Work and Energy
Category : 7th Class
Learning Objectives
Introduction
In everyday language, we often term 'work' and 'energy'. A child sitting and doing his/her homework, a laborer breaking a wall, a man making a presentation on PC, a farmer plugging his fields, all are said to be doing some work. A person who can do more work is said to have more energy. In this chapter we study the actual meaning of work and energy and how they are related to each other.
WORK
When a force is applied on a body, and it displaces the body through some distance, then work is said to be done. But if there is no displacement even when a force is applied, then no work is said to be done.
Thus work depends on the applied force and the displacement made by the body due to that force.
\[Work(\text{W})=Force(\text{F})\times displacement(\text{d})\]
i.e., work is the product of force and displacement.
Do you know?
If nothing is actually moving, no work is done no matter how great the force involved.
Units of Work
The S.I. unit of work is joule (J)
From the equation W = F d
\[\text{1J=1N }\!\!\times\!\!\text{ 1m}\]
i.e. If 1N force displaces a body through 1m in the direction of force, then work done is one joule (1 J).
The C.G.S unit of work is erg.
Again, from the equation
\[\text{W=F}\times \text{d}\]
\[\text{1erg=1dyne }\!\!\times\!\!\text{ 1cm}\]
Work done is said to be one erg, when one dyne of force, displaces a body through 1 cm in the direction of applied force.
\[W=F\times d\]
\[1erg=1dyne\times 1cm\]
Do you know?
The rate at which work is done by a force is called the power delivered by that force
Relation between joule and erg
\[\text{1J=1N}\times \text{1m}\]
\[\text{=1}{{\text{0}}^{\text{5}}}\text{dyne}\times \text{1}{{\text{0}}^{\text{2}}}\text{cm}\]
\[\text{=1}{{\text{0}}^{\text{7}}}\text{dyne cm}\]
\[\therefore 1\,\text{joule=1}{{\text{0}}^{\text{7}}}\text{erg}\]
TYPES OF WORK
Work done can he positive, negative or zero as per the direction of displacement with respect to the force applied.
(1) Positive Work: if displacement occurs in the same direction as that of force applied, then work done is positive
For example:
(a) When a ball falls down due to gravity, the work done is positive .Because force of gravity pulls ball down and it moves also down wards. So the direction of displacement of ball and action of gravity are in the same direction.
(b) On pushing a car, if it moves forward then work done is positive.
(2) Negative Work: If the direction of displacement is opposite to that of the force, then work done is negative.
For example:
(a) A ball thrown upwards makes displacement opposite to the direction of force of gravity pulling it downwards. So the work done by gravity for an object thrown upwards is negative.
(b) The work done by frictional force is negative because frictional force always acts opposite to the direction of motion.
(3) Zero Work: If displacement is zero, then the work done is zero. Also, if applied force and the displacement are perpendicular to each other, the work done is zero.
For example:
(a) Pushing against a wall does zero work because displacement is zero.
(b) The work done by the centripetal force along the radius of the circular path is zero.
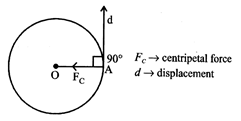
ENERGY
Whenever we do work we need energy. So in simplest term we define energy as ability to do work. Thus work and energy are interrelated.
Since energy is equivalent to work therefore the unit of energy is the same as that of work i.e., joule in SI system and erg in CGS system.
Do you know?
Energy is a promise of work to be done in future. It is the stored ability to do work.
Mechanical Energy
It is the energy possessed by object due to its position potential energy (P.E.) or motion kinetic energy (K.E.) i.e.
\[\text{Mechanical energy, ME}\text{.=P}\text{.E}\text{.+K}\text{.E}\text{.}\]
(a) Potential Energy: The energy possessed by a body by virtue of its position (configuration) is called potential energy. For e.g. A stone at a height, a stretched string of a bow, a compressed spring, etc.
Mathematically
\[\text{P}.\text{E}.=\text{mass}\times \text{acc}.\text{due to gravity}\times \text{height}\left( =\text{mgh} \right).\]
(b) Kinetic Energy: The kinetic energy is the energy possessed by a body due to its state of motion.
Thus only a moving body can have kinetic energy. For e.g. A moving car, a bullet fired from a gun, etc. possess kinetic energy.
\[Mathematically\,K.E.=\frac{1}{2}mass\times {{(velocity)}^{2}}\] \[=\frac{1}{2}m{{v}^{2}}\]
A bird or a plane at a height possesses both kinetic (due to its motion) as well as potential (due to its height from the ground) energy.
Do you know?
Kinetic energy can be passed from one object to another in the form of a collision. Kinetic energy of a body is always positive.
LAW OF CONSERVATION OF ENERGY
It states that energy can neither be created nor be destroyed. It can only be transformed from one form to another form. Most of the phenomena in our daily life are based on the law of conservation of energy. Since we need energy in its different forms for different purposes, various devices are there for their inter conversions.
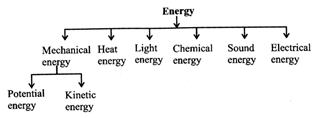
Different Forms of Energy and Their Interconversion
Energy exists in various forms.
Do you know?
One minute of Sunshine supplies enough energy to meet the earth’s needs for a whole year.
You might have observed in daily life that all the above forms of energy are used for different purposes.
Mechanical Energy includes Kinetic and Potential energy. Energy due to muscular force is mechanical energy.
Heat Energy: Heat is a form of energy that flows from a body at higher temperature to a body at lower temperature.
Light Energy: Light is a form of energy that creates the sensation of vision.
Chemical Energy: Energy released during a chemical reaction is called chemical energy. Foods, fuels and batteries possess chemical energy.
Sound Energy: Sound energy is a form of energy caused by vibrations. It causes sensation of hearing. Sound energy travels in the form of sound waves. A vibrating string of guitar produces sound energy.
Electrical Energy: It is more popularly known as electricity. It refers to the flow of charges through a conductor.
Interconversion of Energy:
Heat energy \[\xrightarrow{\text{into}}\]Mechanical energy— Heat engine
Electrical energy \[\xrightarrow{\text{into}}\]Mechanical energy—washing machine
Electrical energy \[\xrightarrow{\text{into}}\]Heat energy— Electric heater
Chemical energy \[\xrightarrow{\text{into}}\]Electrical energy—Battery
Electrical energy \[\xrightarrow{\text{into}}\]Light energy — Electric bulb
Electrical energy \[\xrightarrow{\text{into}}\]Sound energy—Electric bell
Chemical energy \[\xrightarrow{\text{into}}\]Heat energy—burning of fuel
The flow chart below explains conversions of energy from one form to the other.
\[\text{Potenial energy of water at height}\to \text{Kinetic energy of falling water}\]
\[\text{Mechanical energy of turbin }\to \text{Electrical energy}\]\[\to \text{Light energy of bulb}\]
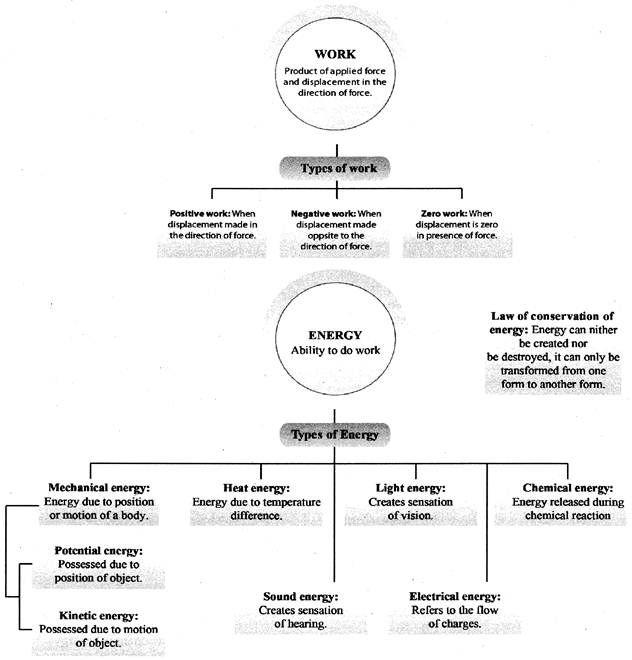
CONCEPT MAP
You need to login to perform this action.
You will be redirected in
3 sec
