
vacuum distillation
done
clear
View Answer play_arrow
question_answer 97) Which of the following is not reducing sugar?
A)
Glucose
done
clear
B)
Fructose
done
clear
C)
Lactose
done
clear
D)
Sucrose
done
clear
View Answer play_arrow
question_answer 98) The monomer of teflon is:
A)
\[CHF=C{{H}_{2}}\]
done
clear
B)
\[C{{F}_{2}}=C{{F}_{2}}\]
done
clear
C)
\[CHCl=CHCl\]
done
clear
D)
\[CHF=CHCl\]
done
clear
View Answer play_arrow
question_answer 99) The hybridisation state of carbon in fullerene is
A)
\[sp\]
done
clear
B)
\[s{{p}^{2}}\]
done
clear
C)
\[s{{p}^{3}}\]
done
clear
D)
\[s{{p}^{3}}d\]
done
clear
View Answer play_arrow
question_answer 100) A fruity smell is produced by the reaction of \[{{\text{C}}_{\text{2}}}{{\text{H}}_{\text{5}}}\text{OH}\,\]with
A)
\[C{{H}_{3}}COC{{H}_{3}}\]
done
clear
B)
\[C{{H}_{3}}COOH\]
done
clear
C)
\[PC{{l}_{5}}\]
done
clear
D)
\[C{{H}_{3}}CHO\]
done
clear
View Answer play_arrow
question_answer 101) Five kingdom classification was proposed by
A)
Linnaeus
done
clear
B)
Aristotle
done
clear
C)
Whittaker
done
clear
D)
Copeland
done
clear
View Answer play_arrow
question_answer 102) According to the Neo-Darwinian theory which of the following is responsible for the origin of new species?
A)
Mutations
done
clear
B)
Useful variations
done
clear
C)
Mutations together with natural selection
done
clear
D)
Hybridization
done
clear
View Answer play_arrow
question_answer 103) Enzymes found attached to inner membrane of mitochondria instead of matrix is/are
A)
succinic dehydrogenase
done
clear
B)
cytochrome oxidase
done
clear
C)
both [a] and [b]
done
clear
D)
malic dehydrogenase
done
clear
View Answer play_arrow
question_answer 104) Chlorosis is caused due to deficiency of
A)
Mg
done
clear
B)
Ca
done
clear
C)
B
done
clear
D)
Mn
done
clear
View Answer play_arrow
question_answer 105) Part of the plant which is cultured to obtain virus free clones is
A)
Leaf
done
clear
B)
root tip
done
clear
C)
shoot tip
done
clear
D)
embryo
done
clear
View Answer play_arrow
question_answer 106) The first stable compound of \[{{\text{C}}_{\text{3}}}\] plants during photosynthesis is
A)
PGA
done
clear
B)
RuBP
done
clear
C)
Oxaloacetate
done
clear
D)
Pyruvic acid
done
clear
View Answer play_arrow
question_answer 107) Double fertilization is fusion of
A)
two eggs
done
clear
B)
two eggs and polar nuclei
done
clear
C)
one male gamete with egg and other with synergid
done
clear
D)
one male gamete with egg and other with secondary nucleus
done
clear
View Answer play_arrow
question_answer 108) Inheritence of ABO blood group shows
A)
polyploidy
done
clear
B)
incomplete dominance
done
clear
C)
multiple allelism
done
clear
D)
polygeny
done
clear
View Answer play_arrow
question_answer 109) Photosynthetic enhancement with flashing light was first observed by
A)
Benson and Calvin
done
clear
B)
Hill and Calvin
done
clear
C)
Hatch and Slack
done
clear
D)
Emerson and Arnold
done
clear
View Answer play_arrow
question_answer 110) Gibberellin was first extracted from
A)
Gibberella fujikuroi
done
clear
B)
Gelidium
done
clear
C)
Gracelaria
done
clear
D)
Aspergillus
done
clear
View Answer play_arrow
question_answer 111) In E. coli. an operator gene combine with
A)
inducer gene to switch on structural gene action
done
clear
B)
inducer gene to switch off structural gene action
done
clear
C)
regulator protein (represser) to switch off structural gene action
done
clear
D)
regulator protein to switch on gene action
done
clear
View Answer play_arrow
question_answer 112) Exarch and polyarch xylem strands are found in
A)
monocot stem
done
clear
B)
dicotstem
done
clear
C)
monocot root
done
clear
D)
dicot root
done
clear
View Answer play_arrow
question_answer 113) The stage of Omithine cycle at which arginase enzyme is used
A)
Omithine\[\to \] citruUine
done
clear
B)
arginine \[\to \]Omithine
done
clear
C)
citrulline \[\to \] arginosuccinic acid
done
clear
D)
Omithine \[\to \] urea
done
clear
View Answer play_arrow
question_answer 114) Photosynthetic food material is transported in the form of
A)
glucose
done
clear
B)
sucrose
done
clear
C)
starch
done
clear
D)
glycogen
done
clear
View Answer play_arrow
question_answer 115) At retrogressive metamorphosis the uro chordate larva
A)
loss notochord
done
clear
B)
loss tail
done
clear
C)
experience reduction of nervous system to a visceral ganglion
done
clear
D)
all of the above
done
clear
View Answer play_arrow
question_answer 116) A species inhabiting different geographical area is known as
A)
Allopatric
done
clear
B)
Sympatric
done
clear
C)
Biospecies
done
clear
D)
Sibling species
done
clear
View Answer play_arrow
question_answer 117) Castor oil is yielded from which of the following?
A)
Brassica compestris
done
clear
B)
Sesamum indicum
done
clear
C)
Ricinus communis
done
clear
D)
Cocos nucifera
done
clear
View Answer play_arrow
question_answer 118) Which type of respiratory organs are present in spiders and scropions?
A)
Book lungs
done
clear
B)
Gills
done
clear
C)
Gill books
done
clear
D)
Lungs
done
clear
View Answer play_arrow
question_answer 119) Energy storage at consumer level is called
A)
gross primary productivity
done
clear
B)
secondry productivity .
done
clear
C)
net primary productivity
done
clear
D)
net productivity
done
clear
View Answer play_arrow
question_answer 120) After examining the blood groups of husband and wife, the doctor advised them not to have more than one child, the blood group of the couple are likely to be
A)
male \[R{{h}^{-}}\]and female \[R{{h}^{+}}\]
done
clear
B)
female \[R{{h}^{-}}\]and male \[R{{h}^{+}}\]
done
clear
C)
male \[R{{h}^{+}}\]and female\[R{{h}^{+}}\]
done
clear
D)
male \[R{{h}^{-}}\]and female\[R{{h}^{-}}\]
done
clear
View Answer play_arrow
question_answer 121) Main component of ribosome are
A)
DNA and RNA
done
clear
B)
RNA and protein
done
clear
C)
DNA and protein
done
clear
D)
protein and lipids
done
clear
View Answer play_arrow
question_answer 122) How many hearts are found in earthworm?
A)
8 (four pairs)
done
clear
B)
2 (one pair)
done
clear
C)
6 (three pairs)
done
clear
D)
12 (six pairs)
done
clear
View Answer play_arrow
question_answer 123) Schizocoelomates and entero coelomates are
A)
acoelomates
done
clear
B)
true coelomates
done
clear
C)
invertebrates
done
clear
D)
echinoderms only
done
clear
View Answer play_arrow
question_answer 124) \[\text{C}{{\text{O}}_{\text{2}}}\]is transported as
A)
dissolved in blood plasma
done
clear
B)
as carbonic acid
done
clear
C)
in carbammo haemoglobin
done
clear
D)
as carbamino haemoglobin and carbonic acid
done
clear
View Answer play_arrow
question_answer 125) Which of the damaged cells can not be repaired?
A)
Liver cells
done
clear
B)
Brain cells
done
clear
C)
Bone cells
done
clear
D)
Epidermal cells
done
clear
View Answer play_arrow
question_answer 126) Superficial meroblastic cleavage occurs in
A)
reptiles
done
clear
B)
birds
done
clear
C)
mammals
done
clear
D)
insects
done
clear
View Answer play_arrow
question_answer 127) One strand of DNA (non-template) has base sequence CAG, TCG GAT. What will be the sequence of bases in mRNA?
A)
AGC CTA CTA
done
clear
B)
GTC AGC CTC
done
clear
C)
CAG UCG GAU
done
clear
D)
GAC TAG CTA
done
clear
View Answer play_arrow
question_answer 128) Krebs cycle was discovered by Krebs in pigeon muscles in 1940. Which step is called gateway step/link reaction/transition reaction in respiration?
A)
Glycoiysis
done
clear
B)
Formation of acetyl-coA
done
clear
C)
citric acid formation
done
clear
D)
ETS terminal oxidation
done
clear
View Answer play_arrow
question_answer 129) When released from ovary, human egg contain
A)
one Y chromosome
done
clear
B)
two X chromosome
done
clear
C)
one X chromosome
done
clear
D)
XY chromosome
done
clear
View Answer play_arrow
question_answer 130) In the body, the membrane surrounding the bone is known as
A)
periosteum
done
clear
B)
endosteum
done
clear
C)
perichondrium
done
clear
D)
chondrocytes
done
clear
View Answer play_arrow
question_answer 131) Polygonum type of embryosac is
A)
7-celled, 8-nucleate
done
clear
B)
8-celled, 7-nucleate
done
clear
C)
9-nucleate
done
clear
D)
16-nucleate
done
clear
View Answer play_arrow
question_answer 132) Some organisms resemble other organisms and thus escape from enemies. This phenomenon is known as
A)
homology
done
clear
B)
variation
done
clear
C)
analogy
done
clear
D)
mimicry
done
clear
View Answer play_arrow
question_answer 133) Recent reports of acid rains in big industrial cities are due to the effect of atmospheric pollution by
A)
more relase of \[\text{N}{{\text{O}}_{\text{2}}}\] and \[\text{S}{{\text{O}}_{\text{2}}}\]by burning of fossil fuels
done
clear
B)
more relase of \[\text{C}{{\text{O}}_{2}}\]by burning of coal/wood cutting of forests and increasing popultions
done
clear
C)
excessive release of \[\text{N}{{\text{H}}_{\text{3}}}\]by coal gas/industries
done
clear
D)
excessive release of CO by incomplete combustion of carbonaceous fuels
done
clear
View Answer play_arrow
question_answer 134) There is more competition for survival between
A)
different animals of same niche
done
clear
B)
same animals of same niche
done
clear
C)
different animals of different niche
done
clear
D)
same animals of different niche
done
clear
View Answer play_arrow
question_answer 135) Which set includes pathogenic Arthropods?
A)
Tse-tse fly, mosquito, flea-plague
done
clear
B)
Crab, \[Culex,\]spider
done
clear
C)
Anopheles, \[Culex,\]cray fish
done
clear
D)
Silver fish, house fly, sandfly
done
clear
View Answer play_arrow
question_answer 136) Ozone layer is being destroyed by
A)
\[S{{O}_{2}}\]
done
clear
B)
\[N{{O}_{2}}\]
done
clear
C)
\[CFCs\]
done
clear
D)
Photochemical smog
done
clear
View Answer play_arrow
question_answer 137) In the blind spot where the optic nerves leaves the eyes
A)
rods and cones are absent
done
clear
B)
only cones are present
done
clear
C)
only rods are present
done
clear
D)
special neurons are present
done
clear
View Answer play_arrow
question_answer 138) Which of the following blood vessels in the circulatory system of frog has more oxygenated blood?
A)
Pulmocutaneous artery
done
clear
B)
Pulmocutaneous vein
done
clear
C)
Pulmonary artery
done
clear
D)
Precaval veins
done
clear
View Answer play_arrow
question_answer 139) The enzyme, which helps to cut one strand of DNA duplex to release tension of coiling of two strands is
A)
DNA ligase
done
clear
B)
DNA pblymerase
done
clear
C)
topo-isomerase
done
clear
D)
swielases (helicase or unwindases)
done
clear
View Answer play_arrow
question_answer 140) Blood vascular system in hemichordata is
A)
open
done
clear
B)
reduced
done
clear
C)
closed
done
clear
D)
absent
done
clear
View Answer play_arrow
question_answer 141) In eukaryotes basic structural unit made of histone and DNA is
A)
nucleosome
done
clear
B)
nucleolus
done
clear
C)
chromosome
done
clear
D)
lysosome
done
clear
View Answer play_arrow
question_answer 142) Anaemia is due to deficiency of
A)
Ca
done
clear
B)
Fe
done
clear
C)
Mg
done
clear
D)
P
done
clear
View Answer play_arrow
question_answer 143) Mechanical support, enzyme circulation, protein synthesis and detoxification of drugs are function of
A)
ER
done
clear
B)
ribosomes
done
clear
C)
dictyosomes
done
clear
D)
chloroplast
done
clear
View Answer play_arrow
question_answer 144) Characteristic of mammalian liver is
A)
Kupffers cells and leucocytes
done
clear
B)
Leucocytes and canaliculae
done
clear
C)
Glissons capsules and Kupffer cells
done
clear
D)
Glissons capsules and leucocytes
done
clear
View Answer play_arrow
question_answer 145) Which of the following required both during photosynthesis and respiration?
A)
Phytochrome
done
clear
B)
Cytochrome
done
clear
C)
Chlorophyll
done
clear
D)
Glucose
done
clear
View Answer play_arrow
question_answer 146) One-gene one enzyme hypothesis was proposed by
A)
Beadle and Tatum
done
clear
B)
Jacob and Monod
done
clear
C)
Lederberg
done
clear
D)
Watson and Crick
done
clear
View Answer play_arrow
question_answer 147) Phytohormone commonly called stress hormone is
A)
auxin
done
clear
B)
abscisic acid
done
clear
C)
gibberellin
done
clear
D)
cytokinins
done
clear
View Answer play_arrow
question_answer 148) Saccharomyces is commonly used in the production of
A)
ethyl alcohol
done
clear
B)
curd
done
clear
C)
citric acid
done
clear
D)
acetic acid
done
clear
View Answer play_arrow
question_answer 149) Food chain is a series of population which starts with producers. It is concerning with
A)
biotic components only
done
clear
B)
energy flow and transfer of nutrients
done
clear
C)
both [a] and [b]
done
clear
D)
biotic and decomposers
done
clear
View Answer play_arrow
question_answer 150) Myxoedema is a disorder of
A)
parathyroid gland
done
clear
B)
adrenal gland
done
clear
C)
pituitary gland
done
clear
D)
thyroid gland
done
clear
View Answer play_arrow
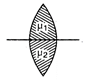
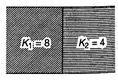 A capacitor having capacitance \[1\,\mu F\]with air, is filled with two dielectrics as shown. How many times capacitance will increase?
A capacitor having capacitance \[1\,\mu F\]with air, is filled with two dielectrics as shown. How many times capacitance will increase?