-
Which of the ores mentioned in Table 6.1 can be concentrated by magnetic separation method?
View Answer play_arrow
-
What is the significance of leaching in the extraction of aluminium?
View Answer play_arrow
-
The reaction, Cr2O3
+ 2Al  Al2O3
+ 2Cr (
Al2O3
+ 2Cr ( G°
= ?421 kJ) is thermodynamically feasible as is apparent from the Gibbs energy
value. Why does it not take place at room temperature?
G°
= ?421 kJ) is thermodynamically feasible as is apparent from the Gibbs energy
value. Why does it not take place at room temperature?
View Answer play_arrow
-
Is it true that under certain conditions, Mg can reduce SiO2 and Si can reduce MgO? What are those conditions?
View Answer play_arrow
-
Copper can be extracted by hydrometallurgy but not zinc. Explain.
View Answer play_arrow
-
What is the role of depreciation in froth floatation process?
View Answer play_arrow
-
Why is the extraction of copper from its pyrite difficult than that from its oxide through reduction?
View Answer play_arrow
-
Explain (i) zone refling (ii) column chromatography.
View Answer play_arrow
-
Out of C or CO, which is a better reducing agent at 673 K?
View Answer play_arrow
-
Name the common elements present in anode mud in the electro-refining of copper, which get collected as anode mud. Why are they so present?
View Answer play_arrow
-
Write the chemical reactions which take place in different zones in the blast furnace during the extraction of iron.
View Answer play_arrow
-
Write the chemical reactions taking place in the extraction of zinc from zinc blende.
View Answer play_arrow
-
State the role of silica in the metallurgy of copper.
View Answer play_arrow
-
What is meant by the term chromatography?
View Answer play_arrow
-
What criterion is followed for the selection of the stationary phase in chromatography?
View Answer play_arrow
-
Describe a method for the refining of nickel.
View Answer play_arrow
-
How can you separate alumina from silica in a bauxite ore associated with silica? Give equations if any.
View Answer play_arrow
-
Giving examples differentiate between calcinations and roasting.
View Answer play_arrow
-
How is 'cast iron' different from 'pig iron'?
View Answer play_arrow
-
Differentiate between minerals and ores.
View Answer play_arrow
-
Why is copper matte put in silica lined converter?
View Answer play_arrow
-
What is the role of cryolite in the metallurgy of aluminium?
View Answer play_arrow
-
How is leaching carried out in case of low grade copper?
View Answer play_arrow
-
Why is zinc not extracted from zinc oxide through reduction using CO?
View Answer play_arrow
-
How
much of 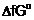 for
the formation of Cr2O3 is ?540 kJ mol?1 and
that of Al2O3 is ?827 kJ mol?1 Is the
reduction The value of Cr2O3 possible with Al ?
for
the formation of Cr2O3 is ?540 kJ mol?1 and
that of Al2O3 is ?827 kJ mol?1 Is the
reduction The value of Cr2O3 possible with Al ?
View Answer play_arrow
-
Out of C and CO, which is a better reducing agent for ZnO?
View Answer play_arrow
-
The choice of a reducing agent in a particular case depends on the thermodynamic factors. How do you agree with this statement? Support your opinion with two examples.
View Answer play_arrow
-
Name the processes from which chlorine is obtained as the by- product. What will happen if an aqueous solution of NaCl is subjected to electrolysis?
View Answer play_arrow
-
What is the role of graphite rod in the electrometallurgy of aluminium?
View Answer play_arrow
-
Outline the principles of refining of metals by following methods :
(i) Zone refining
(ii) Electrolytic refining
(iii) Vapour phase refining.
View Answer play_arrow
-
Predict the conditions under which aluminium might be expected to reduce magnesium oxide.
View Answer play_arrow
-
question_answer32)
In the extraction of
chlorine by electrolysis of brine............
(a) oxidation of 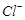 ion to chlorine gas occurs
(b) reduction
of
ion to chlorine gas occurs
(b) reduction
of 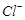 ion to chlorine gas occurs
(C) for
overall reaction
ion to chlorine gas occurs
(C) for
overall reaction 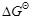 has negative value
(D) a displacement
reaction takes place
has negative value
(D) a displacement
reaction takes place
View Answer play_arrow
-
question_answer33)
When copper ore is mixed
with silica, in a reverberatory furnace copper matte is produced. The copper
matte contains
(a) sulphides of
copper (II) and iron (II)
(b) sulphides
of copper (11) and iron (111)
(c) sulphides
of copper (I) and iron (II)
(d) sulphides of
copper (I) and iron (III)
View Answer play_arrow
-
question_answer34)
Which of the following
reactions is an example of autoreduction?
(a) 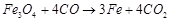 (b)
(b) 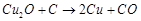 (c)
(c) 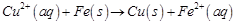 (d)
(d) 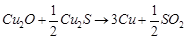
View Answer play_arrow
-
question_answer35)
A number of elements are
available in earth's crust but most abundant elements are..............
(a) Al and
Fe (b) Al and Cu
(c) Fe and
Cu (d) Cu and Ag
View Answer play_arrow
-
question_answer36)
Zone refining is based on
the principle that..........
(a) impurities of
low boiling metals can be separated by distillation.
(b) impurities
are more soluble in molten metal than in solid metal.
(c) different
components of a mixture are differently adsorbed on an adsorbent.
(d) vapours of
volatile compound can be decomposed in pure metal.
View Answer play_arrow
-
question_answer37)
In the extraction of copper
from its sulphide ore, the metal is formed by the reduction of 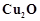 with
(a)
with
(a) 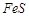 (b)
CO
(c)
(b)
CO
(c) 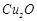 (d)
(d) 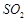
View Answer play_arrow
-
question_answer38)
Brine is electrolysed by
using inert electrodes. The reaction at anode is.........
(a)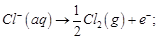
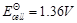 (b)
(b) 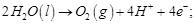
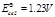 (c)
(c) 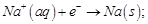
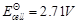 (d)
(d) 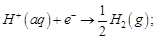
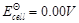
View Answer play_arrow
-
question_answer39)
In the metallurgy of aluminium............
(a) 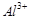 is oxidised to Al (s).
(b) graphide
anode is oxidised to carbon monoxide and carbon dioxide.
(c) oxidation
state of oxygen changes in the reaction at anode.
(d) oxidation state
of oxygen changes in the overall reaction involved in the process.
is oxidised to Al (s).
(b) graphide
anode is oxidised to carbon monoxide and carbon dioxide.
(c) oxidation
state of oxygen changes in the reaction at anode.
(d) oxidation state
of oxygen changes in the overall reaction involved in the process.
View Answer play_arrow
-
question_answer40)
Electrolytic refining is used
to purify which of the following metals?
(a) Cu and Zn (b)
Ge and Si
(c) Zr and Ti (d)
Zn and Hg
View Answer play_arrow
-
question_answer41)
Extraction of gold and silver
involves leaching the metal with 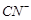 ion. The metal
is recovered by......
(a) displacement of
metal by some other metal from the complex ion.
(b) roasting
of metal complex.
(c) calcination
followed by roasting.
(d) thermal
decomposition of metal complex.
ion. The metal
is recovered by......
(a) displacement of
metal by some other metal from the complex ion.
(b) roasting
of metal complex.
(c) calcination
followed by roasting.
(d) thermal
decomposition of metal complex.
View Answer play_arrow
-
question_answer42)
Choose the correct option of
temperature at which carbon reduces 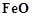 to
iron and produces CO.
(a) Below
temperature at point A
(b) Approximately
at the temperature corresponding to point A
(c) Above temperature at point A but
below temperature at point D
(d) Above
temperature at point A
to
iron and produces CO.
(a) Below
temperature at point A
(b) Approximately
at the temperature corresponding to point A
(c) Above temperature at point A but
below temperature at point D
(d) Above
temperature at point A
View Answer play_arrow
-
question_answer43)
Below point 'A' 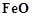 can.............
(a) be reduced by
carbon monoxide only.
(b) be
reduced by both carbon monoxide and carbon.
(c) be
reduced by carbon only.
(d) not be reduced
by both carbon and carbon monoxide.
can.............
(a) be reduced by
carbon monoxide only.
(b) be
reduced by both carbon monoxide and carbon.
(c) be
reduced by carbon only.
(d) not be reduced
by both carbon and carbon monoxide.
View Answer play_arrow
-
question_answer44)
For the reduction of 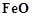 at the temperature
corresponding to point D, which of the following statements is correct?
(a) AC value for the
overall reduction reaction with carbon monoxide is zero.
(b) AC value
for the overall reduction reaction with a mixture of 1 mol carbon and 1 mol
oxygen is positive.
(c) AC value
for the overall reduction reaction with a mixture of 2 mol carbon and 1 mol
oxygen will be positive.
(d) AC value for the
overall reduction reaction with carbon monoxide is negative.
at the temperature
corresponding to point D, which of the following statements is correct?
(a) AC value for the
overall reduction reaction with carbon monoxide is zero.
(b) AC value
for the overall reduction reaction with a mixture of 1 mol carbon and 1 mol
oxygen is positive.
(c) AC value
for the overall reduction reaction with a mixture of 2 mol carbon and 1 mol
oxygen will be positive.
(d) AC value for the
overall reduction reaction with carbon monoxide is negative.
View Answer play_arrow
-
question_answer45)
At the temperature
corresponding to which of the points in Fig. 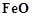 will
be reduced to Fe by coupling the reaction
will
be reduced to Fe by coupling the reaction 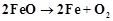 with
all of the following reactions?
1.
with
all of the following reactions?
1. 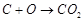 2
2 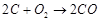 3.
3. 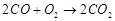 (a) Point A
(b) Point B
(c) Point D (d)
Point £
(a) Point A
(b) Point B
(c) Point D (d)
Point £
View Answer play_arrow
-
question_answer46)
Which of the following
options are correct?
(a) Cast iron is
obtained by remelting pig iron with scrap iron and coke using hot air blast.
(b) In
extraction of silver, silver is extracted as cationic complex.
(c) Nickel
is purified by zone refining.
(d) Zr and Ti are
purified by van Arkel method.
View Answer play_arrow
-
question_answer47)
In the extraction of
aluminium by Hall - Heroult process, purified 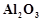 is
mixed with
is
mixed with 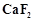 to
(a) lower the
melting point of
to
(a) lower the
melting point of 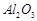 (b) increase
the conductivity of molten mixture.
(c) reduce
(b) increase
the conductivity of molten mixture.
(c) reduce 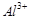 into Al(5)
(d) acts as catalyst
into Al(5)
(d) acts as catalyst
View Answer play_arrow
-
question_answer48)
Which of the following
statements is correct about the role of substances added in the froth
floatation process?
(a) Collectors
enhance the non-wettability of the mineral particles.
(b) Collectors
enhance the wettability of gangue particles.
(c) By using
depressants in the process two sulphide ores can be separated.
(d) Froth
stabilisers decrease wettability of gangue.
View Answer play_arrow
-
question_answer49)
In the froth floatation
process, zinc sulphide and lead sulphide can be separated by...............
(a) using
collectors
(b)
adjusting the proportion of oil to water
(c) using
depressant
(d) using froth
stabilisers
View Answer play_arrow
-
question_answer50)
Common impurities present in
bauxite are........
(a) 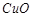 (b)
(b) 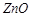 (c)
(c) 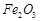 (d)
(d) 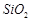
View Answer play_arrow
-
question_answer51)
Which of the following ores are
concentrated by froth floatation?
(a) Haematite (b)
Galena
(c) Copper pyrites (d)
Magnetite
View Answer play_arrow
-
question_answer52)
Which of the following
reactions occur during calcination?
(a) 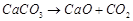 (b)
(b) 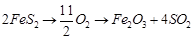 (c)
(c) 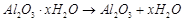 (d)
(d) 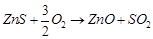
View Answer play_arrow
-
question_answer53)
For the metallurgical
process of which of the ores calcined ore can be reduced by carbon?
(a) Haematite (b)
Calamine
(c) Iron pyrites
(d) Sphalerite
View Answer play_arrow
-
question_answer54)
The main reactions occurring
in blast furnace during extraction of iron from haematite ore ..........
(a) 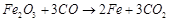 (b)
(b) 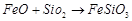 (c)
(c) 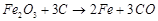 (d)
(d) 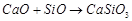
View Answer play_arrow
-
question_answer55)
In which of the following
method of purification, metal is converted to its volatile compound which is
decomposed to give pure metal?
(a) Heating with
stream of carbon monoxide
(b) Heating
with iodine
(c) Liquation
(d) Distillation
View Answer play_arrow
-
question_answer56)
Which of the following
statements are correct?
(a) A depressant
prevents certain type of particle to come to the froth.
(b) Copper
matte contains 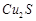 and
and 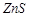 .
(c) The
solidified copper obtained from reverberatory furnace has blistered appearance
due to evolution of
.
(c) The
solidified copper obtained from reverberatory furnace has blistered appearance
due to evolution of 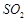 during the extraction.
(d) Zinc can be
extracted by self-reduction.
during the extraction.
(d) Zinc can be
extracted by self-reduction.
View Answer play_arrow
-
question_answer57)
In the extraction of
chlorine from brine............
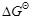 for the overall reaction is
negative.
for the overall reaction is
negative.
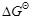 for the overall reaction is
positive.
for the overall reaction is
positive.
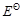 for the overall reaction has
negative value.
for the overall reaction has
negative value.
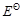 for the overall reaction has
positive value.
for the overall reaction has
positive value.
View Answer play_arrow
-
question_answer58)
Why is an external emf of
more than 2.2V required for the extraction of 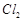 from brine?
from brine?
View Answer play_arrow
-
question_answer59)
At temperature above 1073 K,
coke can be used to reduce 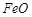 to Fe. How
can you justify this reduction with Ellingham diagram?
to Fe. How
can you justify this reduction with Ellingham diagram?
View Answer play_arrow
-
question_answer60)
Wrought iron is the purest form
of. iron. Write a reaction used for the preparation of wrought iron from cast iron.
How can the impurities of
sulphur, silicon and
phosphorus be removed from cast iron?
View Answer play_arrow
-
question_answer61)
How is copper extracted from
low grade copper ores?
View Answer play_arrow
-
question_answer62)
Write two basic requirements
for refining of a metal by Mond's process and by van Arkel Method.
View Answer play_arrow
-
question_answer63)
Although carbon and hydrogen
are better reducing agents but they are not used to reduce metallic oxides at
high temperatures. Why?
View Answer play_arrow
-
question_answer64)
How do we separate two
sulphide ores, by froth floatation method?
Explain with an
example.
View Answer play_arrow
-
question_answer65)
The purest form of iron is
prepared by oxidising impurities from cast iron in a reverberatory furnace.
Which iron ore is used to line the furnace?
Explain by giving
reaction.
View Answer play_arrow
-
question_answer66)
The mixture of compounds A
and B is passed through a column of 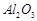 by
using alcohol as eluant. Compound A is eluted in preference to
compound B. Which
of the compounds A or B, is more readily adsorbed on the column?
by
using alcohol as eluant. Compound A is eluted in preference to
compound B. Which
of the compounds A or B, is more readily adsorbed on the column?
View Answer play_arrow
-
question_answer67)
Why is sulphide ore of
copper heated in a furnace after mixing with silica?
View Answer play_arrow
-
question_answer68)
Why are sulphide ores
converted to oxide before reduction?
View Answer play_arrow
-
question_answer69)
Which method is used for
refining Zr and Ti? Explain with equation.
View Answer play_arrow
-
question_answer70)
What should be the
considerations during the extraction of metals by electrochemical method?
View Answer play_arrow
-
question_answer71)
What is the role of flux in
metallurgical processes?
View Answer play_arrow
-
question_answer72)
How are metals used as
semiconductors refined? What is the principle of the method used like
germanium, silicon etc?
View Answer play_arrow
-
question_answer73)
Write down the reactions
taking place in blast furnace related to the metallurgy of iron in the
temperature range 500-800 K.
View Answer play_arrow
-
question_answer74)
Give two requirements for
vapour phase refining.
View Answer play_arrow
-
question_answer75)
Write the chemical reactions
involved in the extraction of gold by cyanide process. Also give the role of
zinc in the extraction.
View Answer play_arrow
-
question_answer76)
Match the items of Column I
with items of Column II and assign the correct code.
|
|
Column 1
|
Column 11
|
|
A. Pendulum
B. Malachite
C. Calamine
D. Cryolite
|
1. Chrome steel
2. Nickel steel
3. 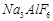 4.
4. 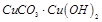 5.
5. 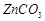
|
Codes
A B
C D A B C D
(a) 1 2 3
4 (b) 2 4 5 3
(c) 2 3 4
5 (d) 4 5 3 2
View Answer play_arrow
-
question_answer77)
Match the items of Column I
with the items of Column II and assign the correct code.
|
Column 1
|
Column 11
|
|
A. Coloured bands
B. Impure metal to
volatile complex
C. Purification of Ge
and Si
D. Purification of
mercury
|
1. Zone refining
2. Fractional
distillation
3. Mond's process
4. Chromatography
5. Liquation
|
Codes
A B
C D A B C D
(a) 1 2 4
5 (b) 4 3 1 2
(c) 3 4 2
1 (d) 5 4 3 2
View Answer play_arrow
-
question_answer78)
Match items of Column I with
the items of Column II .and assign the correct code.
|
Column 1
|
Column 11
|
|
A. Cyanide process
|
1. Ultrapure Ge
|
|
B. Froth floatation
process
|
2. Dressing of ZnS
|
|
C. Electrolytic
reduction
|
3. Extraction of Al
|
|
D. Zone refining
|
4. Extraction of Au
|
|
|
5. Purification of Ni
|
Codes
A B
C D A B C D
(a) 4 2
3 1 (b) 2 3 1 5
(c) 1 2 3
4 (d) 3 4 5 1
View Answer play_arrow
-
question_answer79)
Match the items of Column I
with the items of Column II and assign the correct code.
|
|
Column 1
|
Column 11
|
|
A.
|
Sapphire
|
1. 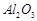
|
|
B.
|
Sphalerite
|
2. 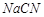
|
|
C.
|
Depressant
|
3. Co
|
|
D.
|
Corundum
|
4. ZnS
|
|
|
|
5. 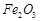
|
Codes
A B
C D A B C D
(a) 3 4 2
1 (b) 5 4 3 2
(b) 2 3 4
5 (d) 1 2 3 4
View Answer play_arrow
-
question_answer80)
Match the items of Column I
with items of Column II and assign the correct code.
|
Column I
|
Column II
|
|
A. Blisterred Cu
B. Blast
furnace
C. Reverberatory
D. Hall ? Heroult
process
|
1. Aluminium
2. 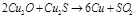
|
3. Iron
4.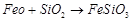 5.
5. 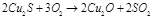
|
Codes
A B C
D A B C D
(a) 2 3
4 1 (b) 1 2 3 5
(c) 5 4 3 2
(d) 4 5 3 2
View Answer play_arrow
-
question_answer81)
Assertion (A) Nickel can
be purified by Mond's process.
Reason (R) Ni
(00)4 is a volatile compound which decomposes at 460 K to give pure Ni.
View Answer play_arrow
-
question_answer82)
Assertion (A) Zirconium
can be purified by van Arkel method.
Reason (R) 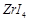 is volatile and decomposes at
1800K.
is volatile and decomposes at
1800K.
View Answer play_arrow
-
question_answer83)
Assertion (A) Sulphide
ores are concentrated by froth flotation method.
Reason (R)
Cresols stabilise the froth in froth floatation method.
View Answer play_arrow
-
question_answer84)
Assertion (A) Zone
refining method is very useful for producing semiconductors.
Reason (R)
Semiconductors are of high purity.
View Answer play_arrow
-
question_answer85)
Assertion (A) Hydro
metallurgy involves dissolving the ore in a suitable reagent followed by
precipitation by a more electropositive metal.
Reason (R)
Copper is extracted by hydro metallurgy.
View Answer play_arrow
-
question_answer86)
Explain the following
(a) 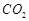 is a better reducing agent below
710 K whereas CO is a better reducing agent above 710 K.
(b)
Generally sulphide ores are converted into oxides before reduction.
(c) Silica
is added to the sulphide ore of copper in the reverberatory furnace.
(d) Carbon
and hydrogen are not used as reducing agents at high temperatures.
(e) Vapour phase
refining method is used for the purification of Ti.
is a better reducing agent below
710 K whereas CO is a better reducing agent above 710 K.
(b)
Generally sulphide ores are converted into oxides before reduction.
(c) Silica
is added to the sulphide ore of copper in the reverberatory furnace.
(d) Carbon
and hydrogen are not used as reducing agents at high temperatures.
(e) Vapour phase
refining method is used for the purification of Ti.
View Answer play_arrow
![]() (b)
(b) ![]() (c)
(c) ![]() (d)
(d) ![]()
![]() (b)
(b) ![]() (c)
(c) ![]() (d)
(d) ![]()
![]() (b)
(b) ![]() (c)
(c) ![]() (d)
(d) ![]()
