Answer:
A reaction in which one reactant is
present in large amount and its concentration does not get altered during the
course of the reaction, behaves as first order reaction. Such reaction
is called pseudo first order reaction.
e.g., (i) hydrolysis of ethyl
acetate
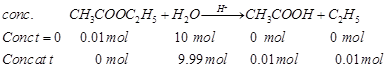 Rate of reaction =
Rate of reaction = ![]() where,
where, ![]() e.g., (ii) inversion of cane
sugar
e.g., (ii) inversion of cane
sugar
![]() Rate of reaction =
Rate of reaction = ![]() where
where ![]()
You need to login to perform this action.
You will be redirected in
3 sec
