question_answer 1) Plancks constant has the dimensions of:
A)
energy
done
clear
B)
power
done
clear
C)
impulse
done
clear
D)
angular momentum
done
clear
View Answer play_arrow
question_answer 2) A particle after starting from rest, experiences constant acceleration for 20 sec. If it covers a distance\[{{S}_{1}}\]in first 10 sec, then the distance covered during next 10 sec will be
A)
\[{{S}_{1}}\]
done
clear
B)
\[2{{s}_{1}}\]
done
clear
C)
\[3{{s}_{1}}\]
done
clear
D)
\[4{{s}_{1}}\]
done
clear
View Answer play_arrow
question_answer 3) The magnitudes of sum and difference of two vectors arc same then the angle between them is
A)
\[90{}^\circ \]
done
clear
B)
\[40{}^\circ \]
done
clear
C)
\[45{}^\circ \]
done
clear
D)
\[60{}^\circ \]
done
clear
View Answer play_arrow
question_answer 4) When two particles each of mass m are dropped from height h and 2h respectively, then the ratio of their times to reach the ground is
A)
\[1:\sqrt{2}\]
done
clear
B)
\[\sqrt{2}:1\]
done
clear
C)
\[1:2\]
done
clear
D)
\[2:1\]
done
clear
View Answer play_arrow
question_answer 5) The escape velocity of a body depends upon
A)
mass of body
done
clear
B)
mass of earth
done
clear
C)
speed of light
done
clear
D)
angle of projection
done
clear
View Answer play_arrow
question_answer 6) A capacitor of 100 \[\mu F\]and resistance of 100\[\Omega \] are connected in series with an a. c source of 50 Hz. The phase angle between the current and source voltage is:
A)
\[{{\tan }^{-1}}(1/\pi )\]
done
clear
B)
\[{{\tan }^{-1}}(1/2\pi )\]
done
clear
C)
\[{{\tan }^{-1}}(\pi /2)\]
done
clear
D)
\[{{\tan }^{-1}}(2\pi /2)\]
done
clear
View Answer play_arrow
question_answer 7) When a horizontal disc is rotated about its vertical axis and sand is dropped on it, then its angular velocity will
A)
decrease
done
clear
B)
increase
done
clear
C)
remain constant
done
clear
D)
first decrease and then increase
done
clear
View Answer play_arrow
question_answer 8) The wavelength of light in air and some other medium are respectively \[{{\lambda }_{a}}\]and\[{{\lambda }_{m}}..\]The refractive index of medium is:
A)
\[{{\lambda }_{a/}}{{\lambda }_{m}}\]
done
clear
B)
\[{{\lambda }_{m}}/{{\lambda }_{a}}\]
done
clear
C)
\[{{\lambda }_{a}}\times {{\lambda }_{m}}\]
done
clear
D)
None of these
done
clear
View Answer play_arrow
question_answer 9) A solid sphere is rolling without slipping on a horizontal surface. The ratio of its rotational kinetic energy and translational kinetic energy is:
A)
\[\frac{2}{9}\]
done
clear
B)
\[\frac{2}{5}\]
done
clear
C)
\[\frac{2}{7}\]
done
clear
D)
\[\frac{7}{2}\]
done
clear
View Answer play_arrow
question_answer 10) A liquid has only
A)
shear modulus
done
clear
B)
Youngs modulus
done
clear
C)
bulk modulus
done
clear
D)
All of the above
done
clear
View Answer play_arrow
question_answer 11) Van der Waals equation for gases is \[\left( P+\frac{a}{{{V}^{2}}} \right)\,(V-b)=RT\] The dimensions of constant a are
A)
\[ML{{\,}^{5}}{{T}^{-2}}\]
done
clear
B)
\[ML{{\,}^{4}}{{T}^{-1}}\]
done
clear
C)
\[ML{{\,}^{3}}{{T}^{-1}}\]
done
clear
D)
\[ML{{\,}^{5}}{{T}^{-1}}\]
done
clear
View Answer play_arrow
question_answer 12) The velocity of a particle in S.H.M. at displacement y from mean position is (\[a=\]amplitude\[\omega =\] angular frequency)
A)
\[\omega \sqrt{{{a}^{2}}+{{y}^{2}}}\]
done
clear
B)
\[\omega \sqrt{{{a}^{2}}-{{y}^{2}}}\]
done
clear
C)
\[\omega y\]
done
clear
D)
\[{{\omega }^{2}}({{a}^{2}}-{{y}^{2}})\]
done
clear
View Answer play_arrow
question_answer 13) Excess pressure in a soap bubble of radius r is proportional to
A)
\[\frac{1}{r}\]
done
clear
B)
\[\frac{1}{{{r}^{2}}}\]
done
clear
C)
\[r\]
done
clear
D)
\[{{r}^{2}}\]
done
clear
View Answer play_arrow
question_answer 14) In a thermodynamic process there is no exchange of heat between the system and surroundings. Then the process is
A)
isothermal
done
clear
B)
adiabatic
done
clear
C)
isochoric
done
clear
D)
isobaric
done
clear
View Answer play_arrow
question_answer 15) The ratio of radii of two spheres of same material is 1 : 4, then the ratio of their heat capacities is
A)
\[\frac{1}{4}\]
done
clear
B)
\[\frac{1}{16}\]
done
clear
C)
\[\frac{1}{32}\]
done
clear
D)
\[\frac{1}{64}\]
done
clear
View Answer play_arrow
question_answer 16) If the critical angle for total internal reflection from a medium to vacuum is \[30{}^\circ \], then the speed of light in the medium is
A)
\[6\times {{10}^{8}}m/s\]
done
clear
B)
\[3\times {{10}^{8}}m/s\]
done
clear
C)
\[2\times {{10}^{8}}m/s\]
done
clear
D)
\[1.5\times {{10}^{8}}m/s\]
done
clear
View Answer play_arrow
question_answer 17) A prism has a refracting angle of \[60{}^\circ \]. If it produces a minimum deviation of \[30{}^\circ \] in an incident ray, the angle of incidence is
A)
\[15{}^\circ \]
done
clear
B)
\[30{}^\circ \]
done
clear
C)
\[45{}^\circ \]
done
clear
D)
\[60{}^\circ \]
done
clear
View Answer play_arrow
question_answer 18) A mass of 30 kg is suspended from a spring of spring constant 15 N/m. The time period of vertical oscillations of mass is
A)
\[\frac{3\pi }{\sqrt{2}}s\]
done
clear
B)
\[2\pi \sqrt{2}\,s\]
done
clear
C)
\[\pi \sqrt{2}\,s\]
done
clear
D)
\[2\sqrt{2}\,s\]
done
clear
View Answer play_arrow
question_answer 19) The focal length of objective and eye piece of a telescope are\[{{f}_{0}}\] and\[{{f}_{e}}\] respectively. Then its magnification will be:
A)
\[{{f}_{0}}+{{f}_{e}}\]
done
clear
B)
\[{{f}_{0}}\times {{f}_{e}}\]
done
clear
C)
\[\frac{{{f}_{0}}}{{{f}_{e}}}\]
done
clear
D)
\[\frac{{{f}_{e}}}{{{f}_{0}}}\]
done
clear
View Answer play_arrow
question_answer 20) The energy radiated by a black body is directly proportional to
A)
\[{{T}^{2}}\]
done
clear
B)
\[{{T}^{-2}}\]
done
clear
C)
\[{{T}^{4}}\]
done
clear
D)
\[T\]
done
clear
View Answer play_arrow
question_answer 21) Huygens wave theory of light could not explain
A)
diffraction
done
clear
B)
interference
done
clear
C)
polarization
done
clear
D)
photoelectric effect
done
clear
View Answer play_arrow
question_answer 22) A thick mirror produces a number of images of an object. The brightest image is
A)
first
done
clear
B)
second
done
clear
C)
third
done
clear
D)
last one
done
clear
View Answer play_arrow
question_answer 23) If the threshold wavelength for sodium is \[5420\,\overset{\text{o}}{\mathop{\text{A}}}\,,\] then the work function a sodium is
A)
4.58 eV
done
clear
B)
2.28 eV
done
clear
C)
1.14 eV
done
clear
D)
0.23 eV
done
clear
View Answer play_arrow
question_answer 24) When a diode shows saturation current its dynamical plate resistance will be
A)
zero
done
clear
B)
infinity
done
clear
C)
indeterminate
done
clear
D)
variable
done
clear
View Answer play_arrow
question_answer 25) The wavelength associated with an electron accelerated through a potential difference of 100V is nearly
A)
\[100\,\overset{\text{o}}{\mathop{\text{A}}}\,\]
done
clear
B)
\[123\,\overset{\text{o}}{\mathop{\text{A}}}\,\]
done
clear
C)
\[1.23\,\overset{\text{o}}{\mathop{\text{A}}}\,\]
done
clear
D)
\[0.123\,\overset{\text{o}}{\mathop{\text{A}}}\,\]
done
clear
View Answer play_arrow
question_answer 26) The equation of motion of a progressive wave is y = 0.3 sin(314 t-1.57x) where and y are in metre and t is in second. The velocity of wave is
A)
100 m/s
done
clear
B)
200 m/s
done
clear
C)
400 m/s
done
clear
D)
5 mm/s
done
clear
View Answer play_arrow
question_answer 27) The minimum orbital angular momentum of the electron in hydrogen atom is
A)
\[h\]
done
clear
B)
\[h/2\]
done
clear
C)
\[h/2\pi \]
done
clear
D)
\[h/\pi \]
done
clear
View Answer play_arrow
question_answer 28) The energy of a hydrogen-like atom in its ground state is -54.4 eV. It may be
A)
hydrogen
done
clear
B)
deuterium
done
clear
C)
helium
done
clear
D)
lithium
done
clear
View Answer play_arrow
question_answer 29) A magnetic field can be produced by
A)
a moving charge
done
clear
B)
a changing electric field
done
clear
C)
a stationary charge
done
clear
D)
both (a) and (b)
done
clear
View Answer play_arrow
question_answer 30) X-rays incident on a metal
A)
exert a force on it
done
clear
B)
transfers energy to it
done
clear
C)
transfers momentum to it
done
clear
D)
All of the above
done
clear
View Answer play_arrow
question_answer 31) The binding energy of the deuterium is L 2.23 MeV. The mass defect in amu is
A)
0.0024
done
clear
B)
0.0012
done
clear
C)
2.23
done
clear
D)
0.024
done
clear
View Answer play_arrow
question_answer 32) An \[\text{-}\]particle is bombarded on \[{{N}^{14}}\]. As a result a \[{{O}^{17}}\]-nucleus is formed and a particle X is emitted. The particle X is
A)
neutron
done
clear
B)
proton
done
clear
C)
electron
done
clear
D)
positron
done
clear
View Answer play_arrow
question_answer 33) In a laser tube all the photons
A)
have same wavelength
done
clear
B)
have same energy
done
clear
C)
move in same direction
done
clear
D)
move with same speed.
done
clear
View Answer play_arrow
question_answer 34) Regarding a semi-conductor which one of the following is wrong?
A)
There are no free electrons at 0 K
done
clear
B)
There are no free electrons at room temperature
done
clear
C)
The number of free electrons increases with rise of temperature
done
clear
D)
The charge carriers are electrons and holes.
done
clear
View Answer play_arrow
question_answer 35) Transformers are used in
A)
DC circuits only
done
clear
B)
AC circuits only
done
clear
C)
in both (a) and (b)
done
clear
D)
neither in (a) nor in (b).
done
clear
View Answer play_arrow
question_answer 36) An organ pipe open at both ends contains
A)
longitudinal stationary waves
done
clear
B)
longitudinal progressive waves
done
clear
C)
transverse stationary waves
done
clear
D)
transverse progressive waves
done
clear
View Answer play_arrow
question_answer 37) Which one of the following acts as a neutron absorber in a nuclear reactor?
A)
Cd-rod
done
clear
B)
Heavy water \[({{D}_{2}}O)\]
done
clear
C)
Graphite
done
clear
D)
Distilled water \[({{H}_{2}}O)\]
done
clear
View Answer play_arrow
question_answer 38) Mass-energy equation\[E=m{{c}^{2}}\] was given
A)
Newton
done
clear
B)
Kepler
done
clear
C)
Einstein
done
clear
D)
Millikan
done
clear
View Answer play_arrow
question_answer 39) For changing an ammeter into a voltmeter, we should connect
A)
low resistance in series
done
clear
B)
low resistance in parallel
done
clear
C)
high resistance in series
done
clear
D)
high resistance in parallel
done
clear
View Answer play_arrow
question_answer 40) The value of R in gas equation \[PV=nRT\] is
A)
universal constant
done
clear
B)
depends on number of moles n
done
clear
C)
4.2 J/mole K
done
clear
D)
depends on nature of gas
done
clear
View Answer play_arrow
question_answer 41) The penetrating power is minimum for
A)
\[\text{-}\]rays
done
clear
B)
\[\text{-}\]rays
done
clear
C)
\[\text{Y-}\]rays
done
clear
D)
\[\text{X-}\]rays
done
clear
View Answer play_arrow
question_answer 42) The breaking stress of a wire depends upon
A)
material of wire
done
clear
B)
length of wire
done
clear
C)
radius of wire
done
clear
D)
shape of cross-section
done
clear
View Answer play_arrow
question_answer 43) Water rises in a vertical capillary tube upto a length of 10 cm. If the tube is inclined at \[45{}^\circ \], the length of water column risen in the tube will be
A)
\[10\,cm\]
done
clear
B)
\[10\,\sqrt{2}\,cm\]
done
clear
C)
\[5\,\sqrt{2}\,cm\]
done
clear
D)
\[20\,cm\]
done
clear
View Answer play_arrow
question_answer 44) The magnetic field at the centre of semi-circular wire carrying current \[i\] is
A)
\[\frac{{{\mu }_{0}}i}{2r}\]
done
clear
B)
\[\frac{{{\mu }_{0}}i}{4r}\]
done
clear
C)
\[\frac{{{\mu }_{0}}i}{r}\]
done
clear
D)
\[\frac{{{\mu }_{0}}i}{2\pi r}\]
done
clear
View Answer play_arrow
question_answer 45) If a wire of resistance R is stretched to double of its length, then the new resistance will be
A)
\[\frac{R}{2}\]
done
clear
B)
\[2R\]
done
clear
C)
\[4R\]
done
clear
D)
\[16R\]
done
clear
View Answer play_arrow
question_answer 46) If currents in two parallel wires flow in opposite directions, the force between the wires will be
A)
attractive
done
clear
B)
repulsive
done
clear
C)
zero
done
clear
D)
attractive or repulsive depending on the material of wires.
done
clear
View Answer play_arrow
question_answer 47) The electric field and potential inside a hollow sphere of radius R and uniformly charged with charge q is
A)
\[0,0\]
done
clear
B)
\[0,\frac{1}{4\pi {{\varepsilon }_{0}}}\frac{q}{R}\]
done
clear
C)
\[\frac{1}{4\pi {{\varepsilon }_{0}}}\frac{q}{{{R}^{2}}}\frac{1}{4\pi {{\varepsilon }_{0}}}\frac{q}{R}\]
done
clear
D)
\[\frac{1}{4\pi {{\varepsilon }_{0}}}\frac{q}{{{R}^{2}}}0\]
done
clear
View Answer play_arrow
question_answer 48) If a glass rod is immersed in a liquid of same refractive index, then it will appear
A)
bent
done
clear
B)
longer
done
clear
C)
shorter
done
clear
D)
invisible
done
clear
View Answer play_arrow
question_answer 49) Two charges of magnitude\[-q\]and\[+4q\] are placed 1 cm apart. The distance between the point on the line joining and point at which the electric field is zero (with respect to\[-q\]), is
A)
1 cm
done
clear
B)
2 cm
done
clear
C)
3cm
done
clear
D)
5cm
done
clear
View Answer play_arrow
question_answer 50) A capacitor is charged by a battery and the charging battery is disconnected and a dielectric slab is inserted in it. Then for the capacitor
A)
charge remains constant
done
clear
B)
charge increases
done
clear
C)
potential difference remains constant
done
clear
D)
potential difference increases
done
clear
View Answer play_arrow
question_answer 51) The dimensional formula of constant a in van der Waals gas equation \[\left( p+\frac{a}{{{V}^{2}}} \right)\,(V-b)\,=RT\] is:
A)
\[[M{{L}^{3}}{{T}^{-2}}]\]
done
clear
B)
\[[M{{L}^{5}}{{T}^{-2}}]\]
done
clear
C)
\[[M{{L}^{2}}{{T}^{-1}}]\]
done
clear
D)
\[[M{{L}^{2}}{{T}^{-5}}]\]
done
clear
View Answer play_arrow
question_answer 52) The angle of projection for which the maximum height and range of the projectile are same is
A)
\[{{\tan }^{-1}}(2)\]
done
clear
B)
\[{{\tan }^{-1}}(4)\]
done
clear
C)
\[{{\tan }^{-1}}\left( \frac{1}{4} \right)\]
done
clear
D)
\[45{}^\circ \]
done
clear
View Answer play_arrow
question_answer 53)
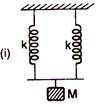
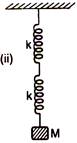
Two springs A and B having force constants k each are arranged (i) in parallel and (ii) in series. A mass M is attached to two arrangements separately. If time period in case (i) is\[{{T}_{1}}\]1 and in case (ii) is \[{{T}_{1}}\] then ratio\[\frac{{{T}_{1}}}{{{T}_{2}}}\] is
A)
\[1\]
done
clear
B)
\[2\]
done
clear
C)
\[0.5\]
done
clear
D)
\[0.25\]
done
clear
View Answer play_arrow
question_answer 54) A particle executing SHM has amplitude m and frequency 60 Hz, The maximum acceleration of the particle is
A)
\[60{{\pi }^{2}}\,m/{{s}^{2}}\]
done
clear
B)
\[80{{\pi }^{2}}\,m/{{s}^{2}}\]
done
clear
C)
\[120{{\pi }^{2}}\,m/{{s}^{2}}\]
done
clear
D)
\[144{{\pi }^{2}}\,m/{{s}^{2}}\]
done
clear
View Answer play_arrow
question_answer 55) In a capillary tube, water rises to a height of 2 cm. The height of water that will rise in another capillary tube of radius one-third that of first is
A)
2 cm
done
clear
B)
3 cm
done
clear
C)
6 cm
done
clear
D)
0.67 cm
done
clear
View Answer play_arrow
question_answer 56) A block of mass 10 kg is moving up an inclined plane of inclination \[30{}^\circ \] with an , initial speed of 5 m/s. It stops after 0.5 s, what is the value of coefficient of kinetic friction?
A)
0.5
done
clear
B)
0.6
done
clear
C)
\[\sqrt{3}\]
done
clear
D)
\[\frac{1}{\sqrt{3}}\]
done
clear
View Answer play_arrow
question_answer 57) The dynamic mass of electron having rest mass \[{{m}_{0}}\] and moving with speed 0.8 c is
A)
\[0.6\,{{m}_{0}}\]
done
clear
B)
\[0.8\,{{m}_{0}}\]
done
clear
C)
\[\frac{5}{3}\,{{m}_{0}}\]
done
clear
D)
\[1.25\,{{m}_{0}}\]
done
clear
View Answer play_arrow
question_answer 58) The potential energy of a satellite of mass and revolving at a height equal to radius of earth from earths surface, is
A)
\[-\,mg{{R}_{e}}\]
done
clear
B)
\[-0.67\,mg{{R}_{e}}\]
done
clear
C)
\[-0.5\,mg{{R}_{e}}\]
done
clear
D)
\[-0.33\,mg{{R}_{e}}\]
done
clear
View Answer play_arrow
question_answer 59) The wavelength of light emitted due to transition of electron from second orbit to first orbit in hydrogen atom is
A)
\[6563\overset{\text{o}}{\mathop{\text{A}}}\,\]
done
clear
B)
\[4102\overset{\text{o}}{\mathop{\text{A}}}\,\]
done
clear
C)
\[4861\overset{\text{o}}{\mathop{\text{A}}}\,\]
done
clear
D)
\[1215\overset{\text{o}}{\mathop{\text{A}}}\,\]
done
clear
View Answer play_arrow
question_answer 60) Which one of the following happens when light ray enters from air to a glass slab?
A)
Wavelength increases
done
clear
B)
Wavelength decreases
done
clear
C)
Frequency increases
done
clear
D)
Frequency decreases
done
clear
View Answer play_arrow
question_answer 61) Minimum wavelength of X-rays produced in a coolidge tube operated at potential difference of 40 kV is
A)
\[0.31\overset{\text{o}}{\mathop{\text{A}}}\,\]
done
clear
B)
\[3.1\overset{\text{o}}{\mathop{\text{A}}}\,\]
done
clear
C)
\[31\overset{\text{o}}{\mathop{\text{A}}}\,\]
done
clear
D)
\[311\overset{\text{o}}{\mathop{\text{A}}}\,\]
done
clear
View Answer play_arrow
question_answer 62) A convex lens of focal length 40 cm is (kept in contact with a concave lens of focal length 25 cm. The power of the combination is
A)
-6.5D
done
clear
B)
+6.5D
done
clear
C)
-1.5D
done
clear
D)
+1.5D
done
clear
View Answer play_arrow
question_answer 63) At the same temperature and pressure the densities of two diatomic gases are\[{{d}_{1,}}\] and \[{{d}_{2,}}\] Then the ratio of velocities of sound in these gases will be
A)
\[\frac{{{d}_{1}}}{{{d}_{2}}}\]
done
clear
B)
\[\sqrt{\frac{{{d}_{2}}}{{{d}_{1}}}}\]
done
clear
C)
\[\sqrt{\frac{{{d}_{1}}}{{{d}_{2}}}}\]
done
clear
D)
\[\frac{{{d}_{2}}^{2}}{{{d}_{1}}^{2}}\]
done
clear
View Answer play_arrow
question_answer 64) In the reaction \[_{92}{{X}^{234}}{{\xrightarrow{{}}}_{87}}{{\Upsilon }^{222}}.\]How many \[\text{-}\]particles and \[\beta \]-particles are emitted?
A)
3 and 5
done
clear
B)
5 and 3
done
clear
C)
3 and 3
done
clear
D)
3 and 1
done
clear
View Answer play_arrow
question_answer 65) When a substance is gradually heated, its initial colour is
A)
red
done
clear
B)
green
done
clear
C)
yellow
done
clear
D)
white
done
clear
View Answer play_arrow
question_answer 66) At which temperature the speed of sound will be three times of its speed at \[0{}^\circ C\]?
A)
\[1100{}^\circ C\]
done
clear
B)
\[1284{}^\circ C\]
done
clear
C)
\[1500{}^\circ C\]
done
clear
D)
\[2184{}^\circ C\]
done
clear
View Answer play_arrow
question_answer 67) Thermocouple, thermometer is based on
A)
Seebeck effect
done
clear
B)
Pettier effect
done
clear
C)
Thomson effect
done
clear
D)
Joule effect
done
clear
View Answer play_arrow
question_answer 68) An ideal gas has volume V0at \[27{}^\circ C\] It is heated at constant pressure, so/ that its volume becomes \[2{{V}_{0}}.\] Then the final temperature is
A)
327 K
done
clear
B)
\[327{}^\circ C\]
done
clear
C)
\[54{}^\circ C\]
done
clear
D)
150K
done
clear
View Answer play_arrow
question_answer 69) The electric field required to keep a water drop of mass in and charge e just to remain suspended is:
A)
\[mg\]
done
clear
B)
\[emg\]
done
clear
C)
\[\frac{mg}{e}\]
done
clear
D)
\[\frac{em}{g}\]
done
clear
View Answer play_arrow
question_answer 70) In a triode valve the plate current will be maximum when
A)
both grid and plate are positive
done
clear
B)
both grid and plate are negative
done
clear
C)
grid is negative and plate is positive
done
clear
D)
grid is positive and plate is negative
done
clear
View Answer play_arrow
question_answer 71) A point charge is located at 0. There is a point P at a distance r from it. The electric field at point P is 500 V/m and a potential of 3000 V. Then the value of r is
A)
6m
done
clear
B)
12m
done
clear
C)
24 m
done
clear
D)
36 m
done
clear
View Answer play_arrow
question_answer 72) A particle having charge 100 times that of an electron is revolving in a circular path of radius 0.8 m at the rate of 1 revolution/second. The magnetic field produced at the centre of orbit is
A)
\[{{10}^{-3}}\,\mu {{ }_{0}}\]
done
clear
B)
\[{{10}^{-7}}\,\mu {{ }_{0}}\]
done
clear
C)
\[{{10}^{-11}}\,\mu {{ }_{0}}\]
done
clear
D)
\[{{10}^{-17}}\,\mu {{ }_{0}}\]
done
clear
View Answer play_arrow
question_answer 73) A body of mass 10 kg is placed in a lift moving upward with an acceleration of 2m/s. The apparent weight of body is \[(g=9.8\,m/{{s}^{2}})\]
A)
118 N
done
clear
B)
78 N
done
clear
C)
98 N
done
clear
D)
198 N
done
clear
View Answer play_arrow
question_answer 74) The work function of a photometal is 6.63 eV. The threshold wavelength is
A)
\[3920\,\overset{o}{\mathop{\text{A}}}\,\]
done
clear
B)
\[1866\,\overset{o}{\mathop{\text{A}}}\,\]
done
clear
C)
\[186.6\,\overset{o}{\mathop{\text{A}}}\,\]
done
clear
D)
\[18666\,\overset{o}{\mathop{\text{A}}}\,\]
done
clear
View Answer play_arrow
question_answer 75) In a transformer the number of turns in primary and secondary coil are 50 and 40 respectively. If 220 V A.C. is applied across the primary coil, the ratio of current in primary and secondary coil is
A)
4 : 5
done
clear
B)
5 : 4
done
clear
C)
16 : 25
done
clear
D)
25 : 16
done
clear
View Answer play_arrow
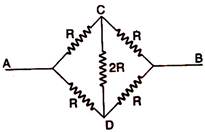
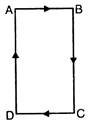
question_answer 76)
The equivalent resistance between A and B in the given circuit will be
A)
R
done
clear
B)
2R
done
clear
C)
\[\frac{R}{2}\]
done
clear
D)
6/R
done
clear
View Answer play_arrow
question_answer 77) The nuclear energy in nuclear reactor is produced by
A)
nuclear fission
done
clear
B)
nuclear fusion
done
clear
C)
electric generator
done
clear
D)
burning of coal
done
clear
View Answer play_arrow
question_answer 78) At which temperature the Fahrenheit and Celcius scale give equal readings?
A)
\[40{}^\circ \]
done
clear
B)
\[37{}^\circ \]
done
clear
C)
\[-40{}^\circ \]
done
clear
D)
\[80{}^\circ \]
done
clear
View Answer play_arrow
question_answer 79) The electric field strength due to a dipole depends upon distance r as
A)
\[E\propto \frac{1}{r}\]
done
clear
B)
\[E\propto \frac{1}{{{r}^{2}}}\]
done
clear
C)
\[E\propto \frac{1}{{{r}^{3}}}\]
done
clear
D)
\[E\propto \frac{1}{{{r}^{4}}}\]
done
clear
View Answer play_arrow
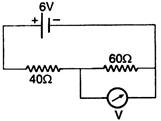
question_answer 80)
The reading of voltmeter in the circuit shown is
A)
2.25 V
done
clear
B)
3.25 V
done
clear
C)
4.25V
done
clear
D)
6.25V
done
clear
View Answer play_arrow
question_answer 81) The Youngs modulus of a wire of length (L) and radius (r) is \[\Upsilon .\] If the length is reduced to \[\frac{L}{2}\] and radius to\[\frac{r}{2}\] then its Youngs modulus will be
A)
\[\frac{\Upsilon }{2}\]
done
clear
B)
\[\Upsilon \]
done
clear
C)
\[2\Upsilon \]
done
clear
D)
\[4\Upsilon \]
done
clear
View Answer play_arrow
question_answer 82) The decay constant of a radioactive substance is\[\lambda .\] The half life and mean life of substance are respectively given by
A)
\[\frac{1}{\lambda }\text{and}\frac{{{\log }_{e}}2}{\lambda }\]
done
clear
B)
\[\frac{{{\log }_{e}}2}{\lambda }\text{and}\frac{1}{\lambda }\]
done
clear
C)
\[\frac{\lambda }{{{\log }_{e}}2}\text{and}\,\frac{1}{\lambda }\]
done
clear
D)
\[\frac{\lambda }{{{\log }_{e}}2}\text{and}\,\text{2}\lambda \]
done
clear
View Answer play_arrow
question_answer 83) A source emitting a note of frequency 240 Hz is moving towards an observer with a speed of 20 m/s. If the observer also moves with a speed of 20 m/s towards the source, the apparent frequency heard by observer is (velocity of sound = 340 m/s)
A)
270 Hz
done
clear
B)
240 Hz
done
clear
C)
268 Hz
done
clear
D)
360 Hz
done
clear
View Answer play_arrow
question_answer 84) In pressure cooker, cooking is fast as compared to open atmosphere due to
A)
decreased boiling point
done
clear
B)
increased boiling point
done
clear
C)
enclosure of lid permanently
done
clear
D)
heat is not lost to surroundings
done
clear
View Answer play_arrow
question_answer 85) The terminal velocity (\[{{\upsilon }_{t}}\]) of a spherical body of radius r falling in a long vertical column of liquid of viscosity \[\eta \] depends on r and \[\eta \] as follows \[{{\upsilon }_{t}}\] is proportional to
A)
\[\eta \,r\]
done
clear
B)
\[\eta \,{{r}^{2}}\]
done
clear
C)
\[{{\eta }^{2}}r\]
done
clear
D)
\[\frac{{{r}^{2}}}{\eta }\]
done
clear
View Answer play_arrow
question_answer 86) An unpolarised light of intensity\[{{I}_{0}}\] on a polaroid whose optic axis is inclined at \[45{}^\circ \] to the incident light. Then the intensity of light emerging from the polaroid is
A)
\[{{I}_{0}}\]
done
clear
B)
\[\frac{{{I}_{0}}}{2}\]
done
clear
C)
\[\frac{{{I}_{0}}}{4}\]
done
clear
D)
zero
done
clear
View Answer play_arrow
question_answer 87) In a triode valve the plate resistance is equal to the toad resistance. Then voltage gain of triode valve is
A)
\[\mu \]
done
clear
B)
\[\frac{\mu }{2}\]
done
clear
C)
\[\frac{\mu }{4}\]
done
clear
D)
\[2\mu \]
done
clear
View Answer play_arrow
question_answer 88) A tube closed at one end produces a note B of frequency 512 Hz. If the tube is opened k at both ends, the frequency will become
A)
512 Hz
done
clear
B)
256 Hz
done
clear
C)
128 Hz
done
clear
D)
1024 Hz
done
clear
View Answer play_arrow
question_answer 89)
A wire EF carrying current \[{{i}_{1}}\] is placed near a current carrying rectangular loop (ABCD as shown. Then the wire EF
A)
remains unaffected
done
clear
B)
is attracted towards loop
done
clear
C)
is repelled away from loop
done
clear
D)
first attracted and then repelled
done
clear
View Answer play_arrow
question_answer 90) The fractional change in internal energy when a gas is cooled from \[927{}^\circ C\] to \[27{}^\circ C\] is
A)
0.25
done
clear
B)
0.75
done
clear
C)
1
done
clear
D)
0.67
done
clear
View Answer play_arrow
question_answer 91) A plane progressive wave is given by \[y=25\cos (2\pi t-\pi x)\] Then the amplitude and frequency are respectively
A)
25, 100
done
clear
B)
25, 1
done
clear
C)
25, 2
done
clear
D)
50\[\pi ,\] 2
done
clear
View Answer play_arrow
question_answer 92) The ratio of specific charge of an \[\text{-}\] particle to that of a proton is
A)
2 : 1
done
clear
B)
1 : 1
done
clear
C)
1 : 2
done
clear
D)
1 : 3
done
clear
View Answer play_arrow
question_answer 93) Two vibrating tuning forks produce waves given by \[{{y}_{1}}=4\sin \,500\pi t,\] \[{{y}_{1}}=2\sin \,506\pi t\] If they are held near the ear of a person, the person will hear
A)
3 beats/second with intensity ratio of maxima to minima equal to 9
done
clear
B)
3 beats/second with intensity ratio of maxima to minima equal to 2
done
clear
C)
6 beats/second with intensity ratio of maxima to minima to equal to 2
done
clear
D)
6 beats/second with intensity ratio of maxima to minima equal to 9
done
clear
View Answer play_arrow
question_answer 94) The half life of a certain radioactive substance is 12 days. The time taken for \[\frac{7}{8}\] of sample to decay is
A)
36 days
done
clear
B)
12 days
done
clear
C)
4 days
done
clear
D)
24 days
done
clear
View Answer play_arrow
question_answer 95) Three bulbs of 40 W, 60 W and 100 W are arranged in series across a 220 V supply. The bulb giving maximum brightness is of
A)
40 W
done
clear
B)
60 W
done
clear
C)
100 W
done
clear
D)
all give equal brightness
done
clear
View Answer play_arrow
question_answer 96) A cubical box contains charge +Q at its centre. The total magnetic flux emerging from the box is :
A)
\[\frac{Q}{{{\varepsilon }_{0}}}\]
done
clear
B)
\[\frac{Q}{6{{\varepsilon }_{0}}}\]
done
clear
C)
\[\frac{Q}{4{{\varepsilon }_{0}}}\]
done
clear
D)
\[{{\varepsilon }_{0}}Q\]
done
clear
View Answer play_arrow
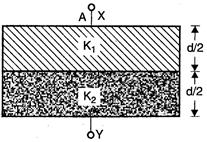
question_answer 97)
A parallel plate capacitor is filled with two dielectrics as shown in figure. If A is area of each plate; then the effective capacitance between X and \[\Upsilon \] is
A)
\[\frac{{{\varepsilon }_{0}}A}{d}\left( \frac{{{K}_{1}}+{{K}_{2}}}{2} \right)\]
done
clear
B)
\[\frac{2{{\varepsilon }_{0}}A}{d}\left( {{K}_{1}}+{{K}_{2}} \right)\]
done
clear
C)
\[\frac{2{{\varepsilon }_{0}}A}{d}\left( \frac{{{K}_{1}}+{{K}_{2}}}{{{K}_{1}}{{K}_{2}}} \right)\]
done
clear
D)
\[\frac{2{{\varepsilon }_{0}}A}{d}\left( \frac{{{K}_{1}}{{K}_{2}}}{{{K}_{1}}+{{K}_{2}}} \right)\]
done
clear
View Answer play_arrow
question_answer 98) A coil has self-inductance 60 mH. If the current in the coil increases from 1.0 A to 1.5 A in 0.1 second and the resistance of coil is \[4\Omega ,\] the current induced in the coil is
A)
1.0A
done
clear
B)
\[\frac{4}{3}\text{A}\]
done
clear
C)
\[\frac{\text{3}}{\text{4}}\text{A}\]
done
clear
D)
\[\frac{3}{40}\text{A}\]
done
clear
View Answer play_arrow
question_answer 99) If a mica sheet of thickness t and refractive index \[\mu .\] is placed in the path of one of interfering beam in a double slit experiment then displacement of fringes will be
A)
\[\frac{D}{d}\mu t\]
done
clear
B)
\[\frac{D}{d}(\mu -1)t\]
done
clear
C)
\[\frac{D}{d}(\mu +1)t\]
done
clear
D)
\[\frac{D}{d}({{\mu }^{2}}-1)t\]
done
clear
View Answer play_arrow
question_answer 100) If a current of \[i\] amp is flowing in the winding of solenoid and ft is the number of turns per unit length, then the magnetic field at the centre of solenoid is
A)
\[{{\mu }_{0}}ni\]
done
clear
B)
\[\frac{{{\mu }_{0}}ni}{2}\]
done
clear
C)
\[\frac{{{\mu }_{0}}i}{2n}\]
done
clear
D)
\[\frac{{{\mu }_{0}}i}{n}\]
done
clear
View Answer play_arrow
question_answer 101) Which one of the following relation is correct for the rate of diffusion of a gas(r)?
A)
\[r\propto \sqrt{d}\]
done
clear
B)
\[r\propto \sqrt{1/d}\]
done
clear
C)
\[r=d\]
done
clear
D)
\[r\propto d\]
done
clear
View Answer play_arrow
question_answer 102) Which of the following species, is planar?
A)
\[CO_{3}^{2-}\]
done
clear
B)
\[N{{H}_{3}}\]
done
clear
C)
\[PC{{l}_{3}}\]
done
clear
D)
None of these
done
clear
View Answer play_arrow
question_answer 103) Which one of the following is not a Lewis acid?
A)
\[AlC{{l}_{3}}\]
done
clear
B)
\[SnC{{l}_{4}}\]
done
clear
C)
\[B{{F}_{3}}\]
done
clear
D)
\[BeC{{l}_{2}}\]
done
clear
View Answer play_arrow
question_answer 104) Which one of the following is important ore of copper?
A)
Calamine
done
clear
B)
Malachite
done
clear
C)
Galena
done
clear
D)
Dolomite
done
clear
View Answer play_arrow
question_answer 105) Which of the following is most commonly used in photo-electrical cells?
A)
Francium
done
clear
B)
Silicon
done
clear
C)
Caesium
done
clear
D)
Lithium
done
clear
View Answer play_arrow
question_answer 106) in the following reaction, what is X? \[HC\equiv CH\xrightarrow[HgS{{O}_{4}}]{{{H}_{2}}O}x\]
A)
\[C{{H}_{3}}COOH\]
done
clear
B)
\[\underset{COOH}{\overset{COOH}{\mathop{|}}}\,\]
done
clear
C)
\[C{{H}_{3}}CHO\]
done
clear
D)
\[\underset{C{{H}_{2}}OH}{\overset{C{{H}_{2}}OH}{\mathop{|}}}\,\]
done
clear
View Answer play_arrow
question_answer 107) The product formed by the reaction of acetamide with \[B{{r}_{2}}\] in presence of \[NaOH\]is:
A)
\[C{{H}_{3}}CN\]
done
clear
B)
\[C{{H}_{3}}CHO\]
done
clear
C)
\[C{{H}_{3}}C{{H}_{2}}OH\]
done
clear
D)
\[C{{H}_{3}}N{{H}_{2}}\]
done
clear
View Answer play_arrow
question_answer 108) An asymmetric carbon atom is that in which
A)
three similar and one dissimilar groups
done
clear
B)
two similar and two dissimilar groups
done
clear
C)
all the different groups attached to it
done
clear
D)
None of these
done
clear
View Answer play_arrow
question_answer 109) How many structural isomers can be obtained from \[{{C}_{2}}HB{{r}_{2}}\]
A)
\[4\]
done
clear
B)
\[3\]
done
clear
C)
\[2\]
done
clear
D)
\[1\]
done
clear
View Answer play_arrow
question_answer 110) When an a-particle is removed from Ra, the new element formed is of
A)
zero group
done
clear
B)
Ist group
done
clear
C)
VIIth group
done
clear
D)
IIIrd group
done
clear
View Answer play_arrow
question_answer 111) Which one of the following metal is used in galvanization?
A)
\[Cu\]
done
clear
B)
\[Ag\]
done
clear
C)
\[Zn\]
done
clear
D)
\[Fe\]
done
clear
View Answer play_arrow
question_answer 112) The process carried out in perfect insulation is
A)
isothermal
done
clear
B)
isobaric
done
clear
C)
isochoric
done
clear
D)
adiabatic
done
clear
View Answer play_arrow
question_answer 113) When potassium dichromate crystals are heated, we get
A)
oxygen
done
clear
B)
hydrogen
done
clear
C)
sulphur dioxide
done
clear
D)
hydrogen sulphide
done
clear
View Answer play_arrow
question_answer 114) In the preparation of sulphuric acid,\[{{V}_{2}}{{O}_{5}}\] is used in the reaction which is
A)
\[S+{{O}_{2}}\to S{{O}_{2}}~~\]
done
clear
B)
\[S{{O}_{2}}+{{H}_{2}}O\to {{H}_{2}}S{{O}_{4}}\]
done
clear
C)
\[2S{{O}_{2}}+{{O}_{2}}\to 2S{{O}_{3}}\]
done
clear
D)
None of these
done
clear
View Answer play_arrow
question_answer 115) Which one of the following configuration is correct for \[F{{e}^{+3}}\] ?
A)
\[1{{s}^{2}}2{{s}^{2}}2{{p}^{6}}3{{s}^{2}}3{{p}^{6}}4{{s}^{2}}3{{d}^{3}}\]
done
clear
B)
\[1{{s}^{2}}2{{s}^{2}}2{{p}^{6}}3{{s}^{2}}3{{p}^{6}}4{{s}^{2}}3{{d}^{7}}\]
done
clear
C)
\[1{{s}^{2}}2{{s}^{2}}2{{p}^{6}}3{{s}^{2}}3{{p}^{6}}4{{s}^{2}}3{{d}^{6}}\]
done
clear
D)
\[1{{s}^{2}}\,2{{s}^{2}}\,2{{p}^{6}}\,3{{s}^{2}}\,3{{p}^{6}}\,3{{d}^{5}}\]
done
clear
View Answer play_arrow
question_answer 116) IUPAC name of tert-butyl alcohol is
A)
Butanol-2
done
clear
B)
2-Methyl propanol-2
done
clear
C)
Butanol-1
done
clear
D)
Propanol-2
done
clear
View Answer play_arrow
question_answer 117) 1-Chlorobutane on reaction with alcoholic potash gives
A)
1-Butanol
done
clear
B)
2-Butene
done
clear
C)
1-Butene
done
clear
D)
2-Butanol
done
clear
View Answer play_arrow
question_answer 118) Which one of the following is good conductor of electricity in solution state?
A)
Metallic solid
done
clear
B)
Covalent
done
clear
C)
Molecular solid
done
clear
D)
Ionic compound
done
clear
View Answer play_arrow
question_answer 119) When \[C{{H}_{3}}C{{H}_{2}}Br\] reacts with sodium acetylide, the main product is
A)
1-butyne
done
clear
B)
1-butene
done
clear
C)
1-butane
done
clear
D)
3-butane
done
clear
View Answer play_arrow
question_answer 120) By increasing the temperature, the vapour pressure of a substance
A)
does not depend on temperature
done
clear
B)
always increases
done
clear
C)
decreases
done
clear
D)
partially depends on temperature
done
clear
View Answer play_arrow
question_answer 121) Which one of the following is Buffer solution?
A)
\[C{{H}_{3}}COON{{H}_{4}}\]
done
clear
B)
\[NaOH\]
done
clear
C)
\[Pb{{(OH)}_{2}}\]
done
clear
D)
\[N{{H}_{4}}Cl+N{{H}_{4}}OH\]
done
clear
View Answer play_arrow
question_answer 122) The correct order of acidic strength is
A)
\[HF<HCl<HBr<HI\]
done
clear
B)
\[HBr<HCl<HI<HF\]
done
clear
C)
\[HCl<HBr<HF<HI\]
done
clear
D)
\[HI<HBr<HCl<HF\]
done
clear
View Answer play_arrow
question_answer 123) Which one of the following has maximum ionization potential?
A)
\[K\]
done
clear
B)
\[Be\]
done
clear
C)
\[Na\]
done
clear
D)
\[Mg\]
done
clear
View Answer play_arrow
question_answer 124) Carbylamine test is performed in alcoholic \[KOH\]by heating a mixture of
A)
chloroform and silver powder
done
clear
B)
an alkyl halide and a pri. Amine
done
clear
C)
an alkyl cyanide and a pri. Amine
done
clear
D)
trihalongenated methane and a primary amine
done
clear
View Answer play_arrow
question_answer 125) The most favourable condition for the manufacture of \[N{{H}_{3}}\]is
A)
low pressure and high temperature
done
clear
B)
low temperature and high pressure
done
clear
C)
high temperature and high pressure
done
clear
D)
low temperature and low pressure
done
clear
View Answer play_arrow
question_answer 126) Which of the following gas is insoluble in water?
A)
\[S{{O}_{2}}\]
done
clear
B)
\[{{H}_{2}}\]
done
clear
C)
\[N{{H}_{3}}\]
done
clear
D)
\[C{{O}_{2}}\]
done
clear
View Answer play_arrow
question_answer 127) Which one of the following shows haloform test?
A)
Butanol-2
done
clear
B)
Butanol-1
done
clear
C)
2-methyl ketone
done
clear
D)
2-methyl propane
done
clear
View Answer play_arrow
question_answer 128) Isobars have
A)
same number of nucleons
done
clear
B)
same number of neutrons
done
clear
C)
same number of protons
done
clear
D)
None of these
done
clear
View Answer play_arrow
question_answer 129) If acetyl chloride is reduced in the presence of \[BaS{{O}_{4}}\]and Pd then
A)
\[C{{H}_{3}}COOH\]is formed
done
clear
B)
\[C{{H}_{3}}CHO\]is formed
done
clear
C)
\[C{{H}_{3}}C{{H}_{2}}OH\]is formed
done
clear
D)
None of these
done
clear
View Answer play_arrow
question_answer 130) The unit of the molality is
A)
mole per litre
done
clear
B)
mole per kilogram
done
clear
C)
per mole per litre
done
clear
D)
mole litre
done
clear
View Answer play_arrow
question_answer 131) Which one of the following does not respond to Cannizzaro reaction?
A)
\[{{C}_{6}}{{H}_{5}}CHO\]
done
clear
B)
\[HCHO\]
done
clear
C)
\[CC{{l}_{3}}CHO\]
done
clear
D)
\[C{{H}_{3}}CHO\]
done
clear
View Answer play_arrow
question_answer 132) \[20\text{ }gm.\]\[CaC{{O}_{3}}\] on decomposition gives \[C{{O}_{2}}\] at STP
A)
\[4.48\text{ }litre\]
done
clear
B)
\[22.4\text{ }litre\]
done
clear
C)
\[2.24\text{ }litre\]
done
clear
D)
None of these
done
clear
View Answer play_arrow
question_answer 133) Lucas reagent is
A)
\[dil\text{ }HCl+\text{ }anhyd.\text{ }ZnC{{l}_{2}}\]
done
clear
B)
\[Pd-BaS{{O}_{4}}\]
done
clear
C)
\[cone.HCl+anhyd.ZuC{{l}_{2}}\]
done
clear
D)
None of the above
done
clear
View Answer play_arrow
question_answer 134) Of the following elements, which one has the same oxidation state in all of its compounds?
A)
carbon
done
clear
B)
hydrogen
done
clear
C)
oxygen
done
clear
D)
fluorine
done
clear
View Answer play_arrow
question_answer 135) When \[{{C}_{2}}{{H}_{4}}\]is reacted with \[{{H}_{2}}\] in presence of \[Ni\], then the product formed is
A)
\[C{{H}_{4}}\]
done
clear
B)
\[{{C}_{2}}{{H}_{6}}\]
done
clear
C)
\[HCOOH\]
done
clear
D)
\[HCHO\]
done
clear
View Answer play_arrow
question_answer 136) Vinegar is
A)
\[HCOOH\]
done
clear
B)
\[C{{H}_{3}}CHO\]
done
clear
C)
\[HCHO\]
done
clear
D)
\[C{{H}_{3}}COOH\]
done
clear
View Answer play_arrow
question_answer 137) At high altitude the boiling point of water lowers because
A)
atmospheric pressure is high
done
clear
B)
atmospheric pressure is low
done
clear
C)
temperature is low
done
clear
D)
None of these
done
clear
View Answer play_arrow
question_answer 138) Catalyst increases rate of reaction by
A)
decreasing equilibrium constant
done
clear
B)
decreasing threshold energy
done
clear
C)
decreasing activation energy
done
clear
D)
none of the above
done
clear
View Answer play_arrow
question_answer 139) \[{{C}_{4}}{{H}_{11}}N\] shows how many primary amines?
A)
\[1\]
done
clear
B)
\[2\]
done
clear
C)
\[3\]
done
clear
D)
\[4\]
done
clear
View Answer play_arrow
question_answer 140) Which one of the following is not a colligative property?
A)
Elevation in boiling point
done
clear
B)
Osmotic pressure
done
clear
C)
Optical activity
done
clear
D)
Relative lowering of vapour pressure
done
clear
View Answer play_arrow
question_answer 141) Grignard reagent reacts with \[HCHO\]to produce
A)
sec. alcohol
done
clear
B)
anhydride
done
clear
C)
an acid
done
clear
D)
primary alcohol
done
clear
View Answer play_arrow
question_answer 142) Which one of the following is slag?
A)
\[CaSi{{O}_{3}}\]
done
clear
B)
\[Si{{O}_{2}}\]
done
clear
C)
\[CaO\]
done
clear
D)
\[Ca{{(OH)}_{2}}\]
done
clear
View Answer play_arrow
question_answer 143) When 2-bromobutane reacts with alcoholic \[KOH,\]the reaction is
A)
hydrogenation
done
clear
B)
halogenation
done
clear
C)
dehydro halogenation
done
clear
D)
none of these
done
clear
View Answer play_arrow
question_answer 144) Adiabatic process involves
A)
\[\Delta q=0\]
done
clear
B)
\[\Delta E=0\]
done
clear
C)
\[\Delta W=0\]
done
clear
D)
\[\Delta V=0\]
done
clear
View Answer play_arrow
question_answer 145) If three unreactive gases having partial pressures \[{{P}_{A}},{{P}_{B}}\] and \[{{P}_{C}};\] their moles are respectively \[1\text{ }mole,\] \[2\text{ }moles\] and \[3moles,\]then their total pressure is
A)
\[P={{P}_{A}}+{{P}_{B}}+{{P}_{C}}\]
done
clear
B)
\[P=\frac{\sqrt{{{P}_{A}}+{{P}_{B}}+{{P}_{C}}}}{3}\]
done
clear
C)
\[P=\frac{{{P}_{A}}+{{P}_{B}}+{{P}_{C}}}{6}\]
done
clear
D)
none of these
done
clear
View Answer play_arrow
question_answer 146) Glucose reacts with bromine water to produce
A)
Glyceraldehyde
done
clear
B)
Gluconic acid
done
clear
C)
saccharic acid
done
clear
D)
Glucaric acid
done
clear
View Answer play_arrow
question_answer 147) If \[0.75\text{ }mole\]of an ideal gas expands isothermally at \[{{27}^{o}}C\]from 15 lit. to\[25\text{ }lit\]. then the maximum work done is
A)
\[9.14\text{ }J\]
done
clear
B)
\[9.23J\]
done
clear
C)
\[9.43J\]
done
clear
D)
\[9.75J\]
done
clear
View Answer play_arrow
question_answer 148) Which does not form linear structure?
A)
\[C{{O}_{2}}\]
done
clear
B)
\[N{{O}_{2}}\]
done
clear
C)
\[{{C}_{2}}{{H}_{2}}\]
done
clear
D)
\[BeC{{l}_{2}}\]
done
clear
View Answer play_arrow
question_answer 149) Zeeman effect explains splitting of lines in
A)
magnetic field
done
clear
B)
electric field
done
clear
C)
both of these
done
clear
D)
none of these
done
clear
View Answer play_arrow
question_answer 150) In Lake test of \[A{{l}^{3+}}\] ion, there is formation of coloured floating-lake. It is due to
A)
adsorption of litmus by \[{{H}_{2}}O\]
done
clear
B)
adsorption of litmus by \[Al{{(OH)}_{3}}\]
done
clear
C)
adsorption of litmus by \[Al{{(OH)}_{4}}\]
done
clear
D)
none of the above.
done
clear
View Answer play_arrow
question_answer 151) In Clemmensens reduction, acetone gives
A)
propane
done
clear
B)
propene
done
clear
C)
propanol
done
clear
D)
propyne
done
clear
View Answer play_arrow
question_answer 152) Chloroform reacts with cone. \[HN{{O}_{3}}\]to give
A)
\[CHC{{l}_{2}}N{{O}_{2}}\]
done
clear
B)
\[CHC{{l}_{2}}N{{O}_{3}}\]
done
clear
C)
\[CC{{l}_{3}}N{{O}_{2}}\]
done
clear
D)
\[CHC{{l}_{2}}.HN{{O}_{3}}\]
done
clear
View Answer play_arrow
question_answer 153) Anhydrous \[AlC{{l}_{3}}\] is prepared from
A)
\[Al+C{{l}_{2}}~\]
done
clear
B)
\[Al+dil.\text{ }HCl\]
done
clear
C)
\[Al+con;\text{ }HCl\]
done
clear
D)
\[Al+dry\text{ }HCl\text{ }gas\]
done
clear
View Answer play_arrow
question_answer 154) Which one of the following responds to Tollens test?
A)
\[C{{H}_{3}}COC{{H}_{3}}\]
done
clear
B)
\[C{{H}_{3}}CHO\]
done
clear
C)
\[HCOOH\]
done
clear
D)
\[{{C}_{2}}{{H}_{5}}O{{C}_{2}}{{H}_{5}}\]
done
clear
View Answer play_arrow
question_answer 155) Marsh gas is
A)
\[C{{H}_{4}}\]
done
clear
B)
\[{{C}_{2}}{{H}_{2}}\]
done
clear
C)
\[{{H}_{2}}S\]
done
clear
D)
\[S{{O}_{2}}\]
done
clear
View Answer play_arrow
question_answer 156) If volume of 2 moles of an ideal gas at \[540\text{ }K\]is \[44.8\text{ }litre,\]its pressure is
A)
\[2\text{ }aim.\]
done
clear
B)
\[4\,atm.\]
done
clear
C)
\[5\text{ }aim.\]
done
clear
D)
\[6\text{ }atm.\]
done
clear
View Answer play_arrow
question_answer 157) Which one of the following is not a Chalcogen?
A)
\[S\]
done
clear
B)
\[Se\]
done
clear
C)
\[O\]
done
clear
D)
\[Na\]
done
clear
View Answer play_arrow
question_answer 158) The number of moles of \[KCl\]in \[1000\text{ }ml\] of \[3\text{ }molar\]solution is
A)
\[2\]
done
clear
B)
\[3\]
done
clear
C)
\[4\]
done
clear
D)
\[5\]
done
clear
View Answer play_arrow
question_answer 159) Tyndall effect is shown by
A)
solution
done
clear
B)
precipitate
done
clear
C)
sol
done
clear
D)
none of these
done
clear
View Answer play_arrow
question_answer 160) Epsom salt is
A)
\[MgS{{O}_{4}}.2{{H}_{2}}O\]
done
clear
B)
\[MgS{{O}_{4}}\text{ }.\text{ }7{{H}_{2}}O\]
done
clear
C)
\[BaS{{O}_{4}}\text{ }.2{{H}_{2}}O\]
done
clear
D)
\[CaS{{O}_{4}}.2{{H}_{2}}O\]
done
clear
View Answer play_arrow
question_answer 161) For the reaction, \[{{N}_{2}}+3{{H}_{2}}\xrightarrow{{}}2N{{H}_{3}}\] which one of the following is correct regarding \[\Delta H\]?
A)
\[\Delta H=\Delta E-RT\]
done
clear
B)
\[\Delta H=\Delta E-2RT\]
done
clear
C)
\[\Delta H=\Delta E+2RT\]
done
clear
D)
\[\Delta H=\Delta E+RT\]
done
clear
View Answer play_arrow
question_answer 162) The best water soluble hydroxide is
A)
\[Ca{{(OH)}_{2}}\]
done
clear
B)
\[Ba{{(OH)}_{2}}\]
done
clear
C)
\[Sr{{(OH)}_{2}}\]
done
clear
D)
\[Mg{{(OH)}_{2}}\]
done
clear
View Answer play_arrow
question_answer 163) On dry heating, calcium formate gives
A)
acetaldehyde
done
clear
B)
formaldehyde
done
clear
C)
acetone
done
clear
D)
acetic acid
done
clear
View Answer play_arrow
question_answer 164) The normality of a solution containing \[31.5\text{ }gm\]. of hydrated oxalic acid \[({{C}_{2}}{{H}_{2}}{{O}_{4}}.2{{H}_{2}}O)\] in\[1250\text{ }ml\]. of solution is
A)
\[0.1N\]
done
clear
B)
\[0.2N\]
done
clear
C)
\[0.4N\]
done
clear
D)
\[0.6N\]
done
clear
View Answer play_arrow
question_answer 165) Both oxidation and reduction takes place in
A)
\[NaBr+HCl\xrightarrow{{}}NaCl+HBr\]
done
clear
B)
\[HBr+AgN{{O}_{3}}\text{ }\xrightarrow{{}}\text{ }AgBr+HN{{O}_{3}}\]
done
clear
C)
\[{{H}_{2}}+B{{r}_{2}}\xrightarrow{{}}2HBr\]
done
clear
D)
\[CaO+{{H}_{2}}S{{O}_{4}}\xrightarrow{{}}CaS{{O}_{4}}+{{H}_{2}}O\]
done
clear
View Answer play_arrow
question_answer 166) The number of atoms in\[4.25\text{ }gm\]. of \[N{{H}_{3}}\]is
A)
\[4.157\times {{10}^{23}}\]
done
clear
B)
\[3.343\times {{10}^{23}}\]
done
clear
C)
\[6.023\times {{10}^{23}}\]
done
clear
D)
\[8.527\times {{10}^{23}}\]
done
clear
View Answer play_arrow
question_answer 167) For the reaction, \[C{{H}_{3}}COCl\xrightarrow{{{H}_{2}}/Pd}X,\] then X is
A)
\[C{{H}_{3}}CHO\]
done
clear
B)
\[C{{H}_{3}}COOH\]
done
clear
C)
\[C{{H}_{3}}COC{{H}_{3}}\]
done
clear
D)
\[C{{H}_{3}}C{{H}_{2}}OH\]
done
clear
View Answer play_arrow
question_answer 168) The freezing point of a solution, prepared from \[1.25\text{ }gm\]. of a non-electrolyte and \[20gm\] of water, is \[271.9\text{ }K\]. If the molar depression constant is \[1.86\text{ }K/mol,\] then molar mass of the solute is
A)
\[103.3\]
done
clear
B)
\[105.7\]
done
clear
C)
\[107.9\]
done
clear
D)
\[109.3\]
done
clear
View Answer play_arrow
question_answer 169) When \[C{{H}_{4}},\] \[{{C}_{2}}{{H}_{2}},\] \[{{C}_{2}}{{H}_{4}}\] and \[{{C}_{2}}{{H}_{6}}\]gasses are passed through the Wolf bottle of \[~AgN{{O}_{3}},\] then which one of the following gas does not come out from the bottle?
A)
\[C{{H}_{4}}\]
done
clear
B)
\[{{C}_{2}}{{H}_{2}}\]
done
clear
C)
\[{{C}_{2}}{{H}_{4}}\]
done
clear
D)
\[{{C}_{2}}{{H}_{6}}\]
done
clear
View Answer play_arrow
question_answer 170) Both \[C{{H}_{4}}\]and \[{{C}_{2}}{{H}_{6}}\] can be prepared in one step by the reaction of
A)
\[C{{H}_{3}}Br\]
done
clear
B)
\[C{{H}_{3}}C{{H}_{2}}OH\]
done
clear
C)
\[C{{H}_{3}}OH\]
done
clear
D)
\[C{{H}_{3}}COC{{H}_{3}}\]
done
clear
View Answer play_arrow
question_answer 171) An ideal solution is formed when its constituents
A)
are converted into ideal gases
done
clear
B)
have zero volume change
done
clear
C)
have zero heat of mixing
done
clear
D)
have both (b) and (c)
done
clear
View Answer play_arrow
question_answer 172) A compound gives a yellow precipitate on warming with a aq. solution of\[NaOH\]. Its vapour density is 29. The compound is
A)
\[C{{H}_{3}}C{{H}_{2}}CHO\]
done
clear
B)
\[C{{H}_{3}}COC{{H}_{3}}\]
done
clear
C)
\[C{{H}_{3}}CHOHC{{H}_{3}}\]
done
clear
D)
\[C{{H}_{3}}C{{H}_{2}}C{{H}_{2}}OH\]
done
clear
View Answer play_arrow
question_answer 173) Which one of the following does not react with \[CuS{{O}_{4}}\]solution?
A)
\[Ag\]
done
clear
B)
\[Fe\]
done
clear
C)
\[Mg\]
done
clear
D)
\[Zn\]
done
clear
View Answer play_arrow
question_answer 174) Glycerol at \[530K\]reacts with oxalic acid to produce
A)
glyceraldehyde
done
clear
B)
allyl alcohol
done
clear
C)
formic acid
done
clear
D)
acetic acid
done
clear
View Answer play_arrow
question_answer 175) On hydrolysis, \[PC{{l}_{3}}\]gives
A)
\[{{H}_{3}}P{{O}_{3}}\]
done
clear
B)
\[{{H}_{3}}P{{O}_{4}}\]
done
clear
C)
\[POC{{l}_{3}}\]
done
clear
D)
\[HP{{O}_{3}}\]
done
clear
View Answer play_arrow
question_answer 176) Bleaching powder is obtained by treating chlorine with
A)
\[CaC{{O}_{3}}\]
done
clear
B)
\[Ca{{(OH)}_{2}}\]
done
clear
C)
\[CaO\]
done
clear
D)
none of these
done
clear
View Answer play_arrow
question_answer 177) \[Mg\] burns in air to produce
A)
\[MgC{{O}_{3}}\]
done
clear
B)
\[MgO\]
done
clear
C)
\[Mg3{{H}_{2}}\]
done
clear
D)
\[MgO+M{{g}_{3}}{{N}_{2}}\]
done
clear
View Answer play_arrow
question_answer 178) Deficiency of vitamin E causes
A)
scurvy
done
clear
B)
beri-beri
done
clear
C)
antifertility
done
clear
D)
rickets
done
clear
View Answer play_arrow
question_answer 179) Total number of electrons contained in all the p-orbitals of bromine is
A)
\[5\]
done
clear
B)
\[17\]
done
clear
C)
\[19\]
done
clear
D)
\[23\]
done
clear
View Answer play_arrow
question_answer 180) How many times a solution of \[pH=2\]has higher acidity than the solution of \[pH=6\]
A)
\[4\]
done
clear
B)
\[12\]
done
clear
C)
\[400\]
done
clear
D)
\[10000\]
done
clear
View Answer play_arrow
question_answer 181) The raw material used in Fischer Tropsch process for manufacture of synthetic petrol is
A)
producer gas
done
clear
B)
coal gas
done
clear
C)
water gas
done
clear
D)
water gas + excess of Hydrogen
done
clear
View Answer play_arrow
question_answer 182) Which one of the following acid possesses oxidizing, reducing and complex forming properties?
A)
\[HN{{O}_{3}}\]
done
clear
B)
\[HCl\]
done
clear
C)
\[{{H}_{2}}S{{O}_{4}}\]
done
clear
D)
\[HN{{O}_{2}}\]
done
clear
View Answer play_arrow
question_answer 183) In the first order reaction, half of the reaction is completed in 100 second. The time for 99% reaction to occur will be
A)
\[664.64\text{ }sec\]
done
clear
B)
\[646.6\text{ }sec\]
done
clear
C)
\[660.9\text{ }sec~\]
done
clear
D)
\[654.5\text{ }sec\]
done
clear
View Answer play_arrow
question_answer 184) Azimuthal quantum number \[(l)=2,\] then the value of principal quantum number (n)is
A)
\[1\]
done
clear
B)
\[2\]
done
clear
C)
\[3\]
done
clear
D)
\[4\]
done
clear
View Answer play_arrow
question_answer 185) The product obtained by heating 3, 3-dimethyl-2-butanol with \[{{H}_{2}}S{{O}_{4}}\]is
A)
3, 3-dimethyl -1-butene
done
clear
B)
2, 3 -dimethyl-1-butene
done
clear
C)
2, 3-dimethyl-2-butene
done
clear
D)
none of these
done
clear
View Answer play_arrow
question_answer 186) \[KI\] reacts with \[CuS{{O}_{4}}\]to produce
A)
\[{{K}_{2}}S{{O}_{4}}+C{{u}_{2}}{{l}_{2}}+{{I}_{2}}\]
done
clear
B)
\[Cu{{I}_{2}}+{{K}_{2}}S{{O}_{4}}\]
done
clear
C)
\[C{{u}_{2}}{{I}_{2}}+{{K}_{2}}S{{O}_{4}}\]
done
clear
D)
\[{{K}_{2}}S{{O}_{4}}+Cu{{I}_{2}}+{{I}_{2}}\]
done
clear
View Answer play_arrow
question_answer 187) Which one of the following have least \[_{p}{{K}_{a}}\] value?
A)
\[CC{{l}_{3}}COOH\]
done
clear
B)
\[C{{H}_{2}}ClCOOH\]
done
clear
C)
\[C{{F}_{3}}COOH\]
done
clear
D)
\[C{{H}_{3}}COOH\]
done
clear
View Answer play_arrow
question_answer 188) lonization potential is maximum for
A)
\[Al\]
done
clear
B)
\[In\]
done
clear
C)
\[Ga\]
done
clear
D)
\[B\]
done
clear
View Answer play_arrow
question_answer 189) Acetylene contains number of bonds
A)
\[5\sigma \] and \[3\pi \]
done
clear
B)
\[5\sigma \] and \[1\pi \]
done
clear
C)
\[3\sigma \] and \[2\pi \]
done
clear
D)
\[4\sigma \] and \[1\pi \]
done
clear
View Answer play_arrow
question_answer 190) Ethylidene dibromide on heating with metallic sodium in ether solution produce
A)
1-Butene
done
clear
B)
Ethylene
done
clear
C)
Ethane
done
clear
D)
2-Butene
done
clear
View Answer play_arrow
question_answer 191) lonization constant of \[C{{H}_{3}}COOH\]is\[1.8\times {{10}^{-5}}\]. The concentration of \[{{H}^{+}}\] ion in \[0.1M\] solution is
A)
\[1.3\times {{10}^{-2}}M\]
done
clear
B)
\[1.8\times {{10}^{-5}}M\]
done
clear
C)
\[1.8\times {{10}^{-6}}M\]
done
clear
D)
\[1.34\times {{10}^{-3}}M\]
done
clear
View Answer play_arrow
question_answer 192) Which one of the following pair of elements has similar properties?
A)
\[10,12\]
done
clear
B)
\[11,20\]
done
clear
C)
\[21,33\]
done
clear
D)
\[13,31\]
done
clear
View Answer play_arrow
question_answer 193) Borazine is represented by the mol. formula
A)
\[{{B}_{3}}{{N}_{3}}{{H}_{6}}\]
done
clear
B)
\[{{B}_{4}}{{N}_{2}}{{H}_{6}}\]
done
clear
C)
\[{{B}_{5}}N{{H}_{6}}\]
done
clear
D)
\[{{B}_{6}}{{H}_{6}}\]
done
clear
View Answer play_arrow
question_answer 194) The increasing order of acidity of \[{{H}_{2}}{{O}_{2}},\]\[{{H}_{2}}O\] and \[C{{O}_{2}}\]is
A)
\[{{H}_{2}}{{O}_{2}}>{{H}_{2}}O>C{{O}_{2}}\]
done
clear
B)
\[{{H}_{2}}{{O}_{2}}>C{{O}_{2}}>{{H}_{2}}O\]
done
clear
C)
\[{{H}_{2}}O>{{H}_{2}}{{O}_{2}}>C{{O}_{2}}\]
done
clear
D)
\[{{H}_{2}}O<{{H}_{2}}{{O}_{2}}>C{{O}_{2}}\]
done
clear
View Answer play_arrow
question_answer 195) Red lead is
A)
\[PbO\]
done
clear
B)
\[Pb{{O}_{2}}\]
done
clear
C)
\[P{{b}_{3}}{{O}_{4}}\]
done
clear
D)
\[P{{b}_{4}}{{O}_{3}}\]
done
clear
View Answer play_arrow
question_answer 196) A compound having mol., \[mass=78\]contains \[C=92.31%\]and \[H=7.69%.\]Its molecular formula is
A)
\[{{C}_{5}}{{H}_{12}}\]
done
clear
B)
\[{{C}_{5}}{{H}_{18}}\]
done
clear
C)
\[{{C}_{4}}{{H}_{30}}\]
done
clear
D)
\[{{C}_{6}}{{H}_{6}}\]
done
clear
View Answer play_arrow
question_answer 197) Anti Markownikoffs rule addition reaction is not observed in:
A)
pent-2-ene
done
clear
B)
but-2-ene
done
clear
C)
1-butene
done
clear
D)
propene
done
clear
View Answer play_arrow
question_answer 198) The hybridization of carbon in \[C{{H}_{3}}-CH=C=C{{H}_{2}}\]is
A)
\[s{{p}^{3}}s{{p}^{3}}s{{p}^{2}}s{{p}^{2}}\]
done
clear
B)
\[s{{p}^{3}}s{{p}^{2}}sp\,s{{p}^{3}}\]
done
clear
C)
\[s{{p}^{3}}s{{p}^{3}}s{{p}^{2}}s{{p}^{3}}\]
done
clear
D)
\[s{{p}^{3}}s{{p}^{2}}sp\text{ }s{{p}^{2}}\]
done
clear
View Answer play_arrow
question_answer 199) Helium is filled in balloons in place of hydrogen as it is
A)
non-inflammable
done
clear
B)
lighter than hydrogen
done
clear
C)
frequently available
done
clear
D)
radioactive
done
clear
View Answer play_arrow
question_answer 200) Hesss law states
A)
energy of formation of a compound does not depend on path
done
clear
B)
energy of formation of a compound depends on path
done
clear
C)
both (a) and (b)
done
clear
D)
none of the above
done
clear
View Answer play_arrow
question_answer 201) Heart sound DUB is caused due to closing of
A)
valve
done
clear
B)
tricuspid valve
done
clear
C)
semilunar valve
done
clear
D)
None of these
done
clear
View Answer play_arrow
question_answer 202) Ornithine cycle is found in
A)
liver
done
clear
B)
kidney
done
clear
C)
heart
done
clear
D)
muscles
done
clear
View Answer play_arrow
question_answer 203) Goitre is a pathological condition associated with:
A)
glucagon
done
clear
B)
thyroxine
done
clear
C)
progesterone
done
clear
D)
testosterone
done
clear
View Answer play_arrow
question_answer 204) Connecting link between reptiles and birds is:
A)
Dodo
done
clear
B)
Dimetrodon
done
clear
C)
Sphenodon
done
clear
D)
Archaeopteryx
done
clear
View Answer play_arrow
question_answer 205) Which one of the following is correct?
A)
all enzymes are proteins
done
clear
B)
all proteins are enzymes
done
clear
C)
some enzymes are proteins
done
clear
D)
all fats are enzymes
done
clear
View Answer play_arrow
question_answer 206) The site of fertilization in mammals is
A)
ovary
done
clear
B)
abdominal
done
clear
C)
uterus
done
clear
D)
fallopian tube
done
clear
View Answer play_arrow
question_answer 207) Natural parthenogenesis is found in?
A)
housefly
done
clear
B)
honey bee
done
clear
C)
Drosopiula
done
clear
D)
all of these
done
clear
View Answer play_arrow
question_answer 208) Sex hormones can work without the help of
A)
insulin
done
clear
B)
placenta
done
clear
C)
pituitary
done
clear
D)
gonadotropins
done
clear
View Answer play_arrow
question_answer 209) Mammals excrete
A)
urea
done
clear
B)
uric acid
done
clear
C)
ammonia
done
clear
D)
all of these
done
clear
View Answer play_arrow
question_answer 210) Correct sequence in cell type is
A)
\[{{\text{G}}_{\text{1}}}\text{,}{{\text{G}}_{\text{2,}}}\text{M}\text{.S}\]
done
clear
B)
\[{{\text{G}}_{\text{1}}}\text{,S,}{{\text{G}}_{\text{2,}}}\text{M}\]
done
clear
C)
\[{{\text{G}}_{\text{1}}}\text{,M,S,}{{\text{G}}_{1}}\]
done
clear
D)
\[\text{S,M,}{{\text{G}}_{1,}}{{G}_{2}}\]
done
clear
View Answer play_arrow
question_answer 211) Vestigeal organ in human being is
A)
incisor
done
clear
B)
canines
done
clear
C)
molar
done
clear
D)
premolar
done
clear
View Answer play_arrow
question_answer 212) Aerobic respiration is related with
A)
mitochondria
done
clear
B)
plasma membrane
done
clear
C)
golgi bodies
done
clear
D)
endoplasmic reticulum
done
clear
View Answer play_arrow
question_answer 213) The connection between two cells is
A)
plasmadesmata
done
clear
B)
microfilaments
done
clear
C)
sieve tube
done
clear
D)
none of these
done
clear
View Answer play_arrow
question_answer 214) Natural pearl is
A)
a mollusc
done
clear
B)
an annelid
done
clear
C)
an arthropod
done
clear
D)
an echinodermate
done
clear
View Answer play_arrow
question_answer 215) Salient feature of Arthropoda is
A)
aquatic and free living
done
clear
B)
chitinous exesrction and jointed appendeges
done
clear
C)
both a and b
done
clear
D)
none of these
done
clear
View Answer play_arrow
question_answer 216) Miller and Urey performed an experiment to prove the origin of life. They took gases \[N{{H}_{3}}\] and \[{{H}_{2}}\] along with
A)
\[{{N}_{2}}\] and \[{{H}_{2}}O\]
done
clear
B)
\[{{H}_{2}}O\] and \[C{{H}_{4}}\]
done
clear
C)
\[C{{H}_{4}}\] and \[{{N}_{2}}\]
done
clear
D)
\[C{{O}_{2}}\] and \[N{{H}_{3}}\]
done
clear
View Answer play_arrow
question_answer 217) Spermatogenesis is induced by
A)
FSH
done
clear
B)
ACTH
done
clear
C)
ICSH
done
clear
D)
ATH
done
clear
View Answer play_arrow
question_answer 218) Which one of the following is the most effective in Ascaris infection?
A)
chloroquin
done
clear
B)
Cinchona
done
clear
C)
Colchicum
done
clear
D)
oil of Chenopodium
done
clear
View Answer play_arrow
question_answer 219) Which one of the following is chemical transmitter for neuro impulse conduction?
A)
glycine
done
clear
B)
GABA
done
clear
C)
adrenalin
done
clear
D)
acetylcholine
done
clear
View Answer play_arrow
question_answer 220) During hibernation, frogs respiration is
A)
cutaneous
done
clear
B)
pulmonary
done
clear
C)
buccopharyngeal
done
clear
D)
pharyngeal
done
clear
View Answer play_arrow
question_answer 221) Blood carries \[C{{O}_{2}}\] mainly in which form?
A)
\[Hb.C{{O}_{2}}\]
done
clear
B)
\[NaHC{{O}_{3}}\]
done
clear
C)
carbonic acid
done
clear
D)
\[Hb.C{{O}_{2}}\] and carbon monoxide
done
clear
View Answer play_arrow
question_answer 222) Which one of the following is necessary to start clotting of blood?
A)
heparin
done
clear
B)
serotonin
done
clear
C)
thromboplastin and \[C{{a}^{++}}\]
done
clear
D)
fibrinogen and prothrombin
done
clear
View Answer play_arrow
question_answer 223) During oxidative phosphorylation, the net gain of ATP is
A)
40
done
clear
B)
38
done
clear
C)
34
done
clear
D)
30
done
clear
View Answer play_arrow
question_answer 224) Which of the following pairs is correct?
A)
annelida-polychaeta-leech
done
clear
B)
arthropoda-crustacea-cockroach
done
clear
C)
mollusca-cephalopoda-Octopus
done
clear
D)
protozoa-Hydra
done
clear
View Answer play_arrow
question_answer 225) Nerve impulse initiates with the movements of
A)
\[{{K}^{+}}\]
done
clear
B)
\[N{{a}^{+}}\]
done
clear
C)
\[C{{a}^{+}}\]
done
clear
D)
\[M{{g}^{+}}\]
done
clear
View Answer play_arrow
question_answer 226) Spindle fibres of a mitotic cells are made Up of
A)
actin
done
clear
B)
myosin
done
clear
C)
collagen
done
clear
D)
tubutin
done
clear
View Answer play_arrow
question_answer 227) The migrating birds rely on the
A)
anaerobic oxidation of proteins
done
clear
B)
highly efficient aerobic oxidation of fats
done
clear
C)
anaerobic oxidation of carbohydrates
done
clear
D)
all of these
done
clear
View Answer play_arrow
question_answer 228) Erythrocytes of adult rabbit and other mammals are formed in
A)
liver
done
clear
B)
spleen
done
clear
C)
kidney
done
clear
D)
red bone marrow
done
clear
View Answer play_arrow
question_answer 229) Pseudocoel is a cavity found in
A)
Hydra
done
clear
B)
Ascaris
done
clear
C)
housefly
done
clear
D)
sponge
done
clear
View Answer play_arrow
question_answer 230) Sarcolemma is a membrane found over
A)
heart
done
clear
B)
cardiac muscle
done
clear
C)
muscle fibre
done
clear
D)
nerve fibre
done
clear
View Answer play_arrow
question_answer 231) Heparin
A)
is antiserum
done
clear
B)
helps in clotting
done
clear
C)
helps in secretion
done
clear
D)
is anticoagulant
done
clear
View Answer play_arrow
question_answer 232) Trichocyst and nematocysts are meant for
A)
defense
done
clear
B)
nutrition
done
clear
C)
respiration
done
clear
D)
excretion
done
clear
View Answer play_arrow
question_answer 233) Digestion and distribution of nutrients both functions are performed by
A)
blastocoels
done
clear
B)
coelom
done
clear
C)
spongocoel
done
clear
D)
gastrovascular cavity
done
clear
View Answer play_arrow
question_answer 234) In mammals the digestion of starch starts from
A)
mouth
done
clear
B)
stomach
done
clear
C)
oesophagus
done
clear
D)
duodenum
done
clear
View Answer play_arrow
question_answer 235) In which of the following, Nissls granules are found?
A)
liver cells
done
clear
B)
nerve cells
done
clear
C)
intestinal cells
done
clear
D)
uriniferous tubules
done
clear
View Answer play_arrow
question_answer 236) In sponges, food is stored in
A)
porocytes
done
clear
B)
choanocytes
done
clear
C)
amoebocytes
done
clear
D)
thesocytes
done
clear
View Answer play_arrow
question_answer 237) Largest egg is of
A)
PPLO
done
clear
B)
Ostrich
done
clear
C)
Hydra
done
clear
D)
Periplanata Americana
done
clear
View Answer play_arrow
question_answer 238) Which one of the following is common in annelida and arthropods?
A)
basal nerve cord
done
clear
B)
ventral nerve cord
done
clear
C)
dorsal nerve cord
done
clear
D)
anterior nerve cord
done
clear
View Answer play_arrow
question_answer 239) When bath-sponges dries up, which of the following is left?
A)
spicules
done
clear
B)
tentacles
done
clear
C)
spongin fibres
done
clear
D)
none of these
done
clear
View Answer play_arrow
question_answer 240) Digestion of cellulose in non-ruminants takes place in
A)
caecum
done
clear
B)
colon
done
clear
C)
small intestine
done
clear
D)
large intestine
done
clear
View Answer play_arrow
question_answer 241) DNA is found in
A)
nucleus only
done
clear
B)
cytoplasm only
done
clear
C)
nucleus and cytoplasm
done
clear
D)
none of these
done
clear
View Answer play_arrow
question_answer 242) Cellular respiration occurs in
A)
nucleus
done
clear
B)
ribosomes
done
clear
C)
golgi body
done
clear
D)
mitochondria
done
clear
View Answer play_arrow
question_answer 243) Joint between humerus and ulna is
A)
saddle joint
done
clear
B)
hinge joint
done
clear
C)
fibrous joint
done
clear
D)
ball and socket
done
clear
View Answer play_arrow
question_answer 244) Who discovered that malaria is transmitted by mosquitoes?
A)
Grassi
done
clear
B)
Laveron
done
clear
C)
Lancisi
done
clear
D)
Ronald Ross
done
clear
View Answer play_arrow
question_answer 245) Which one of the following is a source of energy for muscle contraction
A)
actin
done
clear
B)
myosin
done
clear
C)
ATP
done
clear
D)
actomyosin
done
clear
View Answer play_arrow
question_answer 246) Tube feet are locomotary organs found in
A)
annelids
done
clear
B)
Mollusca
done
clear
C)
arthropods
done
clear
D)
echinoderms
done
clear
View Answer play_arrow
question_answer 247) Anti-A and Anti-B antibodies are not found in which of the following blood group?
A)
AB
done
clear
B)
O
done
clear
C)
A
done
clear
D)
B
done
clear
View Answer play_arrow
question_answer 248) Incubation period of Plasmodium vivax is
A)
4 days
done
clear
B)
6 days
done
clear
C)
14 days
done
clear
D)
30 days
done
clear
View Answer play_arrow
question_answer 249) Sympathetic nervous system induces
A)
heartbeat
done
clear
B)
secretion of semen
done
clear
C)
secretion of saliva
done
clear
D)
secretion of digestive juices
done
clear
View Answer play_arrow
question_answer 250) Organ of Corti is found in
A)
heart
done
clear
B)
kidneys
done
clear
C)
inner ear
done
clear
D)
nasal chamber
done
clear
View Answer play_arrow
question_answer 251) Apical dominance is caused by
A)
\[\text{I}\]AA
done
clear
B)
\[\text{I}\]BA
done
clear
C)
2, 4-D
done
clear
D)
2, 4, 5-T
done
clear
View Answer play_arrow
question_answer 252) Spore of Funnria, on germination, produces
A)
protonema
done
clear
B)
antheridia
done
clear
C)
archegonia
done
clear
D)
vegetative body
done
clear
View Answer play_arrow
question_answer 253) Plasmids are
A)
viral RNA in host cells
done
clear
B)
viral DNA in host cells
done
clear
C)
extra non-genetic element
done
clear
D)
extrachromosomal material of E.coli
done
clear
View Answer play_arrow
question_answer 254) During food chain, maximum energy is stored in
A)
producer
done
clear
B)
primary consumer
done
clear
C)
secondary consumer
done
clear
D)
decomposer
done
clear
View Answer play_arrow
question_answer 255) Cold treatment of seeds is called
A)
vernalization
done
clear
B)
stratification
done
clear
C)
devernalization
done
clear
D)
photophosphorylation
done
clear
View Answer play_arrow
question_answer 256) Bordered pits are generally found in
A)
dicots
done
clear
B)
monocots
done
clear
C)
gymnosperms
done
clear
D)
bryophytes
done
clear
View Answer play_arrow
question_answer 257) One gene-one enzyme hypothesis was given by
A)
Mendel
done
clear
B)
Darwin
done
clear
C)
Robert Hook
done
clear
D)
Beadle and Tatum
done
clear
View Answer play_arrow
question_answer 258) Cell wall of gram positive bacteria is made up of
A)
murein
done
clear
B)
cellulose
done
clear
C)
lipid and protein
done
clear
D)
cellulose and lipid
done
clear
View Answer play_arrow
question_answer 259) Which one of the following is a monocarpic plant?
A)
Pear
done
clear
B)
Citrus
done
clear
C)
Mango
done
clear
D)
Bambusa
done
clear
View Answer play_arrow
question_answer 260) Megasporophyll of Cycas is equivalent to
A)
stamen
done
clear
B)
sepal
done
clear
C)
petal
done
clear
D)
carpel
done
clear
View Answer play_arrow
question_answer 261) The term leptome is used for
A)
xylem
done
clear
B)
phloem
done
clear
C)
endodermis
done
clear
D)
pericycle
done
clear
View Answer play_arrow
question_answer 262) The formation of fruits without fertilization is called
A)
polyspory
done
clear
B)
parthenogenesis
done
clear
C)
polyembryony
done
clear
D)
parthenocarpy
done
clear
View Answer play_arrow
question_answer 263) An alga which is rich in proteins is
A)
Ulothrix
done
clear
B)
Spirogyra
done
clear
C)
Nostoc
done
clear
D)
Chlorella
done
clear
View Answer play_arrow
question_answer 264) Vivipary is seen in
A)
mangroves
done
clear
B)
xerophytes
done
clear
C)
hydrophytes
done
clear
D)
mesophytes
done
clear
View Answer play_arrow
question_answer 265) Which one of the following is a gymnosperm
A)
Mango
done
clear
B)
Walnut
done
clear
C)
Funaria
done
clear
D)
Chilgoza
done
clear
View Answer play_arrow
question_answer 266) Mycorrhiza helps in
A)
respiration
done
clear
B)
water absorption
done
clear
C)
nutrient absorption
done
clear
D)
phosphate absorption
done
clear
View Answer play_arrow
question_answer 267) Bracts enclosing a cluster of flowers are known as
A)
bracteate
done
clear
B)
involucre
done
clear
C)
petaloid
done
clear
D)
polysepalous
done
clear
View Answer play_arrow
question_answer 268) In Funaria, the number of peristomial teeth is
A)
6
done
clear
B)
10
done
clear
C)
16
done
clear
D)
32
done
clear
View Answer play_arrow
question_answer 269) The lateral roots originate from
A)
exodermis
done
clear
B)
endodermis
done
clear
C)
pericycle
done
clear
D)
vascular bundles
done
clear
View Answer play_arrow
question_answer 270) Role of calyx and corolla in fruit formation is
A)
promotory
done
clear
B)
ineffective
done
clear
C)
essential
done
clear
D)
all of these
done
clear
View Answer play_arrow
question_answer 271) Separation of linked genes is called
A)
linkage
done
clear
B)
segregation
done
clear
C)
crossing over
done
clear
D)
genetic mutation
done
clear
View Answer play_arrow
question_answer 272) Anti-viral substance is
A)
bacteria
done
clear
B)
antibody
done
clear
C)
antigen
done
clear
D)
interferon
done
clear
View Answer play_arrow
question_answer 273) The enzyme which is used for replication is
A)
DNA ase
done
clear
B)
aldol ase
done
clear
C)
RNA polymerase
done
clear
D)
DNA polymerase
done
clear
View Answer play_arrow
question_answer 274) \[{{C}_{4}}\]-cycle is found in
A)
fir
done
clear
B)
mango
done
clear
C)
sugarcane
done
clear
D)
none of these
done
clear
View Answer play_arrow
question_answer 275) RuBP is found in
A)
ETS
done
clear
B)
Calvin cycle
done
clear
C)
\[{{C}_{4}}\] plants
done
clear
D)
Krebs cycle
done
clear
View Answer play_arrow
question_answer 276) Incipient nucleus is found in
A)
myxophyceae
done
clear
B)
phaeophyceae
done
clear
C)
rhodophyceae
done
clear
D)
chlorophyceae
done
clear
View Answer play_arrow
question_answer 277) Last electron receptor in respiration is
A)
\[C{{O}_{2}}\]
done
clear
B)
\[{{O}_{2}}\]
done
clear
C)
\[{{H}_{2}}\]
done
clear
D)
NADH
done
clear
View Answer play_arrow
question_answer 278) Diadelphous condition is found in
A)
rosaceae
done
clear
B)
papilionaceae
done
clear
C)
leguminosae
done
clear
D)
cucurbitaceae
done
clear
View Answer play_arrow
question_answer 279) Which of the following algae is parasitic?
A)
Nostoc
done
clear
B)
Rhodomenia
done
clear
C)
Cephaleuros
done
clear
D)
Polysiphonia
done
clear
View Answer play_arrow
question_answer 280) Glycolysis occurs in
A)
grana
done
clear
B)
stroma
done
clear
C)
cytosol
done
clear
D)
mitochondria
done
clear
View Answer play_arrow
question_answer 281) Nucleosides have
A)
purine + pyrimidine
done
clear
B)
purine or pyrimidine + sugar
done
clear
C)
purine + sugar + phosphate
done
clear
D)
purine + pyrimidine + phosphate
done
clear
View Answer play_arrow
question_answer 282) Cladodes are found in
A)
Asparagus
done
clear
B)
Opuntia
done
clear
C)
Parkmsonia
done
clear
D)
Psidium guajava
done
clear
View Answer play_arrow
question_answer 283) R.Q. (respiratory quotient) is less than 1 in
A)
fat
done
clear
B)
glucose
done
clear
C)
fructose
done
clear
D)
organic acid
done
clear
View Answer play_arrow
question_answer 284) Blue-green algae has
A)
chlorophyll b
done
clear
B)
xanthophyll
done
clear
C)
phycocyanin
done
clear
D)
fucoxanthin
done
clear
View Answer play_arrow
question_answer 285) Connecting link between glycolysis and Krebs cycle is
A)
PGA
done
clear
B)
ketone
done
clear
C)
aldehyde
done
clear
D)
acetyl-Co A
done
clear
View Answer play_arrow
question_answer 286) Which of the following fibres is not a plant product?
A)
flax
done
clear
B)
cotton
done
clear
C)
hemp
done
clear
D)
silk
done
clear
View Answer play_arrow
question_answer 287) Which one of the following is not commercially produced by yeast?
A)
enzyme
done
clear
B)
vitamin
done
clear
C)
hormone
done
clear
D)
riboflavin
done
clear
View Answer play_arrow
question_answer 288) Alcoholic fermentation takes place in the presence of
A)
maltase
done
clear
B)
zymnse
done
clear
C)
amylase
done
clear
D)
invertase
done
clear
View Answer play_arrow
question_answer 289) Which one of the following does not grow in artificial media?
A)
TMV
done
clear
B)
bacteria
done
clear
C)
yeast
done
clear
D)
Rhizopus
done
clear
View Answer play_arrow
question_answer 290) The RNA like particle, which causes disease is
A)
virion
done
clear
B)
viroid
done
clear
C)
prion
done
clear
D)
mycoplasma
done
clear
View Answer play_arrow
question_answer 291) Phytochrome is found in
A)
algae
done
clear
B)
flowering plants
done
clear
C)
fungi
done
clear
D)
vascular cryptogams
done
clear
View Answer play_arrow
question_answer 292) Replication of DNA takes place during
A)
S-phase
done
clear
B)
prophase
done
clear
C)
metaphase
done
clear
D)
anaphase
done
clear
View Answer play_arrow
question_answer 293) Ribosomes are particles about \[200\overset{\text{o}}{\mathop{\text{A}}}\,\] units in diameter consisting of protein and RNA. The percentage of protein and RNA respectively is:
A)
60% and 40%
done
clear
B)
40% and 60%
done
clear
C)
80% and 20%
done
clear
D)
50% and 50%
done
clear
View Answer play_arrow
question_answer 294) Which of the following process takes place in mitochondria?
A)
photolysis
done
clear
B)
photophosphorylation
done
clear
C)
carboxylation
done
clear
D)
oxidative phosphorylation
done
clear
View Answer play_arrow
question_answer 295) Fluid mosaic model of cell membrane was given by
A)
Beadle and Tatum
done
clear
B)
Singer and Nicolson
done
clear
C)
Watson and Crick
done
clear
D)
Robertson and Miller
done
clear
View Answer play_arrow
question_answer 296) Late blight of potato is caused by
A)
Rhizopus
done
clear
B)
Ustilago
done
clear
C)
Altemnria solani
done
clear
D)
Phytopthorn infestens
done
clear
View Answer play_arrow
question_answer 297) Cell membrane is composed of
A)
protein
done
clear
B)
phospholipid
done
clear
C)
carbohydrate
done
clear
D)
lipid and potein
done
clear
View Answer play_arrow
question_answer 298) Binomial nomenclature was given by
A)
Darwin
done
clear
B)
Lamarck
done
clear
C)
Linnaeus
done
clear
D)
Theophrastus
done
clear
View Answer play_arrow
question_answer 299) Genetic material of virus is
A)
RNA
done
clear
B)
DNA
done
clear
C)
carbohydrate
done
clear
D)
either RNA or DNA
done
clear
View Answer play_arrow
question_answer 300) Suicidal bags are
A)
lysosomes
done
clear
B)
golgi bodies
done
clear
C)
ribosomes
done
clear
D)
chloroplast
done
clear
View Answer play_arrow