Answer:
'Williamson's
synthesis occurs by ![]() mechanism in
which sodium alkoxide reacts with an alkyi halide.
Now to prepare di-tert-butyl ether, sodium fert-butoxide must be reacted
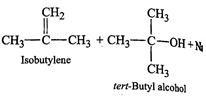
with tert-butyl bromide. Since 3° alkyi halides prefer to undergo elimination
rather than substitution, therefore, sodium fert-butoxid. ?S with tert-butyl
bromide and favours elimination to form isobutylene rather than substitution to
form di-teri- butyl ether
mechanism in
which sodium alkoxide reacts with an alkyi halide.
Now to prepare di-tert-butyl ether, sodium fert-butoxide must be reacted
with tert-butyl bromide. Since 3° alkyi halides prefer to undergo elimination
rather than substitution, therefore, sodium fert-butoxid. ?S with tert-butyl
bromide and favours elimination to form isobutylene rather than substitution to
form di-teri- butyl ether
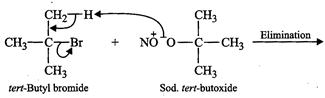
 Tert-Butyl bromide + Sod. Tert-butoxide
Tert-Butyl bromide + Sod. Tert-butoxide
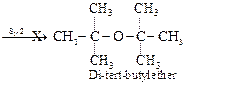
You need to login to perform this action.
You will be redirected in
3 sec
