Answer:
(i) Carbylamine reactionWhen aliphatic and aromatic primary
amines are heated with chloroform, and ethanolicpotassium hydroxide,
carbylamines or isocyanides are formed.These substances produced have foul
smell, Secondary and tertiary amines do not give this reaction. This reaction
is used todetect primary amines.
![]() (ii) Diazotization Aromatic
amines react with nitrous acid at lowtemperatures (273-278K) to form diazonium
salts. This reaction iscalled diazotization reaction.
(ii) Diazotization Aromatic
amines react with nitrous acid at lowtemperatures (273-278K) to form diazonium
salts. This reaction iscalled diazotization reaction.
![]()
 Note When primary
aliphatic amines react with nitrous acid, aliphaticdiazonium salts are formed
(Unstable), which liberate nitrogen gas and alcohols.
Note When primary
aliphatic amines react with nitrous acid, aliphaticdiazonium salts are formed
(Unstable), which liberate nitrogen gas and alcohols.
![]()
![]() (iii) Hofmann's bromamide
reactionThis is a method used forconverting primary amide into primary
amine.
An amide is treated with bromine
in an aqueous or ethanolicsolution of sodium hydroxide. The amine so formed
contains onecarbon less than that present in the amide.
(iii) Hofmann's bromamide
reactionThis is a method used forconverting primary amide into primary
amine.
An amide is treated with bromine
in an aqueous or ethanolicsolution of sodium hydroxide. The amine so formed
contains onecarbon less than that present in the amide.
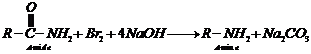
![]() e.g.,
e.g.,![]()
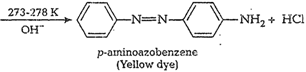
![]() (iv) Coupling reaction When diazonium salts
react with phenols oraromatic amines they form azo compounds of general
formula,
(iv) Coupling reaction When diazonium salts
react with phenols oraromatic amines they form azo compounds of general
formula,![]() . This reaction is called
coupling reaction. Thereaction takes place in alkaline medium (pH 9 to 10) e.g.
e.g.,
(a)
. This reaction is called
coupling reaction. Thereaction takes place in alkaline medium (pH 9 to 10) e.g.
e.g.,
(a)
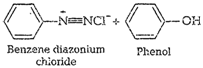
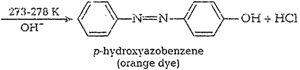 (b)
(b)
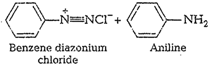
 (v) Ammonolysis The process of cleavage
of C?X bond by ammonia molecule is known as ammonolysis.
(v) Ammonolysis The process of cleavage
of C?X bond by ammonia molecule is known as ammonolysis.

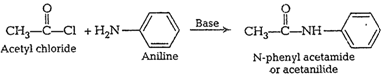
![]() (vi) AcetylationThe
process in which acetyl
(vi) AcetylationThe
process in which acetyl ![]() group isintroduced in a
molecule, is called acetylation, Reagents used forthis purpose are acetyl
chloride or acetic anhydride.
group isintroduced in a
molecule, is called acetylation, Reagents used forthis purpose are acetyl
chloride or acetic anhydride.
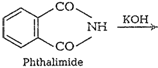
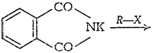
![]() (vii)
Gabriel phthalimide synthesisThis method is used for thepreparation of
aliphatic primary amines. Phthalimide on reactionwith ethanolic potassium
hydroxide forms potassium salt ofphthalimide. This potassium salt on heating
with alkyl halidefollowed by alkaline hydrolysis produces primary amine.
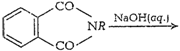
(vii)
Gabriel phthalimide synthesisThis method is used for thepreparation of
aliphatic primary amines. Phthalimide on reactionwith ethanolic potassium
hydroxide forms potassium salt ofphthalimide. This potassium salt on heating
with alkyl halidefollowed by alkaline hydrolysis produces primary amine.
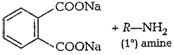
You need to login to perform this action.
You will be redirected in
3 sec
