-
question_answer1)
Observe the rate of evolution of hydrogen gas with five metals P, Q, R, S and T at room temperature. What could be the metals P, Q, R, S and T?
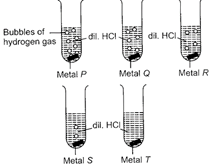
A)
B)
C)
D)
View Solution play_arrow
-
question_answer2)
Four substances have the following electrical properties.
| Substance | Electrical property |
| W | Does not conduct under any condition |
| X | Conducts only in aqueous solution |
| Y | Conducts in both the molten and solid states |
| Z | Conducts in both the molten state and aqueous solution |
What are these four substances?
A)
B)
C)
D)
View Solution play_arrow
-
question_answer3)
|
Fill in the blanks by selecting the option with the correct words.
|
|
The process of removing impurities from impure metal to obtain pure metal is known as ______ of metal. The most widely used method for refining is ______. In this method, the impure metal is taken as ______ and the pure metal is taken as ______.
|
A)
Refining, electrolytic refining, anode, cathode done
clear
B)
Calcination, electrolysis, cathode, anode done
clear
C)
Refining, smelting, cathode, anode done
clear
D)
Smelting, calcination, anode, cathode done
clear
View Solution play_arrow
-
question_answer4)
Shweta was given four squares P, Q, R and S with atomic numbers written on them. She gave the following four statements.

| (i) Element after square Q is a non-metal. |
| (ii) Square R represents a metalloid. |
| (iii) Element just before square R is a metalloid. |
| (iv) Element just before square S is a metal. |
The true statements given by her are ______.
A)
(i), (ii) and (iii) only done
clear
B)
(i), (ii) and (iv) only done
clear
C)
(ii) and (iii) only done
clear
D)
(i), (ii), (iii) and (iv) done
clear
View Solution play_arrow
-
question_answer5)
Match column I (method of concentration) with column II (principle involved) and choose the appropriate option from the codes given below.
| Column I | Column II |
| P. Hydraulic washing | (i) Difference in the wetting properties of ore and gangue particles |
| Q Froth floatation process | (ii) Difference in magnetic properties of ore and gangue particles |
| R. Magnetic separation | (iii) Difference in the densities of ore and gangue particles |
| S. Baeyer?s process | (iv) Difference in chemical properties of ore and gangue particles |
A)
P-(i), Q-(iii), R-(ii), S-(iv) done
clear
B)
P-(iii), Q-(i), R-(ii), S-(iv) done
clear
C)
P-(ii), Q-(iii), R-(iv), S-(i) done
clear
D)
P-(iv), Q-(ii), R-(iii), S-(i) done
clear
View Solution play_arrow
-
question_answer6)
Which of the following statements are incorrect?
| I. Reactivity of Al decreases if it is dipped in nitric acid. |
| II. Carbon can reduce the oxides of Na and \[Mg\]. |
| III. \[NaCl\] is a good conductor of electricity in solid state. |
| IV. Metals like \[K,\,Ca\] and \[Mg\] are always found in free state in nature. |
A)
I, II and III only done
clear
B)
II, III and IV only done
clear
C)
I and III only done
clear
D)
II and III only done
clear
View Solution play_arrow
-
question_answer7)
Read the given statements carefully and find the incorrect match.
A)
A metal used in joining electric wires - Magnesium done
clear
B)
A metal whose oxide is soluble in both acids and bases - Zinc done
clear
C)
A metal unreactive towards oxygen and dilute acids - Gold done
clear
D)
A metal extracted by using electrolytic reduction - Aluminium done
clear
View Solution play_arrow
-
question_answer8)
Read the given statements and mark the correct option.
| Statement 1: Articles made of aluminium do not corrode even though it is an active metal. |
| Statement 2: Aluminium reacts with air to form a layer of oxide due to which it does not react further. |
A)
Both statements 1 and 2 are true and statement 2 is the correct explanation of statement 1. done
clear
B)
Both statements 1 and 2 are true but statement 2 is not the correct explanation of statement 1. done
clear
C)
Statement 1 is true and statement 2 is false. done
clear
D)
Both statements 1 and 2 are false. done
clear
View Solution play_arrow
-
question_answer9)
A non-metal X is an important constituent of our food. It forms two oxides Y and Z. Y is toxic and causes suffocation and even death while Z is responsible for global warming. Identify X, Y and Z.
A)
\[X=C,\,Y=CO,\,Z=C{{O}_{2}}\] done
clear
B)
\[X=S,\,Y=S{{O}_{2}},\,Z=S{{O}_{3}}\] done
clear
C)
\[X=P,\,Y={{P}_{2}}{{O}_{3}},\,Z={{P}_{2}}{{O}_{5}}\] done
clear
D)
\[X=O,\,Y={{O}_{2}},Z={{O}_{3}}\] done
clear
View Solution play_arrow
-
question_answer10)
A brief information about a few elements is given below:
| P: It is used in making electrodes. |
| O: It is required for combustion. |
| R: It is used to preserve food. |
| S: It is used as fungicide. |
| P, Q, R and S are respectively |
A)
Aluminium, carbon, oxygen and phosphorus done
clear
B)
Copper, oxygen, hydrogen and iodine done
clear
C)
Carbon, oxygen, nitrogen and sulphur done
clear
D)
Sulphur, oxygen, nitrogen and hydrogen. done
clear
View Solution play_arrow
-
question_answer11)
In which of the following beakers reaction will take place?

A)
I and III only done
clear
B)
I and IV only done
clear
C)
II and IV only done
clear
D)
III and IV only done
clear
View Solution play_arrow
-
question_answer12)
An ore on heating in air produces sulphur dioxide. The process which is used for its concentration and the two steps involved in the conversion of this ore into related metal are respectively
A)
Hydraulic washing, chemical separation and refining. done
clear
B)
Baeyer's process, electrorefining and reduction done
clear
C)
Froth floatation process, conversion to metal oxide and reduction of metal oxide done
clear
D)
None of these. done
clear
View Solution play_arrow
-
question_answer13)
Which of the following statements about the given reaction is/are incorrect? \[F{{e}_{2}}{{O}_{3}}+2Al\xrightarrow{\lg nited}2Fe+A{{l}_{2}}{{O}_{3}}\]
| I. The mixture of \[F{{e}_{2}}{{O}_{3}}\]and Al is ignited by inserting a magnesium ribbon and then burning it. |
| II. The reaction is used for welding the broken parts of iron machinery, railway tracks, etc. |
| III. The whole process is called aluminothermy. |
| IV. The reaction is highly endothermic. |
A)
I and III only done
clear
B)
I and IV only done
clear
C)
II and III only done
clear
D)
IV only done
clear
View Solution play_arrow
-
question_answer14)
Latin name for X is 'royal water'. It is a mixture of Y and Z in the ratio 3:1. Some of the properties of X are different from Y and Z. What are X, Y and Z respectively?
A)
Aqua regia, \[HCl,\,HN{{O}_{3}}\] done
clear
B)
Aqua regia,\[HN{{O}_{3}},\,HCl\] done
clear
C)
\[{{H}_{2}}S{{O}_{4}},{{H}_{2}}O,S{{O}_{2}}\] done
clear
D)
\[{{H}_{2}}S{{O}_{4}},{{H}_{2}}S,S{{O}_{3}}\] done
clear
View Solution play_arrow
-
question_answer15)
Which of the following elements are metals?
| (i) \[_{12}^{24}X\] |
(ii) \[_{9}^{19}X\] |
| (iii) \[_{15}^{31}X\] |
(iv) \[_{11}^{23}X\] |
A)
(i) and (ii) only done
clear
B)
(ii) and (iii) only done
clear
C)
(i) and (iv) only done
clear
D)
(ii) and (iv) only done
clear
View Solution play_arrow
-
question_answer16)
A)
I and III only done
clear
B)
II and IV only done
clear
C)
I and IV only done
clear
D)
II and III only done
clear
View Solution play_arrow
-
question_answer17)
|
Observe the given figure carefully.
|
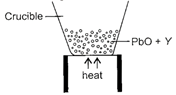
|
|
The residue left behind in the crucible, substance Y and the substance which can replace Y in the above process are respectively
|
A)
White powder of Pb, Cu and K done
clear
B)
Brown crystals of \[P{{b}_{2}}{{O}_{3}},\,C\] and CO done
clear
C)
Shiny globules of Pb, C and Mg done
clear
D)
Black mass of \[P{{b}_{2}}{{O}_{3}},\,CO\] and Cu. done
clear
View Solution play_arrow
-
question_answer18)
Read the given statements carefully.
| I. Metals are generally ductile and metal W is the most ductile metal. |
| II. Metals generally possess high melting points but few metals like X have low melting point. |
| III. Metals are good conductors of electricity but metal Y is very poor conductor of electricity. |
| IV. Among metals, Z is the poorest conductor of heat. |
W, X, Y and Z can be respectively
A)
Aluminium, sodium, lead and silver done
clear
B)
Gold, potassium, mercury and lead done
clear
C)
Copper, zinc, silver and mercury done
clear
D)
Iron, sodium, gold and lead. done
clear
View Solution play_arrow
-
question_answer19)
|
Read the given passage carefully and fill in the blanks by choosing an appropriate option.
|
|
Oxides of ___i___ are reduced to their respective metals by heating with coke and the process is called ___ii___. Before carrying out reduction with carbon, if the calcined or roasted ore still contains infusible impurities of earthy matter, ___iii___, an additional substance, ___iv___ is added which combines with iii to form a fusible material, called ___v___.
|
A)
B)
C)
D)
View Solution play_arrow
-
question_answer20)
|
Flow chart representing extraction of metals from ores is given below:
|
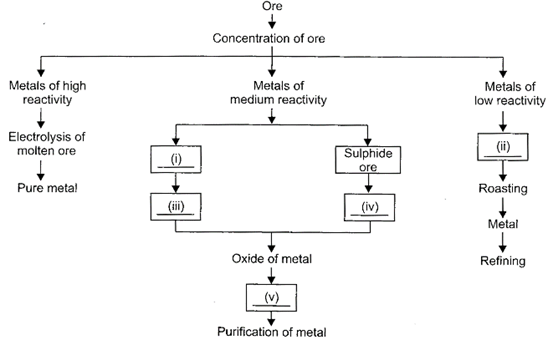
|
|
Fill in the blanks by selecting the appropriate option.
|
A)
B)
C)
D)
View Solution play_arrow

