-
question_answer1)
|
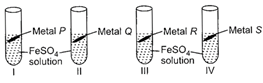
Rohan took 10 mL of freshly prepared iron sulphate solution in four different test tubes and added four different metal strips to each test tube as shown below:
|
|
|
In test tubes I and III, black residue was obtained while in test tubes II and IV, no change was observed. Metals P, Q, R and S could be respectively
|
A)
\[Al,\,Cu,\,Pb,\,Ag\] done
clear
B)
\[Pb,\,Cu,\,Ag,\,Al\] done
clear
C)
\[Zn,\,Al,\,Cu,\,Ag\] done
clear
D)
\[Zn,\,Cu,\,Al,\,Ag\] done
clear
View Solution play_arrow
-
question_answer2)
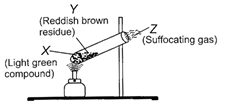
A small amount of light green colored compound X is heated in a test tube. In the beginning, it loses some water and then gas Z with suffocating smell comes out. The vapors of gas are collected and dissolved in water. The solution turns blue litmus red. The residue Y left in the test tube turns reddish brown. Identify X, Y and Z.
A)
B)
C)
D)
View Solution play_arrow
-
question_answer3)
A small amount of calcium oxide (quick lime) is taken in a beaker. Water is slowly added to this. Which of the following observations is/are correct about this activity?
| (i) The beaker becomes hot because it is an endothermic reaction. |
| (ii) A clear solution is obtained at the top after the reaction gets over. |
| (iii) This reaction is a combination reaction in which quick lime \[(CaO)\] is converted into slaked lime, \[Ca{{(OH)}_{2}}.\] |
A)
(i), (ii) and (iii) done
clear
B)
(ii) and (iii) only done
clear
C)
(iii) only done
clear
D)
(i) and (iii) only done
clear
View Solution play_arrow
-
question_answer4)
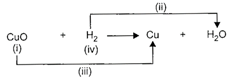
Observe the given reaction carefully and identify (i), (ii), (iii) and (iv).
A)
B)
C)
D)
View Solution play_arrow
-
question_answer5)
Match column I with column II and choose the correct answer from the codes given below.
| Column I | Column II |
| P. Limestone is heated. | (i) Electrolysis |
| Q. Magnesium wire is burnt in air. | (ii) Decomposition reaction |
| R. White ppt. of silver chloride is formed when silver nitrate is added to sodium chloride solution. | (iii) Combination reaction |
| S. Electricity is passed through acidulated water. | (iv) Double displacement reaction |
A)
P-(iv), Q-(ii), R-(i), S-(iii) done
clear
B)
P-(ii), Q-(iii), R-(iv), S-(i) done
clear
C)
P-(ii), Q-(iv), R-(iii), S-(i) done
clear
D)
P-(i), Q-(ii), R-(iv), S-(iii) done
clear
View Solution play_arrow
-
question_answer6)
A student-wrote three statements about rancidity:
| I. When fats and oils are reduced, they become rancid. |
| II. Chips manufacturers usually flush chips bags with oxygen to prevent rancidity. |
| III. Rancidity is prevented by adding substances called antioxidants to food. |
Choose the correct statement(s).
A)
I only done
clear
B)
II and III only done
clear
C)
III only done
clear
D)
All of these done
clear
View Solution play_arrow
-
question_answer7)
|
Metal X is found in earth's crust. This metal forms a reddish brown substance when exposed to moist air. When a blue colored solution Y is stored in a container made of X, the solution turns green and a reddish brown metal Z gets deposited on the container.
|
|
X, Y and Z are respectively
|
A)
\[Cu,CuS{{O}_{4}},Fe\] done
clear
B)
\[Zn,ZnS{{O}_{4}},Fe\] done
clear
C)
\[Fe,CuS{{O}_{4}},Cu\] done
clear
D)
\[Cu,FeS{{O}_{4}},Fe\] done
clear
View Solution play_arrow
-
question_answer8)
Two colorless solutions X and V were mixed together. On mixing, a yellow precipitate Z was formed. Which of the following statements is correct regarding X, Y and Z?
A)
X and Y were lead nitrate and potassium iodide solutions. The yellow precipitate Z was lead iodide. done
clear
B)
X and Y were potassium chloride solution and water. The yellow precipitate Z was of chloride ion. done
clear
C)
X and Y were sodium hydroxide solution and hydrochloric acid and the yellow precipitate Z was sodium chloride. done
clear
D)
X and Y were potassium hydroxide solution and nitric acid and the yellow precipitate Z was potassium nitrate. done
clear
View Solution play_arrow
-
question_answer9)
\[x\]and y in the given reaction are respectively \[xCaC{{O}_{3}}+y{{H}_{3}}P{{O}_{4}}\to C{{a}_{3}}{{(P{{O}_{4}})}_{2}}+x{{H}_{2}}O+xC{{O}_{2}}\]
A)
2, 3 done
clear
B)
3, 3 done
clear
C)
3, 2 done
clear
D)
1, 3 done
clear
View Solution play_arrow
-
question_answer10)
|
Take 5 mL of sodium sulphate solution in a test tube and 5 mL of barium chloride solution in another test tube. When the two solutions are mixed slowly and the test tube is left undisturbed for some time, a white precipitate is formed.
|
|
Which of the following statements is correct about the above reaction?
|
A)
It is a double displacement reaction since exchange of ions occurs between the two reactants. done
clear
B)
It is a combination reaction since barium ions combine with sulphate ions to give barium sulphate. done
clear
C)
It is a displacement reaction since chloride ions are replaced by sulphate ions. done
clear
D)
It is a neutralization reaction since an acid reacts with alkali to give a salt. done
clear
View Solution play_arrow
-
question_answer11)
For the given reaction, match column I with column II and mark the correct option from the codes given below.
|
\[F{{e}_{2}}{{O}_{3}}+xCO\to yFe+xC{{O}_{2}}\]
|
| Column I |
Column II |
| (a) Oxidizing agent |
(i) 2 |
| (b) Reducing agent |
(ii) 3 |
| (c)\[x\] |
(iii)\[F{{e}_{2}}{{O}_{3}}\] |
| (d) y |
(iv) CO |
A)
(a)-(iv), (b)-(iii), (c)-(ii), (d)-(i) done
clear
B)
(a)-(iv), (b)-(iii), (c)-(i), (d)-(ii) done
clear
C)
(a)-(iii), (b)-(iv), (c)-(ii), (d)-(i) done
clear
D)
(a)-(iii), (b)-(iv), (c)-(i), (d)-(ii) done
clear
View Solution play_arrow
-
question_answer12)
|
Read the given passage and fill in the blanks by choosing an appropriate option.
|
|
When a limited quantity of water is sprinkled over a dirty white stone called ___i___, an ___ii___ reaction takes place. After some time, the stone like substance changes into white amorphous powder called, ___iii___ which is used for ___iv___.
|
A)
B)
C)
D)
View Solution play_arrow
-
question_answer13)
Four test tubes were taken and marked 1, 2, 3 and 4 respectively. 2 mL of aqueous solution of aluminium sulphate, \[A{{l}_{2}}{{(S{{O}_{4}})}_{3}}\] was filled in each test tube. A piece of metal zinc was placed in test tube 1, iron in test tube 2, copper in test tube 3 and aluminium in test tube 4. Mark the correct change in colour in the four test tubes.
A)
B)
C)
D)
View Solution play_arrow
-
question_answer14)
Study the given table carefully.
| S. No. | Metal | Reaction with dilute HCI |
| 1. | P | No change |
| 2. | Q | Temperature of reaction mixture rises. |
| 3. | R | Reaction is explosive. |
| 4. | S | Some gas bubbles are seen. |
Metals P, Q, R and S could be respectively
A)
K, Al, Na and Cu done
clear
B)
Cu, Pb, Na and Al done
clear
C)
Ag, Al, Na and Pb done
clear
D)
Cu, Ca, K and AI done
clear
View Solution play_arrow
-
question_answer15)
Read the given statements and mark the correct option.
| Statement 1: When a mixture of hydrogen and chlorine is placed in sunlight, hydrogen chloride is formed. |
| Statement 2: It is an example of displacement reaction. |
A)
Both statements 1 and 2 are true and statement 2 is the correct explanation of statement 1. done
clear
B)
Both statements 1 and 2 are true but statement 2 is not the correct explanation of statement 1. done
clear
C)
Statement 1 is true and statement 2 is false. done
clear
D)
Both statements 1 and 2 are false. done
clear
View Solution play_arrow
-
question_answer16)
Which of the following reactions are exothermic in nature?
| (i) Evaporation of water |
| (ii) Dissolution of sodium hydroxide in water |
| (iii) Dilution of sulphuric acid |
| (iv) Dissolution of ammonium chloride in water |
| (v) Combustion of methane gas |
A)
(i), (iv) and (v) only done
clear
B)
(ii), (iii) and (v) only done
clear
C)
(i), (ii) and (iv) only done
clear
D)
(i), (iii) and (iv) only done
clear
View Solution play_arrow
-
question_answer17)
|
Marble chips or calcium carbonate react with hydrochloric acid as :
|
|
\[CaC{{O}_{3}}+2HCl\to CaC{{l}_{2}}+{{H}_{2}}O+C{{O}_{2}}\]
|
|
The reaction is carried out twice and following graphs were obtained:
|

|
Which of the following statements is incorrect?
A)
Reaction is faster in experiment II. done
clear
B)
Marble chips taken in experiment II are smaller in size than the marble chips taken in experiment I. done
clear
C)
Same amount of marble chips and hydrochloric acid is used for both the experiments. done
clear
D)
None of these done
clear
View Solution play_arrow
-
question_answer18)
Four students P, Q, Rand S noted the initial colour of the solutions kept in beakers I, II, III and IV. After inserting zinc rods in each solution and leaving them undisturbed for two hours, the colour of each solution was again noted in the form of table given below:
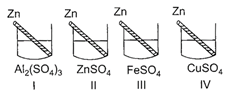
| Student | Colour of the solution | I | II | III | IV |
| P | Initial | Colour- less | Colour- less | Light green | Blue |
| Final | Colour- less | Colour- less | Colour- less | Colour- less |
| Q | Initial | Colour- less | Light yellow | Light green | Blue |
| Final | Colour- less | Colour- less | Light green | Colour- less |
| R | Initial | Colour- less | Colour- less | Light green | Blue |
| Final | Light blue | Colour- less | Colour- less | Light blue |
| S | Initial | Light green | Colour- less | Light green | Blue |
| Final | Colour- less | Colour- less | Dark green | Colour- less |
Which student noted the colour change in all the four beakers correctly?
A)
P done
clear
B)
Q done
clear
C)
R done
clear
D)
S done
clear
View Solution play_arrow
-
question_answer19)
Observe the given figure carefully Which of the following observation(s) is/are correct?
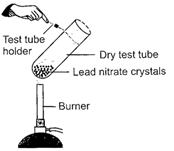
| I. A double decomposition reaction takes place. |
| II. Brown fumes of \[N{{O}_{2}}\]are evolved. |
| III. Red residue is left behind in the test tube. |
A)
I and III only done
clear
B)
II and III only done
clear
C)
I and II only done
clear
D)
II only done
clear
View Solution play_arrow
-
question_answer20)
|
Rupali, a class 10 student has set up the apparatus as shown in the figures.
|

|
|
Which of the following observations is correct?
|
A)
Temperature of beakers I, II and III will be raised as dissolution of salts is an exothermic process. done
clear
B)
Temperature of beakers III and IV will be raised while temperature of beakers I and II will fall. done
clear
C)
Temperature will rise only in beaker IV as redox reactions are exothermic. done
clear
D)
None of these done
clear
View Solution play_arrow

