Other Pathways Of Glucose Oxidation
Category : 11th Class
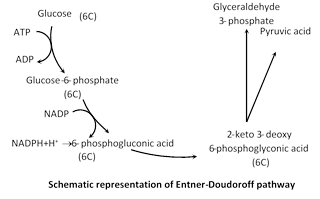
(1) Entner-Doudoroff pathway
Discovery : Entner-Doudoroff path discovered by Entner & Doudoroff. This pathway is also called glycolysis of bacteria.
Certain bacteria such as Pseudomonas sacchorophila, P. fluorescens, P. lindeneri and P. averoginosa lack phosphofructokinase enzyme. They can not degrade glucose by glycolytic process.
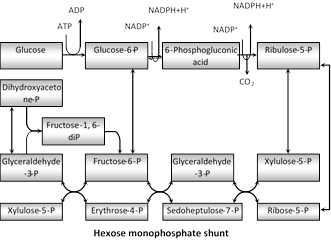
(2) Pentose phosphate pathway
(i) Discovery : It is also called as Hexose monophosphate (HMP) shunt or Warburg Dickens pathway or direct oxidation pathway. It provides as alternative pathway for breakdown of glucose which is independent of EMP pathway (glycolysis) and Krebs cycle. Its existence was suggested for the first time by Warburg et al. (1935) and Dickens (1938). Most of the reaction of this cycle were described by Horecker et al. (1951) and Racker (1954).
(ii) Occurrence : Pentose phosphate pathway that exists in many organisms. This pathway takes place in the cytoplasm and requires oxygen for its entire operation.
(iii) Description : There are two types of evidences is support of the existence of such an alternative pathway-works on the inhibiting action of malonic acid on the Krebs cycle and studies with the radioactive \[({{C}^{14}}).\]
Twelve molecules of \[NAD{{H}_{2}}\] formed in the reaction can be oxidised back to 12 NADP with the help of the cytochrome system and oxygen of the air.
\[12\text{ NADP}{{\text{H}}_{\text{2}}}+6{{O}_{2}}\underset{\text{System}}{\mathop{\xrightarrow{\text{Cytochrome}}}}\,\text{12}{{\text{H}}_{\text{2}}}\text{O}+\text{12NADP}\]
In this electron transfer process, 36 molecules of ATP are synthesized.
(iv) Significance of PPP
(a) It is the only pathway of carbohydrate oxidation that gives \[NADP{{H}_{2}},\]Which is needed for synthetic action like synthesis of fatty acid (in adipose tissues) and amino acids (in liver).
(b) It synthesizes 3C-glyceraldehyde-3-P, 3C-dihydroxy acetone phosphate, 4C-erythrose-4-P, 5C-ribulose phosphate, 5C-xylulose phosphate, 5C-ribose phosphate, 6 C-Fructose 6-phosphate, 7C-sedoheptulose-7-phosphate.
(c) It is the major pathway by which necessary ribose and deoxyribose are supplied in the biosynthesis of nucleotides and nucleic acid.
(d) Erythrose 4 phosphate for the synthesis of lignin, oxine, anthocyanine and aromatic amino acid (phenylalanine, tyrosine, and tryptophan).
(e) Young growing tissues appears to use to the Krebs cycle as the predominant pathway for glucose oxidation, while aerial parts of the plants and other tissues seem to utilise the PPP as well as the Krebs cycle.
(f) It gives \[6C{{O}_{2}},\]required for photosynthesis.
(g) Ribulose five phosphate is used in photosynthesis to produce RuBP which act as primary \[C{{O}_{2}}\]acceptor in \[{{C}_{3}}\]cycle.
(3) Cyanide resistant pathway : Cyanide-resistant respiration seems to be widespread in higher plant tissues. Cyanide prevents flow of electron from Cyt \[{{a}_{3}}\] to oxygen, so called ETC inhibitor. In these plant tissues resistance is due to, a branch point in the ETS preceeding the highly cyanide-sensitive cytochromes. The tissues lacking this branch point, or alternate pathway and blockage of cytochromes by cyanide, inhibits the electron flow.
Significance
(i) The role of alternative pathway is that it may provide a means for the continued oxidation of NADH and operation of the tricarboxylic acid cycle, even through ATP may not be sufficiently drained off.
(ii) It is significant in respiratory climateric of ripening fruits and leads to the production of hydrogen peroxide and super oxide, which in turn enhances the oxidation and breakdown of membranes.
(iii) Necessary activities in the ripening process because peroxides are necessary for ethylene biosynthesis.
Anaerobic respiration
Anaerobic respiration first studied by Kostychev (1902), Anaerobic respiration is an enzyme-controlled, partial break down of organic compounds (food) without using oxygen and releasing only a fraction of the energy. It is also called intra-molecular respiration (Pfluger, 1875). Anaerobic respiration occurs in the roots of some water-logged plants, certain parasitic worms (Ascaris and Taenia), animal muscle and some microorganisms (bacteria, moulds). In microorganisms anaerobic respiration is often called fermentation.
Higher organism like plants can not perform anaerobic respiration for long. It is toxic because accumulation of end products, insufficient amount of available energy and causes stoppage of many active process.
Process of anaerobic respiration : In this process pyruvate which is formed by glycolysis is metabolised into ethyl alcohol or lactic acid and \[C{{O}_{2}}\] in the absence of oxygen. Glycolysis is occurs in cytoplasm so the site of anaerobic respiration is cytoplasm.
\[{{C}_{6}}{{H}_{12}}{{O}_{6}}\to 2{{C}_{2}}{{H}_{5}}OH+2C{{O}_{2}}+52\text{ }Kcal/218.4kJ\]

(1) Formation of ethyl alcohol : When oxygen is not available, yeast and some other microbes convert pyruvic acid into ethyl alcohol.
(2) Production of lactic acid : In this process hydrogen atoms removed from the glucose molecule during glycolysis are added to pyruvic acid molecule and thus lactic acid is formed.
Lactic acid is produced in the muscle cells of human beings and other animals.
Pasteur effect : The process may be defined as "the inhibition of sugar breakdown due to the presence of oxygen under aerobic condition" and the reaction is called Pasteur reaction. Dixon (1937) stated that the Pasteur effect is the action of oxygen is checking the high rate of loss of carbohydrate and in suppressing or diminishing the accumulation of products of fermentation."
Fermentation : Fermentation is a kind of anaerobic respiration carried out by microorganisms fungi and bacteria. In microorganism the term anaerobic respiration is replaced by fermentation (Cruickshank, 1897) ; which is known after the name of its major product, e.g., alcohol fermentation, lactic acid fermentation.
(1) Butyric acid fermentation : It occurs in bacteria Clostridium butyricum.
\[\underset{\text{(hexose)}}{\mathop{{{C}_{6}}{{H}_{12}}{{O}_{6}}}}\,\to \underset{\text{(butyric}\,\text{acid)}}{\mathop{{{C}_{4}}{{H}_{8}}{{O}_{2}}}}\,+2{{H}_{2}}+2C{{O}_{2}}\]
\[\underset{\text{(lactic}\,\text{acid)}}{\mathop{2{{C}_{3}}{{H}_{6}}{{O}_{3}}}}\,\to \underset{\text{(butyric}\,\text{acid)}}{\mathop{{{C}_{4}}{{H}_{8}}{{O}_{2}}}}\,+2{{H}_{2}}+2C{{O}_{2}}\]
(2) Lactic acid fermentation : It occurs in lactic acid bacteria and muscles.
\[\underset{\text{(lactose)}}{\mathop{{{C}_{12}}{{H}_{22}}{{O}_{11}}}}\,+{{H}_{2}}O\to \underset{\text{(glucose)}}{\mathop{{{C}_{6}}{{H}_{12}}{{O}_{6}}}}\,+\underset{\text{(galactose)}}{\mathop{{{C}_{6}}{{H}_{12}}{{O}_{6}}}}\,\]
\[\underset{{}}{\mathop{\underset{(hexose)}{\mathop{{{C}_{6}}{{H}_{12}}{{O}_{6}}}}\,\to \underset{(lactic\,acid)}{\mathop{2{{C}_{3}}{{H}_{6}}{{O}_{3}}}}\,}}\,\]
(3) Acetic acid fermentation : It occurs in acetic acid bacteria.
\[\underset{(\text{ethyl}\,\text{alcohol})}{\mathop{{{C}_{2}}{{H}_{5}}OH}}\,+{{O}_{2}}\to \underset{(\text{acetic}\,\text{acid})}{\mathop{C{{H}_{3}}COOH}}\,+{{H}_{2}}O+\text{energy}\]
You need to login to perform this action.
You will be redirected in
3 sec
