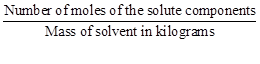-
Calculate the mass percentage of benzene (C6H6) and carbon tetrachloride (CCl4) if 22 g of benzene is dissolved in 122 g of carbon tetrachloride.
View Answer play_arrow
-
Calculate the molarity of each of the following solutions :
(a) 30 g of Co(NO3)2.6H2O in 4.3 L of solution
(b) 30 mL of 0.5 MH2SO4 diluted to 500 mL
View Answer play_arrow
-
Calculate the mole fraction of benzene in solution containing 30% by mass of it in carbon tetrachloride.
View Answer play_arrow
-
Calculate the mass of urea (NH2CONH2) required in making 2.5 kg of 0.25 molal aqueous solution.
View Answer play_arrow
-
Calculate (a) molarity (b) molarity and (c) mole fraction of KI if the density of 20% (mass/mass) aqueous KI is 1.202 g mL–1.
View Answer play_arrow
-
H2S, a toxic gas with rotten egg like smell, is used for qualitative analysis. If the solubility of H2S in water at STP is 0.195 m, calculate Henry's law constant.
View Answer play_arrow
-
Henry's law constant for CO2 in water is 1.67 x 108 Pa at 298 K. Calculate the quantity of CO2 in 500 mL of soda water when packed under 2.5 atm CO2 pressure at 298 K.
View Answer play_arrow
-
The vapour pressure of pure liquids A and B are 450 and 700 mrn Hg at 350 K respectively. Find out the composition of the liquid mixture if total vapour pressure is 600 mm Hg. Also find the composition of the vapour phase.
View Answer play_arrow
-
Vapour pressure of pure water at 298 K is 23.8 mm Hg. 50 g of urea (NH2CONH2) is dissolved in 850 g of water. Calculate the vapour pressure of water for this solution and its relative lowering.
View Answer play_arrow
-
Boiling point of water at 750 mm Hg is 96.63°C. How much sucrose is to be added to 500 g of water such that it boils at 100°C? Molal elevation constant for water is 0.52 K kg mol–1.
View Answer play_arrow
-
Calculate the mass of ascorbic acid (vitamin C, C6H8O6) to be dissolved in 75 g of acetic acid to lower its melting point by 1.5°C. Given Kf = 3.9 K kg mol–1.
View Answer play_arrow
-
Calculate the osmotic pressure in pascals exerted by a solution prepared by dissolving 1.0 g of polymer of molar mass 185,000 in 450 mL of water at 37°C.
View Answer play_arrow
-
Define the term solution. How many types of solutions are formed? Write briefly about each kind of solution with an example.
View Answer play_arrow
-
Given an example of a solid solution in which the solute is a gas.
View Answer play_arrow
-
Define the following terms
(i) Mole fraction (ii) Molality
(iii) Molarity (iv) Mass percentage.
View Answer play_arrow
-
Concentrated nitric acid used in the laboratory work is 68% nitric acid by mass in aqueous solution. What should be the molarity of such a sample of the acid if the density of the solutions is 1.504 g mL–1?
View Answer play_arrow
-
A solution of glucose in water is labeled as 10 percent w/w, what would be the molality and mole fraction of each component in the solution? If the density of the solution is 1.2 g mL–1, then what shall be the molarity of the solution?
View Answer play_arrow
-
How many mL of 0.1 M HCl are required to react completely with 1 g mixture of Na2CO3 and

containing equimolar amounts of the two?
-
A solution is obtained by mixing 300 g of 25% solution and 400 g of 40% solution by mass.
View Answer play_arrow
-
An antifreeze solution is prepared from 222.6 g of ethylene glycol (C2H6O2) and 200 g of water. Calculate at molality of the solution. If the density of the solution is 1.072 g mL–1, then what shall be the molarity of the solution?
View Answer play_arrow
-
A sample of drinking water was found to be severely contaminated with chloroform, CHCl3, supposed to be carcinogen. The level of contamination was 15 ppm (by mass).
(i) Express this in percent by mass.
(ii) Determine the molality of chloroform in the water sample.
View Answer play_arrow
-
What role does the molecular interaction play in solution of alcohol and water?
View Answer play_arrow
-
Why do gases nearly always tend to be less soluble in liquids as the temperature is raised?
View Answer play_arrow
-
State Henry's law. Mention some of its important applications.
View Answer play_arrow
-
The partial pressure of ethane over a saturated solution containing 6.56 x 10–2 g of ethane is 1 bar. If the solution contains 5.00 x 10–2 g of ethane, then what shall be the partial pressure of the gas?
View Answer play_arrow
-
What is meant by positive and negative deviations from Raoult's law and how is the sign of
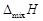
related to positive and negative deviations from Raoult's law?
-
An aqueous solution of 2 percent of non-volatile solute exerts a pressure of 1.004 bar at the boiling point of the solvent. What is the molecular mass of the solute?
View Answer play_arrow
-
Heptane and octane form ideal solution. At 373 K, then vapour pressure of the two liquid components are 105.2 kPa and 46.8 kPa respectively. What will be the vapour pressure, in bar of a mixture of 25.0 g of heptane and 35.0 g of octane?
View Answer play_arrow
-
The vapour pressure of water is 12.3 kPa at 300 K. Calculate the vapour pressure of 1 molal solution of a solute in it.
View Answer play_arrow
-
Calculate the mass of a non-volatile solute (molecular mass 40 g mol–1) which should be dissolved in 114 g octane to reduce its vapour pressure to 80%.
View Answer play_arrow
-
A solution containing 30 g of a non-volatile solute exactly in 90 g water has a vapour pressure of 2.8 kPa at 298 K. Further 18 g of water is then added to the solution, the new vapour pressure becomes 2.9 kPa at 298 K.
Calculate
(i) molecular mass of the solute
(ii) vapour pressure of water at 298 K.
View Answer play_arrow
-
A 5% solution (by mass) of cane sugar in water has freezing point of 271 K. Calculate the freezing point of 5% glucose in water if freezing point of pure water is 273.15 K.
View Answer play_arrow
-
The elements A and B form purely covalent compounds having molecular formulae AB2 and AB4. When dissolved in 20 g of benzene, 1 g of AB2 lowers the freezing point by 2.3 K, whereas 1g of AB4 lowers it by 1.3 K. The molal depression constant for benzene is 5.1 K kg mol–1, calculate the atomic mass of A and atomic mass of B.
View Answer play_arrow
-
At 300 K, 36 g of glucose present per litre in its solution has an osmotic pressure of 4.98 bar. If the osmotic pressure of solution is 1.52 bar at the same temperature, what would be its concentration?
View Answer play_arrow
-
Suggest the most important type of intermolecular interaction in the following pairs:
(i) n-hexane and n-octane
(ii) I2 and CCl4
(iii) NaClO4 and water
(iv) methanol and acetone
View Answer play_arrow
-
Based on solute-solvent interactions, arrange the following in order of increasing solubility in n-octane and explain :
Cyclohexane, KCI, CH3OH, CH3CN
View Answer play_arrow
-
Amongst the following compounds, identify which are insoluble, partially soluble and highly soluble in water.
(i) phenol (ii) toluene
(iii) formic acid (iv) ethylene glycol
(iv) chloroform (vi) pentanol.
View Answer play_arrow
-
If the density of some lake water is 1.25 g mL–1 and contains 92 g of Na+ ions per kg of water, calculate the molality of Na+ ions in the lake.
View Answer play_arrow
-
If the solubility product of CuS is 6 x 10–16, calculate the maximum molarity of CuS in aqueous solution.
View Answer play_arrow
-
Calculate the mass percentage of aspirin (C9H8O4) in acetonitrile (CH3CN) when 6.5 g of C9H8O4 is dissolved in 450 g of CH3CN.
View Answer play_arrow
-
Nalorphene (C19H21NO3), similar to morphine, is used to combat withdrawal symptoms in narcotic users. Dose of nalorphene generally given is 1.5 mg. Calculate the mass of 1.5 x 10–3 m aqueous solution required for the above dose.
View Answer play_arrow
-
Calculate the amount of benzoic acid (C6H5COOH) required for preparing 250 mL of 0.15 M solution in methanol.
View Answer play_arrow
-
The depression in freezing point of water observed for the same amount of acetic acid, trichloroacetic acid and trifluoroacetic acid increases in the order given above. Explain briefly.
View Answer play_arrow
-
Calculate the depression in the freezing point of water when 10 g of CH3CH2CHClCOOH is a dried to 250 g of water, Ka = 1.4 x 10–3, Kf = 1.86 K kg mol–1
View Answer play_arrow
-
19.5 g of CH2FCOOH is dissolved in 500 g of water. The depression in the freezing point observed is 1.0°C. Calculate the van't Hoff factor and dissociation constant of fluoroacetic acid. Kf for water is 1.86 K kg mol–1.
View Answer play_arrow
-
Vapour pressure of water at 293 K is 17.535 mm Hg. Calculate the vapour pressure of water at 293 K when 25 g of glucose is dissolved in 450 g of water.
View Answer play_arrow
-
Henry's law constant for the molarity of methane in benzene at 298 K is 4.27 x 105 mm Hg. Calculate the solubility of methane in benzene at 298 K under 760 mm Hg.
View Answer play_arrow
-
100 g of liquid A (molar mass 140 g mol-1) was dissolved in 1000 g of liquid B (molar mass 180 g mol-1). The vapour pressure of pure liquid B was found to be 500 torr. Calculate the vapour pressure of pure liquid B was found to be 500 torr. Calculate the vapour pressure of pure liquid A and its vapour pressure in the solution if the total vapour pressure c' the solution is 475 torr.
View Answer play_arrow
-
Vapour pressure of pure acetone and
chloroform at 328 K are 632.8 mm Hg and 741.8 nun Hg respectively. Assuming
that they form ideal solution over the entire range of composition, plot ptotal
p, pchloroform and Pacetone as a function of xacetone.
The experimental data observed for different compositions of mixtures is :
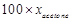
|
0
|
11.8
|
23.4
|
36.0
|
50.8
|
58.2
|
64.5
|
72.1
|
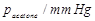
|
0
|
54.9
|
110.1
|
202.4
|
322.7
|
405.9
|
454.1
|
521.1
|
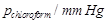
|
632.8
|
548.1
|
469.4
|
359.7
|
257.7
|
193.6
|
161.2
|
120.7
|
Plot this data also on the same graph
paper. Indicate whether it has positive deviation or negative deviation from
the ideal solution.
View Answer play_arrow
-
Benzene and toluene form ideal solution over the entire range of composition. The vapour pressures of pure benzene and toluene at 300 K are 50.71 nun Hg and 32.06 mm Hg respectively. Calculate the mole fraction of benzene in the vapour phase if 80 g of benzene is mixed with 100 g of toluene.
View Answer play_arrow
-
The air is a mixture of a number of gases. The major components are oxygen and nitrogen with approximate proportion of 20% is to 79% by volume at 298 K. The water is in equilibrium with air at a pressure of 10 attn. At 298 K, if the Henry's law constant for oxygen and nitrogen are 3.30 x 107 mm and 6.51 x 107 mm, respectively, calculate the composition of these gases in water.
View Answer play_arrow
-
Determine the amount of CaCl2 (i = 2.47) dissolved in 2.5 litre of water such that its osmotic pressure is 0.75 atm at 27° C.
View Answer play_arrow
-
Determine the osmotic pressure of a solution prepared by dissolving 25 mg of K2SO4 in 2 litre of water at 25° C, assuming that it is completely dissociated.
View Answer play_arrow
-
question_answer54)
Which of the following units is useful in
relating concentration of solution with its vapour pressure?
(a) Mole fraction (b) Parts
per million
(c) Mass percentage (d)
Molality
View Answer play_arrow
-
question_answer55)
On dissolving sugar in water at room temperature
solution feels, cool to touch. Under which of the following cases dissolution
of sugar will be most rapid?
(a) Sugar crystals in cold water
(b) Sugar crystals in hot water
(c) Powdered sugar in cold
water
(d) Powdered sugar in hot water
View Answer play_arrow
-
question_answer56)
At equilibrium the rate of dissolution of a
solid solute in a volatile Liquid solvent is..........
(a) less than the rate of
crystallisation
(b) greater than the rate of
crystallisation
(c) equal to the rate of
crystallisation
(d)
zero
View Answer play_arrow
-
question_answer57)
A beaker contains a solution of substance 'A'.
Precipitation of substance 'A' takes place when small amount of 'A' is added to
the solution. The solution is..........
(a) saturated (b)
supersaturated
(c)
unsaturated (d) concentrated
Thinking Process
This problem includes concept of
saturated, unsaturated, supersaturated and concentrated solution.
View Answer play_arrow
-
question_answer58)
Maximum amount of a solid solute that can be
dissolved in a specified amount of a given liquid solvent does not depend upon..........
(a) temperature (b) nature
of solute
(c) pressure (d)
nature of solvent
View Answer play_arrow
-
question_answer59)
Low concentration of oxygen in the blood and
tissues of people living at high altitude is due to..........
(a) low temperature
(b) low atmospheric pressure
(c) high atmospheric pressure
(d) Both low temperature and
high atmospheric pressure
View Answer play_arrow
-
question_answer60)
Considering the formation, breaking and strength
of hydrogen bond, predict which of the following mixtures will show a positive
deviation from Raoult's law?
(a) Methanol and acetone
(b) Chloroform and acetone
(c) Nitric acid and
water
(d) Phenol and aniline
View Answer play_arrow
-
question_answer61)
Colligative properties depend on..........
(a) the nature of the solute particles
dissolved in solution
(b) the number of solute
particles in solution
(c) the physical properties of
the solute particles dissolved in solution
(d) the nature' of solvent
particles
View Answer play_arrow
-
question_answer62)
Which of the. following aqueous solutions should
have the highest boiling point?
(a) 1.0 M NaOH (b)
1.0 M Na2SC4
(c) 1.0 M N4N03
(d) 1.0 M KN03
View Answer play_arrow
-
question_answer63)
The unit of ebullioscopic constant is
(a) K kg mol-1 or K
(molality)-1
(b) mol kg K-1 or K-1
(molality)
(c) kg mol-1 K-1
or K-1 (molality)-1
(d) K mol kg-1 or K
(molality)
View Answer play_arrow
-
question_answer64)
In comparison to a ?0.01 M solution of glucose,
the depression in freezing point of a 0.01 M MgCl2 solution
is..........
(a) the same (b)
about twice
(c) about three times (d)
about six times
View Answer play_arrow
-
question_answer65)
An unripe mango placed in a concentrated salt
solution to prepare pickle shrivels because..........
(a) it gains water due to osmosis
(b) it loses water due to
reverse osmosis
(c) it gains water due to
reverse osmosis
(d) it loses water due to
osmosis
View Answer play_arrow
-
question_answer66)
At a given temperature, osmotic pressure of a
concentrated solution of a substance..........
(a) is higher than that of a dilute
solution
(b) is lower than that of a
dilute solution
(c) is same as that of a dilute
solution
(d) cannot be compared with
osmotic pressure of dilute solution
View Answer play_arrow
-
question_answer67)
Which of the following statements is false?
(a) Two different solutions of sucrose
of same molality prepared in different solvents will have the same depression
in freezing point.
(b) The osmotic pressure of a
solution is given by the equation 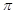 = CRT (where,
C is the molarity of the solution)
(c) Decreasing order of osmotic
pressure for 0.01 M aqueous solutions of barium chloride, potassium chloride,
acetic acid and sucrose is
BaCI2
> KCI > CH3COOH > sucrose
(d) According to Raoult's law,
the vapour pressure exerted by a volatile component of a solution is directly
proportional to its mole fraction in the solution
= CRT (where,
C is the molarity of the solution)
(c) Decreasing order of osmotic
pressure for 0.01 M aqueous solutions of barium chloride, potassium chloride,
acetic acid and sucrose is
BaCI2
> KCI > CH3COOH > sucrose
(d) According to Raoult's law,
the vapour pressure exerted by a volatile component of a solution is directly
proportional to its mole fraction in the solution
View Answer play_arrow
-
question_answer68)
The values of van't Hoff factors for KCl, NaCI
and K2SO4 respectively .are
(a) 2, 2 and 2 (b) 2, 2 and 3
(c) 1,1 and 2 (d)
1, 1 and 1
View Answer play_arrow
-
question_answer69)
Which of the following statements is false?
(a) Units of atmospheric pressure and
osmotic pressure are the same
(b) In reverse osmosis, solvent
molecules move through a semipermeable membrane from a region of lower
concentration of solute to a region of higher concentration
(c) The value of molal
depression constant depends on nature of solvent
(d) Relative lowering of vapour
pressure, is a dimensionless quantity
View Answer play_arrow
-
question_answer70)
Value of Henry's constant KH..........
(a) increases with increase in
temperature
(b) decreases with increase in
temperature
(c) remains
constant
(d) first increases then
decreases
View Answer play_arrow
-
question_answer71)
The value of Henry's constant, K is.........
(a) greater for gases with higher
solubility
(b) greater for gases with lower
solubility
(c) constant for all gases
(d) not related to the
solubility of gases
View Answer play_arrow
-
question_answer72)
Consider the figure and mark the correct option.
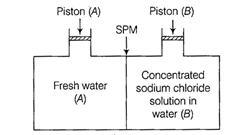 (a) Water will move from side
(A) to side (B) if a pressure lower osmotic pressure is applied on piston (B)
(b) Water will move from side
(B) to side (A) if a pressure greater than osmotic pressure is applied on
piston (B)
(c) Water will move from side (B)
to side (A) if a pressure equal to osmotic pressure is applied on piston (B)
(d) Water will move from side
(A) to side (B) if pressure equal to osmotic pressure is applied on piston (A)
(a) Water will move from side
(A) to side (B) if a pressure lower osmotic pressure is applied on piston (B)
(b) Water will move from side
(B) to side (A) if a pressure greater than osmotic pressure is applied on
piston (B)
(c) Water will move from side (B)
to side (A) if a pressure equal to osmotic pressure is applied on piston (B)
(d) Water will move from side
(A) to side (B) if pressure equal to osmotic pressure is applied on piston (A)
View Answer play_arrow
-
question_answer73)
We have three aqueous solutions of NaCI labelled
as 'A', 'B' and 'C with concentrations 0.1 M, 0.01 M and 0.001 M, respectively.
The value of van't Hoff factor for these solutions will be in the order..........
(a)  (b)
(b)
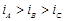 (c)
(c) 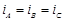 (d)
(d)
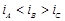
View Answer play_arrow
-
question_answer74)
On the basis of information given below mark the
correct option.
Information
(i) In bromoethane and
chloroethane mixture intermolecular interactions of A?A and B?B type are nearly
same as A?B type interactions.
(ii) In ethanol and acetone
mixture A?A or B?B type intermolecular interactions are stronger than A?B type
interactions.
(iii) In chloroform and acetone
mixture A?A or B?B type intermolecular interactions are weaker than A?B type
interactions.
(a) Solution (ii) and (iii) will
follow Raoult's law
(b) Solution (ij will follow
Raoult's law
(c) Solution (ii) will show
negative deviation from Raoult's law
(d) Solution (iii) will show
positive deviation from Raoult's law
View Answer play_arrow
-
question_answer75)
Two beakers of capacity 500 mL were taken. One
of these beakers, labelled as "A", was filled with 400 mL water
whereas the beaker labelled "B' was filled with 400 mL of 2M solution of
NaCL At the same temperature both the beakers were placed in closed containers
of same material and same capacity as shown in figure.
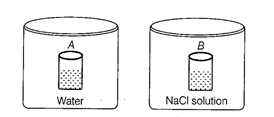 At a given temperature, which of
the following statement is correct about the vapour pressure of pure water and
that of NaCI solution?
(a) Vapour pressure in container
(A) is more than that in container (B)
(b) Vapour pressure in container
(A} is less than that in container (B)
(c) Vapour pressure is equal in
both the containers
(d) Vapour pressure in container
(B) is twice the vapour pressure in container (A)
At a given temperature, which of
the following statement is correct about the vapour pressure of pure water and
that of NaCI solution?
(a) Vapour pressure in container
(A) is more than that in container (B)
(b) Vapour pressure in container
(A} is less than that in container (B)
(c) Vapour pressure is equal in
both the containers
(d) Vapour pressure in container
(B) is twice the vapour pressure in container (A)
View Answer play_arrow
-
question_answer76)
If two liquids A and B form minimum boiling
azeotrope at some specific composition then
(a) A?B interactions are stronger than
those between A?A or B?B
(b) vapour pressure of solution
increases because more number of molecules of liquids A and 6 can escape from
the solution
(c) vapour pressure of solution
decreases because less number of molecules of only one of the liquids escape
from the solution
(d) A?B interactions are weaker
than those between A?A or B?B
View Answer play_arrow
-
question_answer77)
4 L of 0.02 M aqueous solution of NaCI was
diluted by adding 1 L of water The molality of the resultant solution is??
(a)0.004 (b)0.008 (c)0.012 (d)0.016
View Answer play_arrow
-
question_answer78)
On the basis of information given below mark the
correct option.
Information On adding acetone to
methanol some of the hydrogen bonds between methanol molecules break.
(a) At specific composition
methanol ? acetone mixture will from minimum boiling azeotrope and will show
positive deviation from Raoult?s law
(b) At specific composition
methanol ? acetone mixture will from maximum boiling azeotrope and will show
positive deviation from Raoult?s law
(c) At specific composition
methanol ? acetone mixture will from minimum boiling azeotrope and will show
negative deviation from Raoult?s law
(d) At specific composition
methanol ? acetone mixture will from maximum boiling azeotrope and will show
negative deviation from Raoult?s law
View Answer play_arrow
-
question_answer79)
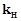 value for
Ar (g), Co2 (g), and CH4 are 40.39, 1.67, 1.83 ´ 10-5 and 0.413 respectively
Arrange these gases in the order of
their increasing solubility
(a)
value for
Ar (g), Co2 (g), and CH4 are 40.39, 1.67, 1.83 ´ 10-5 and 0.413 respectively
Arrange these gases in the order of
their increasing solubility
(a)  (b)
(b) 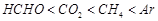 (C)
(C) 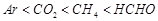 (D)
(D) 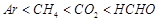
View Answer play_arrow
-
question_answer80)
Which of the following factor(s) affect the
solubility of a gaseous solute in the fixed volume of liquid solvent?
(i) Nature of solute (ii)
Temperature
(iii)Pressure
(a) (i) and (iii) at constant
T
(b) (i) and (ii) at constant p
(c) (ii) and
(iii)
(d) Only (iii)
View Answer play_arrow
-
question_answer81)
Intermolecular forces between two benzene
molecules are nearly of same strength as those between two toluene
molecules. For a mixture of benzene and toluene, which of the following
are not true?
(a) 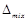 H
= zero
(b)
H
= zero
(b) 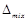 V=zero
(c) These will form minimum
boiling azeotrope
(d) These will not form ideal
solution
V=zero
(c) These will form minimum
boiling azeotrope
(d) These will not form ideal
solution
View Answer play_arrow
-
question_answer82)
Relative lowering of vapour pressure is a colligative
property because
(a) it depends on the concentration of a
non-electrolyte solute in solution and does not depend on the nature of the
solute molecules
(b) it depends on number of
particles of electrolyte solute in solution and does not depend on the nature
of the solute particles
(c) it depends on the
concentration of a non-electrolyte solute in solution as well as on the nature
of the solute molecules
(d) it depends on the
concentration of an electrolyte or non-electrolyte solute in solution as well
as on the nature of solute molecules
View Answer play_arrow
-
question_answer83)
van't Hoff factor (?) is given by the expression
(a) 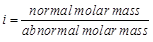 (b)
(b) 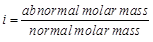 (c)
(c) 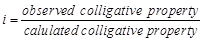 (d)
(d) 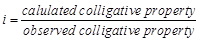
View Answer play_arrow
-
question_answer84)
Isotonic solutions must have the same..........
(a) solute
(b) density
(c) elevation in boiling
point
(d) depression in freezing point
View Answer play_arrow
-
question_answer85)
Which of the following binary mixtures will have
same composition in liquid and vapour phase?
(a) Benzene-toluene
(b) Water-nitric acid
(c) Water-ethanol
(d) n ? hexane ? n-heptane
View Answer play_arrow
-
question_answer86)
In isotonic solutions..........
(a) solute and solvent both are same
(b) osmotic pressure is same
(c) solute and solvent may or
may not be same
(d) solute is always same
solvent may be different
View Answer play_arrow
-
question_answer87)
tor a binary ideal liquid
solution, the variation in total vapour pressure versus composition of solution
is given by which of the curves?
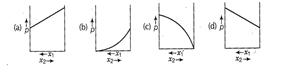
View Answer play_arrow
-
question_answer88)
Colligative properties are observed when..........
(a) a non-volatile solid is dissolved in
a volatile liquid
(b) a non-volatile liquid is
dissolved in another volatile liquid
(c) a gas is dissolved in
non-volatile liquid
(d) a volatile liquid is
dissolved in another volatile liquid
View Answer play_arrow
-
question_answer89)
Components of a binary mixture of two liquids A
and B were being separated by distillation. After some time separation of
components stopped and composition of vapour phase became same as that of
liquid phase. Both the components started coming in the distillate. Explain why
this happened?
View Answer play_arrow
-
question_answer90)
Explain why on addition of 1 mole of NaCl to 1 L
of water, the boiling point of water increases, while addition of 1 mole of
methyl alcohol to 1 L of water decreases its boiling point.
View Answer play_arrow
-
question_answer91)
Explain the solubility rule "like dissolves
like" in terms of intermolecular forces that exist in solutions.
View Answer play_arrow
-
question_answer92)
Concentration terms such as mass percentage,
ppm, mole fraction and molality are independent of temperature, however
molarity is a function of temperature. Explain.
View Answer play_arrow
-
question_answer93)
What is the significance of Henry's law constant
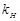 ?
?
View Answer play_arrow
-
question_answer94)
Why are aquatic species more comfortable in cold
water in comparision to warm water?
View Answer play_arrow
-
question_answer95)
(a) Explain the following phenomena with the
help of Henry's law.
(i) Painful condition known as bends.
(ii) Feeling of weakness and
discomfort in breathing at high altitude.
(b) Why soda water bottle
kept at room temperature fizzes on opening?
View Answer play_arrow
-
question_answer96)
Why is the vapour pressure of an aqueous solution
of glucose lower than that of water?
View Answer play_arrow
-
question_answer97)
How does sprinkling of salt help in clearing the
snow covered roads in hilly areas? Explain the phenomenon involved in the
process.
View Answer play_arrow
-
question_answer98)
What is "semipermeable membrane"?
View Answer play_arrow
-
question_answer99)
Give an example of a material used for making
semipermeable membrane for carrying out reverse osmosis.
View Answer play_arrow
-
question_answer100)
Match the items given in Column I and Column
II.
|
|
Column I
|
Column 11
|
|
A.
|
Saturated solution
|
1. Solution having same osmotic pressure at a
given temperature as that of given solution.
|
|
B.
|
Binary solution
|
2. A solution whose osmotic pressure is less than
that of another.
|
|
C.
|
Isotonic solution
|
3. Solution with two components.
|
|
D.
|
Hypotonic solution
|
4. A solution which contains maximum amount of
solute that can be dissolved in a given amount of solvent at a given
temperature.
|
|
E.
|
Solid solution
|
5. A solution whose osmotic pressure is more than
that of another.
|
|
F.
|
Hypertonic solution
|
6. A solution in solid phase.
|
View Answer play_arrow
-
question_answer101)
Match the items given in Column I with the type
of solutions given in Column II.
|
|
Column 1
|
Column 11
|
|
A.
|
Soda water
|
1. A solution of gas in solid
|
|
B.
|
Sugar solution
|
2. A solution of gas in gas
|
|
C.
|
German silver
|
3. A solution of solid in liquid
|
|
D.
|
Air
|
4. A solution of solid in solid
|
|
E.
|
Hydrogen gas in palladium
|
5. A solution of gas in liquid
|
|
|
|
6. A solution of liquid in solid
|
View Answer play_arrow
-
question_answer102)
Match the laws given in Column I with
expressions given in Column II.
|
|
Column
|
Column 11
|
|
A.
|
1 Raoult's law
|
1. 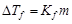
|
|
B.
|
Henry's law
|
2. 
|
|
C.
|
Elevation of boiling point
|
3. 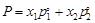
|
|
D.
|
Depression in freezing point
|
4. 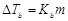
|
|
E.
|
Osmotic pressure
|
5. 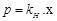
|
View Answer play_arrow
-
question_answer103)
Match the terms given in Column I with
expressions given in Column II
|
Column 1
|
Column 11
|
|
A. Mass percentage
|
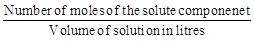
|
|
B. Volume percentage
|
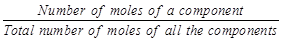
|
|
C. Mole fraction
|
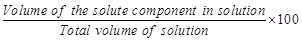
|
|
D. Molality
|
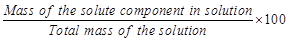
|
|
E. Molarity
|
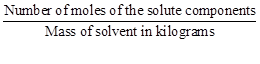
|
View Answer play_arrow
-
question_answer104)
Assertion (A) Molarity of a solution in
Liquid state changes with temperature.
Reason (R) The volume of a
solution changes with change in temperature.
View Answer play_arrow
-
question_answer105)
Assertion (A) When methyl alcohol is added
to water, boiling point of water increases.
Reason (R) When a volatile solute
is added to a volatile solvent elevation in boiling point is observed.
View Answer play_arrow
-
question_answer106)
Assertion (A) When NaCI is added to water a
depression in freezing point is observed.
Reason (R) The lowering of vapour
pressure of a solution causes depression in the freezing point.
View Answer play_arrow
-
question_answer107)
Assertion (A) When a solution is separated
from the pure solvent by a semipermeable membrane, the solvent molecules pass
through it from pure solvent side to the solution side.
Reason (R) Diffusion of solvent
occurs from a region of high concentration solution to a region of low
concentration solution.
View Answer play_arrow
-
question_answer108)
Define the following modes of expressing the
concentration of a solution? Which of these modes are independent of temperature
and
why?
(a) w/w (mass
percentage)
(b) V/V (volume percentage)
(c) w/V (mass by volume
percentage)
(d) ppm (parts per million)
(e) / (mole
fraction)
(f) M (molarity)
(g) m (molality)
View Answer play_arrow
-
question_answer109)
Using Raoult's law explain how the total vapour
pressure over the solution is related to mole fraction of components in the
following solutions.
(a) CHC13 (l) and CH2Cl2
(l)
(b) NaCl(s) and H2O
(l)
View Answer play_arrow
-
question_answer110)
Explain the terms ideal and non-ideal solutions
in the light of forces of interactions operating between molecules in liquid
solutions.
View Answer play_arrow
-
question_answer111)
Why is it not possible to obtain pure ethanol by
fractional distillation?
What general name is given to binary
mixtures which show deviation from Raoult's law and whose components cannot be
separated by fractional distillation. How many types of such mixtures are
there?
View Answer play_arrow
-
question_answer112)
When kept in water, raisin swells in size. Name
and explain the phenomenon involved with the help of a diagram. Give three applications
of the phenomenon.
View Answer play_arrow
-
question_answer113)
Discuss biological and. industrial applications
of osmosis.
View Answer play_arrow
-
question_answer114)
How can you remove the hard calcium carbonate
layer of the egg without damaging its semipermeable membrane? Can this egg be inserted
into a bottle with a narrow neck without distorting its shape?
Explain the process involved.
View Answer play_arrow
-
question_answer115)
Why is the mass determined by measuring a
colligative property in case of some solutes abnormal? Discuss it with the help
of van't Hoff factor.
View Answer play_arrow
![]() (b)
(b) ![]() (c)
(c) ![]() (d)
(d) ![]()
![]() ?
?