Electromagnetic Radiations
Category : JEE Main & Advanced
(1) Light and other forms of radiant energy propagate without any medium in the space in the form of waves are known as electromagnetic radiations. These waves can be produced by a charged body moving in a magnetic field or a magnet in a electric field. e.g. \[\alpha -\]rays, \[\gamma -\]rays, cosmic rays, ordinary light rays etc.
(2) Characteristics
(i) All electromagnetic radiations travel with the velocity of light.
(ii) These consist of electric and magnetic fields components that oscillate in directions perpendicular to each other and perpendicular to the direction in which the wave is travelling.
(3) A wave is always characterized by the following five characteristics,
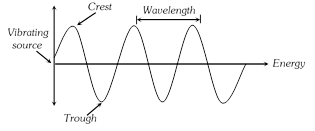
(i) Wavelength : The distance between two nearest crests or nearest troughs is called the wavelength. It is denoted by \[\lambda \](lambda) and is measured is terms of centimeter(cm), angstrom(Å), micron(\[\mu \]) or nanometre (nm).
\[1{\ AA}={{10}^{-8}}\,cm={{10}^{-10}}m\];\[1\mu ={{10}^{-4}}cm={{10}^{-6}}m\];
\[1nm={{10}^{-7}}cm={{10}^{-9}}m\]; \[1cm={{10}^{8}}{\ AA}={{10}^{4}}\mu ={{10}^{7}}nm\]
(ii) Frequency : It is defined as the number of waves which pass through a point in one second. It is denoted by the symbol \[\nu \](nu) and is expressed in terms of cycles (or waves) per second (cps) or hertz (Hz).
\[\lambda \nu =\]distance travelled in one second = velocity =c
\[\nu =\frac{c}{\lambda }\]
(iii) Velocity : It is defined as the distance covered in one second by the wave. It is denoted by the letter ?c?. All electromagnetic waves travel with the same velocity, i.e., \[3\times {{10}^{10}}cm/\sec .\]
\[c=\lambda \nu =3\times {{10}^{10}}\ cm/\sec \]
(iv) Wave number : This is the reciprocal of wavelength, i.e., the number of wavelengths per centimetre. It is denoted by the symbol \[\bar{\nu }\](nu bar). It is expressed in \[c{{m}^{-1}}\,\text{or}\,{{m}^{-1}}\].
\[\bar{\nu }=\frac{1}{\lambda }\]
(v) Amplitude : It is defined as the height of the crest or depth of the trough of a wave. It is denoted by the letter ?A?. It determines the intensity of the radiation.
The arrangement of various types of electromagnetic radiations in the order of their increasing or decreasing wavelengths or frequencies is known as electromagnetic spectrum.
| Name | Wavelength (Å) | Frequency (Hz) |
| Radio wave | \[3\times {{10}^{14}}-3\times {{10}^{7}}\] | \[1\times {{10}^{5}}-1\times {{10}^{9}}\] |
| Microwave | \[3\times {{10}^{7}}-6\times {{10}^{6}}\] | \[1\times {{10}^{9}}-5\times {{10}^{11}}\] |
| Infrared (IR) | \[6\times {{10}^{6}}-7600\] | \[5\times {{10}^{11}}-3.95\times {{10}^{16}}\] |
| Visible | \[7600-3800\] | \[3.95\times {{10}^{16}}-7.9\times {{10}^{14}}\] |
| Ultraviolet (UV) | \[3800-150\] | \[\text{7}\text{.9}\times \text{1}{{\text{0}}^{\text{14}}}-2\times {{10}^{16}}\] |
| X-Rays | \[150-0.1\] | \[2\times {{10}^{16}}-3\times {{10}^{19}}\] |
| \[\gamma -\]Rays | \[0.1-0.01\] | \[\text{3}\times \text{1}{{\text{0}}^{\text{19}}}-3\times {{10}^{20}}\] |
| Cosmic Rays | 0.01- zero | \[3\times {{10}^{20}}-\]infinity |
You need to login to perform this action.
You will be redirected in
3 sec
