Factor Affecting Respiration
Category : NEET
Factors Affecting Rate of Respiration
Many external and internal factors affecting the rate of respiration are as follows:
(1) External factors
(i) Temperature: Temperature is the most important factor for respiration. Most of the plants respire normally between\[{{0}^{o}}Cto\text{ }{{30}^{o}}C\]. With every \[{{10}^{0}}C\] rise of temperature from \[{{0}^{o}}Cto\text{ }{{30}^{o}}C\] the rate of respiration increases 2 to 2.5 times (i.e., temperature coefficient \[\left( {{Q}_{10}}^{o} \right)\] is = 2 to 2.5), following Vant Hoff’s Law. Maximum rate of respiration takes place at 30oC, there is an initial rise, soon followed by a decline. Higher the temperature above this limit, more is the initial rise but more is the decline and earlier is the decline in the rate of respiration. Probably this is due to denaturation of enzymes at high temperature.
Below 0oC the rate of respiration is greatly reduced although in some plants respiration takes place even at\[-{{20}^{o}}C\]. Dormant seeds kept at \[5{{0}^{o}}C\]B survive.
(ii) Supply of oxidisable food : Increase in soluble food content readily available for utilization as respiratory substrate, generally leads to an increase in the rate of respiration upto a certain point when some other factor becomes limiting.
(iii) Oxygen concentration of the atmosphere : Respiration is aerobic or anaerobic depending upon the presence or absence of oxygen. Air has 20.8% oxygen which is more than enough keeping in view the requirements of plants. Due to this if the amount of oxygen in the environment of plants is increased or reduced upto quite low values the rate of respiration is not effected. On decreasing the amount of oxygen to 1.9% in the environment aerobic respiration become negligible (extinction point of aerobic respiration) but anaerobic respiration takes place.
Oxygen poisoning: The significant fall in respiration rate was observed in many tissues in pure O2, even at N.T.P. This inhibiting effect was also observed in green peas when they are exposed to pure oxygen exerting a pressure of 5 atm- the respiration rate fall rapidly. The oxygen poisoning effect was reversible, if the exposure to high oxygen pressure was not too prolonged.
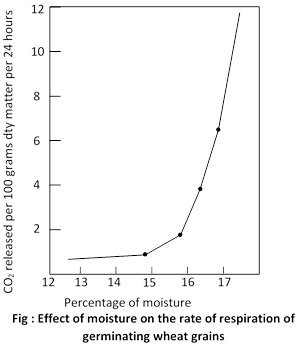
(iv) Water: With increase in the amount of water the rate of respiration increases. In dry seeds, which have 8-12% of water the rate of respiration is very low but as the seeds imbibe water the respiration increases. The life of seeds decreases with increase of water. This increase is slow at first but very rapid later. This is very clearly seen in the tissues of many xerophytes. As the water contents of such plants is increased, often there is no great immediate effect upon the rate of respiration. Minor variation in water content of well-hydrated plant cell do not appear to have very great influence upon the rate of respiration.
Figure shows that in wheat grains rate of respiration increases with increase of water to 16-17%. The rate of respiration of seeds increase with increase of water because water causes hydrolysis, and activity of respiratory enzymes is increased. Also oxygen enters the seed through the medium of water.
(v) Light : Respiration takes place in night also which shows that light is not essential for respiration. But light effects the rate of respiration indirectly by increasing the rate of photosynthesis due to which concentration of respiratory substrates is increased. More the respiratory substrate more is the rate of respiration.
In case of blue green algae (Anabaena) respiration rate was found to depend upon light and the effect was also influenced by O2-concentration.
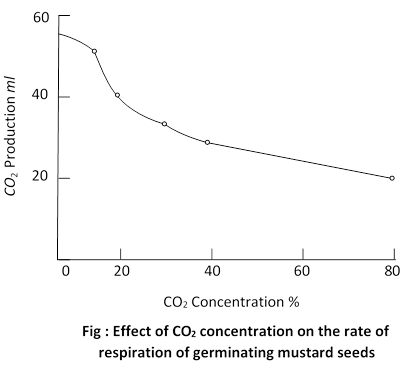
(vi) Carbon dioxide \[\left( C{{O}_{\mathbf{2}}} \right)\] : If the amount of \[\left( C{{O}_{\mathbf{2}}} \right)\] in the air is more than the usual rate of respiration is decreased. Germination of seeds is reduced and rate of growth falls down. Heath, (1950) has shown that the stomata are closed at higher cone. of \[\left( C{{O}_{\mathbf{2}}} \right)\], due to which oxygen does not penetrate the leaf and rate of respiration is lowered.
This fact is made use of in storage of fruits. Air containing 10% CO2 (in atmosphere it is only 0.03%) retards respiratory break down and therefore reduces sugar consumption and thus prolongs the life of the fruit.
(vii) Inorganic salts : The chlorides of alkali cations of Na and K, as also the divalent cations of Li, and Ca and Mg, generally increase the rate of respiration as measured by the amount of \[\left( C{{O}_{\mathbf{2}}} \right)\] evolved. Monovalent chlorides of K and Na increases the rate of respiration, while divalent chlorides of Li, Ca and Mg causes less increase in respiration.
(viii) Injury and effects of mechanical stimulation : Wounding or injury almost invariably results in an increase in the rate of respiration . Whenever a plant tissue is wounded the sugar content of the wounded portion is suddenly increased. This increase in the sugar content is responsible for the observed temporary increase in the respiration rate.
A purely mechanical stimulation of respiration has been demonstrated in leaves of a number of species by Audus (1939,40,46). A gentle rubbing, touching, handling and bending of the leaf blade was sufficient to induce a marked rise in the respiration rate (20 to 183%) which persisted for several days. If the treatment was repeated at intervals, the stimulus gradually lost its effect in increasing the rate of respiration.
(ix) Effect of various chemical substances : Certain enzymatic inhibitors like cyanides, azides, carbon monoxide, iodoacetate, malonate etc. reduce the rate of respiration even if they are present in very low concentration.
However, various chemical substances such as chloroform, ether, acetone, morphine, etc., brings about an increase in respiratory activity. Ripening fruits produce ethylene and this is accompanied by an increase in respiration rate. Other volatile products responsible for the flavour (aroma) e.g., methyl, ethyl, amyl, esters of formic, acetic, caproic and caprylic acid also associated with increased respiration rate, reach a maximum during ripening of fruits.
(x) Pollutants : High concentration of gaseous air pollutants like \[S{{O}_{2}},N{{O}_{X}}and{{O}_{3}}\]inhibit respiration by damaging cell membrane. These gaseous pollutant causes increase in pH which in turn affects the electron transport system thus inhibiting respiration.
Heavy metal pollutant like lead (Pb) and cadmium(Cd) inhibit respiration by inactivating respiratory enzymes.
(2) Internal Factors
(i) Protoplasm : The rate of respiration depends on quality and quantity of protoplasm. The meristematic cells (dividing cells of root and shoot apex) have more protoplasm than mature cells. Hence, the meristematic cells have higher rate of respiration than the mature cells. Respiration rate high at growing regions like floral and vegetative buds, germinating seedlings, young leaves, stem and root apices.
(ii) Respiratory substrate : With the increase of in the amount of respiratory substrate, the rate of respiration increases.
Experiments of respiration
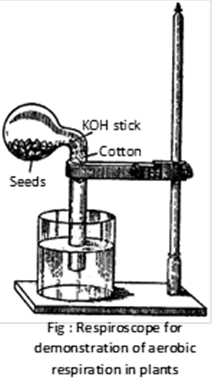
(1) Demonstration of aerobic respiration in plant tissues : Experimentally, aerobic respiration is proved by respiroscope. As a result of this experiment water column arises (also mercury) due to absorption of evolved \[C{{O}_{2}}\]in respiration by KOH.
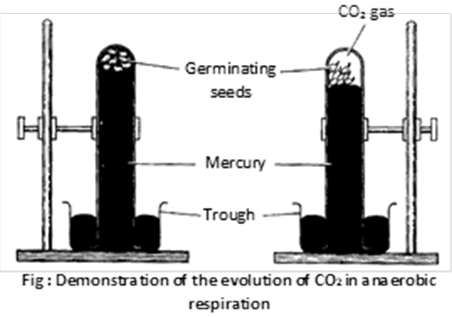
(2) Demonstration of anaerobic respiration : In this experiment anaerobic condition created by filling the test tube with mercury. After completion of experiment the level of mercury goes down, because \[C{{O}_{2}}\] evolved by the respiration push the mercury level down. When KOH is introduced mercury level will again rise up to top.
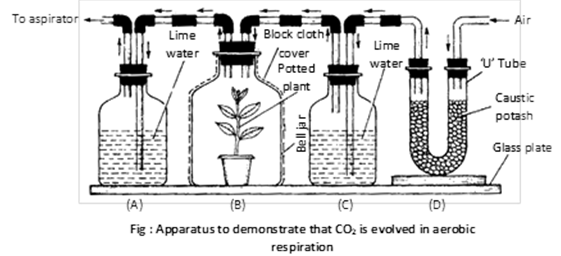
(3) To prove that \[C{{O}_{2}}\] is evolved in aerobic respiration : In following apparatus, when suction pump is started it pulls air through absorbed by KOH of U-tube. \[C{{O}_{2}}\] of air free of \[C{{O}_{2}}\]enters bottles C. If the calcium hydroxide water does not turns milky it is an indication that all \[C{{O}_{2}}\] has been absorbed by KOH. The results shows that lime water in bottle A turns milky proved that liberation of \[C{{O}_{2}}\] takes place in aerobic respiration
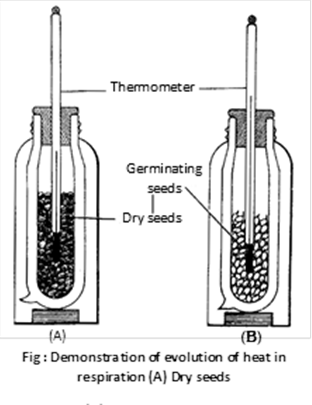
(4) Demonstration of liberation of heat energy during respiration : In this experiment bottle A filled with boiled seed and bottle B filled with germinating seed. After 24 hours temperature of both thermometer noted. Observation shows rise in temperature in bottle B because these seeds are respiring.
(5) Measurement of rate of respiration by Ganong's respirometer : The apparatus consists of three parts:
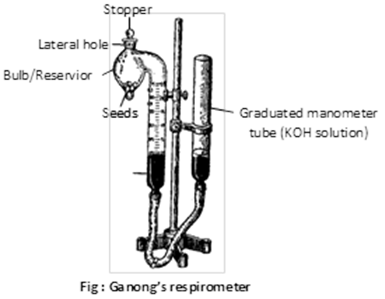
(i) A bulb for the respiring material, with stopper.
(ii) A graduated manometer tube.
(iii) A levelling or reservoir tube connected with manometer tube by a stout rubber tubing.
2 ml of respiring material are put into the bigger bulb of the respirometer and 10% KOH in placed in the manometer tube. Experiment start with the turning the glass stopper at the top. Respiration now takes place in a closed space and the absorption of \[C{{O}_{2}}\] liberated shown by rise in KOH solution in the graduated tube.
[For the measurement of R.Q. saturated solution of NaCl is first place in manometer tube. (Pure water not used, as it absorbed \[C{{O}_{2}}\])].
Different respiratory substrates (carbohydrate, fat seed) are taken. In the graduated tube will remain more or less and unaltered showing that the volumes of \[C{{O}_{2}}\] evolved and oxygen absorbed are the same and R.Q. =1. If solid KOH pellets than added to salt solution in the tube, the accumulated \[C{{O}_{2}}\] is absorbed and can be measured from the reading in the tube.
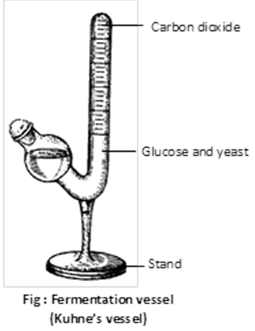
(6) Demonstration of fermentation : For this experiment glucose, baker's yeast and water are taken in Kuhne's tube. As a result the level of solution falls in the upright arm and the solution gives alcoholic smell, proves that alcoholic fermentation of glucose takes place.
Important Tips
You need to login to perform this action.
You will be redirected in
3 sec
