Answer:
(a,
b)
Structure or ![]() is as shown
below
is as shown
below
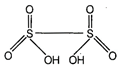 It contains one S?S bond.
(b) In peroxosulphuric acid
It contains one S?S bond.
(b) In peroxosulphuric acid ![]() sulphur
is in + 6 oxidation state.
Structure of g is
sulphur
is in + 6 oxidation state.
Structure of g is![]()
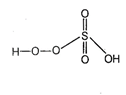 Let oxidation, state of S = x
2 x ( + 1) + x + 3 x (-2) + 2 x (-1) = 0
x - 6 = 0
x = 6
(c) During preparation of ammonia, iron oxide with small
amount of
Let oxidation, state of S = x
2 x ( + 1) + x + 3 x (-2) + 2 x (-1) = 0
x - 6 = 0
x = 6
(c) During preparation of ammonia, iron oxide with small
amount of ![]() and
and ![]() is
used
as a catalyst to increase the rate of attainment of
equilibrium.
(d) Change in enthalpy is negative for preparation of SO3
by catalytic oxidation of SO2.
is
used
as a catalyst to increase the rate of attainment of
equilibrium.
(d) Change in enthalpy is negative for preparation of SO3
by catalytic oxidation of SO2.
You need to login to perform this action.
You will be redirected in
3 sec
