-
For the reaction R

P, the
concentration of a reactant changes from 0.03 M to 0.02 M in 25 minutes.
Calculate the average rate of reaction using units of time both in minutes and
seconds.
-
In a reaction, 2A

Products,
the concentration of A decreases from 0.5 mol L?1 to 0.4 mol L?1
in 10 minutes. Calculate the rate during this interval?
-
For a reaction, A + B

Product;
the rate law, is given by,
r = k [A]1/2 [B]2. What is the order
of the reaction?
-
The conversion of molecules X to Y follows second order kinetics. If concentration of X is increased to three times how will it affect the rate of formation of Y?
View Answer play_arrow
-
A first order reaction has rate constant of 1.15 x 10–3 s–1. How long will 5 g of this reactant take to reduce to 3 g?
View Answer play_arrow
-
Time required to decompose SO2Cl2 to half of its initial concentration is 60 minutes. If the decomposition is a first order reaction, calculate the rate constant of the reaction.
View Answer play_arrow
-
What will be the effect of temperature on rate constant?
View Answer play_arrow
-
The rate of the chemical reaction doubles for an increase of 10 K in absolute temperature from 298 K. Calculate Ea.
View Answer play_arrow
-
The activation energy for the reaction
2HI (g)

H2(g)
+ I2 (g)
is 209.5 kJ mol?1 at
581 K. Calculate the fraction of molecules of reactants having energy equal to
or greater than activation energy.
-
From
the rate expression for the following reactions, determine their order of
reaction and the dimensions of the rate constants.
(i) 3NO (g)
® N2O (g) + NO2 (g); Rate
= k [NO]2
(ii) H2O2
(aq) + 3I? (aq) + 2H+
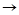
2H2O(I)+
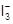
Rate
= k [H2O2] [I?]
(iii) CH3CHO(g)
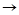
CH4(g)
+ CO (g) ; Rate = k [CH3CHO]3/2
(iv) C2H5Cl(g)
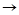
C2H4(g)
+ HCl (g); Rate = k [C2H5Cl]
-
For the reaction : 2A + B

A2B
the rate = k [A] [B]2 with k = 2.0 x 10?6 mol ?2
L2 s?1. Calculate the initial rate of the reaction when
[A] = mol L?1, [B] = 0.2 mol L?1. Calculate the rate of
reaction after [A] is reduced to 0.06 mol L?1.
-
The decomposition of NH3 on platinum surface is zero order reaction. What are the rates of production of N2 and H2 if k = 2.5 × 10–4 mol–1 L s–1?
View Answer play_arrow
-
The
decomposition of dimethyl ether leads to the formation of
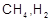
and
CO and the reaction rate is given by Rate = k [CH3OCH3]3/2.
The rate of
reaction is followed by increase in pressure in a closed vessel and the rate
can also be expressed in terms of the partial pressure dimethyl ether i.e. rate
= k (pCH3OCH3)3/2.
If the pressure
is measured in bar and time in minutes, then what are the units of rate and rate
constants?
-
Mention the factors that effect that rate of a chemical reaction.
View Answer play_arrow
-
A raction is second order with respect to a reactant. How is the rate of reaction affected if the concentration of the reactant is (i) doubled (ii) reduced to half?
View Answer play_arrow
-
What is the effect of temperature on the rate constant of reaction? How can this temperature effect on rate constant be represented quantitatively?
View Answer play_arrow
-
In a pseudo first order hydrolysis of ester in
water the following results were obtained :
|
t/s
|
0
|
30
|
60
|
90
|
|
[Ester]/M
|
0.55
|
0.31
|
0.17
|
0.085
|
(i) Calculate the average rate of
reaction between the time interval 30 to 60 seconds.
(ii) Calculate the pseudo first order rate constant for the
hydrolysis of ester.
-
A reaction is first order in A and second order in B.
(i) Write differential rate equation.
(ii) How is the rate affected when the concentration of B is tripled?
(iii) How is the rate affected when the concentration of both A and B is doubled?
View Answer play_arrow
-
View Answer play_arrow
-
View Answer play_arrow
-
View Answer play_arrow
-
Calculate the half life of a first order reaction from their rate constants given below :
(i) 200 s–1 (ii) 2 min–1 (iii) 4 years–1
View Answer play_arrow
-
The half life for radioactive decay of 14C is 5730 y. An archaelogical artefact contained wood had only 80% of the 14C found in a living tree. Estimate the age of the sample.
View Answer play_arrow
-
The experimental
data for decomposition of N2O5
[2N2O5
 4NO2
+ O2]
4NO2
+ O2]
|
t/s
|
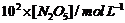
|
|
0
400
800
1200
1600
2000
2400
2800
3200
|
1.63
1.36
1.14
0.93
0.78
0.65
0.53
0.43
0.35
|
(i) Plot [N2O5]
against t.
(ii) Find the
half-life period for the reaction.
(iii) Draw a graph
between log 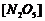 and t.
(iv) What is the
rate law?
(v) Calculate the
rate constant.
(vi)
Calculate the half-life period from k and compare it with (ii).
and t.
(iv) What is the
rate law?
(v) Calculate the
rate constant.
(vi)
Calculate the half-life period from k and compare it with (ii).
View Answer play_arrow
-
The rate constant
for a first order reaction is 60 s?1. How much time will it take to
reduce the initial concentration of the reactant to its 
View Answer play_arrow
-
During nuclear
explosion, one of the products is 90Sr with half life of 28.1 y. If  of
of  was absorbed in
the bones of a newly born baby instead of calcuium, how much of it will remain
after 10 years and 60 years if it is not lost metabolically.
was absorbed in
the bones of a newly born baby instead of calcuium, how much of it will remain
after 10 years and 60 years if it is not lost metabolically.
View Answer play_arrow
-
Show that the time required for 99% completion of a reaction is twice the time required complete 90% of the reaction.
View Answer play_arrow
-
A first order reaction takes 40 minutes for 30% decomposition. Calculate its half life period.
View Answer play_arrow
-
For the decomposition
of azoisopropane to hexane and nitrogen at 543 K, the following data is
obtained.
(CH3)2
CHN = NCH (CH3)2  N2
+ C6H14
N2
+ C6H14
|
t (sec)
|
0
|
360
|
720
|
|
Pressure (atm)
|
35.0
|
54.0
|
63.0
|
Calculate the rate
constant.
View Answer play_arrow
-
The following data
were obtained during first order thermal decomposition of SO2Cl2
at a constant volume.
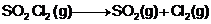
|
t (sec)
|
0
|
100
|
|
Total pressure
(atm)
|
0.5
|
0.6
|
Calculate the rate
of the reaction when the total pressure is 0.65 atm.
View Answer play_arrow
-
The rate constant
for the decomposition of N2O5 at various temperatures is
given below :
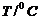
|
0
|
20
|
40
|
60
|
80
|
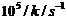
|
0.0787
|
1.70
|
25.7
|
178
|
2140
|
Draw a graph
between in k and 1/T and calculate the value of A and Ea. Predict the rate
constant at 30° and 50°C.
View Answer play_arrow
-
The rate constant for the decomposition of a hydrocarbon is 2.418 x 10–5 s–1 at 546 K. If the energy of activation is 179.9 kJ mol–1, what will be the value of pre-exponential factor?
View Answer play_arrow
-
Consider certain
reaction A  Products with
Products with
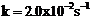 . Calculate the
concentration of A remaining after 100 s if the initial concentration of A is 1.0
mol L?1.
. Calculate the
concentration of A remaining after 100 s if the initial concentration of A is 1.0
mol L?1.
View Answer play_arrow
-
Sucrose decomposes in acid solution into glucose and fructose according to the first order rate law with t1/2 = 3.0 hrs. What fraction the sample of sucrose remains after 8 hours?
View Answer play_arrow
-
The decomposition
of a hydrocarbon follows the equation:
k = (4.5 × 1011
s?1) e?28000K/T.
Calculate the
energy of activation (Ea).
View Answer play_arrow
-
The rate constant for the first order decomposition of H2O2 is given by the following equation :
log k = 14.34 - 1.25 x 104 K/T.
Calculate the Ea for the reaction. At what temperature will the half life period be 256 minutes?
View Answer play_arrow
-
The decomposition of A into product has value of k as 4.5 x 103 s–1 at 10°C and eneqy of activation 60 kJ mol–1. At what temperature would k be 1.5 x 104 s–1?
View Answer play_arrow
-
The time required for 10% completion of a first order reaction at 298 K is equal to that required for its 25% completion at 308 K. If the value of A is 3.56 x 109, calculate k at 318 K and F and Ea.
View Answer play_arrow
-
The rate of a particular reaction quadrules when the temperature changes from 293 K to 313 K. Calculate the energy of activation of the reaction. (R = 8.314 J K–1 mol –1)
View Answer play_arrow
-
question_answer40)
The role of a catalyst is to change..........
(a) Gibbs energy of reaction
(b) enthalpy of reaction
(c) activation energy of
reaction
(d) equilibrium constant
View Answer play_arrow
-
question_answer41)
In the presence of a catalyst, the heat evolved
or absorbed during the reaction..........
(a) increases
(b) decreases
(c) remains
unchanged
(d) may increase or decrease
View Answer play_arrow
-
question_answer42)
Activation energy of a chemical reaction can be
determined by..........
(a) determining the rate constant at
standard temperature
(b) determining the rate
constant at two temperatures
(c) determining probability of
collision
(d) using catalyst
View Answer play_arrow
-
question_answer43)
Consider figure and mark the correct option.
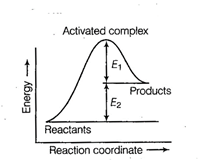 (a) Activation energy of forward
reaction is
(a) Activation energy of forward
reaction is 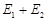 and product is less
stable than reactant
(b) Activation energy of forward
reaction is
and product is less
stable than reactant
(b) Activation energy of forward
reaction is 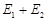 and product is more
stable than reactant
(c) Activation energy of both
forward and backward reaction is
and product is more
stable than reactant
(c) Activation energy of both
forward and backward reaction is 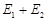 and
reactant is more stable than product
Activation energy of backward
reaction is
and
reactant is more stable than product
Activation energy of backward
reaction is 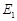 and product is more
stable than reactant
and product is more
stable than reactant
View Answer play_arrow
-
question_answer44)
Consider a first order gas phase decomposition
reaction given below
A(g) ® B(g) + C(g)
`The initial pressure of the
system before decomposition of A was pi. After lapse of time 't?
total pressure of the system increased by x units and became 'pt.?.
The rate constant k for the reaction is given as..........
(a) 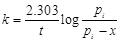 (b)
(b) 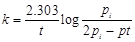 (c)
(c) 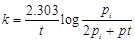 (d)
(d) 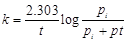
View Answer play_arrow
-
question_answer45)
According to Arrhenius equation rate constant k
is equal to 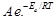 which of
the following options represents the graph of in K
which of
the following options represents the graph of in K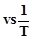 ?
?
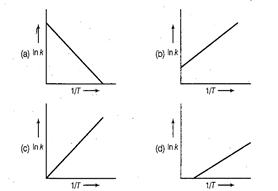
View Answer play_arrow
-
question_answer46)
Consider the Arrhenius equation given below and
mark the correct option.
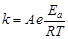 (a) Rate constant increases
exponentially with increasing activation energy and decreasing temperature
(b) Rate constant decreases
exponentially with increasing activation energy and decreasing temperature
(c) Rate constant increases
exponentially with decreasing activation energy and decreasing temperature
(d) Rate constant increases
exponentially with decreasing activation energy and increasing temperature
(a) Rate constant increases
exponentially with increasing activation energy and decreasing temperature
(b) Rate constant decreases
exponentially with increasing activation energy and decreasing temperature
(c) Rate constant increases
exponentially with decreasing activation energy and decreasing temperature
(d) Rate constant increases
exponentially with decreasing activation energy and increasing temperature
View Answer play_arrow
-
question_answer47)
A graph of volume of hydrogen released vs time
for the reaction between zinc and dil. HCl is given in figure. On the basis of
this mark the correct option.
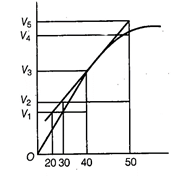 (a) Average rate upto 40 s is
(a) Average rate upto 40 s is 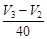 (b) Average rate upto 40 s is
(b) Average rate upto 40 s is 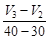 (c) Average rate upto 40 s is
(c) Average rate upto 40 s is 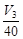 (d) Average rate upto 40 s is
(d) Average rate upto 40 s is 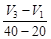
View Answer play_arrow
-
question_answer48)
Which of the following statements is not correct
about order of a reaction?
(a) The order of a reaction can be a
fractional number
(b) Order of a reaction is
experimentally determined quantity
(c) The order of a reaction is
always equal to the sum of the stoichiometric coefficients of reactants in the
balanced chemical equation for a reaction
(d) The order of a reaction is the
sum of the powers of molar concentration of the reactants in the rate law
expression
View Answer play_arrow
-
question_answer49)
Consider the graph given in figure. Which of the
following options does not show instantaneous rate of reaction at 40s?
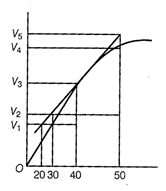 (a)
(a)
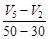 (b)
(b)
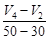 (c)
(c)
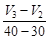 (d)
(d)
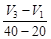
View Answer play_arrow
-
question_answer50)
Which of the following statements is correct?
(a) The rate of a reaction decreases
with passage of time as the concentration of reactants decreases
(b) The rate of a reaction is
same at any time during the reaction
(c) The rate of a reaction is
independent of temperature change
(d) The rate of a reaction
decreases with increase in concentration of reactant (s)
View Answer play_arrow
-
question_answer51)
Which of the following expressions is correct
for the rate of reaction given below?
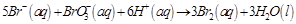 (a)
(a) 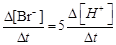 (b)
(b) 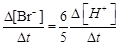 (c)
(c) 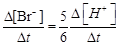 (d)
(d) 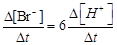
View Answer play_arrow
-
question_answer52)
Which of the following graphs represents
exothermic reaction?
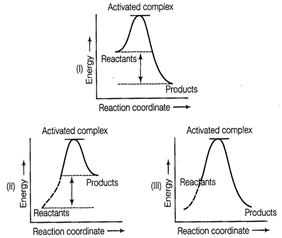 (a) Only (I) (b) Only (ll)
(c) Only (lll) (d)
(I) and (II)
(a) Only (I) (b) Only (ll)
(c) Only (lll) (d)
(I) and (II)
View Answer play_arrow
-
question_answer53)
Rate Law for the reaction A + 2B ® C is found to be
Rate = k [A] [B]
Concentration of reactant 'B?
is doubled, keeping the concentration of 'A' constant, the value of rate
constant will be...........
(a) the same (b)
doubled
(c) quadrupled (d)
halved
View Answer play_arrow
-
question_answer54)
Which of the following statements is incorrect
about the collision theory of chemical reaction?
(a) It considers reacting molecules or
atoms to be hard spheres and ignores their structural features
(b) Number of effective
collisions determines the rate of reaction
(c) Collision of atoms or
molecules possessing sufficient threshold energy results into the product
formation
(d) Molecules should collide
with sufficient threshold energy and proper orientation for the collision to be
effective
View Answer play_arrow
-
question_answer55)
A first order reaction is 50% completed in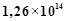 . How much time would
it take for 100% completion?
(a) 1.26x1015 s (b)
252x1014s
(c) 2.52x1028
s (d) Infinite
. How much time would
it take for 100% completion?
(a) 1.26x1015 s (b)
252x1014s
(c) 2.52x1028
s (d) Infinite
View Answer play_arrow
-
question_answer56)
Compounds 'A' and 'B' react according to the
following chemical equation.
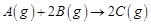 Concentration of either 'A'
or 'B' were changed keeping the concentrations of one of the reactants constant
and rates were measured as a function of initial concentration. Following
results were obtained.
Choose the correct option for the
rate equations for this reaction.
Concentration of either 'A'
or 'B' were changed keeping the concentrations of one of the reactants constant
and rates were measured as a function of initial concentration. Following
results were obtained.
Choose the correct option for the
rate equations for this reaction.
|
Experiment
|
Initial
concentration of [A]/molL-1
|
Initial
concentration of [B]/molL-1
|
Initial
concentration of [CJ/moll-1s-1
|
|
1.
2.
3.
|
0.30
0.30
0.60
|
0.30
0.60
0.30
|
0.10
0.40
0.20
|
(a)
Rate = 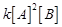 (b) Rate =
(b) Rate =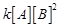 (c) Rate =
(c) Rate =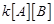 (d) Rate =
(d) Rate = 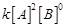 View Answer play_arrow
View Answer play_arrow
-
question_answer57)
Which of the following statement is not correct
for the catalyst?
(a) It catalyses the forward and
backward reactions to the same extent
(b) It alters AC of the reaction
(c) It is a substance that does
not change the equilibrium constant of a reaction
(d) It provides an alternate
mechanism by reducing activation energy between reactants and products
View Answer play_arrow
-
question_answer58)
The value of rate constant of a pseudo first
order reaction..........
(a) depends on the concentration of
reactants present in small amount
(b) depends on the concentration
of reactants present in excess
(c) is independent of the
concentration of reactants
(d) depends only on temperature
View Answer play_arrow
-
question_answer59)
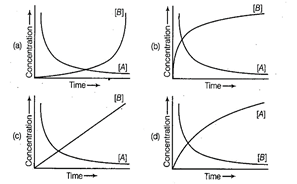
Consider the reaction A ® B. The concentration of both the
reactants and the products varies exponentially with time. Which of the
following figures correctly describes the change in concentration of reactants
and products with time?
View Answer play_arrow
-
question_answer60)
Rate law cannot be determined from balanced
chemical equation if..........
(a) reverse reaction is involved
(b) it is an elementary reaction
(c) it is a sequence of
elementary reactions
(d) any of the reactants is in
excess
Rate law can be determined from
balanced chemical equation if it is an elementary reaction.
View Answer play_arrow
-
question_answer61)
Which of the following statements are applicable
to a balanced chemical equation of an elementary reaction?
(a) Order is same as molecularity
(b) Order is less than the
molecularity
(c) Order is greater than the
molecularity
(d) Molecularity can never be
zero
View Answer play_arrow
-
question_answer62)
In any unimolecular reaction..........
(a) only one reacting species is
involved in the rate determining step
(b) the order and the
molecularity of slowest step are equal to one
(c) the molecularity of the
reaction is one and order is zero
(d) both molecularity and order
of the reaction are one
View Answer play_arrow
-
question_answer63)
For a complex reaction..........
(a) order of overall reaction is same as
molecularity of the slowest step
(b) order of overall reaction is
less than the molecularity of the slowest step
(c) order of overall reaction is
greater than molecularity of the slowest step
(d) molecularity of the slowest
step is never zero or non-integer
View Answer play_arrow
-
question_answer64)
At high pressure the following reaction is zero
order.
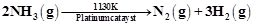 Which of the following
options are correct for this reaction?
(a) Rate of reaction = Rate
constant
(b) Rate of the reaction depends
on concentration of ammonia
(c) Rate of decomposition of
ammonia will remain constant until ammonia disappears completely
(d) Further increase in pressure
will change the rate of reaction
Which of the following
options are correct for this reaction?
(a) Rate of reaction = Rate
constant
(b) Rate of the reaction depends
on concentration of ammonia
(c) Rate of decomposition of
ammonia will remain constant until ammonia disappears completely
(d) Further increase in pressure
will change the rate of reaction
View Answer play_arrow
-
question_answer65)
During decomposition of an activated complex
(a) energy is always released
(b) energy is always absorbed
(c) energy does not
change
(d) reactants may be formed
View Answer play_arrow
-
question_answer66)
According to Maxwell, Boltzmann distribution of
energy...........
(a).the fraction of molecules with most
probable kinetic energy decreases at higher temperatures
(b) the fraction of molecules
with most probable kinetic energy increases at higher temperatures
(c) most probable kinetic energy
increases at higher temperatures
(d) most probable kinetic energy
decreases at higher temperatures
View Answer play_arrow
-
question_answer67)
In the graph showing Maxwell, Boltzmann
distribution of energy.........
(a) area under the curve must not change
with increase in temperature
(b) area under the curve
increases with increase in temperature
(c) area under the curve
decreases with increase in temperature
(d) with increase in temperature
curve broadens and shifts to the right hand side
View Answer play_arrow
-
question_answer68)
Which of the following statements are in
accordance with the Arrhenius equation?
(a) Rate of a reaction increases with
increase in temperature
(b) Rate of a reaction increases
with decrease in activation energy
(c) Rate constant decreases
exponentially with increase in temperature
(d) Rate of reaction decreases
with decrease in activation energy
View Answer play_arrow
-
question_answer69)
Mark the incorrect statements.
(a) Catalyst provides an alternative
pathway to reaction mechanism
(b) Catalyst raises the
activation energy
(c) Catalyst lowers the
activation energy
(d) Catalyst alters enthalpy
change of the reaction
View Answer play_arrow
-
question_answer70)
Which of the following graphs is correct for a
zero order reaction?
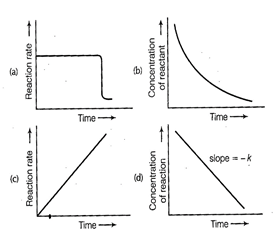
View Answer play_arrow
-
question_answer71)
Which of the following graphs is correct for a
first order reaction?
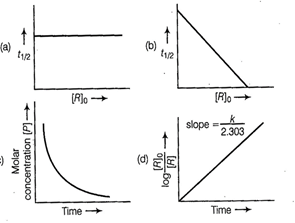
View Answer play_arrow
-
question_answer72)
State a condition under which a bimolecular
reaction is kinetically first order reaction.
View Answer play_arrow
-
question_answer73)
Write the rate equation for the reaction2A + B ® C if the order of the reaction is zero.
View Answer play_arrow
-
question_answer74)
How can you determine the rate law of the
following reaction?
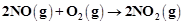
View Answer play_arrow
-
question_answer75)
For which type of reactions, order and
molecularity have the same value?
View Answer play_arrow
-
question_answer76)
In a reaction if the concentration of reactant A
is tripled, the rate of reaction becomes twenty seven times. What is the order
of the reaction?
View Answer play_arrow
-
question_answer77)
Derive an expression to calculate time required
for completion of zero order reaction.
View Answer play_arrow
-
question_answer78)
For a reaction A + B ® Products, the rate law is 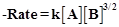 Can the reaction be
an elementary reaction? Explain.
Can the reaction be
an elementary reaction? Explain.
View Answer play_arrow
-
question_answer79)
For a certain reaction large fraction of
molecules has energy more than the threshold energy, yet the rate of reaction
is very slow. Why?
View Answer play_arrow
-
question_answer80)
For a zero order reaction will the molecularity
be equal to zero? Explain.
View Answer play_arrow
-
question_answer81)
For a general reaction A ® B, plot of concentration of A vs time is
given in figure. Answer the following questions on the basis of this graph.
(i) What is the order of the reaction?
(ii) What is the slope of the
curve?
(iii) What are the units of rate
constant?
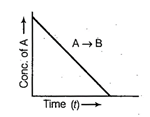
View Answer play_arrow
-
question_answer82)
The reaction between 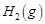 and
and 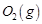 is highly feasible
yet allowing the gases to stand at room temperature in the same vessel does not
lead to the formation of water. Explain.
is highly feasible
yet allowing the gases to stand at room temperature in the same vessel does not
lead to the formation of water. Explain.
View Answer play_arrow
-
question_answer83)
Why does the rate of a reaction increase with
rise in temperature?
View Answer play_arrow
-
question_answer84)
Oxygen is available in plenty in air yet fuels
do not burn by themselves at room temperature. Explain.
View Answer play_arrow
-
question_answer85)
What is the probability of reaction with
molecularity higher than three very rare?
View Answer play_arrow
-
question_answer86)
Why does the rate of any reaction does generally
decreases during the course of the reaction?
View Answer play_arrow
-
question_answer87)
Thermodynamic feasibility of the reaction alone
cannot decide the rate of the reaction. Explain with the help of one example.
View Answer play_arrow
-
question_answer88)
Why in the redox titration ofKMn04 vs oxalic
add, we heat oxalic acid solution before starting the titration?
View Answer play_arrow
-
question_answer89)
Why can't molecularity of any reaction be equal
to zero?
View Answer play_arrow
-
question_answer90)
Why molecularity is applicable only for
elementary reactions and order is applicable for elementary as well as complex
reactions?
View Answer play_arrow
-
question_answer91)
Why can we not determine the order of a reaction
by taking into consideration the balanced chemical equation?
View Answer play_arrow
-
question_answer92)
Match the graph given in Column I with the order
of reaction given in Column II. More than one item in Column I may link to the
same item of Column II.
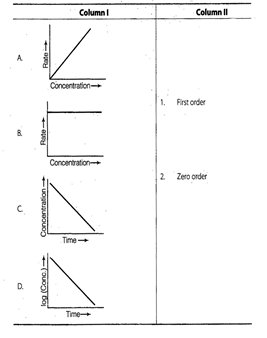
View Answer play_arrow
-
question_answer93)
Match the statements given in Column I and
Column II.
|
|
Column 1
|
Column 11
|
|
A.
B.
C.
D.
E.
F.
|
Catalyst alters the rate of reaction
Molecularity
Second half-life of first order reaction
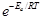 Energetically favorable reactions are sometimes
slow
Area under the Maxwell, Boltzmann curve is
constant
Energetically favorable reactions are sometimes
slow
Area under the Maxwell, Boltzmann curve is
constant
|
1.Cannot be fraction or zero
2. Proper orientation is not there
3.By lowering the activation energy 4.Is same as
the first
5. Total probability is one
6. Refers to the fraction of molecules with
energy equal to or greater than activation energy
|
View Answer play_arrow
-
question_answer94)
Match the items of Column I and Column II.
|
|
Column 1
|
Column 11
|
|
A.
|
Diamond
|
1. Short interval of time
|
|
B.
|
Instantaneous rate
|
2. Ordinarily rate of conversion is imperceptible
|
|
C.
|
Average rate
|
3. Long duration of time
|
View Answer play_arrow
-
question_answer95)
Match the items of Column I and Column II.
|
|
Column 1
|
Column 11
|
|
A.
|
Mathematical expression for rate of reaction
|
1. Rate constant
|
|
B.
|
Rate of reaction for zero order reaction is equal
to
|
2. Rate law
|
|
C.
|
Units of rate constant for zero order reaction is
same as. that of
|
3. Order of slowest step
|
|
D.
|
Order of a complex reaction is determined by
|
4. Rate of reaction
|
View Answer play_arrow
-
question_answer96)
Assertion (A) Order of the reaction can be
zero or fractional.
Reason (R) We cannot determine
order from balanced chemical equation.
View Answer play_arrow
-
question_answer97)
Assertion (A) Order and molecularity are
same.
Reason (R) Order is determined experimentally
and molecularity is the sum of the stoichiometric coefficient of rate
determining elementary step,.
View Answer play_arrow
-
question_answer98)
Assertion (A) The enthalpy of reaction
remains constant in the presence of a catalyst.
Reason (R) A catalyst
participating in the reaction forms different activated complex and lowers down
the activation energy but the difference in energy of reactant and product
remains the same.
View Answer play_arrow
-
question_answer99)
Assertion (A) All collision of reactant
molecules lead to product formation.
Reason (R) Only those collisions
in which molecules have correct orientation and sufficient kinetic energy Lead
to compound formation.
View Answer play_arrow
-
question_answer100)
Assertion (A) Rate constant determined from
Arrhenius equation are fairly accurate for simple as well as complex molecules.
Reason (R) Reactant molecules
undergo chemical change irrespective of their orientation during collision.
View Answer play_arrow
-
question_answer101)
All energetically effective collisions do not
result in a chemical change.
Explain with the help of an example.
View Answer play_arrow
-
question_answer102)
What happens to most probable kinetic energy and
the energy of activation with increase in temperature?
View Answer play_arrow
-
question_answer103)
Describe how does the enthalpy of reaction
remain unchanged when a catalyst is used in the reaction?
View Answer play_arrow
-
question_answer104)
Explain the difference between instantaneous
rate of a reaction and average rate of a reaction.
View Answer play_arrow
-
question_answer105)
With the help of an example explain what is
meant by pseudo first order reaction.
View Answer play_arrow


![]()
