question_answer 1) It is easier to wash clothes in hot water because:
A)
surface tension of hot water is more
done
clear
B)
surface tension of hot water is less
done
clear
C)
less soap is used
done
clear
D)
none of these
done
clear
View Answer play_arrow
question_answer 2) Time period of a satellite ... on decreasing its height:
A)
will decrease
done
clear
B)
will increase
done
clear
C)
will remain same
done
clear
D)
none of these
done
clear
View Answer play_arrow
question_answer 3) A ball is released from certain height which looses 50% of its kinetic energy on striking the ground it will attain a height again:
A)
\[\frac{1}{4}\]th of initial height
done
clear
B)
\[\frac{1}{2}\]th of initial height
done
clear
C)
\[\frac{3}{4}\]th of initial height
done
clear
D)
none of these
done
clear
View Answer play_arrow
question_answer 4) The ratio of escape velocity and orbital velocity of satellite revolving near the earth:
A)
\[\sqrt{2}\]
done
clear
B)
2
done
clear
C)
3
done
clear
D)
4
done
clear
View Answer play_arrow
question_answer 5) When a load of 5 kg is hung on a wire then extension of 3 metre takes place, then work done will be:
A)
\[\text{75}\,\text{J}\]
done
clear
B)
\[60\,\text{J}\]
done
clear
C)
\[50\,\text{J}\]
done
clear
D)
\[100\,\text{J}\]
done
clear
View Answer play_arrow
question_answer 6) The relation between time period (T) of a body and radius of circular orbit (r) is:
A)
\[{{T}^{2}}\propto {{r}^{3}}\]
done
clear
B)
\[T\propto {{r}^{3}}\]
done
clear
C)
\[T\propto r\]
done
clear
D)
\[T\propto {{r}^{4}}\]
done
clear
View Answer play_arrow
question_answer 7) If applied torque on a system is zero, i.e., \[\tau =0\], then for that system:
A)
\[\omega =0\]
done
clear
B)
\[\alpha =0\]
done
clear
C)
\[J=0\]
done
clear
D)
\[F=0\]
done
clear
View Answer play_arrow
question_answer 8) A steel wire of 1 m long and 1 mm2 cross section area is hanged from rigid end when weight of 1 kg is hung from it then change in length will be (for wire Youngs coefficient \[Y=2\times {{10}^{11}}\,N/{{m}^{2}}\]):
A)
0.5 mm
done
clear
B)
0.25 mm
done
clear
C)
0.05 mm
done
clear
D)
5 mm
done
clear
View Answer play_arrow
question_answer 9) Reason for weightlessness in satellite:
A)
zero gravity
done
clear
B)
centre of gravity
done
clear
C)
zero reaction force on plane of satellite
done
clear
D)
none of these
done
clear
View Answer play_arrow
question_answer 10) If solid sphere and solid cylinder of same mass and density rotate about their own axis, the moment of inertia will be greater for:
A)
solid sphere
done
clear
B)
cylinder
done
clear
C)
equal for both
done
clear
D)
none of these
done
clear
View Answer play_arrow
question_answer 11) A satellite of mass\[{{m}_{s}}\] is moving in an orbit of radius R, then its angular momentum:
A)
\[\sqrt{G{{M}_{e}}{{m}_{s}}R}\]
done
clear
B)
\[\sqrt{G{{M}_{e}}{{m}_{s}}^{2}R}\]
done
clear
C)
\[\frac{\sqrt{G{{M}_{e}}{{m}_{s}}^{2}}}{R}\]
done
clear
D)
\[\frac{\sqrt{G{{M}_{e}}{{m}_{s}}}}{R}\]
done
clear
View Answer play_arrow
question_answer 12) Contact angle of kerosen oil with solid surface is:
A)
\[0{}^\circ \]
done
clear
B)
\[90{}^\circ \]
done
clear
C)
\[45{}^\circ \]
done
clear
D)
\[33{}^\circ \]
done
clear
View Answer play_arrow
question_answer 13) True relation for triode valve is:
A)
\[\mu ={{r}_{P}}\times {{g}_{m}}\]
done
clear
B)
\[\mu =\frac{{{g}_{m}}}{{{r}_{P}}}\]
done
clear
C)
\[\mu =2{{g}_{m}}\times {{r}_{p}}\]
done
clear
D)
\[\mu =\frac{{{g}_{m}}}{{{r}_{p}}}\]
done
clear
View Answer play_arrow
question_answer 14) When plate voltage in diode valve is increased from 100 volt to 150 volt. Then plate current increases from 7.5 mA to 12 mA, then dynamic plate resistance will be:
A)
10 \[k\Omega \]
done
clear
B)
11\[k\Omega \]
done
clear
C)
15\[k\Omega \]
done
clear
D)
11.1\[k\Omega \]
done
clear
View Answer play_arrow
question_answer 15) A larger drop is made from two small drops each having 100 V, then voltage on the bigger drop is:
A)
100 V
done
clear
B)
200 V
done
clear
C)
156 V
done
clear
D)
\[100\times {{2}^{-2/3}}V\]
done
clear
View Answer play_arrow
question_answer 16) Which of the following is a correct relation:
A)
\[\alpha =\frac{\beta }{1-\beta }\]
done
clear
B)
\[\beta =\frac{\alpha }{1-\alpha }\]
done
clear
C)
\[\beta =\frac{\alpha }{1+\alpha }\]
done
clear
D)
\[\alpha =\frac{\beta }{1+\alpha }\]
done
clear
View Answer play_arrow
question_answer 17) If the sun rays are incidenting at \[60{}^\circ \] angle and intensity is \[I\]. If sun rays are made incident at \[30{}^\circ \] angle, then what will be the intensity?
A)
\[\frac{I}{\sqrt{3}}\]
done
clear
B)
\[\sqrt{3}I\]
done
clear
C)
\[3I\]
done
clear
D)
\[\frac{I}{3}\]
done
clear
View Answer play_arrow
question_answer 18) Value of forbidden energy gap for semi conductor is:
A)
1 eV
done
clear
B)
6 eV
done
clear
C)
0 eV
done
clear
D)
3 eV
done
clear
View Answer play_arrow
question_answer 19) The reason of current flow in p-n junction in forward bias is:
A)
drifting of charge carriers
done
clear
B)
drifting of minority charge carriers
done
clear
C)
diffusion of charge carriers
done
clear
D)
all the above
done
clear
View Answer play_arrow
question_answer 20) A current of 1 mA is flowing in conductor, then number of electrons passing through per second in conductor is:
A)
\[6.25\times {{10}^{17}}\]
done
clear
B)
\[6.25\times {{10}^{15}}\]
done
clear
C)
\[1.6\,\times {{10}^{16}}\]
done
clear
D)
\[1.6\,\times {{10}^{-16}}\]
done
clear
View Answer play_arrow
question_answer 21) In a coil current changes from 2A to 4A in 0.05 second. If the average induced e.m.f. is 8 volt, then coefficient of self inductance is:
A)
0.2H
done
clear
B)
0.1H
done
clear
C)
0.8H
done
clear
D)
0.4 H
done
clear
View Answer play_arrow
question_answer 22) A battery having e.m.f. 5 volt and internal resistance 0.5\[\Omega \] is connected with a resistance of 4.5\[\Omega \], then the voltage at the terminals of battery is:
A)
4.5 V
done
clear
B)
4 V
done
clear
C)
0 V
done
clear
D)
2 V
done
clear
View Answer play_arrow
question_answer 23) A charged particle is accelerated by a potential of 200 volt. If its velocity is \[8.4\times {{10}^{8}}m/s,\] then value of e/m for that particle is:
A)
\[17.6\times {{10}^{16}}\]
done
clear
B)
\[14.5\times {{10}^{12}}\]
done
clear
C)
\[1.76\times {{10}^{15}}\]
done
clear
D)
\[1.45\times {{10}^{15}}\]
done
clear
View Answer play_arrow
question_answer 24) Thermoionic current is directly proportional to:
A)
\[{{T}^{1/2}}\]
done
clear
B)
\[{{T}^{3/2}}\]
done
clear
C)
\[{{T}^{2}}\]
done
clear
D)
\[T\]
done
clear
View Answer play_arrow
question_answer 25) Zener diode is used as:
A)
an amplifier
done
clear
B)
a rectifier
done
clear
C)
a oscillator
done
clear
D)
a voltage regulator
done
clear
View Answer play_arrow
question_answer 26) Ga As is a/an:
A)
element semiconductor
done
clear
B)
alloy semiconductor
done
clear
C)
bad conductor
done
clear
D)
metalic semiconductor
done
clear
View Answer play_arrow
question_answer 27) Junction diode is used as:
A)
a rectifier
done
clear
B)
an amplifier
done
clear
C)
an indicator
done
clear
D)
a modulator
done
clear
View Answer play_arrow
question_answer 28) On giving 220 V to a resistor the power dissipated is 40 watt, then the value of resistance is:
A)
1210\[\Omega \]
done
clear
B)
2000\[\Omega \]
done
clear
C)
1000\[\Omega \]
done
clear
D)
none of these
done
clear
View Answer play_arrow
question_answer 29)
If each resistance in the figure is of 9\[\Omega \] then reading of ammeter is:
A)
5 A
done
clear
B)
8 A
done
clear
C)
2 A
done
clear
D)
9 A
done
clear
View Answer play_arrow
question_answer 30) Intensity of light depends upon:
A)
amplitude
done
clear
B)
frequency
done
clear
C)
wavelength
done
clear
D)
none of these
done
clear
View Answer play_arrow
question_answer 31) Velocity of sound is maximum in:
A)
\[{{H}_{2}}\]
done
clear
B)
\[{{N}_{2}}\]
done
clear
C)
He
done
clear
D)
\[{{O}_{2}}\]
done
clear
View Answer play_arrow
question_answer 32) If refractive index of a medium is 1.5, then velocity of light in that medium will be:
A)
\[10\times {{10}^{8}}\]
done
clear
B)
\[2\times {{10}^{8}}\]
done
clear
C)
\[3\times {{10}^{8}}\]
done
clear
D)
\[4\times {{10}^{8}}\]
done
clear
View Answer play_arrow
question_answer 33) Decible is the unit of:
A)
intensity of light
done
clear
B)
radiating power of X-rays
done
clear
C)
intensity of sound
done
clear
D)
energy of radiation
done
clear
View Answer play_arrow
question_answer 34) Light enters into glass from air then its:
A)
frequency increases
done
clear
B)
frequency decreases
done
clear
C)
wavelength increases
done
clear
D)
wavelength decreases
done
clear
View Answer play_arrow
question_answer 35) A real gas behaves like an ideal gas at:
A)
high temperature and low pressure
done
clear
B)
low temperature and high pressure
done
clear
C)
high temperature and high pressure
done
clear
D)
low temperature and low pressure
done
clear
View Answer play_arrow
question_answer 36) Relation between pressure (P) and energy (E) for a unit volume of gas :
A)
\[E=\frac{3}{2}p\]
done
clear
B)
\[E=\frac{2}{3}p\]
done
clear
C)
\[E=\frac{p}{3}\]
done
clear
D)
\[E=3p\]
done
clear
View Answer play_arrow
question_answer 37) Ratio of rms velocities of 02 and H2 at equal temperature will be :
A)
1 : 1
done
clear
B)
1 : 4
done
clear
C)
2 : 1
done
clear
D)
4 : 1
done
clear
View Answer play_arrow
question_answer 38) The colour of red glass in a dark room will be:
A)
green
done
clear
B)
indigo
done
clear
C)
black
done
clear
D)
yellow
done
clear
View Answer play_arrow
question_answer 39) What will be the wave velocity, if the radar gives 5.4 waves per minute and wavelength of the given wave is 10 m?
A)
4m/s
done
clear
B)
6m/s
done
clear
C)
0.9m/s
done
clear
D)
5m/s
done
clear
View Answer play_arrow
question_answer 40) Which of the following will remain constant in the phenomenon of refraction of light?
A)
Wavelength
done
clear
B)
Velocity
done
clear
C)
Frequency
done
clear
D)
None
done
clear
View Answer play_arrow
question_answer 41) If distance between the molecules of a gas is doubled, then pressure will become (if initial presure is P):
A)
\[\frac{p}{4}\]
done
clear
B)
\[\frac{p}{2}\]
done
clear
C)
\[\frac{p}{8}\]
done
clear
D)
\[p\]
done
clear
View Answer play_arrow
question_answer 42) When an open organ is dipped in water upto half of its height, then its frequency will become:
A)
half
done
clear
B)
double
done
clear
C)
remain same
done
clear
D)
four time
done
clear
View Answer play_arrow
question_answer 43) On basis of which property light waves can be differentiated from sound waves:
A)
interference
done
clear
B)
diffraction
done
clear
C)
polarisaton
done
clear
D)
reflection
done
clear
View Answer play_arrow
question_answer 44) If position of a particle X is directly proportional to t2 then:
A)
velocity will be constant
done
clear
B)
acceleration will be constant
done
clear
C)
acceleration will be variable
done
clear
D)
none of these
done
clear
View Answer play_arrow
question_answer 45) Which of the following required no medium for propagation?
A)
Cathode rays
done
clear
B)
Electromagnetic rays
done
clear
C)
Sound waves
done
clear
D)
None of these
done
clear
View Answer play_arrow
question_answer 46) If wavelength of photon is \[6000\overset{\text{o}}{\mathop{\text{A}}}\,\], then its energy will be:
A)
0.66 eV
done
clear
B)
1.66 eV
done
clear
C)
2.66 eV
done
clear
D)
3.5 eV
done
clear
View Answer play_arrow
question_answer 47) Which of the following particle has similar mass to electron?
A)
Proton
done
clear
B)
Neutron
done
clear
C)
Positron
done
clear
D)
Neutrino
done
clear
View Answer play_arrow
question_answer 48) 1 amu is equivalent to:
A)
931 MeV
done
clear
B)
0.51 eV
done
clear
C)
9.31 MeV
done
clear
D)
1-02 MeV
done
clear
View Answer play_arrow
question_answer 49) If half life of a substance is 38 days and its quantity is 10.38 g. Then quantity remaining left after 19 days will be:
A)
0.151 g
done
clear
B)
0.32g
done
clear
C)
0.51g
done
clear
D)
0.16g
done
clear
View Answer play_arrow
question_answer 50) Boron rods in nuclear reactor are used as a:
A)
moderator
done
clear
B)
control rods
done
clear
C)
coolent
done
clear
D)
protectivc shield
done
clear
View Answer play_arrow
question_answer 51) The number of free electrons in Si at normal temperature is:
A)
\[2.5\times {{10}^{6}}\,per\,c{{m}^{3}}\]
done
clear
B)
\[1.5\times {{10}^{10}}\,per\,c{{m}^{3}}\]
done
clear
C)
\[1.5\times {{10}^{13}}\,per\,c{{m}^{3}}\]
done
clear
D)
\[2.5\times {{10}^{13}}\,per\,c{{m}^{3}}\]
done
clear
View Answer play_arrow
question_answer 52) Zener breakdown will occur if:
A)
impurity level is high
done
clear
B)
impurity level is low
done
clear
C)
impurity level is less is n side
done
clear
D)
impurity level is less in p side
done
clear
View Answer play_arrow
question_answer 53) A train is stopped by applying breaks, then it stops after covering 50 m. If initial velocity of train is double than before and train is stopped with same force, then the train will stop at a distance of:
A)
50 metre
done
clear
B)
100 metre
done
clear
C)
200 metre
done
clear
D)
400 metre
done
clear
View Answer play_arrow
question_answer 54) A shell having a hole of radius r is dipped in water. It holds the water upto a depth h, then the value of r is:
A)
\[r=\frac{T}{2hdg}\]
done
clear
B)
\[r=\frac{\sqrt{2}T}{hdg}\]
done
clear
C)
\[r=\frac{T}{hdg}\]
done
clear
D)
\[r=\frac{2T}{hdg}\]
done
clear
View Answer play_arrow
question_answer 55) The energy of an electron in the excited state of H-atom is - 1.5 eV, then according to Bohrs model, its momentum will be:
A)
\[3.15\times {{10}^{-34}}\,J-\sec \]
done
clear
B)
\[1.82\times {{10}^{-34}}\,J-\sec \]
done
clear
C)
\[5.01\times {{10}^{-30}}\,J-\sec \]
done
clear
D)
\[7.14\times {{10}^{-31}}\,J-\sec \]
done
clear
View Answer play_arrow
question_answer 56) A force of 10 N displaces an object by 1 m. If work done is 50 J, then direction of force makes an angle with direction of displacement:
A)
\[0{}^\circ \]
done
clear
B)
\[60{}^\circ \]
done
clear
C)
\[90{}^\circ \]
done
clear
D)
\[30{}^\circ \]
done
clear
View Answer play_arrow
question_answer 57) The work done against gravity in taking 10 kg mass at 1 metre height in 1 sec will be:
A)
98 J
done
clear
B)
150 J
done
clear
C)
600 J
done
clear
D)
980 J
done
clear
View Answer play_arrow
question_answer 58) A swing is revolving in vertical plane, its height from ground in lowermost and uppermost position or 1 metre and 5 metre. Then its velocity at uppermost point will be:
A)
\[\sqrt{2}g\]
done
clear
B)
\[2\sqrt{g}\]
done
clear
C)
\[2g\]
done
clear
D)
\[4g\]
done
clear
View Answer play_arrow
question_answer 59) \[dU+dW=0,\] is valid for:
A)
cyclic process
done
clear
B)
isochoric process
done
clear
C)
adiabatic process
done
clear
D)
isobaric process
done
clear
View Answer play_arrow
question_answer 60) If mass number for an element is M and atomic number is Z, then number of neutrons will be:
A)
M - Z
done
clear
B)
Z - M
done
clear
C)
M + Z
done
clear
D)
Z
done
clear
View Answer play_arrow
question_answer 61) If initial temperature of a gas is \[25{}^\circ C\] and its temperature is raised upto \[250{}^\circ C\], then its rms velocity:
A)
will decrease by a factor \[\sqrt{1.754}\]
done
clear
B)
will increase by a factor \[\sqrt{1.754}\]
done
clear
C)
will decrease by a factor \[\sqrt{10}\]
done
clear
D)
will increase by a factor \[\sqrt{10}\]
done
clear
View Answer play_arrow
question_answer 62) Work function of a oxide coated metal surface will be:
A)
1.1 eV
done
clear
B)
2.63 eV
done
clear
C)
4.52 eV
done
clear
D)
75 eV
done
clear
View Answer play_arrow
question_answer 63) Positron was discovered ill the year:
A)
1898
done
clear
B)
1902
done
clear
C)
1925
done
clear
D)
1932
done
clear
View Answer play_arrow
question_answer 64)
In the following figure distance of the point from A, when electric field intensity is zero:
A)
20 cm
done
clear
B)
10 cm
done
clear
C)
33 cm
done
clear
D)
none of these
done
clear
View Answer play_arrow
question_answer 65) Prism of which material is used for study of infrared spectrum :
A)
rock salt
done
clear
B)
flint glass
done
clear
C)
crown glass
done
clear
D)
quartz
done
clear
View Answer play_arrow
question_answer 66) Three capacitors of \[2\mu F,\]\[3\mu F,\] and \[6\mu F,\]are joined in scries with n battery of 10 volt, then amount of charge in\[3\mu F,\] capacitor will be:
A)
\[5\mu C\]
done
clear
B)
\[10\mu C\]
done
clear
C)
\[11\mu C\]
done
clear
D)
\[15\mu C\]
done
clear
View Answer play_arrow
question_answer 67) An \[\alpha -\]particle moving perpendicularly to a magnetic field of \[0.2\,wb/metr{{e}^{2}},\] experiences a force of \[3.84\times {{10}^{-24}}N\]. Then speed of a particle will be:
A)
\[6\times {{10}^{5}}\,m/s\]
done
clear
B)
\[5\times {{10}^{-5}}\,m/s\]
done
clear
C)
\[1.2\times {{10}^{6}}\,m/s\]
done
clear
D)
\[3.8\,\times {{10}^{6}}\,m/s\]
done
clear
View Answer play_arrow
question_answer 68) If a satellite revolving round the earth decreases its orbital radius, then its time period:
A)
decreases
done
clear
B)
unchanged
done
clear
C)
increases
done
clear
D)
none of these
done
clear
View Answer play_arrow
question_answer 69) Unit of magnetic flux is:
A)
ampere/metre2
done
clear
B)
weber
done
clear
C)
gauss
done
clear
D)
orested
done
clear
View Answer play_arrow
question_answer 70) Equation of a progressive wave is \[y=a\,\sin \left( 400\pi t-\frac{\pi x}{0.85} \right),\]then its velocity will be:
A)
34 m/s
done
clear
B)
40 m/s
done
clear
C)
340 m/s
done
clear
D)
400 m/s
done
clear
View Answer play_arrow
question_answer 71) Magnetic moment of Ne will be:
A)
0
done
clear
B)
\[{{\mu }_{B}}\]
done
clear
C)
\[\sqrt{2}{{\mu }_{B}}\]
done
clear
D)
\[{{\sqrt{2\mu }}_{B}}\]
done
clear
View Answer play_arrow
question_answer 72) To obtain beats, the minimum difference in the frequencies of two sources is:
A)
4 Hz
done
clear
B)
10 Hz
done
clear
C)
15 Hz
done
clear
D)
20 Hz
done
clear
View Answer play_arrow
question_answer 73) Remaining quantity (in %) of radioactive element after 5 half lives:
A)
4.125%
done
clear
B)
3.125%
done
clear
C)
31.1%
done
clear
D)
42.125%
done
clear
View Answer play_arrow
question_answer 74) When neutrons are bombarded on \[_{5}{{B}^{10}}\]then\[_{5}{{B}^{10}}+{{1}_{{{n}_{0}}}}\xrightarrow{{}}X{{+}_{2}}H{{e}^{4}},X\] is:
A)
\[_{3}L{{i}^{7}}\]
done
clear
B)
\[_{3}L{{i}^{6}}\]
done
clear
C)
\[_{5}B{{e}^{8}}\]
done
clear
D)
\[_{5}B{{e}^{12}}\]
done
clear
View Answer play_arrow
question_answer 75) 75. Work function of a metal is \[5.26\text{ }\times {{10}^{-18}}\] then its threshold wavelength will be:
A)
\[736.7\,\overset{\text{o}}{\mathop{\text{A}}}\,\]
done
clear
B)
\[760.7\,\overset{\text{o}}{\mathop{\text{A}}}\,\]
done
clear
C)
\[301\,\overset{\text{o}}{\mathop{\text{A}}}\,\]
done
clear
D)
\[344.4\,\overset{\text{o}}{\mathop{\text{A}}}\,\]
done
clear
View Answer play_arrow
question_answer 76) The resistors whose ratio is 1 : 2, are connected in parallel, the ratio of power dissipated is:
A)
1 : 2
done
clear
B)
1 : 4
done
clear
C)
4 : 1
done
clear
D)
2 : 1
done
clear
View Answer play_arrow
question_answer 77) Hole are the charge carriers in:
A)
semiconductor
done
clear
B)
ionic solids
done
clear
C)
p-type semiconductor
done
clear
D)
metals
done
clear
View Answer play_arrow
question_answer 78) A radio station is transmitting the waves of wavelength 300m. Radiation capacity of the transmitter is 10 kW. Find out the number of photons which are emitting in per unit time:
A)
\[1.5\times {{10}^{35}}\]
done
clear
B)
\[1.5\times {{10}^{31}}\]
done
clear
C)
\[1.5\times {{10}^{29}}\]
done
clear
D)
\[1.5\times {{10}^{33}}\]
done
clear
View Answer play_arrow
question_answer 79) Which of the following has maximum energy?
A)
Radio waves
done
clear
B)
Infrared waves
done
clear
C)
Ultravoilet waves
done
clear
D)
Micro waves
done
clear
View Answer play_arrow
question_answer 80) On increasing the temperature by \[1{}^\circ C\] of sound, its velocity increases by:
A)
0.6 m/s
done
clear
B)
1.2 m/s
done
clear
C)
4 m/s
done
clear
D)
0
done
clear
View Answer play_arrow
question_answer 81) A cylindrical wire is stretched upto twice of its length, then its resistance becomes :
A)
4 times
done
clear
B)
unchanged
done
clear
C)
\[\frac{1}{2}\]times
done
clear
D)
2 times
done
clear
View Answer play_arrow
question_answer 82) A person is standing on a moving platform spreading his arms. He suddenly contracts his rms, his:
A)
angular momentum will increase.
done
clear
B)
moment of inertia will increase.
done
clear
C)
moment of inertia will decrease.
done
clear
D)
angular momentum remains same.
done
clear
View Answer play_arrow
question_answer 83) A larger drop is made from two smaller drops, then energy :
A)
emitted
done
clear
B)
absorbed
done
clear
C)
no exchange of heat
done
clear
D)
none of these
done
clear
View Answer play_arrow
question_answer 84) A particle is moving with constant speed in horizontal circle of radius R. Then in completing half cycle:
A)
change in momentum is \[m\upsilon r\]
done
clear
B)
change in kinetic energy is\[\frac{1}{2}m\upsilon r\]
done
clear
C)
change in kinetic energy is \[m{{\upsilon }^{2}}\]
done
clear
D)
change in kinetic energy is zero
done
clear
View Answer play_arrow
question_answer 85) A lift is ascending by acceleration g/3. What will be the time period of a simple pendulum, suspended from its cilling? If its time period in stationary life is T:
A)
\[\frac{T}{2}\]
done
clear
B)
\[\frac{\sqrt{3T}}{2}\]
done
clear
C)
\[\frac{\sqrt{3T}}{4}\]
done
clear
D)
\[\frac{T}{4}\]
done
clear
View Answer play_arrow
question_answer 86) Equation for an ideal gas is:
A)
PV = RT
done
clear
B)
\[P{{V}^{\gamma }}\] = constant
done
clear
C)
\[{{C}_{P}}-{{C}_{P}}=R\]
done
clear
D)
none of these
done
clear
View Answer play_arrow
question_answer 87) A child is running towards a plane mirror with a velocity 5 m/s, then his image appears coming towards him with velocity:
A)
10 m/s
done
clear
B)
2.5 m/s
done
clear
C)
5m/s
done
clear
D)
4m/s
done
clear
View Answer play_arrow
question_answer 88) Which of the colour of light is maximum scattered?
A)
Red
done
clear
B)
Voilet
done
clear
C)
Blue
done
clear
D)
Green
done
clear
View Answer play_arrow
question_answer 89) Image formed by a concave mirror of focal length 6 cm, is 3 times of the object, then the distance of object from mirror is:
A)
- 4 cm
done
clear
B)
8 cm
done
clear
C)
6 cm
done
clear
D)
12 cm
done
clear
View Answer play_arrow
question_answer 90) Focal length of a plane mirror is:
A)
zero
done
clear
B)
infinite
done
clear
C)
very less
done
clear
D)
indefinite
done
clear
View Answer play_arrow
question_answer 91) Magnetic; field of earth is H = 0.3g, A magnet is vibrating 5 oscillations/ minute the deppreciation required in the magnetic field of earth of increase time period uoto 10 oscillations minute:
A)
2.25 g
done
clear
B)
0.6g
done
clear
C)
0.9g
done
clear
D)
0.12g
done
clear
View Answer play_arrow
question_answer 92) Torr is the unit of:
A)
pressure
done
clear
B)
density
done
clear
C)
volume
done
clear
D)
flux
done
clear
View Answer play_arrow
question_answer 93) Who first discovered that white light consists of seven colours?
A)
Einestein
done
clear
B)
Raman
done
clear
C)
Faraday
done
clear
D)
Newton
done
clear
View Answer play_arrow
question_answer 94) The energy obtained by accelerating an electron by a potential difference of 1 V:
A)
1 eV
done
clear
B)
2 J
done
clear
C)
1 W
done
clear
D)
1 erg
done
clear
View Answer play_arrow
question_answer 95) Angular momentum is:
A)
axial vector
done
clear
B)
polar vector
done
clear
C)
scalar
done
clear
D)
vector
done
clear
View Answer play_arrow
question_answer 96) The displacement (s) of a body is directly proportional to square of time (r), then its acceleration:
A)
increases
done
clear
B)
remain constant
done
clear
C)
decreases
done
clear
D)
zero
done
clear
View Answer play_arrow
question_answer 97) Kinetic energy of a gas is:
A)
\[\frac{3}{2}KT\]
done
clear
B)
\[KT\]
done
clear
C)
\[RT\]
done
clear
D)
\[3KT\]
done
clear
View Answer play_arrow
question_answer 98) Kinetic energy of a liquid on freezing:
A)
increases
done
clear
B)
decreases
done
clear
C)
remains same
done
clear
D)
none of these
done
clear
View Answer play_arrow
question_answer 99) Ratio among linear expansion coefficient (\[\alpha \]), areal expansion coefficient (\[\beta \]) and volume expansion coefficient \[(\gamma )\] is:
A)
1: 2 : 3
done
clear
B)
3 : 2 : 1
done
clear
C)
4 : 3 : 2
done
clear
D)
none of these
done
clear
View Answer play_arrow
question_answer 100) The force acting on unit length on unit parallel current carrying conductor:
A)
zero
done
clear
B)
\[\frac{{{\mu }_{0}}}{2\pi }\frac{{{i}_{1}}{{i}_{2}}}{r}\]
done
clear
C)
\[{{\mu }_{0}}\frac{^{i}{{1}^{i}}2}{{{r}^{2}}}\]
done
clear
D)
\[\frac{^{i}{{1}^{i}}2}{r}\]
done
clear
View Answer play_arrow
question_answer 101) \[C{{H}_{3}}CHO\]and \[C{{H}_{3}}COC{{H}_{3}}\]from addition products with:
A)
Phenyl hydrazine
done
clear
B)
\[{{H}_{2}}N.N{{H}_{2}}\]
done
clear
C)
Semi carbazide
done
clear
D)
\[HCN\]
done
clear
View Answer play_arrow
question_answer 102) Carbinol is the trivial name for:
A)
Methyl alcohol
done
clear
B)
\[C{{H}_{3}}C{{H}_{2}}CHOHC{{H}_{3}}\]
done
clear
C)
Ethyl alcohol
done
clear
D)
\[{{(C{{H}_{3}})}_{3}}COH\]
done
clear
View Answer play_arrow
question_answer 103) The order of polarity of \[C{{H}_{3}}I,\]\[C{{H}_{3}}Br\] and \[C{{H}_{3}}Cl\]molecule is:
A)
\[C{{H}_{3}}Cl>C{{H}_{3}}I>C{{H}_{3}}Br\]
done
clear
B)
\[C{{H}_{3}}I>C{{H}_{3}}Br>C{{H}_{3}}Cl\]
done
clear
C)
\[C{{H}_{3}}Cl>C{{H}_{3}}Br>C{{H}_{3}}I\]
done
clear
D)
\[C{{H}_{3}}Br>C{{H}_{3}}Cl>C{{H}_{3}}I\]
done
clear
View Answer play_arrow
question_answer 104) Phenol reacts with \[B{{r}_{2}}\] in \[CC{{l}_{4}}\] at low temperature to give:
A)
O and p-bromo phenol
done
clear
B)
p-bromo phenol
done
clear
C)
2, 4, 6-tribromo phenol
done
clear
D)
m-bromo phenol
done
clear
View Answer play_arrow
question_answer 105) Which of these is gem-dihalide?
A)
\[C{{H}_{3}}.\,CHB{{r}_{2}}\]
done
clear
B)
\[C{{H}_{2}}Br.\,C{{H}_{2}}Br\]
done
clear
C)
\[C{{H}_{3}}CHBr.C{{H}_{2}}Br\]
done
clear
D)
None of these
done
clear
View Answer play_arrow
question_answer 106) The nitro group in nitrobenzene is:
A)
Para directing
done
clear
B)
Ortho directing
done
clear
C)
Meta directing
done
clear
D)
Ortho and para directing
done
clear
View Answer play_arrow
question_answer 107) Which of the following group combined and formed carboxylic group?
A)
2-carbonyl group
done
clear
B)
Carbonyl group + hydroxyl group
done
clear
C)
Both (a) and (b)
done
clear
D)
None of these
done
clear
View Answer play_arrow
question_answer 108) In methyl sprit:
A)
\[C{{H}_{3}}OH+pyridine+poision\text{ }product\]
done
clear
B)
\[C{{H}_{3}}OH+{{H}_{2}}O\]
done
clear
C)
\[{{C}_{2}}{{H}_{5}}OH+{{H}_{2}}O\]
done
clear
D)
None of these
done
clear
View Answer play_arrow
question_answer 109) In which of the following L.P.G. gas is:
A)
Lower pressure scale
done
clear
B)
Liquid petrolium gas
done
clear
C)
Low pressure gas
done
clear
D)
None of these
done
clear
View Answer play_arrow
question_answer 110) Halogenation of alkanes is a example of:
A)
Nucleophilic substitution
done
clear
B)
Oxidation
done
clear
C)
Electrophilic substitution
done
clear
D)
Free radical substitution
done
clear
View Answer play_arrow
question_answer 111) Which of the molecule of minimum dipole moment?
A)
\[CO\]
done
clear
B)
\[{{H}_{2}}O\]
done
clear
C)
\[HCl\]
done
clear
D)
\[N{{H}_{3}}\]
done
clear
View Answer play_arrow
question_answer 112) Compound formed by \[ds{{p}^{2}}\]hybridisation will have configuration:
A)
Octahedral
done
clear
B)
Tetrahedral
done
clear
C)
Square planar
done
clear
D)
Trigonal
done
clear
View Answer play_arrow
question_answer 113) Which of the following colour give methyl orange indicator in acidic medium:
A)
Red
done
clear
B)
Yellow
done
clear
C)
Blue
done
clear
D)
Colourless
done
clear
View Answer play_arrow
question_answer 114) The correct relationship between \[{{K}_{P}}\] and \[{{K}_{c}}\] in gaseous equilibrium is:
A)
\[{{K}_{p}}={{K}_{p}}{{(RT)}^{\Delta n}}\]
done
clear
B)
\[{{K}_{c}}=RT{{({{K}_{p}})}^{\Delta n}}\]
done
clear
C)
\[{{K}_{p}}={{K}_{c}}{{(RT)}^{\Delta n}}\]
done
clear
D)
\[{{K}_{p}}=RT{{({{K}_{c}})}^{\Delta n}}\]
done
clear
View Answer play_arrow
question_answer 115) Which species should be least likely to act as a Lewis base?
A)
\[C{{N}^{-}}\]
done
clear
B)
\[PC{{l}_{3}}\]
done
clear
C)
\[{{I}^{+}}\]
done
clear
D)
\[SC{{l}_{2}}\]
done
clear
View Answer play_arrow
question_answer 116) Optical isomerism arises due to the presence of:
A)
a plane of symmetry
done
clear
B)
a centre of symmetry
done
clear
C)
a line symmetry
done
clear
D)
an asymmetric carbon atom
done
clear
View Answer play_arrow
question_answer 117) The dissociation constants of two acid AB and \[A{{B}_{2}}\]are \[3.4\times {{10}^{-4}}\]and \[1.8\times {{10}^{5}}\] respectively. The relative strengths of the acids will approximately be:
A)
\[16:1\]
done
clear
B)
\[1:4\]
done
clear
C)
\[1:16\]
done
clear
D)
\[4:1\]
done
clear
View Answer play_arrow
question_answer 118) Which of the following statements is correct?
A)
A small anion undergoes a high degree of polarisation
done
clear
B)
Small sized of cation minimizes the polarisation
done
clear
C)
A small anion can cause large degree of polarisation
done
clear
D)
Polarisation of an anion is maximum by high charged cation.
done
clear
View Answer play_arrow
question_answer 119) Baking soda is:
A)
\[NaHC{{O}_{3}}\]
done
clear
B)
\[NaN{{O}_{3}}\]
done
clear
C)
\[NaOH\]
done
clear
D)
\[N{{a}_{2}}C{{O}_{3}}.10{{H}_{2}}O\]
done
clear
View Answer play_arrow
question_answer 120) In which of the following chemical reduction is not possible?
A)
Hematite change into iron
done
clear
B)
cupratite change into copper
done
clear
C)
Bauxite change into aluminium
done
clear
D)
Zinc oxide change into zinc
done
clear
View Answer play_arrow
question_answer 121) For hypothetical equlibrium \[4A+5B4X+6Y\] The unit of \[{{K}_{c}}\] will be:
A)
\[litre\text{ }mol{{e}^{-1}}\]
done
clear
B)
\[mole\,\,litr{{e}^{-1}}\]
done
clear
C)
\[litre\text{ }mol{{e}^{-2}}\]
done
clear
D)
\[mol{{e}^{2}}\text{ }litr{{e}^{-2}}\]
done
clear
View Answer play_arrow
question_answer 122) In the following sequence of reaction the end product is: \[HC\equiv CH\xrightarrow[{{H}_{2}}S{{O}_{4}}]{H{{g}^{2+}}}A\xrightarrow[{{H}_{2}}O]{C{{H}_{3}}MgX}B\xrightarrow{O}C\]
A)
\[C{{H}_{3}}COC{{H}_{3}}\]
done
clear
B)
\[C{{H}_{3}}CHO\]
done
clear
C)
\[{{C}_{2}}{{H}_{5}}OH\]
done
clear
D)
\[C{{H}_{3}}C{{H}_{2}}C{{H}_{2}}OH\]
done
clear
View Answer play_arrow
question_answer 123) Hybridisation of carbon in \[-CHO\] group is:
A)
\[s{{p}^{3}}\]
done
clear
B)
\[sp\]
done
clear
C)
\[s{{p}^{2}}\]
done
clear
D)
none of these
done
clear
View Answer play_arrow
question_answer 124) The boiling point of \[{{C}_{2}}{{H}_{5}}OH\] is much higher than that of dimethyl ether, though both have the same molecular weight The reason for this is:
A)
Dipole moment of \[{{C}_{2}}{{H}_{5}}OH\]is less
done
clear
B)
Ether is insoluble in water
done
clear
C)
\[{{C}_{2}}{{H}_{5}}OH\]Shows hydrogen bonding
done
clear
D)
Methyl group are attached to oxygen in ether
done
clear
View Answer play_arrow
question_answer 125) The starch change into glucose from this enzyme:
A)
amylase
done
clear
B)
invertase
done
clear
C)
zymase
done
clear
D)
urease
done
clear
View Answer play_arrow
question_answer 126)
A)
\[CH=CH\]
done
clear
B)
\[{{C}_{6}}{{H}_{5}}N{{H}_{2}}\]
done
clear
C)
\[{{H}_{6}}{{H}_{5}}Cl\]
done
clear
D)
phenyl isocyanide
done
clear
View Answer play_arrow
question_answer 127) When \[{{C}_{6}}{{H}_{5}}N{{H}_{2}}\] heated with \[{{C}_{6}}{{H}_{5}}CHO\] the product is:
A)
schiffs base
done
clear
B)
benzoin
done
clear
C)
azoxy benzene
done
clear
D)
unsaturated acid
done
clear
View Answer play_arrow
question_answer 128) In which of the following dry ice is:
A)
solid \[{{H}_{2}}O\]
done
clear
B)
solid \[C{{O}_{2}}\]
done
clear
C)
both (a) and (b)
done
clear
D)
none of these
done
clear
View Answer play_arrow
question_answer 129) Nitration of benzene followed by reduction with \[(Sn+HCl)\] will give:
A)
\[{{C}_{6}}{{H}_{5}}OH\]
done
clear
B)
\[{{C}_{6}}{{H}_{5}}N{{H}_{2}}\]
done
clear
C)
\[{{C}_{6}}{{H}_{5}}N{{O}_{2}}\]
done
clear
D)
\[{{C}_{6}}{{H}_{5}}CN\]
done
clear
View Answer play_arrow
question_answer 130) Which of the following will have least hindered rotation about carbon-carbon?
A)
\[{{C}_{2}}{{H}_{5}}\]
done
clear
B)
\[C{{H}_{2}}=C{{H}_{2}}\]
done
clear
C)
\[CH\equiv CH\]
done
clear
D)
Hexa chloro ethane
done
clear
View Answer play_arrow
question_answer 131) \[{{C}_{6}}{{H}_{5}}N{{H}_{2}}+HN{{O}_{2}}\xrightarrow{(NaN{{O}_{2}}/HCl)}X\] Product X is:
A)
nitrobenzene
done
clear
B)
benzene diazonium chloride
done
clear
C)
phenol
done
clear
D)
ethyl alcohol
done
clear
View Answer play_arrow
question_answer 132) \[A+{{C}_{6}}{{H}_{5}}COCl\xrightarrow[\text{(Priedel}\,\text{Craft}\,\text{reaction)}]{Anhy\,AlC{{l}_{3}}}\] \[{{C}_{6}}{{H}_{5}}CO{{C}_{6}}{{H}_{5}}\]
A)
\[{{C}_{6}}{{H}_{6}}\]
done
clear
B)
\[C{{H}_{3}}-C=C-C{{H}_{3}}\]
done
clear
C)
\[C{{H}_{3}}CH=C{{H}_{2}}\]
done
clear
D)
\[CH=CH\]
done
clear
View Answer play_arrow
question_answer 133) When excess of ethyl alcohol heated at \[{{140}^{o}}C\]with concentrate sulphuric acid the compound that distills is:
A)
\[{{C}_{2}}{{H}_{5}}-O-{{C}_{2}}{{H}_{5}}\]
done
clear
B)
ethyl hydrogen sulphate
done
clear
C)
\[C{{H}_{2}}=C{{H}_{2}}\]
done
clear
D)
di ethyl sulphate
done
clear
View Answer play_arrow
question_answer 134) Acetamide and urea can be distinguished by employing:
A)
Biurete test
done
clear
B)
Lucas reagent
done
clear
C)
Victor Maeyers test
done
clear
D)
All of these
done
clear
View Answer play_arrow
question_answer 135) When aniline react with \[HN{{O}_{2}}\] \[(NaN{{O}_{2}}+HCl)\] formed diazonium chloride which react \[{{H}_{3}}P{{O}_{2}}\] gives:
A)
\[CH=CH\]
done
clear
B)
\[{{C}_{6}}{{H}_{6}}\]
done
clear
C)
\[C{{H}_{2}}=C{{H}_{2}}\]
done
clear
D)
\[C{{H}_{3}}-C{{H}_{3}}\]
done
clear
View Answer play_arrow
question_answer 136) Paraldehyde is:
A)
a trimer of \[HCHO\]
done
clear
B)
a trimer of \[C{{H}_{3}}CHO\]
done
clear
C)
a hexamer of \[HCHO\]
done
clear
D)
a hexamer of \[C{{H}_{3}}CHO\]
done
clear
View Answer play_arrow
question_answer 137) Phosgene is the common name for:
A)
\[CC{{l}_{4}}\]
done
clear
B)
\[C{{O}_{2}}+P{{H}_{3}}\]
done
clear
C)
phosphoryl chloride
done
clear
D)
carbonyl chloride
done
clear
View Answer play_arrow
question_answer 138) Which of the following reagent is used to distinguish between \[C{{H}_{3}}CHO\]and \[C{{H}_{3}}COC{{H}_{3}}\]?
A)
Semicarbazide
done
clear
B)
Phenyl hydrazine
done
clear
C)
Hydroxy amine
done
clear
D)
Ammonical \[AgN{{O}_{3}}\]
done
clear
View Answer play_arrow
question_answer 139) If chloroform is left open in air in presence of sun-rays:
A)
phosgene gas is formed
done
clear
B)
explosion takes place
done
clear
C)
polymerisation take place
done
clear
D)
no reaction take place
done
clear
View Answer play_arrow
question_answer 140) Which of the following is used to distinguish between acetamide and ammonium acetate?
A)
Reaction with \[FeC{{l}_{3}}\]
done
clear
B)
Reaction with Fehling solution
done
clear
C)
Reaction with \[~NaOH\]
done
clear
D)
None of these
done
clear
View Answer play_arrow
question_answer 141) Which of the following is the weakest base?
A)
\[N{{H}_{3}}\]
done
clear
B)
\[{{(C{{H}_{3}})}_{2}}NH\]
done
clear
C)
\[C{{H}_{3}}N{{H}_{2}}\]
done
clear
D)
\[{{(C{{H}_{3}})}_{3}}N\]
done
clear
View Answer play_arrow
question_answer 142) \[{{C}_{6}}{{H}_{5}}N{{H}_{2}}\xrightarrow[{{0}^{o}}-{{5}^{o}}C]{NaN{{O}_{2}}/HCl}{{C}_{6}}{{H}_{5}}{{\overset{+}{\mathop{N}}\,}_{2}}Cl\] \[\xrightarrow[CuS{{O}_{4}}]{KCN}A\] product A is:
A)
\[{{C}_{6}}{{H}_{5}}OH\]
done
clear
B)
\[{{C}_{6}}{{H}_{5}}HS{{O}_{3}}\]
done
clear
C)
\[{{C}_{6}}{{H}_{5}}CN\]
done
clear
D)
\[{{C}_{6}}{{H}_{5}}Cl\]
done
clear
View Answer play_arrow
question_answer 143) The bond present in methane is:
A)
\[4\sigma \] bond and zero \[\pi \] bond
done
clear
B)
\[4\pi \] bond \[4\pi \] bond
done
clear
C)
zero \[\sigma \] bond \[4\pi \] bond
done
clear
D)
none of these
done
clear
View Answer play_arrow
question_answer 144) The possible isomer of \[{{C}_{3}}{{H}_{6}}O\] is:
A)
\[2\]
done
clear
B)
\[1\]
done
clear
C)
\[3\]
done
clear
D)
\[8\]
done
clear
View Answer play_arrow
question_answer 145) Which of the following passage in \[C{{O}_{2}}\] whose does milky of solution?
A)
\[CaO\]
done
clear
B)
\[Ca{{(OH)}_{2}}\]
done
clear
C)
\[CaC{{O}_{3}}\]
done
clear
D)
None of these
done
clear
View Answer play_arrow
question_answer 146) Which electronic configuration of neutral atom will have the highest first potential:
A)
\[1{{s}^{2}},2{{s}^{2}}\text{ }2{{p}^{4}}\]
done
clear
B)
\[1{{s}^{2}},2{{s}^{2}}\text{ }2{{p}^{1}}\]
done
clear
C)
\[1{{s}^{2}},2{{s}^{2}}\text{ }2{{p}^{3}}\]
done
clear
D)
\[1{{s}^{2}},2{{s}^{2}}\text{ }2{{p}^{2}}\]
done
clear
View Answer play_arrow
question_answer 147) Acid group is:
A)
\[O-\overset{O}{\mathop{\overset{||}{\mathop{C}}\,}}\,-H\]
done
clear
B)
done
clear
C)
\[H-C=O\]
done
clear
D)
none of these
done
clear
View Answer play_arrow
question_answer 148) The Cannizzaros reaction is not given by:
A)
\[{{C}_{6}}{{H}_{5}}OH\]
done
clear
B)
\[HCHO\]
done
clear
C)
\[C{{H}_{3}}CHO\]
done
clear
D)
trimethyl aldehyde
done
clear
View Answer play_arrow
question_answer 149) Maximum paramagnetism in 3d series is shown by:
A)
\[Co\]
done
clear
B)
\[Fe\]
done
clear
C)
\[Ni\]
done
clear
D)
\[Mn\]
done
clear
View Answer play_arrow
question_answer 150) At \[{{90}^{o}}C\]pure water has \[[{{H}_{3}}{{O}^{+}}]={{10}^{-6}}\] \[mole\text{ }litr{{e}^{-1}},\]the value of \[{{K}_{\omega }}\] at \[{{90}^{o}}C\]is:
A)
\[{{10}^{-14}}\]
done
clear
B)
\[{{10}^{-12}}\]
done
clear
C)
\[{{10}^{-8}}\]
done
clear
D)
\[{{10}^{-6}}\]
done
clear
View Answer play_arrow
question_answer 151) Electron occupy the available sub level which has lower \[(n+l)\] value. This is called:
A)
Aufbau rule
done
clear
B)
Faults exclusion principle
done
clear
C)
Hunds rule
done
clear
D)
Heisenberg uncertainty principle
done
clear
View Answer play_arrow
question_answer 152) The oxidation number of \[Cr\] in \[{{K}_{2}}C{{r}_{2}}{{O}_{7}}\] is:
A)
\[+6\]
done
clear
B)
\[-6\]
done
clear
C)
\[+4\]
done
clear
D)
\[-4\]
done
clear
View Answer play_arrow
question_answer 153) Which represents nitrolium?
A)
\[CaC{{N}_{2}}+C\]
done
clear
B)
\[Ca{{C}_{2}}+{{N}_{2}}\]
done
clear
C)
\[Ca{{(CN)}_{2}}+Ca{{(N{{O}_{3}})}_{2}}\]
done
clear
D)
None of these
done
clear
View Answer play_arrow
question_answer 154) For \[n=3\]the value of I and m respectively are:
A)
\[3,9\]
done
clear
B)
\[3,6\]
done
clear
C)
\[9,3\]
done
clear
D)
\[6,3\]
done
clear
View Answer play_arrow
question_answer 155) The solvent which is neither proton donor nor proton accepter is called:
A)
protonic
done
clear
B)
aprotic
done
clear
C)
amphoteric
done
clear
D)
neutral
done
clear
View Answer play_arrow
question_answer 156) Which is not permissible sub shell:
A)
\[3s\]
done
clear
B)
\[4f\]
done
clear
C)
\[6p\]
done
clear
D)
\[2d\]
done
clear
View Answer play_arrow
question_answer 157) Which have high penetrating power?
A)
\[\alpha \] rays
done
clear
B)
\[\beta \] rays
done
clear
C)
\[\gamma \] rays
done
clear
D)
none of these
done
clear
View Answer play_arrow
question_answer 158) Alkali metals are characterized by:
A)
high melting points
done
clear
B)
high ionization potential
done
clear
C)
low oxidation potential
done
clear
D)
good conductor of heat and electricity
done
clear
View Answer play_arrow
question_answer 159) How many \[\pi \] bond are there in \[C{{O}_{2}}\]molecule?
A)
\[2\pi \] bond
done
clear
B)
\[4\pi \] bond
done
clear
C)
\[3\pi \]bond
done
clear
D)
zero
done
clear
View Answer play_arrow
question_answer 160) In benzene \[C-C\]bond length than acetylene will be:
A)
higher
done
clear
B)
lower
done
clear
C)
middle
done
clear
D)
none of these
done
clear
View Answer play_arrow
question_answer 161) \[[{{H}^{+}}]\] ion concentration in water is:
A)
\[0\]
done
clear
B)
\[1\times {{10}^{-7}}\]
done
clear
C)
\[7\]
done
clear
D)
\[1/4\]
done
clear
View Answer play_arrow
question_answer 162) Which of the following is paramagnetic?
A)
\[{{O}_{2}}^{-}\]
done
clear
B)
\[C{{H}_{3}}\]
done
clear
C)
\[CO\]
done
clear
D)
\[N{{O}^{+}}\]
done
clear
View Answer play_arrow
question_answer 163) The IUPAC name of \[C{{H}_{3}}-\overset{C{{H}_{2}}}{\mathop{\overset{|}{\mathop{C}}\,}}\,H-C{{H}_{2}}-\overset{Cl}{\mathop{\overset{|}{\mathop{C}}\,}}\,H-\overset{O}{\mathop{\overset{||}{\mathop{C}}\,}}\,-C{{H}_{3}}\]is:
A)
3-chloro-5-methyl 2-hexanon
done
clear
B)
5-methyl -3 chloro 2-hexanon
done
clear
C)
both (a) and (b)
done
clear
D)
none of these
done
clear
View Answer play_arrow
question_answer 164) Select the correct order of the strength of acids given below:
A)
\[HCl{{O}_{4}}<HCl{{O}_{3}}<HClO<HCl{{O}_{2}}\]
done
clear
B)
\[HCl{{O}_{4}}<HCl{{O}_{3}}<HCl{{O}_{2}}<HClO\]
done
clear
C)
\[HClO<HCl{{O}_{2}}<HCl{{O}_{3}}<HCl{{O}_{4}}\]
done
clear
D)
none of these
done
clear
View Answer play_arrow
question_answer 165) Effect of catalyst in any reaction is:
A)
the rate of reaction increases
done
clear
B)
reactant increases
done
clear
C)
product increases
done
clear
D)
none of these
done
clear
View Answer play_arrow
question_answer 166) Which of the following has fractional bond order?
A)
\[{{H}_{2}}^{2+}\]
done
clear
B)
\[{{O}_{2}}^{2-}\]
done
clear
C)
\[{{O}_{2}}^{2+}\]
done
clear
D)
\[{{F}_{2}}^{2-}\]
done
clear
View Answer play_arrow
question_answer 167) Which of the base of \[C{{H}_{3}}COOH\]?
A)
\[C{{H}_{3}}CO{{O}^{-}}\]
done
clear
B)
\[C{{H}_{3}}COOH\]
done
clear
C)
both (a) and (b)
done
clear
D)
None of these
done
clear
View Answer play_arrow
question_answer 168) \[2\text{ }moles\]of \[PC{{l}_{5}}\] were heated in a closed vessels of 2 litre capacity at equilibrium 40% of \[PC{{l}_{5}}\] dissociated in \[PC{{l}_{3}}\] and \[C{{l}_{2}}\]. The value of the equilibrium constant is:
A)
\[2.67\]
done
clear
B)
\[5.3\]
done
clear
C)
\[0.53\]
done
clear
D)
\[0.267\]
done
clear
View Answer play_arrow
question_answer 169) \[\pi \] bond in \[{{N}_{2}}\] is:
A)
\[2\]
done
clear
B)
\[3\]
done
clear
C)
\[1\]
done
clear
D)
zero
done
clear
View Answer play_arrow
question_answer 170) The screening effect of d-electrons is:
A)
less than p-electron
done
clear
B)
more than p-electron
done
clear
C)
equal to the p-electron
done
clear
D)
same as p-electron
done
clear
View Answer play_arrow
question_answer 171) The purpose of smelting of an ore is:
A)
to separate volatile impurities
done
clear
B)
to reduce it
done
clear
C)
to oxidize it
done
clear
D)
to obtain an alloy
done
clear
View Answer play_arrow
question_answer 172) When \[FeC{{l}_{3}}\] react with acetate which colour is obtained?
A)
Blood colour
done
clear
B)
Colorless
done
clear
C)
Red
done
clear
D)
Yellow
done
clear
View Answer play_arrow
question_answer 173) One among the following is incorrect order in increasing ionisation energy:
A)
\[Ka<Ca<Cs\]
done
clear
B)
\[Au<Ag<Cu\]
done
clear
C)
\[C{{l}^{-}}<Ar<{{K}^{+}}\]
done
clear
D)
\[Cs<RB<K\]
done
clear
View Answer play_arrow
question_answer 174) The equilibrium constants for equilibria: \[S{{O}_{2}}(g)+\frac{1}{2}{{O}_{2}}(g)S{{O}_{3}}(g)\] and \[2S{{O}_{2}}(g)+{{O}_{2}}(g)2S{{O}_{3}}(g)\] are \[{{K}_{1}}\] and \[{{K}_{2}}\] respectively, then:
A)
\[{{K}_{2}}={{K}_{1}}^{2}\]
done
clear
B)
\[{{K}_{2}}=\frac{1}{{{K}_{{{1}^{2}}}}}\]
done
clear
C)
\[{{K}_{2}}={{K}_{1}}\]
done
clear
D)
\[{{K}_{2}}=\frac{1}{{{K}_{1}}}\]
done
clear
View Answer play_arrow
question_answer 175) The bond angle in \[P{{H}_{3}}\] is:
A)
slightly more than \[N{{H}_{3}}\]
done
clear
B)
much greater than \[N{{H}_{3}}\]
done
clear
C)
equal to that of \[N{{H}_{3}}\]
done
clear
D)
much less than \[N{{H}_{3}}\]
done
clear
View Answer play_arrow
question_answer 176) Of the solubility product \[{{K}_{sp}}\] of a sparingly soluble salt \[A{{X}_{2}}\] at \[{{25}^{o}}C\]is \[1\times {{10}^{-11}}\]. The solubility of the salt in mole \[litr{{e}^{-1}}\]at this temperature will be:
A)
\[1.20\times {{10}^{-10}}\]
done
clear
B)
\[1.36\times {{10}^{-4}}\]
done
clear
C)
\[2.60\times {{10}^{-7}}\]
done
clear
D)
\[2.46\times {{10}^{14}}\]
done
clear
View Answer play_arrow
question_answer 177) In acidic medium phenolphthalien gives:
A)
red colour
done
clear
B)
yellow colour
done
clear
C)
colorless
done
clear
D)
blue colour
done
clear
View Answer play_arrow
question_answer 178) Heavy water is:
A)
\[{{H}_{2}}O\]
done
clear
B)
\[{{D}_{2}}O\]
done
clear
C)
both (a) and (b)
done
clear
D)
none of these
done
clear
View Answer play_arrow
question_answer 179) Hydrogen can be placed in halogen group because:
A)
It contains one electron only
done
clear
B)
It forms hydrides like chloride
done
clear
C)
It is light
done
clear
D)
It has isotopes D and T
done
clear
View Answer play_arrow
question_answer 180) The correct representation of solubility product of \[Sn{{S}_{2}}\] is:
A)
\[[S{{n}^{4+}}]\,[2{{S}^{2-}}]\]
done
clear
B)
\[[S{{n}^{4+}}]\,[{{S}^{2-}}]\]
done
clear
C)
\[[S{{n}^{4+}}]\,{{[2{{S}^{2+}}]}^{2}}\]
done
clear
D)
\[[S{{n}^{4+}}]\,{{[{{S}^{2-}}]}^{2}}\]
done
clear
View Answer play_arrow
question_answer 181) The minimum real charge on any particle which can exist is:
A)
\[4.8\times {{10}^{-10}}\]coulomb
done
clear
B)
\[1.6\times {{10}^{-19}}\]coulomb
done
clear
C)
\[1.6\times {{10}^{-10}}\]coulomb
done
clear
D)
zero
done
clear
View Answer play_arrow
question_answer 182) Hydrolysis of product which formed reaction between ketone and G.R. gives:
A)
\[(C{{H}_{3}})CHO\]
done
clear
B)
\[{{C}_{2}}{{H}_{5}}OH\]
done
clear
C)
\[C{{H}_{3}}OH\]
done
clear
D)
none of these
done
clear
View Answer play_arrow
question_answer 183) The first organic compound urea was synthesised by in the laboratory:
A)
Berzilius
done
clear
B)
Wohler
done
clear
C)
Kolbe
done
clear
D)
None of these
done
clear
View Answer play_arrow
question_answer 184) In d-subshell electron is:
A)
less than p
done
clear
B)
equal to p
done
clear
C)
more than s
done
clear
D)
more than f
done
clear
View Answer play_arrow
question_answer 185)
When
A)
\[C{{H}_{3}}COO{{C}_{2}}{{H}_{5}}\]
done
clear
B)
\[C{{H}_{3}}COOH\]
done
clear
C)
\[{{C}_{2}}{{H}_{5}}OH\]
done
clear
D)
None of these
done
clear
View Answer play_arrow
question_answer 186) Diethyl ether decomposes on heating with:
A)
\[{{H}_{2}}O\]
done
clear
B)
\[KMn{{O}_{4}}\]
done
clear
C)
\[HI\]
done
clear
D)
\[NaOH\]
done
clear
View Answer play_arrow
question_answer 187) Alkynes shows:
A)
substitution reaction
done
clear
B)
polymerization
done
clear
C)
electrophilic addition reaction
done
clear
D)
all of the above
done
clear
View Answer play_arrow
question_answer 188) The hydrogen ion concentration in a solution of weak acid of dissociation constant \[{{K}_{a}}\] and concentration C is nearly equal to:
A)
\[{{K}_{a}}.C\]
done
clear
B)
\[C/{{K}_{a}}\]
done
clear
C)
\[\sqrt{{{K}_{a}}.C}\]
done
clear
D)
\[\sqrt{{{K}_{a}}/C}\]
done
clear
View Answer play_arrow
question_answer 189) Alkaline earth metals salts are:
A)
paramagnetic
done
clear
B)
diamagnetic
done
clear
C)
ferromagnetic
done
clear
D)
all of these
done
clear
View Answer play_arrow
question_answer 190) The weakest acid among the following is:
A)
\[HF\]
done
clear
B)
\[HBr\]
done
clear
C)
\[HI\]
done
clear
D)
\[HCl\]
done
clear
View Answer play_arrow
question_answer 191) Magnetic quantum number specifies:
A)
orientation of orbitals in space
done
clear
B)
size of orbitals
done
clear
C)
shape of orbitals
done
clear
D)
spin of the electron
done
clear
View Answer play_arrow
question_answer 192) Which of the following is less soluble in water?
A)
\[Bi{{S}_{3}}(Ksp=1\times {{10}^{-70}})\]
done
clear
B)
\[A{{g}_{2}}S(Ksp=6\times {{10}^{-51}})\]
done
clear
C)
\[ZnS(Ksp=7\times {{10}^{-16}})\]
done
clear
D)
\[MnS(KSP=8\times {{10}^{-37}})\]
done
clear
View Answer play_arrow
question_answer 193) Rusting of iron m moist air involves:
A)
loss of electron by \[Fe\]
done
clear
B)
gain of electron by \[Fe\]
done
clear
C)
hydration of \[Fe\]
done
clear
D)
neither gain nor loss electron by \[Fe\]
done
clear
View Answer play_arrow
question_answer 194) The mass of neutron is of the order of:
A)
\[{{10}^{-27}}kg\]
done
clear
B)
\[{{10}^{-26}}kg\]
done
clear
C)
\[{{10}^{-24}}kg\]
done
clear
D)
\[{{10}^{-23}}kg\]
done
clear
View Answer play_arrow
question_answer 195) The isomerism exhibited by n-butyl alcohol and isobutyl alcohol is:
A)
metamerism
done
clear
B)
position isomerism
done
clear
C)
chain isomerism
done
clear
D)
functional isomerism
done
clear
View Answer play_arrow
question_answer 196)
A)
\[C{{H}_{3}}CO{{C}_{2}}{{H}_{5}}\]
done
clear
B)
\[C{{H}_{3}}COOC{{H}_{3}}\]
done
clear
C)
\[C{{H}_{3}}C{{H}_{2}}COO{{C}_{2}}{{H}_{5}}\]
done
clear
D)
\[C{{H}_{2}}C{{H}_{2}}COOH\]
done
clear
View Answer play_arrow
question_answer 197) \[X\xrightarrow[{{O}_{3}}]{Ozonolysis}C{{H}_{3}}CHO+C{{H}_{3}}C{{H}_{2}}CHO.\] X is:
A)
2-butene
done
clear
B)
2-pentene
done
clear
C)
1-pentene
done
clear
D)
1-butene
done
clear
View Answer play_arrow
question_answer 198) Urea is not used:
A)
in the preparation of medicines
done
clear
B)
in purification of water
done
clear
C)
as fertilizer
done
clear
D)
in manufacture of plastic
done
clear
View Answer play_arrow
question_answer 199) Which pair represents chain isomers?
A)
2-methyl butane and neopentane
done
clear
B)
\[C{{H}_{3}}CHC{{l}_{2}}\]and \[ClC{{H}_{2}}C{{H}_{2}}Cl\]
done
clear
C)
Dethyl ether and propyl ether
done
clear
D)
Propyl alcohol and isopropyl alcohol
done
clear
View Answer play_arrow
question_answer 200) Hydrogen bromide is added to \[C{{H}_{3}}CH=C{{H}_{2}}\]in the presence of peroxide. The resultant compound formed is:
A)
\[C{{H}_{3}}C{{H}_{2}}C{{H}_{2}}Br\]
done
clear
B)
\[C{{H}_{3}}CHBr.C{{H}_{3}}\]
done
clear
C)
\[{{C}_{2}}{{H}_{5}}C{{H}_{2}}Br\]
done
clear
D)
none of these
done
clear
View Answer play_arrow
question_answer 201) Giraffe is common animal of:
A)
desert
done
clear
B)
savannah
done
clear
C)
rain forest
done
clear
D)
praire
done
clear
View Answer play_arrow
question_answer 202) Conversion of PEP to pyruvate is influenced by:
A)
\[M{{g}^{+++\,}}\]\[C{{a}^{+\,}}\]
done
clear
B)
\[N{{a}^{++\,}}+C{{a}^{++}}\]
done
clear
C)
\[{{K}^{+\,}}+C{{1}^{-}}\]
done
clear
D)
\[M{{g}^{++\,}}+{{K}^{+}}\]
done
clear
View Answer play_arrow
question_answer 203) Dihydroxy acetone is formed by catalysis of the following enzymes :
A)
aldolase
done
clear
B)
PFK
done
clear
C)
PEPCK
done
clear
D)
phosphoglucoisomerase
done
clear
View Answer play_arrow
question_answer 204) Which one have lophophore?
A)
Waldeimia
done
clear
B)
Wallago
done
clear
C)
Wall lizard
done
clear
D)
Heart urchin
done
clear
View Answer play_arrow
question_answer 205) Which animal is roofed terrapin?
A)
Trionys
done
clear
B)
Kachuga tectum
done
clear
C)
Chrysemys
done
clear
D)
Chelone
done
clear
View Answer play_arrow
question_answer 206) In man erythrocytes are found in:
A)
0.5 million per microliter
done
clear
B)
8.0 million per microliter
done
clear
C)
5.0 million per microliter
done
clear
D)
10-0 million per microliter
done
clear
View Answer play_arrow
question_answer 207) Which one of the following acts on local messenger?
A)
Carrier protein
done
clear
B)
Glycoprotein
done
clear
C)
Phospholipid
done
clear
D)
Nitric oxide
done
clear
View Answer play_arrow
question_answer 208) Oxytocin is released from :
A)
hypothalamus
done
clear
B)
adenohypophysis (posterior lobe)
done
clear
C)
adenohypophysis (anterior lobe)
done
clear
D)
neurohypophysis
done
clear
View Answer play_arrow
question_answer 209) What is the molecular weight to insulin?
A)
6000
done
clear
B)
5000
done
clear
C)
4000
done
clear
D)
15000
done
clear
View Answer play_arrow
question_answer 210) Parathormone controls :
A)
calcium and phosphate metabolism
done
clear
B)
Na and K metabolism
done
clear
C)
fatty acid metabolism
done
clear
D)
protein metabolism
done
clear
View Answer play_arrow
question_answer 211) Which one is detritus feeder?
A)
Parrot
done
clear
B)
Unio
done
clear
C)
Dung beetle
done
clear
D)
Sheep
done
clear
View Answer play_arrow
question_answer 212) Cortical granules are associated with :
A)
fertilization
done
clear
B)
sperma to genesis
done
clear
C)
oogenes is
done
clear
D)
cleavage
done
clear
View Answer play_arrow
question_answer 213) Which of the following type of egg is found in Frog?
A)
Isolecithal
done
clear
B)
Centrolecithal
done
clear
C)
Oligolecithal
done
clear
D)
Teleolecithal
done
clear
View Answer play_arrow
question_answer 214) The number of eggs in ootheca of Cockroach is :
A)
8
done
clear
B)
16
done
clear
C)
32
done
clear
D)
64
done
clear
View Answer play_arrow
question_answer 215) Mentomeckelian is specially characteristic bone of:
A)
Raw tigrina
done
clear
B)
Equns
done
clear
C)
Bos indicus
done
clear
D)
Felis domesticus
done
clear
View Answer play_arrow
question_answer 216) Which one of the following animal contains idiophores in its integument?
A)
Dog fish
done
clear
B)
Parrot
done
clear
C)
Siren
done
clear
D)
Orangeutan
done
clear
View Answer play_arrow
question_answer 217) In both chordates and non-choradates intestine develops from :
A)
ectoderm
done
clear
B)
pharyngeal pouch
done
clear
C)
endoderm
done
clear
D)
mesoderm
done
clear
View Answer play_arrow
question_answer 218) In which of the following mosaic cleavage occurs?
A)
Echinococcus granulosus
done
clear
B)
Camel
done
clear
C)
Whale
done
clear
D)
Dog fish
done
clear
View Answer play_arrow
question_answer 219) In which one moeroblastic segmentation is found:
A)
Dog
done
clear
B)
Monkey
done
clear
C)
Owl
done
clear
D)
Rhinoceros
done
clear
View Answer play_arrow
question_answer 220) Km is related to :
A)
morphometry
done
clear
B)
ABO blood group
done
clear
C)
ES complex
done
clear
D)
chroma tography
done
clear
View Answer play_arrow
question_answer 221) Platelet plug is associated with :
A)
hcmostasis
done
clear
B)
clot retraction
done
clear
C)
fibrin formation
done
clear
D)
homcostasis
done
clear
View Answer play_arrow
question_answer 222) In Rabbit what is the number of spinal nerves?
A)
37
done
clear
B)
30
done
clear
C)
32
done
clear
D)
38
done
clear
View Answer play_arrow
question_answer 223) Which one of the following mouth parts arc found in Cockroach?
A)
Cutting and chewing
done
clear
B)
Piercing
done
clear
C)
Sucking
done
clear
D)
Drilling
done
clear
View Answer play_arrow
question_answer 224) In Periplaneta the cuticular lipid is secreted by:
A)
hypudermal cells
done
clear
B)
oenocyte cells
done
clear
C)
dermal gland cells
done
clear
D)
basal ceils
done
clear
View Answer play_arrow
question_answer 225) In Pheretima blood glands are found in which of the following segments?
A)
1, 2, 3
done
clear
B)
7, 8, 9
done
clear
C)
4, 5, 6
done
clear
D)
10, 11, 12
done
clear
View Answer play_arrow
question_answer 226) How many lateral hearts are in Pheretima?
A)
12
done
clear
B)
16
done
clear
C)
8
done
clear
D)
4
done
clear
View Answer play_arrow
question_answer 227) Which one of the following is holotype ?
A)
Oesophagostomum apiostomum
done
clear
B)
Osophagostomum columbianum
done
clear
C)
Oesophagostomum radiatum
done
clear
D)
Oesophygostomum venulosm
done
clear
View Answer play_arrow
question_answer 228) Which one of the following is first nematode parasite to be discovered ?
A)
Enterobius vermicularis
done
clear
B)
Ancylostoma duodenale
done
clear
C)
Ascaris lumbicoides
done
clear
D)
Dracunculus medinensis
done
clear
View Answer play_arrow
question_answer 229) Excretory system of Ascaris lumbricoides is made up of:
A)
single cell
done
clear
B)
many cells
done
clear
C)
four cells
done
clear
D)
two cells
done
clear
View Answer play_arrow
question_answer 230) What is the length of male Ascaris lumbricoides is ?
A)
50-60 mm
done
clear
B)
100-200 mm
done
clear
C)
150-310 mm
done
clear
D)
160-315 mm
done
clear
View Answer play_arrow
question_answer 231) Cyclin is associated with which one of the following?
A)
Haemolysis
done
clear
B)
Mitosis
done
clear
C)
Glycolysis
done
clear
D)
Cyclosis
done
clear
View Answer play_arrow
question_answer 232) In which one of the following white ghost are found?
A)
Lymphocytes
done
clear
B)
Thrombocytes
done
clear
C)
Choanocytes
done
clear
D)
Erythrocytes
done
clear
View Answer play_arrow
question_answer 233) Which one of the following is energy supplier ?
A)
GALT
done
clear
B)
MALT
done
clear
C)
MAB
done
clear
D)
UTP
done
clear
View Answer play_arrow
question_answer 234) Which one of the following is pernicious quotidian, malignant and tropical malarial parasite?
A)
Plasmodium vivax
done
clear
B)
Plasmodium falciparum
done
clear
C)
Plasmodium berghei
done
clear
D)
Plasmodium yolii
done
clear
View Answer play_arrow
question_answer 235) Which one of the following is associated with osmoregulation in Amoeba ?
A)
Plasma membrane
done
clear
B)
Contractile vacuole
done
clear
C)
Mitochondria
done
clear
D)
Endoplasm
done
clear
View Answer play_arrow
question_answer 236) In Entamoeba histolytica cytochromes are found in which one of the following?
A)
Ectoplasm
done
clear
B)
Mitochondria
done
clear
C)
Endoplasmic reticulum
done
clear
D)
Endoplasm
done
clear
View Answer play_arrow
question_answer 237) In case of Amoeba proteus which temperature is optimum ?
A)
\[5{}^\circ C\text{ }to\text{ }10{}^\circ C\]
done
clear
B)
\[20{}^\circ C\text{ }to\text{ }25{}^\circ C\]
done
clear
C)
\[15{}^\circ C\text{ }to\text{ }16{}^\circ C\]
done
clear
D)
\[30{}^\circ C\text{ }to\text{ }35{}^\circ C\]
done
clear
View Answer play_arrow
question_answer 238) Apparatus reticulo interno was discovered by Golgi in which year :
A)
1898
done
clear
B)
1850
done
clear
C)
1900
done
clear
D)
1925
done
clear
View Answer play_arrow
question_answer 239) Synaptonemal complex was discovered in :
A)
1955
done
clear
B)
1950
done
clear
C)
1980
done
clear
D)
1935
done
clear
View Answer play_arrow
question_answer 240) How many ATP are required during anaphase to move chromosomes from equator to the poles?
A)
38 ATP
done
clear
B)
5 ATP
done
clear
C)
30 ATP
done
clear
D)
76 ATP
done
clear
View Answer play_arrow
question_answer 241) What is the diameter of stenotypes?
A)
16 microns
done
clear
B)
25 microns
done
clear
C)
10 microns
done
clear
D)
5 microns
done
clear
View Answer play_arrow
question_answer 242) Hydra belongs to which of the following?
A)
Biradially symmetrical
done
clear
B)
Radially symmetrical and coelomate
done
clear
C)
Coelomate and bilaterally symmetrical
done
clear
D)
Radially symmetrical and acoelomate
done
clear
View Answer play_arrow
question_answer 243) In Plasmodium which one of the following is not distinct?
A)
Mitrochondria
done
clear
B)
Golgi body
done
clear
C)
Cilia
done
clear
D)
Nucleus
done
clear
View Answer play_arrow
question_answer 244) Which one of the following is ionic detergent?
A)
PEG
done
clear
B)
HAT
done
clear
C)
SDS
done
clear
D)
RPM
done
clear
View Answer play_arrow
question_answer 245) Which one of the following is dominant intracellular anion?
A)
Chloride
done
clear
B)
Potassium
done
clear
C)
Phosphate
done
clear
D)
Calcium
done
clear
View Answer play_arrow
question_answer 246) Zinc is associated with:
A)
Immunization
done
clear
B)
Glycosylation
done
clear
C)
Sulfation
done
clear
D)
Transcription
done
clear
View Answer play_arrow
question_answer 247) Which one of the following percentage is of bound water in protoplasm ?
A)
1-2%
done
clear
B)
4-5%
done
clear
C)
60-65%
done
clear
D)
90-95%
done
clear
View Answer play_arrow
question_answer 248) Which animal is Surinam toad?
A)
Pipa
done
clear
B)
Bufo
done
clear
C)
Bombimntor
done
clear
D)
Alyte
done
clear
View Answer play_arrow
question_answer 249) Membrane attack complex (MAC) is formed by :
A)
B-lymphocytes
done
clear
B)
compliments
done
clear
C)
macrophages
done
clear
D)
T-lymphocytes
done
clear
View Answer play_arrow
question_answer 250) Which animal is eukaryotic unicellular?
A)
Synapta
done
clear
B)
Sun animalcule
done
clear
C)
Mitre-Jelly fish
done
clear
D)
Sea lemon
done
clear
View Answer play_arrow
question_answer 251) In which of the following class polymorphism is very distinct?
A)
Class-Euccstoda
done
clear
B)
Class-Hydrozoa
done
clear
C)
Class-Crinoidea
done
clear
D)
Class-Gastropoda
done
clear
View Answer play_arrow
question_answer 252) Which of the following annelid is most (modified?
A)
Arenicola
done
clear
B)
Chaetopterus
done
clear
C)
Pontobdella
done
clear
D)
Hirudomedicmalis
done
clear
View Answer play_arrow
question_answer 253) Which one of the following character (differentiate man from other mammals?
A)
Social life
done
clear
B)
Greater cranial capacity
done
clear
C)
Superior intelligence
done
clear
D)
Power of speech
done
clear
View Answer play_arrow
question_answer 254) Which one of the following animal is protostome?
A)
Aconworm
done
clear
B)
Sping skinned animal
done
clear
C)
Amphitrehus
done
clear
D)
(e) Echidna
done
clear
View Answer play_arrow
question_answer 255) Which one of the following animal is not deuterostome?
A)
Ambystoma
done
clear
B)
Ambhilina
done
clear
C)
Anguilla
done
clear
D)
Amphiuma
done
clear
View Answer play_arrow
question_answer 256) From substrate level phosphortyation ATP are produced :
A)
2
done
clear
B)
6
done
clear
C)
10
done
clear
D)
8
done
clear
View Answer play_arrow
question_answer 257) Which pollutant accumulates in liver and kidney
A)
Cadmium
done
clear
B)
Lead
done
clear
C)
Mercury
done
clear
D)
Copper
done
clear
View Answer play_arrow
question_answer 258) Which one is a commensalism?
A)
Euspongia
done
clear
B)
Jelly fish
done
clear
C)
Silkworm
done
clear
D)
Bonellia female
done
clear
View Answer play_arrow
question_answer 259) Norepinephrin is secreted from:
A)
medulla of adrenal
done
clear
B)
zona fasciculate
done
clear
C)
zona reticularis
done
clear
D)
zona glomerulosa
done
clear
View Answer play_arrow
question_answer 260) Thymosine stimulates:
A)
milk secretion
done
clear
B)
erythrocytes
done
clear
C)
T-lymphocytes
done
clear
D)
melanocytes
done
clear
View Answer play_arrow
question_answer 261) FSH is:
A)
glycoprotein
done
clear
B)
phospholipid
done
clear
C)
glycolipid
done
clear
D)
metalloprotein
done
clear
View Answer play_arrow
question_answer 262) Prostaglandins affect:
A)
blood pressure
done
clear
B)
defecation
done
clear
C)
osmoregulation
done
clear
D)
oxygen metabolism
done
clear
View Answer play_arrow
question_answer 263) What is the molecular weight of phosvitin?
A)
3500
done
clear
B)
4000
done
clear
C)
6000
done
clear
D)
10000
done
clear
View Answer play_arrow
question_answer 264) Which one of the following scientist has written latest book on developmental biology?
A)
Balinansky
done
clear
B)
Gilbert
done
clear
C)
Baer
done
clear
D)
Lilly
done
clear
View Answer play_arrow
question_answer 265) Which one of the following egg is found in Cockroach?
A)
Alecithal
done
clear
B)
Microlecithal
done
clear
C)
Centrolecithal
done
clear
D)
Oligolecithal
done
clear
View Answer play_arrow
question_answer 266) Reten mirabiles are found in which animal?
A)
Elephant
done
clear
B)
Whale
done
clear
C)
Ox
done
clear
D)
Tiger
done
clear
View Answer play_arrow
question_answer 267) Nervous system develops from :
A)
ectoderm
done
clear
B)
endomesoderm
done
clear
C)
mesoderm
done
clear
D)
ectomesoderm
done
clear
View Answer play_arrow
question_answer 268) In indeterminate cleavage the fate of blastomeres is fixed at which of (the following stage?
A)
Blastula
done
clear
B)
Gastrulation
done
clear
C)
32-celled stage
done
clear
D)
64-celled stage
done
clear
View Answer play_arrow
question_answer 269) With which of the following first quarter of micromeres are associated?
A)
determinate cleavage
done
clear
B)
larvation
done
clear
C)
hatching
done
clear
D)
indeterminate cleavage
done
clear
View Answer play_arrow
question_answer 270) Which one of the following is both hormone and enzyme?
A)
ADH hormone
done
clear
B)
Acetycholinesterase
done
clear
C)
Angiotensinogen
done
clear
D)
Rennin
done
clear
View Answer play_arrow
question_answer 271) Which of the following hormones induce secretion of succus entericus?
A)
Insulin
done
clear
B)
Secretin and cholecystokinin
done
clear
C)
Glucagon
done
clear
D)
Secretin
done
clear
View Answer play_arrow
question_answer 272) Which of the following nerve innervates upper jaw of frog?
A)
Maxillary
done
clear
B)
Pathetic
done
clear
C)
Palatine
done
clear
D)
Occulomotor
done
clear
View Answer play_arrow
question_answer 273) How many Malpighian tubules in Cockroach are present?
A)
80-90
done
clear
B)
100-150
done
clear
C)
50-60
done
clear
D)
200-250
done
clear
View Answer play_arrow
question_answer 274) In pheretian lymph gland produces :
A)
lymphocyte cells
done
clear
B)
phagocyte cells
done
clear
C)
oxyntic cells
done
clear
D)
amoeboryte cells
done
clear
View Answer play_arrow
question_answer 275) In Pheretima mouth develops from which of the following?
A)
Ectoderm
done
clear
B)
Mesoderm
done
clear
C)
Blastopore
done
clear
D)
Kndoderm
done
clear
View Answer play_arrow
question_answer 276) In Pheretima which nephridia is present?
A)
Protonephridia
done
clear
B)
Coelom duct
done
clear
C)
Micro-metanephridia
done
clear
D)
Solenocytes
done
clear
View Answer play_arrow
question_answer 277) Muscle cells of Ascaris are arranged in :
A)
coclomyarian
done
clear
B)
platymydrian
done
clear
C)
polymynrian
done
clear
D)
holomyarian
done
clear
View Answer play_arrow
question_answer 278) What is the length of female Ascaris lumbricoides?
A)
200-350 mm
done
clear
B)
150-320 mm
done
clear
C)
100-150 mm
done
clear
D)
50-80 mm
done
clear
View Answer play_arrow
question_answer 279) How many nucteosomes are found in helical coil of 30 nm chromatin fibre ?
A)
6
done
clear
B)
10
done
clear
C)
12
done
clear
D)
9
done
clear
View Answer play_arrow
question_answer 280) Temin worked on which virus?
A)
Rhinovirus
done
clear
B)
Herpesvirus
done
clear
C)
Retrovirus
done
clear
D)
Dengurvirus
done
clear
View Answer play_arrow
question_answer 281) Which one of the following peak absorption of ultraviolet light by heterocyclic bases (nitrogen bases)?
A)
26 nm
done
clear
B)
1500 nm
done
clear
C)
75 nm
done
clear
D)
260 nm
done
clear
View Answer play_arrow
question_answer 282) Which stage of Plasmodium contains rhoptries?
A)
Sporozoite
done
clear
B)
Merozoite
done
clear
C)
Signetring stage
done
clear
D)
Ookinete
done
clear
View Answer play_arrow
question_answer 283) What is the chromosome number of Plasmodium?
A)
9
done
clear
B)
14
done
clear
C)
10
done
clear
D)
4
done
clear
View Answer play_arrow
question_answer 284) In Amoeba who gave contraction hydraulic theory?
A)
Schultze
done
clear
B)
Claude
done
clear
C)
Simpson
done
clear
D)
Swan
done
clear
View Answer play_arrow
question_answer 285) In Entamoeba histolytica intestinal ulceration is caused by which one of the following?
A)
Metacystic trophozoites
done
clear
B)
Binucleate stage
done
clear
C)
Tetranucleate stage
done
clear
D)
Trophozoites
done
clear
View Answer play_arrow
question_answer 286) Which one of the following energy storing compound is formed when succinyl CoA is converted into succinic acid ?
A)
ATP
done
clear
B)
ADP
done
clear
C)
AMP
done
clear
D)
GTP
done
clear
View Answer play_arrow
question_answer 287) In which one of the following mitochondrial cristae are in concentric form?
A)
Spirogyra
done
clear
B)
Euglena
done
clear
C)
Retina of tree shrew
done
clear
D)
Salivary gland of Limnea
done
clear
View Answer play_arrow
question_answer 288) Recombinant nodules are found during which of the following?
A)
Prophase
done
clear
B)
Anaphase
done
clear
C)
Telophase
done
clear
D)
Metaphase
done
clear
View Answer play_arrow
question_answer 289) Which type of reproduction is found in Hydra?
A)
Polyembryony
done
clear
B)
Sexual and asexual
done
clear
C)
Parthenogenesis
done
clear
D)
Encystment
done
clear
View Answer play_arrow
question_answer 290) Which one of the following scientist coined the name Hydra ?
A)
Rudulphi
done
clear
B)
Hyman
done
clear
C)
Linnaeus
done
clear
D)
Grobben
done
clear
View Answer play_arrow
question_answer 291) In which city Ronald Ross worked for malarial parasite?
A)
Delhi
done
clear
B)
Calcutta
done
clear
C)
Hyderabad
done
clear
D)
Madras
done
clear
View Answer play_arrow
question_answer 292) Manganese is associated which enzymes?
A)
Lipase
done
clear
B)
Transformylase
done
clear
C)
Peptidase
done
clear
D)
Carboxyhydrolase
done
clear
View Answer play_arrow
question_answer 293) Which one of the following percentage of inorganic salt in the protoplasm?
A)
5-7%
done
clear
B)
2-3%
done
clear
C)
0.1-2.5%
done
clear
D)
50-60%
done
clear
View Answer play_arrow
question_answer 294) Which one of the following is a primary phosphate in a cell?
A)
\[{{H}_{2}}PO\,{{_{4}^{+}}^{++}}\]
done
clear
B)
\[{{H}_{2}}PO\,{{_{4}^{-}}^{{}}}\]
done
clear
C)
\[{{H}_{2}}PO\,{{_{4}^{-}}^{-}}\]
done
clear
D)
\[{{H}_{2}}PO\,{{_{4}^{+}}^{-}}\]
done
clear
View Answer play_arrow
question_answer 295) Iron is present in which one of the following?
A)
Catalyze
done
clear
B)
Glycoprotein
done
clear
C)
Muck-in
done
clear
D)
Lectin
done
clear
View Answer play_arrow
question_answer 296) National bird of India is:
A)
Pavo cristatus
done
clear
B)
Flamingo
done
clear
C)
Cohimba livia
done
clear
D)
Psittncitia
done
clear
View Answer play_arrow
question_answer 297) What is the molecular weight of IgG antibody?
A)
146, 000
done
clear
B)
160, 000
done
clear
C)
190. 000
done
clear
D)
200, 000
done
clear
View Answer play_arrow
question_answer 298) Which one of the following animal has rhabdites?
A)
Syllis
done
clear
B)
May fly
done
clear
C)
tse-tse fly
done
clear
D)
Dugesia
done
clear
View Answer play_arrow
question_answer 299) Which animal has pseudocoelom ?
A)
Dragon fly
done
clear
B)
Amia
done
clear
C)
Wuchereria bancrftii
done
clear
D)
Lapisma
done
clear
View Answer play_arrow
question_answer 300) Which animal Aristotles lantern found?
A)
Strongylocentrotus
done
clear
B)
Sea urchin
done
clear
C)
Sea mouse
done
clear
D)
Sea horse
done
clear
View Answer play_arrow
question_answer 301) Oxidative phosphorylation occurs in which of the following?
A)
Outer membrane of mitochondria
done
clear
B)
Matrix of mitochondria
done
clear
C)
Inner membrane of mitochondria
done
clear
D)
Inter membrane
done
clear
View Answer play_arrow
question_answer 302) The zygote of Ulothrix is:
A)
quadriflagellate
done
clear
B)
biflagellate
done
clear
C)
uniflagellate
done
clear
D)
aflagellate
done
clear
View Answer play_arrow
question_answer 303) Heterochromatin remains condensed in which part of chromosome?
A)
Secondary construction-I
done
clear
B)
Secondary construction-II
done
clear
C)
Telomeres
done
clear
D)
Both (a) and (b)
done
clear
View Answer play_arrow
question_answer 304) The maximum growth of plants occurs during:
A)
summer
done
clear
B)
winter
done
clear
C)
autumn
done
clear
D)
spring
done
clear
View Answer play_arrow
question_answer 305) Mitochondria remains absent in:
A)
Chlorophyceac
done
clear
B)
Myxophyceae
done
clear
C)
Fungal cell
done
clear
D)
Mesophyll cell
done
clear
View Answer play_arrow
question_answer 306) Trachcids differs from vessels in having :
A)
thick wall
done
clear
B)
bordered pits
done
clear
C)
discontinuous intercalary wall
done
clear
D)
spiral thickening
done
clear
View Answer play_arrow
question_answer 307) The vascular bundles in Cucurbita are:
A)
radial
done
clear
B)
collateral
done
clear
C)
bicollateral
done
clear
D)
concentric
done
clear
View Answer play_arrow
question_answer 308) Which of the following causes delay in senescence?
A)
Cytokinin
done
clear
B)
Auxins
done
clear
C)
Gibberellins
done
clear
D)
ABA
done
clear
View Answer play_arrow
question_answer 309) Commercial cork is obtained from:
A)
Quercus
done
clear
B)
Oak
done
clear
C)
Pine
done
clear
D)
Peepal
done
clear
View Answer play_arrow
question_answer 310) Bost fibres obtains from which part of the stem?
A)
Cork
done
clear
B)
Cortex
done
clear
C)
Xylem
done
clear
D)
Phloem
done
clear
View Answer play_arrow
question_answer 311) The growth in diameter of stem is due to:
A)
cambium
done
clear
B)
xylem
done
clear
C)
phloem
done
clear
D)
shoot apex
done
clear
View Answer play_arrow
question_answer 312) The non porous wood is present in which of the following?
A)
Dicots
done
clear
B)
Monocots
done
clear
C)
Gymnosperms
done
clear
D)
Cactus
done
clear
View Answer play_arrow
question_answer 313) In dorsiventral leaf phloem is situated towards the:
A)
adaxial side
done
clear
B)
abaxial side
done
clear
C)
lateral side
done
clear
D)
both (a) and (b)
done
clear
View Answer play_arrow
question_answer 314) Monothecous anther is the characteristic of:
A)
Malvaceae
done
clear
B)
Liliaceae
done
clear
C)
Brassicaceae
done
clear
D)
Solanaceae
done
clear
View Answer play_arrow
question_answer 315) The onagrad type embryo development is found in :
A)
Solanum
done
clear
B)
Capsella
done
clear
C)
Lilium
done
clear
D)
Hibiscus
done
clear
View Answer play_arrow
question_answer 316) In Capsella embryosac is :
A)
diploid
done
clear
B)
haploid
done
clear
C)
tetrasporic
done
clear
D)
trisporic
done
clear
View Answer play_arrow
question_answer 317) Rauwolfin obtains from which part of the plant?
A)
Stem
done
clear
B)
Root
done
clear
C)
Fruit
done
clear
D)
Leaf
done
clear
View Answer play_arrow
question_answer 318) In polygonum type embryosac, all the cells are :
A)
haploid
done
clear
B)
diploid
done
clear
C)
both (a) and (b)
done
clear
D)
haploid and polyploidy
done
clear
View Answer play_arrow
question_answer 319) Which of the following is not visible through electron microscope in Chlamydomonas?
A)
Contractile vacuole
done
clear
B)
Stigma
done
clear
C)
Neuromotor apparatus
done
clear
D)
All of the above
done
clear
View Answer play_arrow
question_answer 320) The inner most membrane of gram (-) negative bacteria is consist of:
A)
mucopeptide
done
clear
B)
lipoprotein
done
clear
C)
phospholipid
done
clear
D)
lipopolysaccharides
done
clear
View Answer play_arrow
question_answer 321) Which type of placentation is found in family Fabaceae?
A)
Axile
done
clear
B)
Marginal
done
clear
C)
Perietal
done
clear
D)
Basal
done
clear
View Answer play_arrow
question_answer 322) The floral formula of family Malvaceae is:
A)
\[{{K}_{4}}{{C}_{4}}{{A}_{5}}{{G}_{(2)}}\]
done
clear
B)
\[{{K}_{2+2}}{{C}_{4}}{{A}_{2+6}}{{G}_{(2)}}\]
done
clear
C)
\[{{K}_{(5)}}{{C}_{4}}{{A}_{4}}{{G}_{(5)}}\]
done
clear
D)
\[{{K}_{(5)}}{{\overset\frown{{{C}_{5}}A}}_{(5-\infty )}}{{G}_{(\infty )}}\]
done
clear
View Answer play_arrow
question_answer 323) Ferula assafoetida is the main source of:
A)
Haldi
done
clear
B)
Heeng
done
clear
C)
Heroin
done
clear
D)
Opium
done
clear
View Answer play_arrow
question_answer 324) In meiotic division the DNA synthesis takes place in :
A)
S-phase
done
clear
B)
S-phase or leptotene phase
done
clear
C)
M-phase and zygotene
done
clear
D)
all of the above
done
clear
View Answer play_arrow
question_answer 325) Thigmotropism occurs due to the:
A)
light
done
clear
B)
contact
done
clear
C)
temperature
done
clear
D)
pressure
done
clear
View Answer play_arrow
question_answer 326) The sporogenous tissue is originated from amphithecium in which of the following ?
A)
Riccia
done
clear
B)
Marchantia
done
clear
C)
Anthoceros
done
clear
D)
Funaria
done
clear
View Answer play_arrow
question_answer 327) The number of chromosome in an egg is 10. The number of chromosomes in anaphase will be :
A)
10
done
clear
B)
20
done
clear
C)
30
done
clear
D)
40
done
clear
View Answer play_arrow
question_answer 328) Microsporangium of Cycas is placed in which of the following manner on microsporophyll?
A)
Marginal
done
clear
B)
Adaxial
done
clear
C)
Abaxial
done
clear
D)
Lateral
done
clear
View Answer play_arrow
question_answer 329) The process of DNA-synthesis is called as:
A)
translation
done
clear
B)
transformation
done
clear
C)
transduction
done
clear
D)
replication
done
clear
View Answer play_arrow
question_answer 330) The longest plant cell is:
A)
fibre
done
clear
B)
vessel
done
clear
C)
tracheid
done
clear
D)
sieve tube
done
clear
View Answer play_arrow
question_answer 331) Nucleolar chromatic have gene for which of the following?
A)
r-DNA
done
clear
B)
Z-DNA
done
clear
C)
SAT-DNA
done
clear
D)
B-DNA
done
clear
View Answer play_arrow
question_answer 332) The maximum amount of calcium pectate is present in:
A)
(n) primary cell wall
done
clear
B)
secondary cell wall
done
clear
C)
middle lamella
done
clear
D)
cell membrane
done
clear
View Answer play_arrow
question_answer 333) The plant cell wall is:
A)
semi-permeable
done
clear
B)
permeable
done
clear
C)
impermeable
done
clear
D)
selectively permeable
done
clear
View Answer play_arrow
question_answer 334) Cell organelle are present in:
A)
nucleoplasm
done
clear
B)
cytoplasm
done
clear
C)
hyluplasm
done
clear
D)
none of these
done
clear
View Answer play_arrow
question_answer 335) Cell theory was proposed by:
A)
Robert Hook
done
clear
B)
Schneider and Schwann
done
clear
C)
Brawn
done
clear
D)
Leeuwenhoek
done
clear
View Answer play_arrow
question_answer 336) The membrane less cell organelle is:
A)
golgi body
done
clear
B)
chloroplast
done
clear
C)
endoplasmic reticulum
done
clear
D)
ribosome
done
clear
View Answer play_arrow
question_answer 337) The enzyme ATP are remains active in:
A)
head piece of \[{{F}_{1}}\]
done
clear
B)
base piece of \[{{F}_{1}}\]
done
clear
C)
circle of \[{{F}_{2}}\]
done
clear
D)
all of the above
done
clear
View Answer play_arrow
question_answer 338) Cellulose is a polymer of:
A)
glucose
done
clear
B)
fructose
done
clear
C)
galactose
done
clear
D)
mannose
done
clear
View Answer play_arrow
question_answer 339) Which of the following has their own DNA?
A)
Ribosome
done
clear
B)
Dictyosome
done
clear
C)
Microsome
done
clear
D)
Chloroplast
done
clear
View Answer play_arrow
question_answer 340) Krebs cycle takes place in:
A)
inner membrane of mitochondria
done
clear
B)
outer membrane of mitochondria
done
clear
C)
matrix of mitochondria
done
clear
D)
inter membranous space
done
clear
View Answer play_arrow
question_answer 341) Plant cell is differ from animal cell due to the presence of:
A)
chlorophyll
done
clear
B)
mitochondria
done
clear
C)
cellulosic cell wall
done
clear
D)
large size
done
clear
View Answer play_arrow
question_answer 342) The mosaic condition of cell membrane can be proved by :
A)
electroforetic study
done
clear
B)
auto radiography
done
clear
C)
french-inch-technology
done
clear
D)
none of the above
done
clear
View Answer play_arrow
question_answer 343) Which of the following is not a micronutrient?
A)
Cu
done
clear
B)
Mg
done
clear
C)
B
done
clear
D)
\[C{{l}_{2}}\]
done
clear
View Answer play_arrow
question_answer 344) The element which is found in the structure of chlorophyll molecule ?
A)
Fe
done
clear
B)
Mg
done
clear
C)
K
done
clear
D)
Mn
done
clear
View Answer play_arrow
question_answer 345) Photophosphorylation is found in:
A)
outer membrane of chloroplast
done
clear
B)
inner membrane of chloroplast
done
clear
C)
matrix of chloroplast
done
clear
D)
inter membranous space
done
clear
View Answer play_arrow
question_answer 346) Chlorosis in leaf occurs due to die deficiency of:
A)
chloride
done
clear
B)
magnesium
done
clear
C)
phosphate
done
clear
D)
molebdenum
done
clear
View Answer play_arrow
question_answer 347) The first \[C{{O}_{2}}\] acceptor in \[{{C}_{3}}\]-plants is:
A)
RuBP carboxylase
done
clear
B)
PEP carboxylase
done
clear
C)
dehydrogenase
done
clear
D)
hydrolases
done
clear
View Answer play_arrow
question_answer 348) The sun light in PS-II causes:
A)
reduction in \[C{{O}_{2}}\]
done
clear
B)
activation of chlorophyll
done
clear
C)
photolysis of water
done
clear
D)
all of the above
done
clear
View Answer play_arrow
question_answer 349) It is belived that origin of mitochondria and chloroplast through endo symbiotic theory because :
A)
both have same nucleic acid
done
clear
B)
both have same plasma membrane
done
clear
C)
both have same RNA
done
clear
D)
all of the above
done
clear
View Answer play_arrow
question_answer 350) When genes are located on the same locus the condition is known as :
A)
homozygous
done
clear
B)
heterozygous
done
clear
C)
hybrid
done
clear
D)
all of the above
done
clear
View Answer play_arrow
question_answer 351) Which of the following is not involved with the movement of stomata?
A)
High concentration of \[{{K}^{+}}\] in guard cell
done
clear
B)
High light intensity
done
clear
C)
High concentration of IAA
done
clear
D)
High concentration of ABA
done
clear
View Answer play_arrow
question_answer 352) When one gene expresses itself in more than one stages then it is called :
A)
heterozygous
done
clear
B)
genotype
done
clear
C)
complementory gene
done
clear
D)
alleles
done
clear
View Answer play_arrow
question_answer 353) Which of the following is an exception of Mendels law of independent assortment?
A)
Multiple alleles
done
clear
B)
Crossing over
done
clear
C)
Linkage
done
clear
D)
Dominance and recessive
done
clear
View Answer play_arrow
question_answer 354) The anti transpirant is:
A)
PMA
done
clear
B)
ABA
done
clear
C)
Both (a) and (b)
done
clear
D)
None of these
done
clear
View Answer play_arrow
question_answer 355) Which of the following is autotrophic prokaryote?
A)
Sulphur bacteria
done
clear
B)
Cyanobacteria
done
clear
C)
Archebacteria
done
clear
D)
None of these
done
clear
View Answer play_arrow
question_answer 356) Gibberellin was obtained from:
A)
Gibberellin fujikuroi
done
clear
B)
Aspergillus
done
clear
C)
Yeast
done
clear
D)
none of the above
done
clear
View Answer play_arrow
question_answer 357) Pneumatophores are found in:
A)
Orchid
done
clear
B)
Piper
done
clear
C)
Rhizophorn
done
clear
D)
Ficus
done
clear
View Answer play_arrow
question_answer 358) The common pathway between aerobic and anaerobic respiration is :
A)
Krebs cycle
done
clear
B)
ETS
done
clear
C)
Glycolysis
done
clear
D)
Glycogenesis
done
clear
View Answer play_arrow
question_answer 359) The father of cohesion theory is:
A)
Dixon and Jolly
done
clear
B)
Boss
done
clear
C)
Prestelay
done
clear
D)
None of these
done
clear
View Answer play_arrow
question_answer 360) The energy is liberated during coal burning and respiration both, but respiration is step wise process because of the:
A)
enzyme control
done
clear
B)
hormone control
done
clear
C)
combustion
done
clear
D)
respiratory substrate
done
clear
View Answer play_arrow
question_answer 361) Annual rings are most clear in:
A)
temperate regions
done
clear
B)
tropical regions
done
clear
C)
xerophytes
done
clear
D)
hydrophyte
done
clear
View Answer play_arrow
question_answer 362) The peristome teeth of moss shows:
A)
hydrochasy
done
clear
B)
zerochasy
done
clear
C)
chemotropism
done
clear
D)
none of these
done
clear
View Answer play_arrow
question_answer 363) Plants grow on sand and stone is called as:
A)
lithophyte?
done
clear
B)
mesophytos
done
clear
C)
phenerophytes
done
clear
D)
oxalophytes
done
clear
View Answer play_arrow
question_answer 364) Cytochromes are found in:
A)
chloroplast and ribosome
done
clear
B)
mitochondria and chloroplast
done
clear
C)
mitochondria and ribosome
done
clear
D)
chloroplast and golgi body
done
clear
View Answer play_arrow
question_answer 365) In an ecosystem:
A)
primary producers are higher than primary consumers
done
clear
B)
primary consumers are higher than primary producers
done
clear
C)
primary consumers are higher than secondary consumers
done
clear
D)
secondary consumers are higher than primary producers
done
clear
View Answer play_arrow
question_answer 366) Which of the following is involved in the structure CoA FAD and FMN?
A)
Vitamin A
done
clear
B)
Vitamin C
done
clear
C)
Vitamin \[{{B}_{1}}\]
done
clear
D)
Vitamin \[{{B}_{2}}\]
done
clear
View Answer play_arrow
question_answer 367) The enzyme nitrogenase catalyzes:
A)
\[{{N}_{3}}\] to \[N{{N}_{3}}\]
done
clear
B)
\[{{N}_{2}}\] to \[N{{O}_{3}}\]
done
clear
C)
\[N{{O}_{2}}\] to \[{{N}_{2}}\]
done
clear
D)
\[{{N}_{2}}\] to \[N{{O}_{2}}\]
done
clear
View Answer play_arrow
question_answer 368) The hydrolysis of starch takes place by:
A)
sucrose
done
clear
B)
amylase
done
clear
C)
cellulose
done
clear
D)
in vertase
done
clear
View Answer play_arrow
question_answer 369) Ribozyme is:
A)
RNA without sugar
done
clear
B)
RNA without phosphate
done
clear
C)
RNA which acts as an enzyme
done
clear
D)
none of the above
done
clear
View Answer play_arrow
question_answer 370) The transition zone between two different communities is known as :
A)
ecotype
done
clear
B)
ecosystem
done
clear
C)
ecotone
done
clear
D)
biome
done
clear
View Answer play_arrow
question_answer 371) Which of the following belongs to same trophic level?
A)
Tiger and Bear
done
clear
B)
Deer and Bee
done
clear
C)
Crow and Cow
done
clear
D)
Snake and Pig
done
clear
View Answer play_arrow
question_answer 372) The correct food chain is:
A)
Dead leaves \[\to \] Bacteria\[\to \]Insect\[\to \]Bird
done
clear
B)
Phytoplankton\[\to \]Zooplankton\[\to \]Fish
done
clear
C)
Grass\[\to \] Fox \[\to \] Rabbit
done
clear
D)
none of these
done
clear
View Answer play_arrow
question_answer 373) Virus contains
A)
Only DNA
done
clear
B)
Only RNA
done
clear
C)
DNA or RNA
done
clear
D)
None of these
done
clear
View Answer play_arrow
question_answer 374) Initiation of protein synthesis in eukaryotes occurs by the codon :
A)
AUG
done
clear
B)
GCA
done
clear
C)
UAA
done
clear
D)
UAG
done
clear
View Answer play_arrow
question_answer 375) Maize is differ from Wheat in:
A)
phenotype
done
clear
B)
genotype
done
clear
C)
mesotype
done
clear
D)
halophytic
done
clear
View Answer play_arrow
question_answer 376) The plant with red flower is crossed with white flower in \[{{F}_{1}}\] generation all red plants are obtained this shows:
A)
law of segregation
done
clear
B)
law of dominant and recessive
done
clear
C)
law of independence assortment
done
clear
D)
none of the above
done
clear
View Answer play_arrow
question_answer 377) Plant which used by Hugo de Vries for mutation experiment was ?
A)
Oenothera Iamarkiana
done
clear
B)
Solanum tuberosom
done
clear
C)
Ficus elastic
done
clear
D)
None of the above
done
clear
View Answer play_arrow
question_answer 378) In the classification of Whittaker the kingdom Monera include:
A)
bacteria, cyanobacteria, and fungus
done
clear
B)
bacteria and cyanobacteria
done
clear
C)
fungus and alga
done
clear
D)
lichens and bacteria
done
clear
View Answer play_arrow
question_answer 379) The disease caused by mycoplasma is:
A)
Papaya bunchy top
done
clear
B)
Brinjal little leaf
done
clear
C)
Witches broom
done
clear
D)
All of the above
done
clear
View Answer play_arrow
question_answer 380) Vinegar is obtained from alcohol by the:
A)
Bacillus aceti
done
clear
B)
Lactobacillus
done
clear
C)
Azotobactor
done
clear
D)
None of these
done
clear
View Answer play_arrow
question_answer 381) Agar-agar is obtained from:
A)
Chlorella
done
clear
B)
Chlamydomonas
done
clear
C)
Nostoc
done
clear
D)
Gracilaria and Gellidium
done
clear
View Answer play_arrow
question_answer 382) Lichens are the association of:
A)
green algae and Phycomycetes
done
clear
B)
green algae and Ascomycetes
done
clear
C)
glue-green algae and Phycomycetes
done
clear
D)
none of these
done
clear
View Answer play_arrow
question_answer 383) Aflatoxins are obtained from:
A)
bacteria
done
clear
B)
fungus
done
clear
C)
algae
done
clear
D)
moss
done
clear
View Answer play_arrow
question_answer 384) Which of the following have gametuphytic plant body and sporophytic remains as a parasite on it?
A)
Funaria
done
clear
B)
Pteris
done
clear
C)
Cycas
done
clear
D)
Neem
done
clear
View Answer play_arrow
question_answer 385) During development of embryo in archegonium of bryophyte, its posterior part form protective embryo cover which is called:
A)
calyptra
done
clear
B)
apophysis
done
clear
C)
paraphysis
done
clear
D)
hypophysis
done
clear
View Answer play_arrow
question_answer 386) The fern prothallus is:
A)
heart shaped
done
clear
B)
kidney shaped
done
clear
C)
liver shaped
done
clear
D)
none of these
done
clear
View Answer play_arrow
question_answer 387) The vascular cryptogames are:
A)
bryophytes
done
clear
B)
pteridophytes
done
clear
C)
angiosperms
done
clear
D)
none of these
done
clear
View Answer play_arrow
question_answer 388) The living fossil is:
A)
Ginkgo biloba
done
clear
B)
Pinus
done
clear
C)
Ephedra
done
clear
D)
none of these
done
clear
View Answer play_arrow
question_answer 389) The plants in which naked seed is formed without flower and fruit formation is grouped under?
A)
Fungus
done
clear
B)
Gymnosperm
done
clear
C)
Angiospenn
done
clear
D)
Pteridophyta
done
clear
View Answer play_arrow
question_answer 390) Hypanthodium inflorescence is found in:
A)
Ficus
done
clear
B)
Tulsi
done
clear
C)
Cedrus
done
clear
D)
Callotropis
done
clear
View Answer play_arrow
question_answer 391) Flower which never opens in their life cycle is known as:
A)
hetcrogamous
done
clear
B)
cleistogamous
done
clear
C)
dichogamous
done
clear
D)
homogamous
done
clear
View Answer play_arrow
question_answer 392) The inflorescence is edible part in:
A)
Brassica campestris
done
clear
B)
Brassica oleracea botrytis
done
clear
C)
Raphanus sativus
done
clear
D)
Brassica nigra
done
clear
View Answer play_arrow
question_answer 393) The study of tree is called:
A)
Cytology
done
clear
B)
Dendrology
done
clear
C)
Embryology
done
clear
D)
Physiology
done
clear
View Answer play_arrow
question_answer 394) The flow of water from one cell to another adjacent cell takes place by :
A)
osmotic pressure
done
clear
B)
diffusion
done
clear
C)
DPD
done
clear
D)
osmosis
done
clear
View Answer play_arrow
question_answer 395) Root hairs originates from:
A)
endodermis
done
clear
B)
epidermis
done
clear
C)
outer cortex
done
clear
D)
none of these
done
clear
View Answer play_arrow
question_answer 396) The rate of transpiration is measured by:
A)
auxanometer
done
clear
B)
photometer
done
clear
C)
perimeter
done
clear
D)
none of these
done
clear
View Answer play_arrow
question_answer 397) How much energy is used in photosynthesis?
A)
50%
done
clear
B)
35%
done
clear
C)
10%
done
clear
D)
1 to 2%
done
clear
View Answer play_arrow
question_answer 398) The soluble food material is translocated through phloem in:
A)
acropetal manner
done
clear
B)
basipetal manner
done
clear
C)
both (a) and (b)
done
clear
D)
lateral
done
clear
View Answer play_arrow
question_answer 399) Which of the following is long day plant (LDP)?
A)
Sugarcane
done
clear
B)
Guldabdi
done
clear
C)
Tobacco and Soybean
done
clear
D)
Wheat and Radish
done
clear
View Answer play_arrow
question_answer 400) Greenhouse gases are:
A)
CFCS, \[C{{O}_{2}}\,N{{H}_{4}}\] and \[N{{O}_{2}}\]
done
clear
B)
\[{{O}_{2}},\,{{N}_{3}},\,No;\]
done
clear
C)
\[{{N}_{3}},\,C{{O}_{2}}\,N{{H}_{4}}\]
done
clear
D)
None of the above
done
clear
View Answer play_arrow
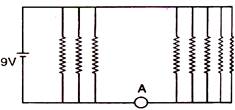
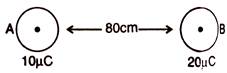
 bad smelling substance. The bad smelling substance is:
bad smelling substance. The bad smelling substance is:
 react with \[{{H}_{2}}O\] gives:
react with \[{{H}_{2}}O\] gives:
