Answer:
(a) \[MgC{{l}_{2}}.6{{H}_{2}}O\xrightarrow{Heat}MgO+5{{H}_{2}}O+2HCl\]
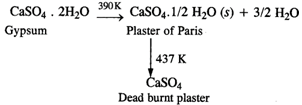
(b)
 (c) \[Ca{{(HC{{O}_{3}})}_{2}}\xrightarrow{Heat}\,CaC{{O}_{3}}+{{H}_{2}}O+C{{O}_{2}}\]
(d) \[\underset{\text{Epsom}\,\,\text{salt}}{\mathop{MgS{{O}_{4}}}}\,.\,7{{H}_{2}}O\xrightarrow[-6{{H}_{2}}O]{423\,K}\,MgS{{O}_{4}}.{{H}_{2}}O\xrightarrow[-{{H}_{2}}O]{503\,K}\,MgS{{O}_{4}}\xrightarrow[\text{heating}]{\text{Strong}}\,MgO+S{{O}_{3}}\]
(c) \[Ca{{(HC{{O}_{3}})}_{2}}\xrightarrow{Heat}\,CaC{{O}_{3}}+{{H}_{2}}O+C{{O}_{2}}\]
(d) \[\underset{\text{Epsom}\,\,\text{salt}}{\mathop{MgS{{O}_{4}}}}\,.\,7{{H}_{2}}O\xrightarrow[-6{{H}_{2}}O]{423\,K}\,MgS{{O}_{4}}.{{H}_{2}}O\xrightarrow[-{{H}_{2}}O]{503\,K}\,MgS{{O}_{4}}\xrightarrow[\text{heating}]{\text{Strong}}\,MgO+S{{O}_{3}}\]
You need to login to perform this action.
You will be redirected in
3 sec
