-
Classify the following as primary,
secondary and tertiary alcohols.
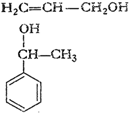
(i)
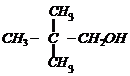
(ii)
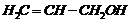
(iii)
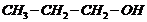
(iv)
(v) (vi)
If
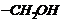
group
is present, alcohol is
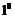
(primary),
if
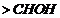
group
is present is
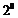
(secondary) and if
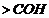
group
is present, alcohol is
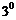
.
So classify on this basis.
-
Identify allylic alcohols in the above
examples.
Following groups are included in
allylic alcohols.
-
Name the following compounds according to the IUPAC system.
(i)
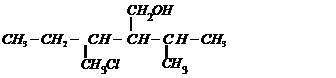
(ii)
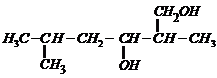
(iii)
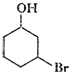
(iv)
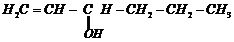
(v)
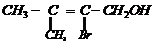
Prefix according to the C-chain;
for alcohol add suffix ?ol; for double bond ?ene; tell the position of
substituents, double bond and functional group (s).Then, given the name in the
following manner.
Name of substituent with position (in
alphabetical order) + parent C-chain name + primary suffix (antiene etc.) +
secondary suffix with position (for main functional group).
-
Show how are the following alcohols prepared by the reaction
of a suitable Grignard reagent on methanol?
(i)
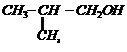
(ii)
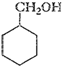
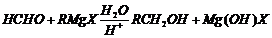
. From h
here, it is clear that HCHO gives
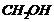
groups,
so R of Grignard reagent is the remaining part of given alcohols. Thus, select
the suitable Grignard reagent by substituting the value of R.
-
Write structures of the products of the
following reaction.
(i)
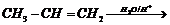
(ii)
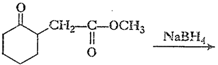
(iii)
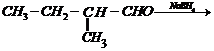
(i) Acidic hydration of propene
i.e., addition of
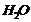
(according
to Markownikoff?s rule) result in the formation of alcohol.
(ii) Reduction of
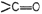
group (not is
ester group) into ?OH.
(iii) Reduction of ?CHO group into
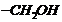
.
-
Give structure of the products you would expect when each
of the following alcohol reacts with
(a)
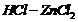
(b)
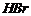
and
(c)
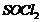
(i) Butan-1-ol (ii)
2-methylbutan-2-ol
Replacement of ?OH by (a)
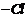
(b) ?Br
(c)
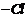
-
Predict the major product of acid catalyzed dehydration of
(i) 1-methylcyclohexanol (ii) butan-1-ol
In acid catalyzed dehydration process a molecule of water is liberatedaccording to Saytzeff'srule i.e., results in the formation of moresubstituted alkene.
View Answer play_arrow
-
Ortho and para nitrophenols are more acidic than phenol.Draw the resonance structures of the corresponding phenoxide ions.
Acidic strength depenas on the relative stabilities of correspondingphenoxide ions based on resonance.
View Answer play_arrow
-
Write the
equations involved in the following reactions :
(i)
Reimer-Tiemann reaction
(ii)
Kolbe's reaction
(i) It
is the reaction of phenol with chloroform in the presence of sodium hydroxide,
As a result, 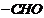 group is
introduced at o-position of benzene ring.
(ii) Reaction
of phenol and
group is
introduced at o-position of benzene ring.
(ii) Reaction
of phenol and 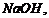 then
with
then
with 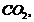 a weak
electrolyte orthooenzoic acid is formed.
a weak
electrolyte orthooenzoic acid is formed.
View Answer play_arrow
-
Write the reactions of Williamson synthesis of2-ethoxy-3-methyipentane starting from ethanol and 3-methyl pentan-2-ol.
(i) Reaction of3-methylpentan-2-ol with sodium.
(ii) Reaction of ethanol with hydrogen bromide.
(iii) Reaction of product formed in (i) reaction with the product of (ii).
View Answer play_arrow
-
Which of the following is an appropriate
set of reactants for the preparation of 1-methoxy-4-nitrobenzene and why?
(i)
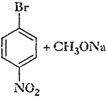
(ii)
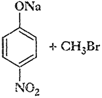
This reaction involves preparation of
ether by Williamson synthesis it takes place by
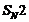
attack
of an alkoxide ion on primary alkyl halide.
-
Predict the products of the following
reactions.
(i)
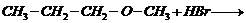
(ii)
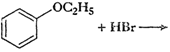
(iii)
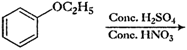
(iv)
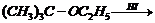
(i) and (ii)
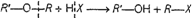
i.e.,
primary halide is formed.
(iii) Nitration (introduction of
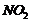
group)
takes place at o- and p-positions.
(iv) Tertiary iodide and primary alcohol
are formed.
-
1.
Write IUPAC names of the following compounds.
(i)
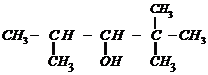
(ii)
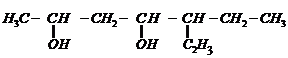
(iii)
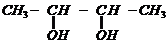
(iv)
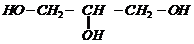
(v)
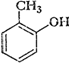
(vi)

(vii)
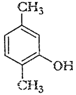
(viii)
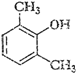
(ix)
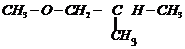
(x)
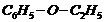
(xi)
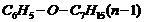
(xii)
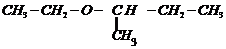
Prefix according to the carbon
chain: for alcohol (?OH) use suffix ?ol; tell the position of substitutents and
functional group. For ether (?O?), prefix alkoxy is used. For ?OH group
attached directly to benzene ring,
suffix phenol is used.
So, give the name of alcohol as substituent
with position (In alphabetical order) + prefix of carbon chain + prefix of
carbon chain + primary suffix (ane, ene, yne) + ?ol? with position and give the
name of other as alkoxy alkane (where alkoxy smaller R group and alkane is main
chain)
-
Write structures of the compounds whose IUPAC names are as follows.
(i) 2-methylbutan-2-ol (ii) 1-phenylpropan-2-ol
(iii) 3, 5-dimethylhexane-1, 3, 5-triol (iv) 2, 3-diethylphenol
(v) 1-ethoxypropane (vi) 2-ethoxy-3-methylpentane
(vii) Cyclohexylmethanol (viii) 3-cyclohexylpentan-3-ol
(ix) Cyclopent-3-en-1-ol (x) 3-chloromethylpentan-1-ol
(i) Draw the main carbon chain according to the root word of carbon chain and then attach the other groups according to the positions mentioned.
(ii) Suffix ‘al’ represents -OH group and prefix 'alkoxy' represents 'RO'croup.
View Answer play_arrow
-
(i) Draw the structures of all isomeric
alcohols of molecular formula
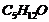
and give
their IUPAC names.
(ii) Classify the isomers of
alcohols in question 3. (i) as primary, secondary arid tertiary alcohols.
(i) Make different isomers by
varying the number or C-atoms in continuous chain, the position of
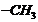
and
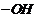
groups.
(ii) Primary
alcohols
have
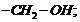
group
Secondary (2°) have group
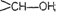
and Tertiary (3°)
have
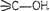
,
-
Explain why propanol has higher boiling point than that ofthe hydrocarbon, butane?
Boiling point is directly proportional to the intermolecular forces existing in a compound.
View Answer play_arrow
-
Alcohols are comparatively more soluble in water thanhydrocarbons of comparable molecular masses. Explain this fact.
Consider polar nature of water as well as alcohol; presence of hydrogen bonding.
View Answer play_arrow
-
What is meant by hydroboration-oxidation reaction?Illustrate it with an example.
Addition of diborane to alkenes followed by oxidation.
View Answer play_arrow
-
Give the structures and IUPAC names of
monohydric phenols of molecular formula,
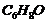
.
Ortho (1, 2 ); para (1, 4); meta (1, 3)
Draw these isomers according to the mentioned positions of ?OH and
groups.
-
While separating a mixture ofortho and para nitrophenols bysteam distillation, name the isomer which will be steam volatile. Give reason.
Isomer, which is stema volatiles less strongly bonded. The other one is strongly bonded by H-bonding.
View Answer play_arrow
-
Give the equations of reactions for the preparation of phenol from cumence.
Oxidation of cumece into cumenehydroperoxide, then acidic hydrolysis.
View Answer play_arrow
-
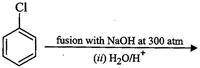
Write chemical reaction for the preparation of phenol from chlorobenzene.
Reaction of chlorobenzene with sodium hydroxide folio-wed byacidification of the product formed.
View Answer play_arrow
-
Write the mechanism of hydration of ethane to yield ethanol.
The steps involved in this process are
(1) Protonation of alkene.
(2) Nucleophilic attack of water on carbocation.
(3) Deprotonation to form an alcohol
View Answer play_arrow
-
You are given
benzene, cone. 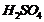 and
and 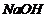 . Write
theequations for the preparation of phenol using these reagents,
(i)
Sulphonation of benzene.
(ii) Reaction with
. Write
theequations for the preparation of phenol using these reagents,
(i)
Sulphonation of benzene.
(ii) Reaction with 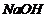 and
water.
and
water.
View Answer play_arrow
-
Show how will you synthesis:
(i) 1-phenylethanol form a
suitable alkene.
(ii) Cyclohexylmethanol using an
alkyl halide by an
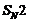
reactions.
(iii) pentan-1-ol using a
suitable alkyl halide?
(i) Acidic hydration of ethenyl
benzene.
(ii) Alkaline hydrolysis of
cyclohexyl methyl bromide.
(iii) Alkaline hydrolysis of
1-bromopentane.
-
Give two reactions that show the acidic
nature of phenol.Compare acidity of phenol with that of ethanol.
Reaction of phenol with Na and

comparing
of acidity of phenoland ethanol,
-
Explain why is ortho nitrophenol more
acidic than orthomethoxyphenol?
(i)
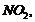
electron
withdrawing group,
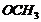
-electron
releasing group,
(ii) Presence of electron
withdrawing group increases the stability ofphenoxide ion while presence of
electron releasing group decreasesits stability.
(iii) Higher is the stability of
phenoxide ion formed, more is the acidiccharacter.
-
Explain how does the —OH group attached to a carbon of benzene ring activate if towards electrophilic substitution?
Resonance effect clue to —OH group.
View Answer play_arrow
-
Give equation of the following
reactions.
(i) Oxidation ofpropan-1-ol with
alkaline
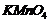
solution.
(ii) Bromine in
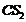
with
phenol.
(iii) Dilute
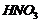
with
phenol.
(iv) Treating phenol with chloroform
in presence of aqueous
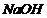
.
(i) Alcohol oxidises to carboxylic
acid.
(ii) o-and p- bromophenol are formed,
(iii) Nitration (introduction of
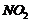
group)
ato-andp- positions.
(v) Reimer-Tiemann reaction involves
introduction of
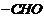
group
atortho position, sailcylaldehyde is formed.
-
Explain the following with an example.
(i) Kolbe's reaction.
(ii) Reimer-Tiernann reaction.
(iii) Williamson ether
synthesis.
(iv) Unsymmetrical ether.
(i) (Reaction of phenol with
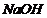
and
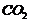
),
Salicylic acid is formed.
(ii) Reaction of phenol with
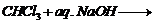
salicylaldehyde.
(iii) AlkyI halide + sod. alkoxide

Ether +
Sod. halide.
(iv) Ethers containing two different
alkyl/aryl groups.
-
Write the mechanism of acid catalysed dehydration ofethanol to yield ethene.
Step involved in acid catalysed dehydration are
(i) Protonation of alcohol,
(ii) Formation of carbocation.
(iii) Elimination of a proton.
View Answer play_arrow
-
How are the following conversion carried
out?
(i) Propene

propan-2-ol.
(ii) Benzyl chloride

Benzyl
alcohol.
(iii) Ethyl magnesium chloride

Propan-1-ol.
(iv) Methyl magnesium bromide

2-methylpropan-2-ol.
(i) Acidic hydration of propene.
(ii) Alkaline hydrolysis of
benzyl chloride.
(iii) Reaction with methanal in
dry ether followed by acid hydrolysis.
(iv) Reaction with propanone in dry ether
followed by acid hydrolysis.
-
1.
Name the reagents used in the following reactions.
(i) Oxidation of a primary
alcohol to carboxylic acid.
(ii) Oxidation of a primary
alcohol to aldehyde.
(iii) Bromination of phenol to
2,4,6- tribromophenol.
(iv) Benzyl alcohol to benzole
acid.
(v) Dehydration of propan -2-ol
to propene.
(vi) Butan-2-one to butan-2-ol.
(i) Oxidising agent (strong)
(ii) Weak oxidising agent
(iii)
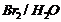
(iv) Strongoxidising agent
(v) Dehydrating agent
(vi) Reducing agent.
-
Give reason for the higher point of ethanol in comparison to methoxymethane.
Presence of intermolecular H-bonding in ethanol, but not in ether.
View Answer play_arrow
-
Give IUPAC names of the following
ethers.
(i)
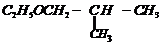
(ii)
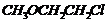
(iii)
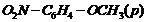
(iv)
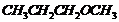
(v)
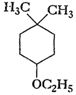
(vi)
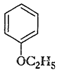
Recall the rules of IUPAC nomenclature
see solution 1 of exercise.
-
Write the names of reagents and
equations for the preparation of the following ethers by Williamson's
synthesis.
(i) 1-propoxypropane
(ii) Ethoxybenzene
(iii) 2-methoxy-2-methylpropane
(iv) 1-methoxyethane
(i)
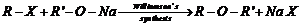
(ii) Alkyl halide should be primary.
-
Illustrate the examples the limitations of Williamson'ssynthesis for the preparation of certain types of ethers.
Tertiary alkyl halides readily give elimination product with strong baseslike alkoxide ions. The C—X bond of alkyl halide does not possess doublebond character i.e., should not be in conjugation with multiple bond.
View Answer play_arrow
-
How is 1-propoxypropane synthesized from propan-1-ol? Write mechanism of this reaction.
(i) Williamson’s synthesis or
(ii) Dehydration of propan-1-ol
View Answer play_arrow
-
Preparation of
ethers by acid dehydration of secondary ortertiary alcohols is not a suitable
method. Give reason.
Due to steric
hindrance, alkenes are formed by  mechanism.
mechanism.
View Answer play_arrow
-
Write the equation of the reaction of hydrogen iodidewith: (i) 1-propoxypropane (ii) methoxybenzene and (iii) benzyl ethyl ether.
Alcohols and alkyl of aryl iodides are formed. Generally primary halide (or lower halide) is formed in this reaction.
View Answer play_arrow
-
Explain the fact that in aryl alkyl ethers
(i) the alkoxy group activates the benzene ring towards electrophilic substitution.
(ii) it directs the incoming substituents to ortho and para positions in benzene ring.
Consider +R effect of the alkoxy group and more electron density at o-and p-positions.
View Answer play_arrow
-
Write the mechanism of the reaction of HI withmethoxymethane.
Steps involved in this process are
(i) protonation
(ii) formation of methanol and iodomethane.
View Answer play_arrow
-
Write equations of the following
reactions.
(i) Friedel Crafts
reaction-alkylation ofanisole.
(ii) Nitration of anisole.
(iii) Bromination ofanisole in
ethanoic acid medium,
(iv) Priedel Crafts acetylation
of anisole.
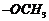
group is
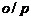
directing.
(i) Adulation (i.e.,
introduction of R group) at- o and p-positions. (By anhyd.
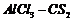
),
(ii) Nitration (i.e..
introduction of
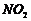
group) at
o-and p-positions (B
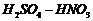
).
(iii) Bromination (introduction
of
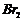
group)
ato-ando-positions (By
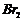
in
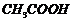
).
(iv) Acetylation (introduction of
RCO-group) at o-and p-position (By
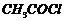
-anhydrous
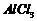
).
-
Show how would you synthesis the
following alcohols from appropriate alkenes?
(i) (ii)
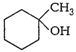

(iii) (iv)

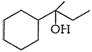
By hydration (addition of
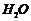
)
appropriate alkenes in accordance with Markownkoff?s rule.
-
When 3-methylbutane-2-ol is treated with
HBr, the following reaction takes place
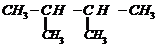
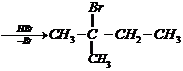
Give a mechanism for this
reaction.
The secondary carbocation formed in the
process rearranges to a more stable tertiary carbocation by a hydride ion shift
from 3rd carbon atom.
-
question_answer46)
Mono
chlorination of toluene in sunlight followed by hydrolysis with aq. NaOH
yields.
(a) o-Cresol (b) m-Cresol
(c) 2,4-Dihydroxytoluene
(d) Benzyl alcohol
View Answer play_arrow
-
question_answer47)
How
many alcohols with molecular formula
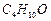 are chiral in nature ?
(a) 1 (b) 2
(c) 3 (d) 4
are chiral in nature ?
(a) 1 (b) 2
(c) 3 (d) 4
View Answer play_arrow
-
question_answer48)
What
is the correct order of reactivity of alcohols in the following reaction ?
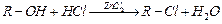 (a)
(a)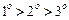 (b)
(b)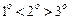 (c)
(c)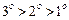 (d)
(d)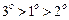
View Answer play_arrow
-
question_answer49)
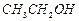 can be
converted into
can be
converted into 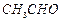 by....... .
(a) catalytic hydrogenadon
(b) treatment with
by....... .
(a) catalytic hydrogenadon
(b) treatment with 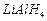 (c) treatment with pyridinium chlorochromate
(d) treatment with
(c) treatment with pyridinium chlorochromate
(d) treatment with 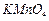
View Answer play_arrow
-
question_answer50)
The
process of converting alkyl halides into alcohols involves.......
(a) addition reaction
(b) substitution reaction
(c) dehydrohalogenation reaction
(d) rearrangement reaction
View Answer play_arrow
-
question_answer51)
Which
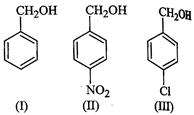
of the following compounds is aromatic alcohol?
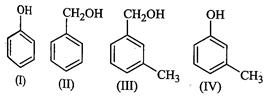 (a) I, II, III, IV (b) I, IV
(c) II, III (d) I
(a) I, II, III, IV (b) I, IV
(c) II, III (d) I
View Answer play_arrow
-
question_answer52)
Give
IUPAC name of the compound given
below.
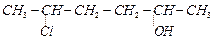 (a) 2-ChIoro-5-hydroxyhexane
(b) 2-Hydroxy-5-chlorohexane
(c) 5-Chlorohexan-2-ol
(d) 2-Chlorohexan-5-ol
(a) 2-ChIoro-5-hydroxyhexane
(b) 2-Hydroxy-5-chlorohexane
(c) 5-Chlorohexan-2-ol
(d) 2-Chlorohexan-5-ol
View Answer play_arrow
-
question_answer53)
IUPAC
name of m-cresol is .......
(a) 3-methylphenol (b) 3-chlorophenol
(c) 3-methoxyphenol (d) benzene-1, 3-diol
View Answer play_arrow
-
question_answer54)
IUPAC
name of the compound 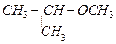 is
..........
(a) 1-methoxy-l-methylethane
(b) 2-methoxy-2-methylethane
(c) 2-methoxypropane
(d) isopropylmethyl ether
is
..........
(a) 1-methoxy-l-methylethane
(b) 2-methoxy-2-methylethane
(c) 2-methoxypropane
(d) isopropylmethyl ether
View Answer play_arrow
-
question_answer55)
Which
of the following species can act as the strongest base ?
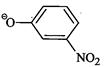 (a)
(a)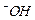 (b)
(b)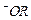 (c)
(c)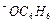 (d)
(d)
View Answer play_arrow
-
question_answer56)
Which
of the following compounds will react
with sodium hydroxide solution in water ?
(a)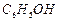 (b)
(b)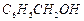 (c)
(c)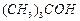 (d)
(d)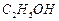
View Answer play_arrow
-
question_answer57)
Phenol
is less acidic than .......
(a) ethanol (b) o-nitrophenol
(c) o-methylphenol (d) o-methoxyphenol
View Answer play_arrow
-
question_answer58)
Which
of the following is most acidic ?
(a) Benzyl alcohol (b) Cyclohexanol
(c) Phenol (d) w-Chlorophenol
View Answer play_arrow
-
question_answer59)
Make
the correct order of decreasing acid strength of the following compounds.
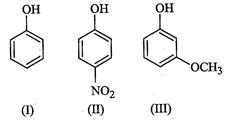
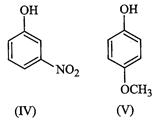 (a)
(a)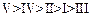 (b)
(b)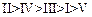 (c)
(c)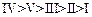 (d)
(d)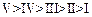
View Answer play_arrow
-
question_answer60)
Mark
the correct increasing order of reactivity of the following compounds with HBr/HCl.
 (a)
(a)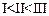 (b)
(b)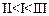 (c)
(c)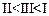 (d)
(d)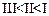
View Answer play_arrow
-
question_answer61)
Arrange
the following compounds in increasing order of boiling point.
Propan-1-ol, butan-1-ol, butan-2-ol, pentan-1-ol
(a) Propan-l-ol, butan-2-ol, butan-1-ol, pentan-1-ol
(b) Propan-l-ol, butan-1-ol, butan-2-ol, pentan-ol
(c) Pentan-l-ol, butan-2-ol, butan-1-ol, propan-l-ol
(d) Pentan-1-ol, butan-1-ol, butan-2-ol, propan-l-ol
View Answer play_arrow
-
question_answer62)
Which
of the following are used to convert RCHO in to 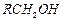 ?
(a)
?
(a)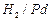 (b)
(b)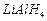 (c)
(c)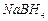 (d) Reaction with RMgX followed by hydrolysis
(d) Reaction with RMgX followed by hydrolysis
View Answer play_arrow
-
question_answer63)
Which
of the following reactions will yield phenol?
(a)
 (b)
(b)
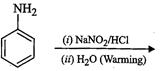 (c)
(c)
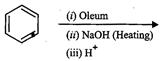 (d)
(d)
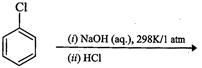
View Answer play_arrow
-
question_answer64)
Which
of the following reagents can be used to oxidise primary alcohols to aldehydes
?
(a)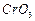 in anhydrous medium-
(b)
in anhydrous medium-
(b)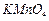 in
acidic medium
(c) Pyridinium chlorochromate
(d) Heat in the presence of Cu at 573 K.
in
acidic medium
(c) Pyridinium chlorochromate
(d) Heat in the presence of Cu at 573 K.
View Answer play_arrow
-
question_answer65)
Phenol
can be distinguished from ethanol by the reactions with.......
(a) 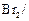 water (b)
Na
(c) Neutral
water (b)
Na
(c) Neutral 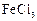 (d)
All the above
(d)
All the above
View Answer play_arrow
-
question_answer66)
Which
of the following are benzylic alcohols.
(a)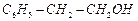 (b)
(b)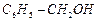 (c)
(c)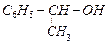 (d)
(d)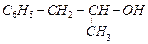
View Answer play_arrow
-
question_answer67)
What
is the structure and IUPAC name of glycerol ?
View Answer play_arrow
-
question_answer68)
Write
the lUPAC name of the following compounds.
(a)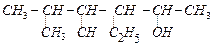 (b)
(b)
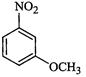
View Answer play_arrow
-
question_answer69)
Write
the IUPAC name of the compound given below.
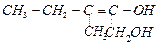
View Answer play_arrow
-
question_answer70)
Name
the factors responsible for the solubility of alcohols in water.
View Answer play_arrow
-
question_answer71)
What
is denatured alcohol?
View Answer play_arrow
-
question_answer72)
Suggest
a reagent for the following conversion.

View Answer play_arrow
-
question_answer73)
Out
of 2-chloroethanol and ethanol which is more acidic and why ?
View Answer play_arrow
-
question_answer74)
Suggest
a reagent for conversion of ethanol to ethanal.
View Answer play_arrow
-
question_answer75)
Suggest
a reagent for conversion of ethanol to ethanoic acid.
View Answer play_arrow
-
question_answer76)
Out
of o-nitrophenol andp-nitrophenol, which is more volatile ? Explain.
View Answer play_arrow
-
question_answer77)
Out
of o-nitrophenol and o-cresol which is more acidic ?
View Answer play_arrow
-
question_answer78)
When
phenol is treated with bromine water, white precipitate is obtained. Give the
structure and the name of the compound formed.
View Answer play_arrow
-
question_answer79)
Arrange
the following compounds in increasing order of acidity and give a suitable
explanation. Phenol, o-nitrophenol, o-cresol.
View Answer play_arrow
-
question_answer80)
Alcohols
react with active metals, e.g., Na, K, etc. to give corresponding alkoxides.
Write down the decreasing order of reactivity of sodium metal towards primary,
secondary and tertiary alcohols.
View Answer play_arrow
-
question_answer81)
What
happens when benzenediazonium chloride is heated with water?
View Answer play_arrow
-
question_answer82)
Arrange
the following compounds in decreasing order of
acidity.
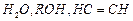
View Answer play_arrow
-
question_answer83)
Name
the enzymes and write the reactions involved in the preparation of ethanol from
sucrose fermentation.
View Answer play_arrow
-
question_answer84)
How
can propan-2-one be converted into tert- butyl alcohol ?
View Answer play_arrow
-
question_answer85)
Write
the structures of the isomers of alcohols with molecular formula 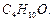 Which of these exhibits
optical activity ?
Which of these exhibits
optical activity ?
View Answer play_arrow
-
question_answer86)
Explain
why is OH group in phenols more strongly held as compared to OH group in
alcohols.
View Answer play_arrow
-
question_answer87)
Explain
why nucleophilic substitution reactions are not very common in phenols.
View Answer play_arrow
-
question_answer88)
Preparation
of alcohols from alkenes involves the electrophilic attack on alkene carbon
atom. Explain its mechanism.
View Answer play_arrow
-
question_answer89)
Explain
why is O = C = O non polar while R?O?R is polar.
View Answer play_arrow
-
question_answer90)
Why
is the reactivity of all the three classes of alcohols with cone. HCl and 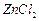 (Lucas reagent) different
?
(Lucas reagent) different
?
View Answer play_arrow
-
question_answer91)
Write
steps to carry out the conversion of phenol to aspirin.
View Answer play_arrow
-
question_answer92)
Nitration
is an example of aromatic electrophilic substitution and its rate depends upon
the group already present in the benzene ring. Out of benzene and phenol, which
one is more easily nitrated and why ?
View Answer play_arrow
-
question_answer93)
In
Kolbe's reaction, instead of phenol, phenoxide ion is treated with carbon
dioxide. Why ?
View Answer play_arrow
-
question_answer94)
Dipole
moment of phenol is smaller than that of methanol. Why ?
View Answer play_arrow
-
question_answer95)
Ethers
can be prepared by Williamson synthesis in which an alkyl halide is reacted
with sodium alkoxide. Di-tert-butyl ether can't be prepared by this method.
Explain.
View Answer play_arrow
-
question_answer96)
Why
is the C?O?H bond angle in alcohols slightly less than the tetrahedral angle
where the C?O?C bond angle in ether is slightly greater ?
View Answer play_arrow
-
question_answer97)
Explain
why low molecular mass alcohols are soluble in water.
View Answer play_arrow
-
question_answer98)
Explain
why p-nitrophenol is more acidic than phenol.
View Answer play_arrow
-
question_answer99)
Explain
why alcohols and ethers of comparable molecular mass have different boiling
points ?
View Answer play_arrow
-
question_answer100)
The
carbon-oxygen bond in phenol is slightly stronger than that in methanol. Why ?
View Answer play_arrow
-
question_answer101)
Arrange
water, ethanol and phenol in increasing order of acidity and give reason for
your answer.
View Answer play_arrow
-
question_answer102)
Match
the structures of the compounds given in Column I with the name of the
compounds given in Column II
|
|
Column I
|
|
Column II
|
|
(a)
|
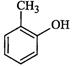
|
(i)
|
Hydroquinone
|
|
(b)
|
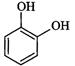
|
(ii)
|
Phenetole
|
|
(c)
|
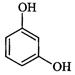
|
(iii)
|
Catechol
|
|
(d)
|
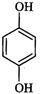
|
(iv)
|
o-Cresol
|
|
(e)
|
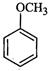
|
(v)
|
Quinone
|
|
(f)
|
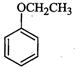
|
(vi)
|
Resorcinol
|
|
|
|
(vii)
|
Anisole
|
View Answer play_arrow
-
question_answer103)
Match
the starting materials given in Column I with the products formed by these
(Column II) in the reaction with HI.
|
|
Column I
|
|
Column II
|
|
(a)
|
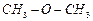
|
(i)
|
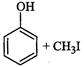
|
|
(b)
|
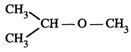
|
(ii)
|
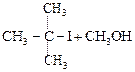
|
|
(c)
|
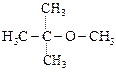
|
(iii)
|
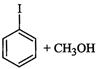
|
|
(d)
|
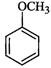
|
(iv)
|
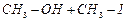
|
|
|
(v)
|
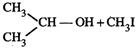
|
|
(vi)
|
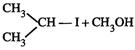
|
|
(vii)
|
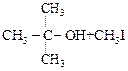
|
View Answer play_arrow
-
question_answer104)
Match
the items of column I with items of column n.
|
|
Column I
|
|
Column II
|
|
(a)
|
(a)
Antifreeze used in car engine
|
(i)
|
Neutral
ferric chloride
|
|
(b)
|
Solvent used in perfumes
|
(ii)
|
Glycerol
|
|
(c)
|
Starting material for picric acid
|
(iii)
|
Methanol
|
|
(d)
|
Wood spirit
|
(iv)
|
Phenol
|
|
(e)
|
Reagent
used for detection of phenolic group
|
(v)
|
Ethlene
glycol
|
|
(f)
|
By
product of soap industry used in cosmetics
|
(vi)
|
Ethanol
|
View Answer play_arrow
-
question_answer105)
Match
the items of Column I with items of Column II.
|
|
Column I
|
|
Column II
|
|
(a)
|
Methanol
|
(i)
|
Conversion
of phenol to o-hydroxysalicylic acid
|
|
(b)
|
Kolbe's reaction
|
(ii)
|
Ethyl
alcohol
|
|
(c)
|
Williamson's synthesis
|
(iii)
|
Conversion
of phenol to salicyaldehyde
|
|
(d)
|
Conversion
of 2° alcohol to ketone
|
(iv)
|
Wood
spirit
|
|
(e)
|
Reimer-Tiemann reaction
|
(v)
|
Heated
copper at 573 K
|
|
(f)
|
Fermentation
|
(vi)
|
Reaction
of alkyl halide with sodium alkoxide
|
View Answer play_arrow
-
question_answer106)
Assertion
: Addition reaction of water to but-1-ene in acidic medium yields butan-1-ol.
View Answer play_arrow
-
question_answer107)
Assertion
: p-Nitrophenol is more acidic than phenol.
View Answer play_arrow
-
question_answer108)
Assertion
: IUPAC name of the compound 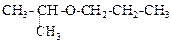 is Ethoxy-2-methlethane
is Ethoxy-2-methlethane
View Answer play_arrow
-
question_answer109)
Assertion
: Bond angle in ethers is slightly less than the tetrahedral angle.
View Answer play_arrow
-
question_answer110)
Assertion
: Boiling points of alcohols and ethers are high.
View Answer play_arrow
-
question_answer111)
Assertion:
Like bromination of benzene, bromination of phenol is also carried out in the
presence of Lewis acid.
View Answer play_arrow
-
question_answer112)
Assertion
: o-Nitrophenol is less soluble in water than the m- and p-isomers.
View Answer play_arrow
-
question_answer113)
Assertion
: Ethanol is a weaker acid than phenol.
View Answer play_arrow
-
question_answer114)
Assertion
: Phenol forms 2, 4, 6-tribromophenol on treatment with 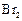 in carbon disulphide at
273 K.
in carbon disulphide at
273 K.
View Answer play_arrow
-
question_answer115)
Assertion
: Phenols give o- and p-nitrophenol on nitration with .cone. 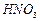 and
and 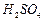 mixture.
mixture.
View Answer play_arrow
-
question_answer116)
Write
the mechanism of the reaction of HI with methoxybenzene.
View Answer play_arrow
-
question_answer117)
(a)
Name the starting material used in the industrial preparation of phenol.
(b) Write complete reaction for the bromination of phenol in
aqueous and non aqueous medium.
(c) Explain why Lewis acid is not required in
bromination of phenol.
View Answer play_arrow
-
question_answer118)
How
can phenol be converted to aspirin ?
View Answer play_arrow
-
question_answer119)
Explain
a process in which a biocatalyst is used in industrial preparation of a
compound known to you-
View Answer play_arrow
 (i)
(i)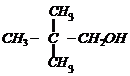 (ii)
(ii)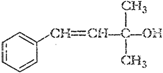
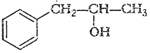

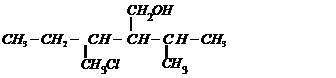 (ii)
(ii)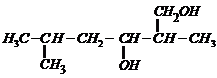 (iii)
(iii)
 (iv)
(iv)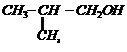 (ii)
(ii)

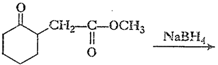 (iii)
(iii) 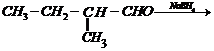 (i) Acidic hydration of propene
i.e., addition of
(i) Acidic hydration of propene
i.e., addition of  (ii)
(ii)
 This reaction involves preparation of
ether by Williamson synthesis it takes place by
This reaction involves preparation of
ether by Williamson synthesis it takes place by 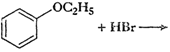 (iii)
(iii)
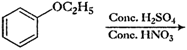 (iv)
(iv)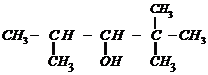 (ii)
(ii) 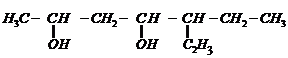 (iii)
(iii)  (vi)
(vi)
 (viii)
(viii)
 (ix)
(ix) (vi)
(vi)
 Recall the rules of IUPAC nomenclature
see solution 1 of exercise.
Recall the rules of IUPAC nomenclature
see solution 1 of exercise.

 By hydration (addition of
By hydration (addition of 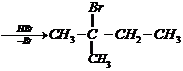 Give a mechanism for this
reaction.
The secondary carbocation formed in the
process rearranges to a more stable tertiary carbocation by a hydride ion shift
from 3rd carbon atom.
Give a mechanism for this
reaction.
The secondary carbocation formed in the
process rearranges to a more stable tertiary carbocation by a hydride ion shift
from 3rd carbon atom.
 (a)
(a)![]() (b)
(b)![]() (c)
(c)![]() (d)
(d)
 (b)
(b)
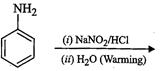 (c)
(c)
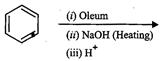 (d)
(d)

![]() (b)
(b)![]() (c)
(c)![]() (d)
(d)![]()
![]() (b)
(b)

![]()

![]()





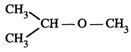
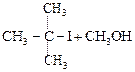
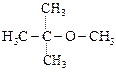


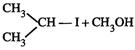
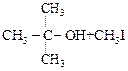
![]() is Ethoxy-2-methlethane
is Ethoxy-2-methlethane
