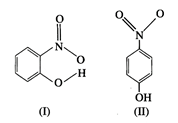
 (a) Which of the two compounds
will have intermolecular hydrogen bonding and which compound is expected to show
intramolecular hydrogen bonding?
(b) The melting point of a
compound depends on, among other things, the extent of hydrogen bonding. On
this basis explain which of the above two compounds will show higher melting
point?
(c) Solubility of compounds in
water depends on power toform hydrogen bonds with water. Which of the
abovecompounds will form hydrogen bond with water easilyand be more soluble in
it?
(a) Which of the two compounds
will have intermolecular hydrogen bonding and which compound is expected to show
intramolecular hydrogen bonding?
(b) The melting point of a
compound depends on, among other things, the extent of hydrogen bonding. On
this basis explain which of the above two compounds will show higher melting
point?
(c) Solubility of compounds in
water depends on power toform hydrogen bonds with water. Which of the
abovecompounds will form hydrogen bond with water easilyand be more soluble in
it?
Answer:
(a)
Intramolecular hydrogen bonding is observed in compound \[(I)\] because \[N{{O}_{2}}\]
and OH groups are nearer to each other.
In
compound \[(II)\] intermolecular hydrogen bonding is observed as different
molecules are involved.
(b)
Compound \[(II)\] will have higher melting point as it is an associated
species.
(c)
Compound \[(I)\] is less soluble than compound \[(II)\]. Compound \[(II)\]
easily forms hydrogen bonds with water.
You need to login to perform this action.
You will be redirected in
3 sec
