Answer:
1. J.J. Thomson's model of atom
(i) An atom consists of a sphere of positive
charge in which electrons are embedded just like seeds in the water melon
(ii) Total positive charge on the sphere is equal
to the total negative charge present on the electrons so that atom as a whole
is electrically neutral,
(iii) It could not explain the results of
Rutherford's scatter experiments.
2. Rutherford's model of atom
(i) An atom consists of a small positively
charged nucleus in the centre and the electrons are revolving around it.
(ii) There is very large empty space between the
nucleus and the electrons.
(iii) All the mass of the atom is mainly
concentrated in the nucleus.
(iv) It could not explain the stability of the
atom because the revolving electron will be accelerated towards nucleus.
Hence it will lose energy. Its orbit will become smaller and smaller and
ultimately the electron will fall into the nucleus.
3. Bohr's model of atom
The main points of this model of atom (called postulates
of Bohr's model of atom) are as follows:
(i) An atom consists of a small heavy positively
charged nucleus in the centre and the electrons revolve around it in circular
paths called orbits.
(ii) In a particular atom, the orbits in which
the electrons revolve are the discrete orbits having fixed radii and energy.
These discrete orbits are, therefore, also called energy levels or shells.
The term shell is used to indicate that atom is three-dimensional i.e., it is
not like a plate but is like a ball. As energy of the orbits is fixed these
are also called stationary states. These are numbered as 1, 1, 3, 4, etc. as
we move outwards from the nucleus or they are represented by the letters K,
L, M, N etc. as shown in Fig. The energy of these shells increases as we move
outward from the nucleus. Thus, representing the energies of 1st,
2nd, 3rd, 4th, shell etc. by
![]() etc.,
we have :
etc.,
we have : ![]() However, the gap between the successive energy
shells decreases as we move outwards from the nucleus.
However, the gap between the successive energy
shells decreases as we move outwards from the nucleus.
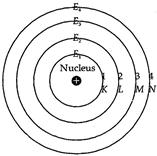 Circular orbits or energy
levels or shells around the nucleus
(iii) As long as an electron is revolving in a
particular orbit, it can neither lose energy nor gain energy. Thus, the atom
is stable and does not collapse. This state of the atom with lowest energy is
called ground state of the atom.
(iv) Energy is lost or gained by an electron only
when it jumps from one orbit to the other. The energy falls on an electron
and it absorbs this energy, it will jump to some outer shell. The atom is
then said to be in the excited state. In the excited state, the atom is not
stable. It loses or emits energy and jumps back to some inner energy level.
In other words, an electron jumps from inner shell to outer shell by
absorbing energy whereas energy is emitted when an electron jumps from an
outer shell to an inner.
Circular orbits or energy
levels or shells around the nucleus
(iii) As long as an electron is revolving in a
particular orbit, it can neither lose energy nor gain energy. Thus, the atom
is stable and does not collapse. This state of the atom with lowest energy is
called ground state of the atom.
(iv) Energy is lost or gained by an electron only
when it jumps from one orbit to the other. The energy falls on an electron
and it absorbs this energy, it will jump to some outer shell. The atom is
then said to be in the excited state. In the excited state, the atom is not
stable. It loses or emits energy and jumps back to some inner energy level.
In other words, an electron jumps from inner shell to outer shell by
absorbing energy whereas energy is emitted when an electron jumps from an
outer shell to an inner.
You need to login to perform this action.
You will be redirected in
3 sec
