-
Why are substances like platinum and palladium often used for carrying out electrolysis of aqueous solutions?
View Answer play_arrow
-
Why does physisorption decrease with increase of temperature?
View Answer play_arrow
-
Why are powdered substances more effective adsorbents as compared to their crystalline forms?
View Answer play_arrow
-
Why is it necessary to remove CO when ammonia is obtained by Haber's process?
View Answer play_arrow
-
Why is ester hydrolysis slow in the beginning and becomes fast after some time?
View Answer play_arrow
-
What is the role of desorption in the process of catalysi?
View Answer play_arrow
-
What modification can you suggest in the Hardy Schulze law?
View Answer play_arrow
-
Why is it essential to wash the precipitate with water before estimating it quantitatively?
View Answer play_arrow
-
Distinguish between the meaning of the terms adsorption and absorption. Give one example of each.
View Answer play_arrow
-
What is the different between physical adsorption and chemisorption?
View Answer play_arrow
-
Give reason why a finally divided substance more effective as an adsorbent.
View Answer play_arrow
-
What are the factors which influence the adsorption of a gas on a solid?
View Answer play_arrow
-
What is an adsorption isotherm? Describe Freundlich adsorption isotherm.
View Answer play_arrow
-
What do you understand by activation of adsorbent? How is it achieved?
View Answer play_arrow
-
What role does adsorption play in heterogeneous catalysis?
View Answer play_arrow
-
Why is adsorption always exothermic?
View Answer play_arrow
-
How are the colloidal solutions classified on the basis of physical states of the dispersed phase and dispersion medium?
View Answer play_arrow
-
Discuss the effect of pressure and temperature on the adsorption of gases on solids.
View Answer play_arrow
-
What are lyophilic and lyophobic sols? Give an example of each type. Why is hydrophobic sols easily coagulated?
View Answer play_arrow
-
What is the difference between multimolecular and macromolecular colloids? Give one example of each. How are associated colloids different from these two types of colloids?
View Answer play_arrow
-
What are enzymes? Write in brief the mechanism of enzyme catalysis.
View Answer play_arrow
-
How are colloids classified on the basis of (a) physical states of components, (b) nature of dispersion medium and (c) interaction between dispersed phase and dispersion medium.
View Answer play_arrow
-
Explain what is observed when (i) a beam of light is passed through a colloidal sol; (ii) an electrolyte i.e. NaCI is added to hydrated ferric oxide sol; (iii) electric current is passed through acolloidal sol?
View Answer play_arrow
-
What are emulsions? What are their different types? Give example of each type.
View Answer play_arrow
-
What is demulsification? Name two demulsifiers.
View Answer play_arrow
-
Action of soap is due to emulsification and micelle formation. Comment.
View Answer play_arrow
-
Give four examples of heterogeneous catalysis.
View Answer play_arrow
-
What do you mean by activity and selectivity of catalyst?
View Answer play_arrow
-
Describe some features of catalysis by zeolites.
View Answer play_arrow
-
What is shape selective catalysis?
View Answer play_arrow
-
Explain the terms:
(i) Electrophoresis (ii) Coagulation
(iii) Dialysis (iv) Tyndall effect.
View Answer play_arrow
-
Give four uses of emulsions.
View Answer play_arrow
-
What are micelles? Give an example of miceller system.
View Answer play_arrow
-
Explain the terms with suitable examples
(i) Alcosol (ii) Aerosol and (iii) Hydroso
View Answer play_arrow
-
Comment on the statement that “colloid is nota substance but a state of substance”.
View Answer play_arrow
-
question_answer36)
Which of the following process does not occur at
the interface of phases?
(a) Crystallisation
(b) Heterogeneous catalysis
(c) Homogeneous
catalysis
(d) Corrosion
View Answer play_arrow
-
question_answer37)
At the equilibrium position in the process of adsorption..........
(a)  H > 0 (b)
H > 0 (b) H =
H =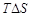 (c)
(c)  H >
H > 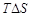 (d)
(d) H <
H < 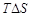
View Answer play_arrow
-
question_answer38)
Which of the following interface cannot be
obtained?
(a) Liquid-liquid
(b) Solid-liquid
(c) Liquid-gas
(d) Gas-gas
View Answer play_arrow
-
question_answer39)
The term 'sorption' stands for........ .
(a) absorption
(b) adsorption
(c) Both absorption and
adsorption
(d) desorption
View Answer play_arrow
-
question_answer40)
Extent of physisorption of a gas increases with..........
(a) increase in temperature
(b) decrease in temperature
(c) decrease in surface area of
adsorbent
(d) decrease in strength of van der
Waals' forces
View Answer play_arrow
-
question_answer41)
Extent of adsorption of adsorbate from solution
phase increases with??..
(a) increase in amount of adsorbate in
solution
(b) decrease in surface area of
adsorbent
(c) increase in temperature of
solution
(d) decrease in amount of adsorbate in
solution
View Answer play_arrow
-
question_answer42)
Which one of the following is
not applicable to the phenomenon of adsorption?
(a) 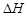 > 0 (b)
> 0 (b) 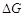 < 0 (c)
< 0 (c) 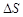 < 0 (d)
< 0 (d) 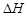 < 0
< 0
View Answer play_arrow
-
question_answer43)
Which of the following is not a favourable
condition for physical adsorption?
(a) High pressure
(b) Negative AH
(c) Higher critical temperature
of adsorbate
(d) High temperature
View Answer play_arrow
-
question_answer44)
Physical adsorption of a gaseous species may
change to chemical adsorption with...........
(a) decrease in temperature
(b) increase in temperature
(c) increase in surface area of
adsorbent
(d) decrease in surface area of
adsorbent
View Answer play_arrow
-
question_answer45)
In physisorption adsorbent does not show
specificity for any particular gas because.........
(a) involved van der Waals' forces are
universal
(b) gases involved behave like
ideal gases
(c) enthalpy of adsorption is
low
(d) it is a reversible process
View Answer play_arrow
-
question_answer46)
Which of the following is an example of
absorption?
(a) Water on silica gel
(b) Water on calcium chloride
(c) Hydrogen on finely divided
nickel
(d) Oxygen on metal surface
View Answer play_arrow
-
question_answer47)
On the basis of data given below predict which
of the following gases shows least adsorption on a definite amount of charcoal?
|
Gas
|
Co2
|
SO2
|
CH4
|
H2
|
|
Critical temp./k
|
304
|
630
|
190
|
33
|
(a) CO2 (b) S02
(c) CH4 (d) H2
View Answer play_arrow
-
question_answer48)
In which of the following reactions
heterogeneous catalysis is involved?
(i) 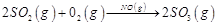 (ii)
(ii) 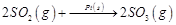 (iii)
(iii) 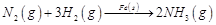 (iv)
(iv) 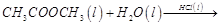
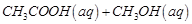 (a)'(ii), (iii) (b)
(ii), (iii) and (iv)
(c) (i), (ii) and (iii) (d)
(iv)
(a)'(ii), (iii) (b)
(ii), (iii) and (iv)
(c) (i), (ii) and (iii) (d)
(iv)
View Answer play_arrow
-
question_answer49)
At high concentration of soap in water, soap
behaves as..........
(a) molecular colloid
(b) associated colloid
(c) macromolecular
colloid
(d) lyophilic colloid
View Answer play_arrow
-
question_answer50)
Which of the following will show Tyndall effect?
(a) Aqueous solution of soap below
critical micelle concentration
(b) Aqueous solution of soap
above critical micelle concentration
(c) Aqueous solution of sodium
chloride
(d) Aqueous solution of sugar
View Answer play_arrow
-
question_answer51)
Method by which lyophobic sol can be protected.
(a) By addition of oppositely charged
sol
(b) By addition of an
electrolyte
(c) By addition of lyophilic sol
(d) By boiling
View Answer play_arrow
-
question_answer52)
Freshly prepared precipitate sometimes gets
converted to colloidal solution by..........
(a) coagulation (b)
electrolysis
(c) diffusion (d)
peptisation
View Answer play_arrow
-
question_answer53)
Which of the following electrolytes will have
maximum coagulating value for Ag/Ag+ sol?
(a) Na2S (b)
Na3PO4
(c) Na2SO4 (d)
NaCI
View Answer play_arrow
-
question_answer54)
A colloidal system having a solid substance as a
dispersed phase and a liquid as a dispersion medium is classified as..........
(a) solid sol (b)
gel
(c) emulsion (d)
sol
View Answer play_arrow
-
question_answer55)
The values of colligative properties of
colloidal solution are of small order in comparison to those shown by true
solutions of same concentration because of colloidal particles..........
(a) exhibit enormous surface area
(b) remain suspended in the
dispersion medium
(c) form lyophilic colloids
(d) are comparatively less in number
View Answer play_arrow
-
question_answer56)
Arrange the following diagrams in correct
sequence of steps involved in the mechanism of catalysis, in accordance with
modern adsorption theory.
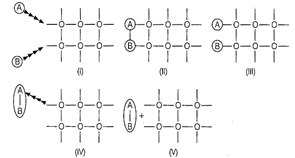 (a) l®ll®lll®IV®V
(b) l®|||®||®|V®V
(c) l®lll®ll®V®IV
(d) |®||®|||®V®IV
(a) l®ll®lll®IV®V
(b) l®|||®||®|V®V
(c) l®lll®ll®V®IV
(d) |®||®|||®V®IV
View Answer play_arrow
-
question_answer57)
Which of the following process is responsible
for the formation of delta at a place where rivers meet the sea?
(a) Emulsification (b)
Colloid formation
(c) Coagulation (d)
Peptisation
View Answer play_arrow
-
question_answer58)
Which of the following curves is in according
with Freundlich adsorption isotherm?
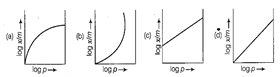
View Answer play_arrow
-
question_answer59)
Which of the following process is not
responsible for the presence of electric charge on the sol particles?
(a) Electron capture by sol particles
(b) Adsorption of ionic species
from solution
(c) Formation of Helmholtz
electrical double layer
(d) Absorption of ionic species from
solution
View Answer play_arrow
-
question_answer60)
Which of the following phenomenon is applicable
to the process shown in the figure?
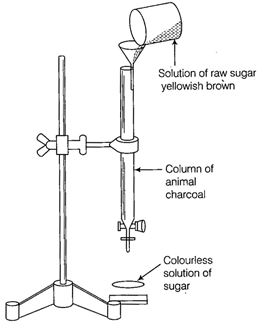 (a) Absorption (b)
Adsorption
(c) Coagulation (d)
Emulsification
(a) Absorption (b)
Adsorption
(c) Coagulation (d)
Emulsification
View Answer play_arrow
-
question_answer61)
Which of the following options are correct?
(a) Micelle formation by soap in aqueous
solution is possible at all temperatures
(b) Micelle formation by soap in
aqueous solution occurs above a particular concentration
(c) On dilution of soap solution
micelles may revert to individual ions
(d) Soap solution behaves as a normal
strong electrolyte at all concentrations
View Answer play_arrow
-
question_answer62)
Which of the following statements are correct
about solid catalyst?
(a) Same reactants may give different
products by using different catalysts
(b) Catalyst does not change  of reaction
(c) Catalyst is required in
large quantities to catalyse reactions
(d) Catalytic activity of a solid
catalyst does not depend upon the strength of chemisorption
of reaction
(c) Catalyst is required in
large quantities to catalyse reactions
(d) Catalytic activity of a solid
catalyst does not depend upon the strength of chemisorption
View Answer play_arrow
-
question_answer63)
Freundlich adsorption isotherm is given by the
expression 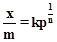 Which of
the following conclusions can be drawn from this expression?
(a) When
Which of
the following conclusions can be drawn from this expression?
(a) When 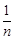 = 0, the adsorption is
independent of pressure
(b) When
= 0, the adsorption is
independent of pressure
(b) When 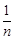 = 0, the adsorption is
directly proportional to pressure
(c) When
= 0, the adsorption is
directly proportional to pressure
(c) When 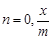 vs p graph is a line
parallel to x - axis
(d) When n = 0, plot of
vs p graph is a line
parallel to x - axis
(d) When n = 0, plot of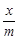 vs p is a curve
vs p is a curve
View Answer play_arrow
-
question_answer64)
H2 gas is adsorbed on activated
charcoal to a very little extent in comparison to easily liquefiable gases due to..........
(a) very strong van der Waals'
interaction
(b) very weak van der Waals'
forces
(c) very low critical
temperature
(d) very high critical temperature
View Answer play_arrow
-
question_answer65)
Which of the following statements are correct?
(a) Mixing two oppositely charged sols
neutralises their charges and stabilises the colloid
(b) Presence of equal and
similar charges on colloidal particles provides stability to the colloids
(c) Any amount of dispersed
liquid can be added to emulsion without destabilising it
(d) Brownian movement stabilises sols
View Answer play_arrow
-
question_answer66)
An emulsion cannot be broken by ......... and.........
.
(a) heating
(b) adding more amount of
dispersion medium
(c) freezing
(d) adding emulsifying agent
View Answer play_arrow
-
question_answer67)
Which of the following substances will
precipitate the negatively charged emulsions?
(a) KCI (b)
Glucose
(c) Urea (d)
NaCI
View Answer play_arrow
-
question_answer68)
Which of the following colloids cannot be
coagulated easily?
(a) Lyophobic colloids
(b) Irreversible colloids
(c) Reversible
colloids
(d) Lyophilic colloids
View Answer play_arrow
-
question_answer69)
What happens when a Lyophilic sol is added to a
Lyophobic sol?
(a) Lyophobic sol is protected
(b) Lyophilic sol is protected
(c) Film of lyophilic sol is
formed over lyophobic sol
(d) Film of lyophobic sol is formed over
lyophilic sol
View Answer play_arrow
-
question_answer70)
Which phenomenon occurs when an electric field
is applied to a colloidal solution and electrophoresis is prevented?
(a) Reverse osmosis takes place
(b) Electroosmosis takes place
(c) Dispersion medium begins to
move
(d) Dispersion medium becomes stationary
View Answer play_arrow
-
question_answer71)
In a reaction, catalyst changes..........
(a) physically (b)
qualitatively
(c) chemically (d)
quantitatively
View Answer play_arrow
-
question_answer72)
Which of the following phenomenon occurs when a
chalk stick is dipped in ink?
(a) Adsorption of coloured substance
(b) Adsorption of solvent
(c) Absorption and adsorption
both of solvent
(d) Absorption of solvent
View Answer play_arrow
-
question_answer73)
Why is it important to have clean surface in
surface studies?
View Answer play_arrow
-
question_answer74)
Why is chemisorption referred to as activated
adsorption?
View Answer play_arrow
-
question_answer75)
What type of solutions are formed on dissolving
different concentrations of soap in water?
View Answer play_arrow
-
question_answer76)
What happens when gelatin is mixed with gold
sol?
View Answer play_arrow
-
question_answer77)
How does it become possible to cause artificial
rain by spraying silver iodide on the clouds?
View Answer play_arrow
-
question_answer78)
Gelatin which is a peptide is added in
ice-creams. What can be its role?
View Answer play_arrow
-
question_answer79)
What is collodion?
View Answer play_arrow
-
question_answer80)
Why do we add alum to purify water?
View Answer play_arrow
-
question_answer81)
What happens when electric field is applied to
colloidal solution?
View Answer play_arrow
-
question_answer82)
What causes Brownian motion in colloidal
dispersion?
View Answer play_arrow
-
question_answer83)
A colloid .is formed by adding 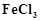 in excess of hot
water. What will happen if excess sodium chloride is added to this colloid?
in excess of hot
water. What will happen if excess sodium chloride is added to this colloid?
View Answer play_arrow
-
question_answer84)
How do emulsifying agents stabilise the
emulsion?
View Answer play_arrow
-
question_answer85)
Why are some medicines more effective in the
colloidal form?
View Answer play_arrow
-
question_answer86)
Why does leather get hardened after tanning?
View Answer play_arrow
-
question_answer87)
How does the precipitation of colloidal smoke
take place in Cottrell precipitator?
View Answer play_arrow
-
question_answer88)
How will you distinguish between dispersed phase
and dispersion medium in an emulsion?
View Answer play_arrow
-
question_answer89)
On the basis of Hardy-schuize rule explain why
the coagulating power of phosphate is higher than chloride?
View Answer play_arrow
-
question_answer90)
Why does bleeding stop by rubbing moist alum?
View Answer play_arrow
-
question_answer91)
Why is 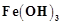 colloid positively
charged, when prepared by adding
colloid positively
charged, when prepared by adding 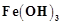 to
hot water?
to
hot water?
View Answer play_arrow
-
question_answer92)
Why do physisorption and chemisorption behave
differently with rise in temperature?
View Answer play_arrow
-
question_answer93)
What happens when dialysis is prolonged?
View Answer play_arrow
-
question_answer94)
Why does the white precipitate of silver halide
become coloured in the presence of dye eosin?
View Answer play_arrow
-
question_answer95)
What is the role of activated charcoal in gas
mask used in coal mines?
View Answer play_arrow
-
question_answer96)
How does a delta form at the meeting place of
sea and river water?
View Answer play_arrow
-
question_answer97)
Give an example where physisorption changes to
chemisorption with rise in temperature. Explain the reason for change.
View Answer play_arrow
-
question_answer98)
Why is desorption important for a substance to
act as good catalyst?
View Answer play_arrow
-
question_answer99)
What is the role of diffusion in heterogeneous
catalyst?
View Answer play_arrow
-
question_answer100)
How does a solid catalyst enhance the rate of
combination of gaseous molecules?
View Answer play_arrow
-
question_answer101)
Do the vital functions of the body such as
digestion get affected during fever? Explain your answer.
View Answer play_arrow
-
question_answer102)
Method of formation of solution is given in Column
I. Match it with the type of solution given in Column II.
|
|
Column 1
|
Column 11
|
|
A.
|
Sulphur vapours passed through cold water.
|
1. Normal electrolyte solution
|
|
B.
|
Soap mixed with water above critical micelle
concentration.
|
2. Molecular colloids
|
|
C.
|
White of egg whipped with water.
|
3. Associated colloid
|
|
P
|
With water below critical
|
4. Macromolecular colloids
|
View Answer play_arrow
-
question_answer103)
Match the statement given in Column I with the
phenomenon given in Column II.
|
|
Column 1
|
Column 11
|
|
A.
B.
C.
D.
|
Dispersion medium moves in an electric field.
Solvent molecules pass through semipermeable membrane
towards solvent side.
Movement of charged colloidal particles under the
influence of applied electric potential towards oppositely charged
electrodes.
Solvent molecules pass through semipermeable
membranes towards solution side.
|
1. Osmosis
2. Electrophoresis
3. Electroosmosis
4. Reverse-osmosis
|
View Answer play_arrow
-
question_answer104)
Match the items given in Column I and
Column II.
|
|
Column 1
|
Column 11
|
|
A.
B.
C.
D.
|
Protective colloid Liquid-liquid colloid
Positively charged colloid Negatively charged colloid
|
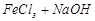 Lyophilic colloids Emulsion
Lyophilic colloids Emulsion
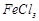 +
hot water +
hot water
|
View Answer play_arrow
-
question_answer105)
Match
the types of colloidal systems given in Column I with the name given in Column
II.
|
|
Column
1
|
Column
11
|
|
A.
B.
C.
D.
|
Solid
in liquid
Liquid
in solid
Liquid
in liquid
Gas
in liquid
|
1. Foam
2.
Sol
3.
Gel
4.
Emulsion
|
View Answer play_arrow
-
question_answer106)
Match the items of Column I and Column II.
|
|
Column 1
|
Column 11
|
|
A.
B.
C.
D.
|
Dialysis Peptisation Emulsification
Electrophoresis
|
Cleansing action of soap Coagulation Colloidal
sol formation Purification
|
View Answer play_arrow
-
question_answer107)
Match the items of Column I and Column Il
|
|
Column 1
|
Column 11
|
|
A.
B.
C.
D.
|
Butter
Pumice stone
Milk
Paints
|
1. Dispersion of liquid in liquid
2.Dispersion of solid in liquid
3.Dispersion of gas in solid
4.Dispersion of liquid in solid
|
View Answer play_arrow
-
question_answer108)
Assertion (A) An ordinary filter paper
impregnated with collodion solution stops the flow of colloidal particles.
Reason (R) Pore size of the
filter paper becomes more than the size of colloidal particle.
View Answer play_arrow
-
question_answer109)
Assertion. (A) Colloidal solution show
colligative properties.
Reason (R) Colloidal particles are
large in size.
View Answer play_arrow
-
question_answer110)
Assertion (A) Colloidal solutions do not
show Brownian motion.
Reason (R) Brownian motion is
responsible for stability of sols.
View Answer play_arrow
-
question_answer111)
Assertion (A) Coagulation power of 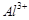 is more than
is more than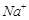 .
Reason (R) Greater the valency of
the flocculating ion added, greater is its power to cause precipitation
(Hardy-SchuIze rule) .
.
Reason (R) Greater the valency of
the flocculating ion added, greater is its power to cause precipitation
(Hardy-SchuIze rule) .
View Answer play_arrow
-
question_answer112)
Assertion (A) Detergents with low CMC are
more economical to use.
Reason (R) Cleansing action of
detergents involves the formation of micelles. These are formed when the
concentration of detergents
becomes equal to CMC.
View Answer play_arrow
-
question_answer113)
What is the role of adsorption in heterogeneous
catalysis?
View Answer play_arrow
-
question_answer114)
What are the applications of adsorption in
chemical analysis?
View Answer play_arrow
-
question_answer115)
What is the role of adsorption in froth
floatation process used especially for concentration of sulphide ores?
View Answer play_arrow
-
question_answer116)
What do you understand by shape selective
catalysis? Why are zeolites good shape selective catalysts?
View Answer play_arrow
![]() colloid positively
charged, when prepared by adding
colloid positively
charged, when prepared by adding ![]() to
hot water?
to
hot water?
