General Organic Chemistry
Category : Banking
General Organic Chemistry


Prefix - word root - Suffix
Word root: A word root indicates the nature of basic carbon skeleton.
|
Functional group |
Prefix/Suffix |
Functional group |
Example |
IUPAC Name |
|
1. Halogen |
Chloro, bromo, lodo |
-Cl,-Br,-I |
|
-Bromopropane |
|
2. Alcohol |
-ol |
-OH |
|
-Ethanol |
|
3. Aldehyde |
-al |
-CHO |
\[C{{H}_{3}}C{{H}_{2}}C{{H}_{2}}CHO\] |
-Butanal |
|
4. Ketone |
-one |
-CO |
\[C{{H}_{3}}COC{{H}_{3}}\] |
-Propanone |
|
5. Carboxylic arid |
-oic acid |
-COOH |
\[C{{H}_{3}}C{{H}_{2}}COO{{H}_{{}}}\] |
-Propanoicacid |
|
6. Amine |
Ammo |
\[-N{{H}_{2}}\] |
\[C{{H}_{3}}C{{H}_{2}}N{{H}_{2}}\] |
-Amino ethane |
|
7. Ester |
oate- |
-COOR |
\[C{{H}_{3}}COOC{{H}_{3}}\] |
-Methyl ethanoate |
|
8. Double bond |
ene |
|
\[C{{H}_{3}}-CH=C{{H}_{2}}\] |
-Propene |
|
9. Triple bond |
yne |
|
\[C{{H}_{3}}-C{{H}_{2}}-C\equiv CH\] |
-Butyne |
(i) Combustion: Carbon compound undergo combustion reaction to produce \[C{{O}_{2}}\] and \[{{H}_{2}}O\] with the evolution of heat and light \[C{{H}_{4}}+{{O}_{2}}\to C{{O}_{2}}+{{H}_{2}}O+\]heat and light
(ii) Oxidation: \[\underset{ethanol}{\mathop{C{{H}_{3}}C{{H}_{2}}OH}}\,\xrightarrow{alk.\,\,KMn{{O}_{4}}/\Delta }\underset{ethanoic\,\,acid}{\mathop{C{{H}_{3}}COOH}}\,\]
The substance which are used for oxidation are known as oxidising agent, e.g alkaline\[KMn{{O}_{4}}\], acidified\[{{K}_{2}}C{{r}_{2}}{{O}_{7}}\].
(iii) Addition reaction:
Unsaturated hydrocarbons (alkenes and alkynes undergo addition reaction in presence of catalysts e.g.
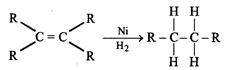
(iv) Substitution reaction: Saturated hydrocarbons give substitution reaction e.g. methane in presence of sunlight undergo chlorination.
You need to login to perform this action.
You will be redirected in
3 sec
