Answer:
(i)
(ii)
Formation of Na2O:
The atomic number of sodium is 11 and it has only one
valence electron. Hence, electronic configuration of 11Na is 2, 8,
1.
The atomic
number of oxygen is 8 and it has 6 electrons in its valence shell. Hence,
electronic configuration of 8O is 2, 6.
Sodium
has a tendency to lose the valence electron and oxygen has a tendency to gain
the electron lost by sodium. Since, sodium can lose only one electron of the
valence shell and oxygen atom needs two electrons to complete its octet in the
valence shell, two atoms of sodium combine with one atom of oxygen. By losing
valence electron, sodium is changed into Na+ and by gaining two
electrons lost by two sodium atoms, oxygen atom is changed into an oxide anion,
O2-. In this process, both the atoms, sodium and oxygen, obtain the
stable electronic configuration of the noble gas neon.
Metal
Symbol
Atomic number
Electronic configuration
K, L, M, N
No. of outermost electrons
Electron dot structures
Sodium
Na
11
2, 8, 1
1
Na.
Oxygen
O
8
2, 6
6
![]()
Magnesium
Mg
12
2, 8, 2
2
![]()
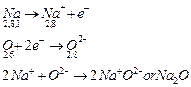 The
oppositely charged sodium ion, Na+ and oxide ion, O2- are
now held together by electrostatic force of attraction or by ionic or electrovalent
bond. Na2O is, therefore, an ionic or electrovalent compound.
The
oppositely charged sodium ion, Na+ and oxide ion, O2- are
now held together by electrostatic force of attraction or by ionic or electrovalent
bond. Na2O is, therefore, an ionic or electrovalent compound.
![]() Formation
of MgO:
The
atomic number of magnesium =12
Its
electronic configuration is K, L, M.
2, 8, 2
It has two
electrons in its outermost shell. So, the magnesium atom donates its 2 valence
electrons and forms a stable magnesium ion, Mg2+ to attain the electronic
arrangement of neon atom.
Mg ® Mg2+ + 2e-
2,
8, 2 2, 8
The
atomic number of oxygen = 8
Electronic
configuration = K, L
2, 6
It has 6
electrons in its valence shell. Therefore, it requires 2 more electrons to
attain the stable electronic arrangement of neon gas. Thus, oxygen accepts 2
electrons donated by magnesium atom and forms a stable oxide ion, O2-
O + 2e- ®
O2-
2, 6
2, 8
The
oppositely charged magnesium ions, Mg2+, and oxide ions, O2-
are held together by a strong force of electrostatic attraction to form magnesium
oxide compound Mg 2 +O2- or MgO.
Formation
of MgO:
The
atomic number of magnesium =12
Its
electronic configuration is K, L, M.
2, 8, 2
It has two
electrons in its outermost shell. So, the magnesium atom donates its 2 valence
electrons and forms a stable magnesium ion, Mg2+ to attain the electronic
arrangement of neon atom.
Mg ® Mg2+ + 2e-
2,
8, 2 2, 8
The
atomic number of oxygen = 8
Electronic
configuration = K, L
2, 6
It has 6
electrons in its valence shell. Therefore, it requires 2 more electrons to
attain the stable electronic arrangement of neon gas. Thus, oxygen accepts 2
electrons donated by magnesium atom and forms a stable oxide ion, O2-
O + 2e- ®
O2-
2, 6
2, 8
The
oppositely charged magnesium ions, Mg2+, and oxide ions, O2-
are held together by a strong force of electrostatic attraction to form magnesium
oxide compound Mg 2 +O2- or MgO.
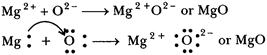 MgO is an ionic compound.
(iii)
The ions present in Na2O are sodium ions (2Na+) and oxide
ion O2-.
The
ions present in MgO are magnesium ion (Mg 2 +) and oxide ion O2-
.
MgO is an ionic compound.
(iii)
The ions present in Na2O are sodium ions (2Na+) and oxide
ion O2-.
The
ions present in MgO are magnesium ion (Mg 2 +) and oxide ion O2-
.
You need to login to perform this action.
You will be redirected in
3 sec
