-
What are polymers?
View Answer play_arrow
-
How are polymers classified on the basis of structure?
View Answer play_arrow
-
Write the names of monomers of the
following polymers ;
(i)
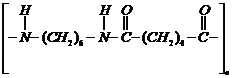
(ii)
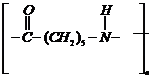
(iii)
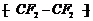
(i) Since amide linkage
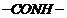
is
present, it is a polymer of diaminehaving 6 C atom and diacid having 6 C atoms.
(ii) It also contains amide linkage
but with in the same molecule, so itcontains>CO and amine group within a
ring.
(iii) It contains the same monomer and
repeating .unit so it is an additionpolymer of a monomer containing double
bond. Moreover, absence offunctional group also shows the unsaturation.
-
Classify following as addition and condensation polymers:Terylene, bakelite, polyvinyl chloride, polythene.
(i) Addition polymers are obtained from unsaturated monomers andcondensation polymers are obtained from the monomerscontaining by functional groups.
(ii) Monomers of terylene, bakelite, PVC and polythene are phthalicacid + ethylene glycol, phenol + HCHO, vinyl chloride and ethene.
View Answer play_arrow
-
Explain the difference between buna-N and buna-S.
View Answer play_arrow
-
Arrange the following polymers in increasing order of theirintermolecular forces.
(i) Nylon-6, 6, buna-S, polythene.
(ii) Nylon-6, neoprene, polyinyl chloride
The order of intermolecular forces in different polymers is Easterners <plastics < fibres. So first classify the given polymer into the aboveclasses and then arrange them in increasing order of forces.
View Answer play_arrow
-
Explain the terms polymer and monomer.
View Answer play_arrow
-
What are natural and synthetic polymers? Give twoexamples of each type.
View Answer play_arrow
-
Distinguish between the terms homopolymer andcopolymer and given an example of each.
Homopolymer: Single monomer
Copolymer: More than one monomers. Give definition on this basis.
View Answer play_arrow
-
How do you explain the functionality of a monomer?
View Answer play_arrow
-
Define the term polymerization.
View Answer play_arrow
-
Is
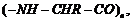
a homopolymer or
copolymer?
-
In which classes, the polymers are classified on the basis ofmolecular forces?
View Answer play_arrow
-
How can you differentiate between addition andcondensation polymerization?
View Answer play_arrow
-
Explain the term copolymerization and give two examples.
It includes involvement of two or more monomers, for polymerization.
View Answer play_arrow
-
Write the free radical mechanism for the polymerizationof ethene.
View Answer play_arrow
-
Define thermoplastics and thermosetting polymers withtwo examples of each.
Effect of heat on these polymers is the basis of this classification.
View Answer play_arrow
-
Write the monomers used for getting the followingpolymers.
(i) Polyvinyl chloride (ii) Teflon (iii) Bakelite
View Answer play_arrow
-
Write the name and structure of one of the common initiators used in free radical addition polymerization.
Peroxides play this role.
View Answer play_arrow
-
How does the presence of double bonds in rubbermolecules influence their structure and reactivity?
View Answer play_arrow
-
Discuss the main purpose of vulcanization of rubber.
View Answer play_arrow
-
What are the monomeric repeating units of nylon-6 andnylon-6,6?
View Answer play_arrow
-
Write the name and structures of the monomers of thefollowing polymers.
(i) Buna-S (ii) Buna-N (iii) Dacron (iv) Neoprene
View Answer play_arrow
-
Identify the monomer in the following
polymeric structures.
(i)
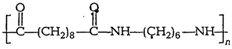
c
(ii)
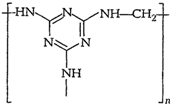
(i) Break ?CO ?NH ? bond and add
to
get the structure of monomers.
(ii) Break
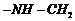
bond and add
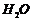
to
get the structure of monomers.
-
How is Dacron obtained from
ethylene glycol and terephthalic acid?
Formula of
ethylene glycol is 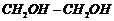 and of
terephthalicacid
and of
terephthalicacid . The process involves
condensation polymerization. i.e., removal of water.
. The process involves
condensation polymerization. i.e., removal of water.
View Answer play_arrow
-
What is a biodegradable polymer? Give an example of abiodegradable aliphatic polyester.
View Answer play_arrow
-
question_answer27)
Which
of the following polymers of glucose is
stored by animals ?
(a) Cellulose (b) Amylose
(c) Amylopectin (a) Glycogen
View Answer play_arrow
-
question_answer28)
Which
of the following is not a semi synthetic polymer ?
(a) cis-Polyisoprene (b) Cellulose nitrate
(c) Cellulose acetate (d) Vulcanised rubber
View Answer play_arrow
-
question_answer29)
The
commercial name of polyacrylonitrile is
(a) Dacron (b) Orion (acrilan)
(c) PVC (d) Bakelite
View Answer play_arrow
-
question_answer30)
Which
of the following polymer is biodegradable ?
(a) (b)
(b) (c)
(c) (d)
(d)
View Answer play_arrow
-
question_answer31)
In
which of the following polymers, ethylene
glycol is one of the momoner units ?
(a)
 (b)
(b) (c)
(c)
 (d)
(d)
View Answer play_arrow
-
question_answer32)
Which
of the following statements is not about low density polythene ?
(a) Tough (b) Hard
(c) Poor conductor of electricity
(d) Highly branched structure
View Answer play_arrow
-
question_answer33)
 is a polymer having
unit
is a polymer having
unit
(a) (b)
(c) (d)
View Answer play_arrow
-
question_answer34)
Which
of the following polymer can be for by using the following monomer unit?
 (a) Nylon 6, 6 (b) Nylon 2-nylon 6
(c) Melamine polymer (d) Nylon-6
(a) Nylon 6, 6 (b) Nylon 2-nylon 6
(c) Melamine polymer (d) Nylon-6
View Answer play_arrow
-
question_answer35)
Which
of the following polymers, need atleast one diene monomer for their preparation
?
(a) Dacron (b) Buna-S
(c) Neoprene (d) No volac
View Answer play_arrow
-
question_answer36)
Which
of the following are characteristics of
thermosetting polymers ?
(a) Heavily branched cross linked polymers
(b) Linear slightly branched long chain molecules
(c) Become infusible on moulding so cannot be reused
(d) Soften on heating and harden on cooling, can be reused
View Answer play_arrow
-
question_answer37)
Which
of the following polymers are thermoplastic ?
(a)Teflon (b) Natural rubber
(c) Neoprene (d) Polystyrene
View Answer play_arrow
-
question_answer38)
Which
of the following polymers are used as
fibre?
(a) Polytetrafluoroethane
(b) Polychloroprene
(c) Nylon (a)Terylene
View Answer play_arrow
-
question_answer39)
Which
of the following are addition polymers?
(a) Nylon
(b) Melamine formaldehyde resin
(c) Orion (d) Polystyrene
View Answer play_arrow
-
question_answer40)
Which
of the following polymers are condensation polymers ?
(a) Bakelite (b) Teflon
(c) Butyl rubber
(d) Melamine formaldehyde resin
View Answer play_arrow
-
question_answer41)
Which
of the following monomers form biodegradable polymers ?
(a) 3-hydroxybutanoic acid + 3-hydroxypentanoic acid
(b) Glycine + aminocaproic acid
(c) Ethylene glycol + phthalic acid
(d) Caprolactum
View Answer play_arrow
-
question_answer42)
Which
of the following are exmples of synthetic rubber ?
(a) Polychloroprene (b) Polyacrylonitrile
(c) Buna-N (d) cis-Polyisoprene
View Answer play_arrow
-
question_answer43)
Which
of the following polymers can have strong intermolecular forces ?
(a) Nylon (b) Polystyrene
(c) Rubber (d) Polyesters
View Answer play_arrow
-
question_answer44)
Which
of the following polymers have vinylic
monomer units ?
(a) Acrilan (b) Polystyrene
(c) Nylon (d) Teflon
View Answer play_arrow
-
question_answer45)
Vulcanisation
makes rubber.........
(a) more elastic
(b) soluble in inorganic solvent
(c) crystalline (d) more stiff
View Answer play_arrow
-
question_answer46)
A
natural linear polymer of2-methyl-l, 3-butadiene becomes hard on treatment with
sulphur between 373 to 415 K and ?S?S? bonds are formed between chains. Write
the structure of the product of this treatment ?
View Answer play_arrow
-
question_answer47)
Identify
the type of polymer.
?A?A?A?A?A?A?
View Answer play_arrow
-
question_answer48)
Identify
the type of polymer.
?A?B?B?A?A?A?B?A?
View Answer play_arrow
-
question_answer49)
Out
of chain growth polymerisation and step growth polymer is ation, in which type
will you place the " following :

View Answer play_arrow
-
question_answer50)
Identify
the type of polymer given in the following figure.

View Answer play_arrow
-
question_answer51)
Identify
the polymer given below :

View Answer play_arrow
-
question_answer52)
Why
are rubbers called elastomers ?
View Answer play_arrow
-
question_answer53)
Can
enzyme be called a polymer ?
View Answer play_arrow
-
question_answer54)
Can
nucleic acids, proteins and starch be considered as step growth polymers ?
View Answer play_arrow
-
question_answer55)
How
is the following resin intermediate prepared and which polymer is formed by
this monomer unit ?

View Answer play_arrow
-
question_answer56)
To
have practical applications why are cross links required in rubber ?
View Answer play_arrow
-
question_answer57)
Why
does cis-polyisoprene possess elastic property ?
View Answer play_arrow
-
question_answer58)
What
is the structural difference between HDP and LDP? How does the structure
account for different behaviour and nature, hence the use of a polymer ?
View Answer play_arrow
-
question_answer59)
What
is the role of benzoyl peroxide in addition polymerisation of alkenes ? Explain
its mode of action with the help of an example.
View Answer play_arrow
-
question_answer60)
Which
factor imparts crystalline nature to a polymer like nylon ?
View Answer play_arrow
-
question_answer61)
Name
the polymers used in laminated sheets and give the name of monomeric units
involved in its formation.
View Answer play_arrow
-
question_answer62)
Which
type of bio molecules have some structural similarity with synthetic polyamides
? What is this similarity ?
View Answer play_arrow
-
question_answer63)
Why
should the monomers used in addition polymerisation through free radical
pathway be very pure ?
View Answer play_arrow
-
question_answer64)
Match
the polymer of column I with correct monomer of column II.
|
|
Column I
|
|
Column II
|
|
(i)
|
High density polythene
|
(a)
|
Isoprene
|
|
(ii)
|
Neoprene
|
(b)
|
Tetrafluoroethene
|
|
(iii)
|
Natural rubber
|
(c)
|
Chloroprene
|
|
(iv)
|
Teflon
|
(d)
|
Acrylonitrile
|
|
(v)
|
Acrilan
|
(e)
|
Ethene
|
View Answer play_arrow
-
question_answer65)
Match
the polymers given in Column I with their chemical names given in Column II.
|
|
Column I
|
|
Column II
|
|
(i)
|
Nylon 6
|
(a)
|
Polyvinyl chloride
|
|
(ii)
|
PVC
|
(b)
|
Polyacrylonitrile
|
|
(iii)
|
Acrilan
|
(c)
|
Polycaprolactum
|
|
(iv)
|
Natural rubber
|
(d)
|
Low density polythene
|
|
(v)
|
LDP
|
(e)
|
Cis-Polyisoprene
|
View Answer play_arrow
-
question_answer66)
Match
the polymers given in Column I with their commercial names given in Column n.
|
|
Column I
|
|
Column II
|
|
(i)
|
Polyester
of glycol and phthalic acid
|
(a)
|
Nvolac
|
|
(ii)
|
Copolymer
of 1,3-butadiene and styrene
|
(b)
|
Glyptal
|
|
(iii)
|
Phenol
and formaldehyde resin
|
(c)
|
Buna-S
|
|
(iv)
|
Polyester
of glycol and terephthalic acid
|
(d)
|
Buna-N
|
|
(v)
|
Copolymer
of 1, 3-butadiene and acrylonitrile
|
(e)
|
Dacron
|
View Answer play_arrow
-
question_answer67)
Match
the polymers given in Column I with their main applications given in Column II.
|
|
Column I
|
|
Column II
|
|
(i)
|
Bakelite
|
(a)
|
Unbreakable
crockery
|
|
(ii)
|
Low
density polythene
|
(b)
|
Non-stick
cook wares
|
|
(iii)
|
Melamine-formaldehyde
resin
|
(c)
|
Packaging
material for shock absorbance
|
|
(iv)
|
Nylon
6
|
(d)
|
Electrical switches
|
|
(v)
|
Polytetrafluoroethene
|
(e)
|
Squeeze bottles
|
|
(vi)
|
Polystyrene
|
(f)
|
Tyre, cords
|
View Answer play_arrow
-
question_answer68)
Match
the polymers given in Column I with the preferred mode of polymerisation
followed by their monomers.
|
|
Column I
|
|
Column II
|
|
(i)
|
Nylon- 6,6
|
(a)
|
Free radical polymerization
|
|
(ii)
|
PVC
|
(b)
|
Ziegler-Natta polymerization or coordination
polymerization
|
|
(iii)
|
HDP
|
(c)
|
Anionic polymerization
|
|
|
|
(d)
|
Condensation polymerisation
|
View Answer play_arrow
-
question_answer69)
Match
the polymers given in Column I with the type of linkage present an them given
in Column II.
|
|
Column I
|
|
Column II
|
|
(i)
|
Terylene
|
(a)
|
Glycosidic linkage
|
|
(ii)
|
Nylon
|
(b)
|
Ester linkage
|
|
(iii)
|
Cellulose
|
(c)
|
Phosphodiester linkage
|
|
(iv)
|
Protein
|
(d)
|
Amide linkage
|
|
(v)
|
RNA
|
|
|
View Answer play_arrow
-
question_answer70)
Match
materials given in Column I with the polymers given in Column II.
|
|
Column I
|
|
Column II
|
|
(i)
|
Natural rubber latex
|
(a)
|
Nylon
|
|
(ii)
|
Wood laminates
|
(b)
|
Neoprene
|
|
(iii)
|
Ropes and fibres
|
(c)
|
Dacron
|
|
(iv)
|
Polyester fabric
|
(d)
|
Melamine formaldehyde resins
|
|
(v)
|
Synthetic rubber
|
(e)
|
Urea-formaldehyde resins
|
|
(vi)
|
Unbreakable crockery
|
(f)
|
Cis-polyisoprene
|
View Answer play_arrow
-
question_answer71)
Match
the polymers given in Column I with their repeating units given in Column II.
|
|
|
Column I
|
|
Column II
|
|
(i)
|
|
Acrilan
|
(a)
|

|
|
(ii)
|
|
Polystyrene
|
(b)
|

|
|
(iii)
|
|
Neoprene
|
(c)
|
 
|
|
(iv)
|
|
No volac
|
(d)
|

|
|
(v)
|
|
Buna-N
|
(f)
|

|
View Answer play_arrow
-
question_answer72)
Assertion
: Rayon is a semi synthetic polymer and is taken as a better choice than
cotton fabric.
View Answer play_arrow
-
question_answer73)
Assertion
: Most of the Synthetic polymers are not biodegradable.
View Answer play_arrow
-
question_answer74)
Assertion
: Olefinic monomers undergo addition polymerisation.
View Answer play_arrow
-
question_answer75)
Assertion:
Polyamides are best used as fibres because of high tensile strength.
View Answer play_arrow
-
question_answer76)
Assertion:
For making rubber synthetically, isoprene molecules are polymerised.
View Answer play_arrow
-
question_answer77)
Assertion:
Network polymers are thermosetting.
View Answer play_arrow
-
question_answer78)
Assertion:
Polytetrafluoroethene is used in making non-stick cook wares.
View Answer play_arrow
-
question_answer79)
Synthetic
polymers do not degrade in the environment for a long time. How can
biodegradable synthetic polymers be made. Differentiate between biopolymers and
biodegradable polymers and give examples of each type.
View Answer play_arrow
-
question_answer80)
Differentiate
between rubbers and plastics on the basis of intermolecular forces.
View Answer play_arrow
-
question_answer81)
Phenol
and formaldehyde undergo condensation to give a polymer (A) which on heating
with formaldehyde gives a thermosetting polymer (B). Name the polymers. Write
the reactions involved in the formation of (A). What is the structural difference
between two polymers?
View Answer play_arrow
-
question_answer82)
Low
density polythene and high density polythene, both are polymers of ethene but
there is marked difference in their properties. Explain.
View Answer play_arrow
-
question_answer83)
Which
of the following polymers soften on heating and harden on cooling? What are the
polymers with this property collectively called ? What are the structural
similarities between such polymers ?
Bakelite, urea-formaldehyde resin, polythene, polyvinyl, polystyrene.
View Answer play_arrow
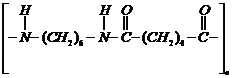 (ii)
(ii) 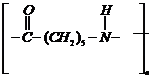 (iii)
(iii)  (i) Break ?CO ?NH ? bond and add
(i) Break ?CO ?NH ? bond and add
![]() (b)
(b)![]() (c)
(c)![]() (d)
(d)
 is a polymer having
unit
is a polymer having
unit

![]()
