-
question_answer1)
Be's 4th electron will have four quantum numbers [MNR 1985]
A)
\[n\]- 1 \[l\]- 0 \[m\]- 0 \[s\]- +1/2 done
clear
B)
\[n\]- 1 \[l\]- 1 \[m\]- +1 \[s\]- +1/2 done
clear
C)
\[n\]- 2 \[l\]- 0 \[m\]- 0 \[s\]- -1/2 done
clear
D)
\[n\]- 2 \[l\]- 1 \[m\]- 0 \[s\]- +1/2 done
clear
View Solution play_arrow
-
question_answer2)
The quantum number which specifies the location of an electron as well as energy is [DPMT 1983]
A)
Principal quantum number done
clear
B)
Azimuthal quantum number done
clear
C)
Spin quantum number done
clear
D)
Magnetic quantum number done
clear
View Solution play_arrow
-
question_answer3)
The shape of an orbital is given by the quantum number [NCERT 1984; MP PMT 1996]
A)
\[n\] done
clear
B)
\[l\] done
clear
C)
\[m\] done
clear
D)
\[s\] done
clear
View Solution play_arrow
-
question_answer4)
In a given atom no two electrons can have the same values for all the four quantum numbers. This is called [BHU 1979; AMU 1983; EAMCET 1980, 83; MADT Bihar 1980; CPMT 1986, 90, 92; NCERT 1978, 84; RPMT 1997; CBSE PMT 1991; MP PET 1986, 99]
A)
Hund's rule done
clear
B)
Aufbau's principle done
clear
C)
Uncertainty principle done
clear
D)
Pauli's exclusion principle done
clear
View Solution play_arrow
-
question_answer5)
Nitrogen has the electronic configuration \[1{{s}^{2}},2{{s}^{2}}2p_{x}^{1}2p_{y}^{1}2p_{z}^{1}\] and not \[1{{s}^{2}},2{{s}^{2}}2p_{x}^{2}2p_{y}^{1}2p_{z}^{0}\] which is determined by [DPMT 1982, 83, 89; MP PMT/PET 1988; EAMCET 1988]
A)
Aufbau's principle done
clear
B)
Pauli's exclusion principle done
clear
C)
Hund's rule done
clear
D)
Uncertainty principle done
clear
View Solution play_arrow
-
question_answer6)
Which one of the following configuration represents a noble gas [CPMT 1983, 89, 93; NCERT 1973; MP PMT 1989; DPMT 1984]
A)
\[1{{s}^{2}},2{{s}^{2}}2{{p}^{6}},3{{s}^{2}}\] done
clear
B)
\[1{{s}^{2}},2{{s}^{2}}2{{p}^{6}},3{{s}^{1}}\] done
clear
C)
\[1{{s}^{2}},2{{s}^{2}}2{{p}^{6}}\] done
clear
D)
\[1{{s}^{2}},2{{s}^{2}}s{{p}^{6}},3{{s}^{2}}3{{p}^{6}},4{{s}^{2}}\] done
clear
View Solution play_arrow
-
question_answer7)
The electronic configuration of silver atom in ground state is [CPMT 1984, 93]
A)
\[[Kr]3{{d}^{10}}\,4{{s}^{1}}\] done
clear
B)
\[[Xe]\,4{{f}^{14}}5{{d}^{10}}6{{s}^{1}}\] done
clear
C)
\[[Kr]\,4{{d}^{10}}5{{s}^{1}}\] done
clear
D)
\[[Kr]\,4{{d}^{9}}5{{s}^{2}}\] done
clear
View Solution play_arrow
-
question_answer8)
Principal, azimuthal and magnetic quantum numbers are respectively related to [CPMT 1988; AIIMS 1999]
A)
Size, shape and orientation done
clear
B)
Shape, size and orientation done
clear
C)
Size, orientation and shape done
clear
D)
None of the above done
clear
View Solution play_arrow
-
question_answer9)
Correct set of four quantum numbers for valence electron of rubidium (Z = 37) is [IIT 1984; JIPMER 1999; UPSEAT 2003]
A)
\[5,\,0,\,0,\,+\frac{1}{2}\] done
clear
B)
\[5,\,1,\,0,\,+\frac{1}{2}\] done
clear
C)
\[5,\,1,\,1,\,+\frac{1}{2}\] done
clear
D)
\[6,\,0,\,0,\,+\,\frac{1}{2}\] done
clear
View Solution play_arrow
-
question_answer10)
The correct ground state electronic configuration of chromium atom is [IIT 1989, 94; MP PMT 1993; EAMCET 1997; ISM Dhanbad 1994; AFMC 1997; Bihar MEE 1996; MP PET 1995, 97; CPMT 1999; Kerala PMT 2003]
A)
\[[Ar]\,3{{d}^{5}}\,4{{s}^{1}}\] done
clear
B)
\[[Ar]\,3{{d}^{4}}4{{s}^{2}}\] done
clear
C)
\[[AR]3{{d}^{6}}4{{s}^{0}}\] done
clear
D)
\[[Ar]4{{d}^{5}}4{{s}^{1}}\] done
clear
View Solution play_arrow
-
question_answer11)
2p orbitals have [NCERT 1981; MP PMT 1993, 97]
A)
\[n=1,\,l=2\] done
clear
B)
\[n=1,\,l=0\] done
clear
C)
\[n=2,\,l=1\] done
clear
D)
\[n=2,\,l=0\] done
clear
View Solution play_arrow
-
question_answer12)
Electronic configuration of \[{{H}^{-}}\] is [CPMT 1985]
A)
\[1{{s}^{0}}\] done
clear
B)
\[1{{s}^{1}}\] done
clear
C)
\[1{{s}^{2}}\] done
clear
D)
\[1{{s}^{1}}\,2{{s}^{1}}\] done
clear
View Solution play_arrow
-
question_answer13)
The quantum numbers for the outermost electron of an element are given below as \[n=2,\,l=0,\,m=0,\,s=+\frac{1}{2}\]. The atoms is [EAMCET 1978]
A)
Lithium done
clear
B)
Beryllium done
clear
C)
Hydrogen done
clear
D)
Boron done
clear
View Solution play_arrow
-
question_answer14)
Principal quantum number of an atom represents [EAMCET 1979; IIT 1983; MNR 1990;UPSEAT 2000, 02]
A)
Size of the orbital done
clear
B)
Spin angular momentum done
clear
C)
Orbital angular momentum done
clear
D)
Space orientation of the orbital done
clear
View Solution play_arrow
-
question_answer15)
An element has the electronic configuration \[1{{s}^{2}},2{{s}^{2}}2{{p}^{6}},3{{s}^{2}}3{{p}^{2}}\]. Its valency electrons are [NCERT 1973]
A)
6 done
clear
B)
2 done
clear
C)
3 done
clear
D)
4 done
clear
View Solution play_arrow
-
question_answer16)
The magnetic quantum number specifies [MNR 1986; BHU 1982; CPMT 1989, 94; MP PET 1999; AFMC 1999; AMU (Engg.) 1999]
A)
Size of orbitals done
clear
B)
Shape of orbitals done
clear
C)
Orientation of orbitals done
clear
D)
Nuclear stability done
clear
View Solution play_arrow
-
question_answer17)
Which of the following sets of quantum numbers represent an impossible arrangement [IIT 1986; MP PET 1995]
A)
n ? 3 l ? 2 m - - 2 \[{{m}_{s}}\]- (+)\[\frac{1}{2}\] done
clear
B)
n ? 4 l ? 0 m ? 0 \[{{m}_{s}}\]- (?)\[\frac{1}{2}\] done
clear
C)
n ? 3 l ? 2 m - -3 \[{{m}_{s}}\] - (+)\[\frac{1}{2}\] done
clear
D)
n ? 5 l ? 3 m ? 0 \[{{m}_{s}}\] - (?) \[\frac{1}{2}\] done
clear
View Solution play_arrow
-
question_answer18)
If \[n=3\], then the value of \['l'\] which is incorrect [CPMT 1994]
A)
0 done
clear
B)
1 done
clear
C)
2 done
clear
D)
3 done
clear
View Solution play_arrow
-
question_answer19)
Which orbital is dumb-bell shaped [MP PMT 1986; MP PET/PMT 1998]
A)
\[s\]-orbital done
clear
B)
\[p\]-orbital done
clear
C)
\[d\]-orbital done
clear
D)
\[f\]-orbital done
clear
View Solution play_arrow
-
question_answer20)
The total number of unpaired electrons in \[d\]- orbitals of atoms of element of atomic number 29 is [CPMT 1983]
A)
10 done
clear
B)
1 done
clear
C)
0 done
clear
D)
5 done
clear
View Solution play_arrow
-
question_answer21)
The shape of \[2p\]orbital is [CPMT 1983; NCERT 1979]
A)
Spherical done
clear
B)
Ellipsoidal done
clear
C)
Dumb-bell done
clear
D)
Pyramidal done
clear
View Solution play_arrow
-
question_answer22)
The magnetic quantum number for an electron when the value of principal quantum number is 2 can have [CPMT 1984]
A)
3 values done
clear
B)
2 values done
clear
C)
9 values done
clear
D)
6 values done
clear
View Solution play_arrow
-
question_answer23)
Which one is the correct outer configuration of chromium [AIIMS 1980, 91; BHU 1995]
A)
B)
C)
D)
View Solution play_arrow
-
question_answer24)
The following has zero valency [DPMT 1991]
A)
Sodium done
clear
B)
Beryllium done
clear
C)
Aluminium done
clear
D)
Krypton done
clear
View Solution play_arrow
-
question_answer25)
The number of electrons in the valence shell of calcium is [IIT 1975]
A)
6 done
clear
B)
8 done
clear
C)
2 done
clear
D)
4 done
clear
View Solution play_arrow
-
question_answer26)
The valence electron in the carbon atom are [MNR 1982]
A)
0 done
clear
B)
2 done
clear
C)
4 done
clear
D)
6 done
clear
View Solution play_arrow
-
question_answer27)
For the dumb-bell shaped orbital, the value of \[l\] is [CPMT 1987, 2003]
A)
3 done
clear
B)
1 done
clear
C)
0 done
clear
D)
2 done
clear
View Solution play_arrow
-
question_answer28)
Chromium has the electronic configuration \[4{{s}^{1}}3{{d}^{5}}\] rather than \[4{{s}^{2}}3{{d}^{4}}\] because
A)
\[4s\] and \[3d\] have the same energy done
clear
B)
\[4s\] has a higher energy than \[3d\] done
clear
C)
\[4{{s}^{1}}\] is more stable than \[4{{s}^{2}}\] done
clear
D)
\[4{{s}^{1}}3{{d}^{5}}\] half-filled is more stable than \[4{{s}^{2}}3{{d}^{4}}\] done
clear
View Solution play_arrow
-
question_answer29)
The electronic configuration of calcium ion \[(C{{a}^{2+}})\] is [CMC Vellore 1991]
A)
\[1{{s}^{2}},2{{s}^{2}}2{{p}^{6}},3{{s}^{2}}3{{p}^{6}},4{{s}^{2}}\] done
clear
B)
\[1{{s}^{2}},2{{s}^{2}}s{{p}^{6}},3{{s}^{2}}3{{p}^{6}},4{{s}^{1}}\] done
clear
C)
\[1{{s}^{2}},2{{s}^{2}}2{{p}^{6}},3{{s}^{2}}3{{p}^{6}}3{{d}^{2}}\] done
clear
D)
\[1{{s}^{2}},2{{s}^{2}}s{{p}^{6}},3{{s}^{2}}3{{p}^{6}}3{{d}^{5}}\] done
clear
E)
\[1{{s}^{2}},2{{s}^{2}}2{{p}^{6}},3{{s}^{2}}3{{p}^{6}},4{{s}^{0}}\] done
clear
View Solution play_arrow
-
question_answer30)
The structure of external most shell of inert gases is [JIPMER 1991]
A)
\[{{s}^{2}}{{p}^{3}}\] done
clear
B)
\[{{s}^{2}}{{p}^{6}}\] done
clear
C)
\[{{s}^{1}}{{p}^{2}}\] done
clear
D)
\[{{d}^{10}}{{s}^{2}}\] done
clear
View Solution play_arrow
-
question_answer31)
The two electrons in K sub-shell will differ in [MNR 1988; UPSEAT 1999, 2000; Kerala PMT 2003]
A)
Principal quantum number done
clear
B)
Azimuthal quantum number done
clear
C)
Magnetic quantum number done
clear
D)
Spin quantum number done
clear
View Solution play_arrow
-
question_answer32)
A completely filled \[d\]-orbital \[({{d}^{10}})\] [MNR 1987]
A)
Spherically symmetrical done
clear
B)
Has octahedral symmetry done
clear
C)
Has tetrahedral symmetry done
clear
D)
Depends on the atom done
clear
View Solution play_arrow
-
question_answer33)
If magnetic quantum number of a given atom represented by ?3, then what will be its principal quantum number [BHU 2005]
A)
2 done
clear
B)
3 done
clear
C)
4 done
clear
D)
5 done
clear
View Solution play_arrow
-
question_answer34)
The total number of orbitals in an energy level designated by principal quantum number \[n\] is equal to [AIIMS 1997; J&K CET 2005]
A)
\[2n\] done
clear
B)
\[2{{n}^{2}}\] done
clear
C)
\[n\] done
clear
D)
\[{{n}^{2}}\] done
clear
View Solution play_arrow
-
question_answer35)
The number of orbitals in the fourth principal quantum number will be
A)
4 done
clear
B)
8 done
clear
C)
12 done
clear
D)
16 done
clear
View Solution play_arrow
-
question_answer36)
Which set of quantum numbers are not possible from the following
A)
\[n=3,\,l=2,\,m=0,\,s=-\frac{1}{2}\] done
clear
B)
\[n=3,\,l=2,\,m=-2,\,s=-\frac{1}{2}\] done
clear
C)
\[n=3,\,l=3,\,m=-3,\,s=-\frac{1}{2}\] done
clear
D)
\[n=3,\,l=0,\,m=0,\,s=-\frac{1}{2}\] done
clear
View Solution play_arrow
-
question_answer37)
The four quantum number for the valence shell electron or last electron of sodium (Z = 11) is [MP PMT 1999]
A)
\[n=2,\,l=1,\,m=-1,\,s=-\frac{1}{2}\] done
clear
B)
\[n=3,\,l=0,\,m=0,\,s=+\frac{1}{2}\] done
clear
C)
\[n=3,\,l=2,\,m=-2,\,s=-\frac{1}{2}\] done
clear
D)
\[n=3,\,l=2,\,m=2,\,s=+\frac{1}{2}\] done
clear
View Solution play_arrow
-
question_answer38)
The explanation for the presence of three unpaired electrons in the nitrogen atom can be given by [NCERT 1979; RPMT 1999; DCE 1999, 2002; CPMT 2001; MP PMT 2002; Pb. PMT / CET 2002]
A)
Pauli's exclusion principle done
clear
B)
Hund's rule done
clear
C)
Aufbau's principle done
clear
D)
Uncertainty principle done
clear
View Solution play_arrow
-
question_answer39)
The maximum energy is present in any electron at
A)
Nucleus done
clear
B)
Ground state done
clear
C)
First excited state done
clear
D)
Infinite distance from the nucleus done
clear
View Solution play_arrow
-
question_answer40)
The electron density between \[1s\]and \[2s\]orbital is
A)
High done
clear
B)
Low done
clear
C)
Zero done
clear
D)
None of these done
clear
View Solution play_arrow
-
question_answer41)
For \[ns\] orbital, the magnetic quantum number has value
A)
2 done
clear
B)
4 done
clear
C)
? 1 done
clear
D)
0 done
clear
View Solution play_arrow
-
question_answer42)
The maximum number of electrons that can be accommodated in the \[{{M}^{th}}\] shell is
A)
2 done
clear
B)
8 done
clear
C)
18 done
clear
D)
32 done
clear
View Solution play_arrow
-
question_answer43)
For a given value of quantum number \[l\], the number of allowed values of \[m\] is given by
A)
\[l+2\] done
clear
B)
\[2l+2\] done
clear
C)
\[2l+1\] done
clear
D)
\[l+1\] done
clear
View Solution play_arrow
-
question_answer44)
The number of radial nodes of 3s and 2p orbitals are respectively. [IIT-JEE 2005]
A)
2, 0 done
clear
B)
0, 2 done
clear
C)
1, 2 done
clear
D)
2, 1 done
clear
View Solution play_arrow
-
question_answer45)
Which of the sub-shell is circular
A)
\[4s\] done
clear
B)
\[4f\] done
clear
C)
\[4p\] done
clear
D)
\[4d\] done
clear
View Solution play_arrow
-
question_answer46)
Which electronic configuration for oxygen is correct according to Hund's rule of multiplicity
A)
\[1{{s}^{2}},2{{s}^{2}}2p_{x}^{2}2p_{y}^{1}2p_{z}^{1}\] done
clear
B)
\[1{{s}^{2}},2{{s}^{2}}2p_{x}^{2}2p_{y}^{2}2p_{z}^{0}\] done
clear
C)
\[1{{s}^{2}},2{{s}^{2}}2p_{x}^{3}2p_{y}^{1}2p_{z}^{0}\] done
clear
D)
None of these done
clear
View Solution play_arrow
-
question_answer47)
If value of azimuthal quantum number \[l\] is 2, then total possible values of magnetic quantum number will be
A)
7 done
clear
B)
5 done
clear
C)
3 done
clear
D)
2 done
clear
View Solution play_arrow
-
question_answer48)
The type of orbitals present in \[Fe\] is
A)
\[s\] done
clear
B)
\[s\] and \[p\] done
clear
C)
\[s,\,p\] and \[d\] done
clear
D)
\[s,\,p,\,d\] and \[f\] done
clear
View Solution play_arrow
-
question_answer49)
The shape of \[{{d}_{xy}}\] orbital will be
A)
Circular done
clear
B)
Dumb-bell done
clear
C)
Double dumb-bell done
clear
D)
Trigonal done
clear
View Solution play_arrow
-
question_answer50)
In any atom which sub-shell will have the highest energy in the following
A)
\[3p\] done
clear
B)
\[3d\] done
clear
C)
\[4s\] done
clear
D)
\[3s\] done
clear
View Solution play_arrow
-
question_answer51)
Which electronic configuration is not observing the (\[n+l\]) rule
A)
\[1{{s}^{2}},2{{s}^{2}}2{{p}^{6}},3{{s}^{2}}3{{p}^{6}}3{{d}^{1}},4{{s}^{2}}\] done
clear
B)
\[1{{s}^{2}},2{{s}^{2}}s{{p}^{6}},3{{s}^{2}}3{{p}^{6}}3{{d}^{7}},4{{s}^{2}}\] done
clear
C)
\[1{{s}^{2}},2{{s}^{2}}2{{p}^{6}},3{{s}^{2}}3{{p}^{6}}3{{d}^{5}},4{{s}^{1}}\] done
clear
D)
\[1{{s}^{2}},2{{s}^{2}}2{{p}^{6}},3{{s}^{2}}3{{p}^{6}}3{{d}^{8}},4{{s}^{2}}\] done
clear
View Solution play_arrow
-
question_answer52)
The four quantum numbers of the outermost orbital of \[K\] (atomic no. =19) are [MP PET 1993, 94]
A)
\[n=2,\,l=0,\,m=0,\,s=+\frac{1}{2}\] done
clear
B)
\[n=4,\,l=0,\,m=0,\,s=+\frac{1}{2}\] done
clear
C)
\[n=3,\,l=1,\,m=1,\,s=+\frac{1}{2}\] done
clear
D)
\[n=4,\,l=2,\,m=-1,\,s=+\frac{1}{2}\] done
clear
View Solution play_arrow
-
question_answer53)
The angular momentum of an electron depends on [BHU 1978; NCERT 1981]
A)
Principal quantum number done
clear
B)
Azimuthal quantum number done
clear
C)
Magnetic quantum number done
clear
D)
All of these done
clear
View Solution play_arrow
-
question_answer54)
The electronic configuration of copper \[{{(}_{29}}Cu)\] is [DPMT 1983; BHU 1980; AFMC 1981; CBSE PMT 1991; MP PMT 1995]
A)
\[1{{s}^{2}},2{{s}^{2}}2{{p}^{6}},3{{s}^{2}}3{{p}^{6}}3{{d}^{9}},4{{s}^{2}}\] done
clear
B)
\[1{{s}^{2}},2{{s}^{2}}2{{p}^{6}},3{{s}^{2}}3{{p}^{6}}3{{d}^{10}},4{{s}^{1}}\] done
clear
C)
\[1{{s}^{2}}.2{{s}^{2}}2{{p}^{6}},3{{s}^{2}}3{{p}^{6}},4{{s}^{2}}4{{p}^{6}}\] done
clear
D)
\[1{{s}^{2}},2{{s}^{2}}2{{p}^{6}},3{{s}^{2}}3{{p}^{6}}3{{d}^{10}}\] done
clear
View Solution play_arrow
-
question_answer55)
The number of orbitals in \[2p\] sub-shell is [NCERT 1973; MP PMT 1996]
A)
6 done
clear
B)
2 done
clear
C)
3 done
clear
D)
4 done
clear
View Solution play_arrow
-
question_answer56)
The number of orbitals in \[d\] sub-shell is [MNR 1981]
A)
1 done
clear
B)
3 done
clear
C)
5 done
clear
D)
7 done
clear
View Solution play_arrow
-
question_answer57)
A sub-shell \[l=2\] can take how many electrons [NCERT 1973, 78]
A)
3 done
clear
B)
10 done
clear
C)
5 done
clear
D)
6 done
clear
View Solution play_arrow
-
question_answer58)
Pauli's exclusion principle states that [MNR 1983; AMU 1984]
A)
Two electrons in the same atom can have the same energy done
clear
B)
Two electrons in the same atom cannot have the same spin done
clear
C)
The electrons tend to occupy different orbitals as far as possible done
clear
D)
Electrons tend to occupy lower energy orbitals preferentially done
clear
E)
None of the above done
clear
View Solution play_arrow
-
question_answer59)
For \[d\]electrons, the azimuthal quantum number is [MNR 1983; CPMT 1984]
A)
0 done
clear
B)
1 done
clear
C)
2 done
clear
D)
3 done
clear
View Solution play_arrow
-
question_answer60)
For \[p\]-orbital, the magnetic quantum number has value
A)
2 done
clear
B)
4, ? 4 done
clear
C)
? 1, 0, +1 done
clear
D)
0 done
clear
View Solution play_arrow
-
question_answer61)
For \[n=3\] energy level, the number of possible orbitals (all kinds) are [BHU 1981; CPMT 1985; MP PMT 1995]
A)
1 done
clear
B)
3 done
clear
C)
4 done
clear
D)
9 done
clear
View Solution play_arrow
-
question_answer62)
Which of the following ions is not having the configuration of neon
A)
\[{{F}^{-}}\] done
clear
B)
\[M{{g}^{+2}}\] done
clear
C)
\[N{{a}^{+}}\] done
clear
D)
\[C{{l}^{-}}\] done
clear
View Solution play_arrow
-
question_answer63)
Elements upto atomic number 103 have been synthesized and studied. If a newly discovered element is found to have an atomic number 106, its electronic configuration will be [AIIMS 1980]
A)
\[[Rn]5{{f}^{14}},6{{d}^{4}},7{{s}^{2}}\] done
clear
B)
\[[Rn]5{{f}^{14}},6{{d}^{1}},7{{s}^{2}}7{{p}^{3}}\] done
clear
C)
\[[Rn]5{{f}^{14}},6{{d}^{6}},7{{s}^{0}}\] done
clear
D)
\[[Rn]5{{f}^{14}},6{{d}^{5}},7{{s}^{1}}\] done
clear
View Solution play_arrow
-
question_answer64)
Ions which have the same electronic configuration are those of
A)
Lithium and sodium done
clear
B)
Sodium and potassium done
clear
C)
Potassium and calcium done
clear
D)
Oxygen and chlorine done
clear
View Solution play_arrow
-
question_answer65)
When the azimuthal quantum number has a value of \[l=0\], the shape of the orbital is [MP PET 1995]
A)
Rectangular done
clear
B)
Spherical done
clear
C)
Dumbbell done
clear
D)
Unsymmetrical done
clear
View Solution play_arrow
-
question_answer66)
The magnetic quantum number for valency electrons of sodium is [CPMT 1988; MH CET 1999]
A)
3 done
clear
B)
2 done
clear
C)
1 done
clear
D)
0 done
clear
View Solution play_arrow
-
question_answer67)
The electronic configuration of an element with atomic number 7 i.e. nitrogen atom is [CPMT 1982, 84, 87]
A)
\[1{{s}^{2}},2{{s}^{1}},2p_{x}^{3}\] done
clear
B)
\[1{{s}^{2}},2{{s}^{2}}2p_{x}^{2}2p_{y}^{1}\] done
clear
C)
\[1{{s}^{2}},2{{s}^{2}}2p_{x}^{1}2p_{y}^{1}2p_{z}^{1}\] done
clear
D)
\[1{{s}^{2}},2{{s}^{2}}2p_{x}^{1}2p_{y}^{2}\] done
clear
View Solution play_arrow
-
question_answer68)
In a multi-electron atom, which of the following orbitals described by the three quantum members will have the same energy in the absence of magnetic and electric fields [AIEEE 2005] (1) \[n=1,\,l=0,\,m=0\] (2) \[n=2,\,l=0,\,m=0\] (3) \[n=2,\,l=1,\,m=1\] (4) \[n=3,\,l=2,\,m=0\] (5) \[n=3,\,l=2,\,m=0\]
A)
(1) and (2) done
clear
B)
(2) and (3) done
clear
C)
(3) and (4) done
clear
D)
(4) and (5) done
clear
View Solution play_arrow
-
question_answer69)
Which of the following represents the electronic configuration of an element with atomic number 17 [AMU 1982]
A)
\[1{{s}^{2}},2{{s}^{2}}2{{p}^{6}},3{{s}^{1}}3{{p}^{6}}\] done
clear
B)
\[1{{s}^{2}},2{{s}^{2}}2{{p}^{6}},3{{s}^{2}}3{{p}^{4}},4{{s}^{1}}\] done
clear
C)
\[1{{s}^{2}},2{{s}^{2}}2{{p}^{6}},3{{s}^{2}}3{{p}^{5}}\] done
clear
D)
\[1{{s}^{2}},2{{s}^{2}}2{{p}^{6}},3{{s}^{1}}3{{p}^{4}},4{{s}^{2}}\] done
clear
View Solution play_arrow
-
question_answer70)
The shape of \[s\]-orbital is [NCERT 1978I]
A)
Pyramidal done
clear
B)
Spherical done
clear
C)
Tetrahedral done
clear
D)
Dumb-bell shaped done
clear
View Solution play_arrow
-
question_answer71)
When \[3d\] orbital is complete, the new electron will enter the [EAMCET 1980; MP PMT 1995]
A)
\[4p\]-orbital done
clear
B)
\[4f\]-orbital done
clear
C)
\[4s\]-orbital done
clear
D)
\[4d\]-orbital done
clear
View Solution play_arrow
-
question_answer72)
In a potassium atom, electronic energy levels are in the following order [EAMCET 1979; DPMT 1991]
A)
\[4s>3d\] done
clear
B)
\[4s>4p\] done
clear
C)
\[4s<3d\] done
clear
D)
\[4s<3p\] done
clear
View Solution play_arrow
-
question_answer73)
\[Fe\] (atomic number = 26) atom has the electronic arrangement [NCERT 1974; MNR 1980]
A)
2, 8, 8, 8 done
clear
B)
2, 8, 16 done
clear
C)
2, 8,14, 2 done
clear
D)
2, 8, 12, 4 done
clear
View Solution play_arrow
-
question_answer74)
\[C{{u}^{2+}}\] will have the following electronic configuration [MP PMT 1985]
A)
\[1{{s}^{2}},2{{s}^{2}}2{{p}^{6}},3{{s}^{2}}3{{p}^{6}}3{{d}^{10}}\] done
clear
B)
\[1{{s}^{2}},2{{s}^{2}}2{{p}^{6}},3{{s}^{2}}3{{p}^{6}}3{{d}^{9}},4{{s}^{1}}\] done
clear
C)
\[1{{s}^{2}},2{{s}^{2}}2{{p}^{6}},3{{s}^{2}}3{{p}^{6}}3{{d}^{9}}\] done
clear
D)
\[1{{s}^{2}},2{{s}^{2}}2{{p}^{6}},3{{s}^{2}}3{{p}^{6}}3{{d}^{10}},4{{s}^{1}}\] done
clear
View Solution play_arrow
-
question_answer75)
Which one is the electronic configuration of \[F{{e}^{+2}}\] [MADT Bihar 1982; AIIMS 1989]
A)
\[1{{s}^{2}},2{{s}^{2}}2{{p}^{6}},3{{s}^{2}}3{{p}^{6}}3{{d}^{6}}\] done
clear
B)
\[1{{s}^{2}},2{{s}^{2}}2{{p}^{6}},3{{s}^{2}}3{{p}^{6}}3{{d}^{4}},4{{s}^{2}}\] done
clear
C)
\[1{{s}^{2}},2{{s}^{2}}2{{p}^{6}},3{{s}^{2}}3{{p}^{6}}3{{d}^{5}},4{{s}^{1}}\] done
clear
D)
None of these done
clear
View Solution play_arrow
-
question_answer76)
How many electrons can be fit into the orbitals that comprise the \[{{3}^{rd}}\] quantum shell \[n=3\] [MP PMT 1986, 87; Orissa JEE 1997]
A)
2 done
clear
B)
8 done
clear
C)
18 done
clear
D)
32 done
clear
View Solution play_arrow
-
question_answer77)
Which element is represented by the following electronic configuration
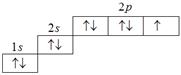
[MP PMT 1987]
A)
Nitrogen done
clear
B)
Oxygen done
clear
C)
Fluorine done
clear
D)
Neon done
clear
View Solution play_arrow
-
question_answer78)
If the value of azimuthal quantum number is 3, the possible values of magnetic quantum number would be [MP PMT 1987; RPMT 1999; AFMC 2002; KCET 2002]
A)
0, 1, 2, 3 done
clear
B)
0, ? 1, ? 2, ? 3 done
clear
C)
0, \[\pm \]1, \[\pm \]2, \[\pm \] 3 done
clear
D)
\[\pm \]1, \[\pm \]2, \[\pm \]3 done
clear
View Solution play_arrow
-
question_answer79)
Krypton \[{{(}_{36}}Kr)\] has the electronic configuration \[{{(}_{18}}Ar)\] \[4{{s}^{2}},3{{d}^{10}},4{{p}^{6}}\]. The \[{{37}^{th}}\] electron will go into which one of the following sub-levels [CBSE PMT 1989; CPMT 1989; EAMCET 1991]
A)
\[4f\] done
clear
B)
\[4d\] done
clear
C)
\[3p\] done
clear
D)
\[5s\] done
clear
View Solution play_arrow
-
question_answer80)
If an electron has spin quantum number of \[+\frac{1}{2}\] and a magnetic quantum number of \[-1\], it cannot be presented in an [CBSE PMT 1989; UPSEAT 2001]
A)
\[d\]-orbital done
clear
B)
\[f\]-orbital done
clear
C)
\[p\]-orbital done
clear
D)
\[s\]-orbital done
clear
View Solution play_arrow
-
question_answer81)
The azimuthal quantum number is related to [BHU 1987, 95]
A)
Size done
clear
B)
Shape done
clear
C)
Orientation done
clear
D)
Spin done
clear
View Solution play_arrow
-
question_answer82)
The total number of electrons that can be accommodated in all the orbitals having principal quantum number 2 and azimuthal quantum number 1 is [CPMT 1971, 89, 91]
A)
2 done
clear
B)
4 done
clear
C)
6 done
clear
D)
8 done
clear
View Solution play_arrow
-
question_answer83)
Electronic configuration of \[C\] is [CPMT 1975]
A)
\[1{{s}^{2}},2{{s}^{2}}2{{p}^{2}}\] done
clear
B)
\[1{{s}^{2}},2{{s}^{2}}2{{p}^{3}}\] done
clear
C)
\[1{{s}^{2}},2{{s}^{2}}\] done
clear
D)
\[1{{s}^{2}},2{{s}^{2}}2{{p}^{6}}\] done
clear
View Solution play_arrow
-
question_answer84)
There is no difference between a \[2p\] and a \[3p\]orbital regarding [BHU 1981]
A)
Shape done
clear
B)
Size done
clear
C)
Energy done
clear
D)
Value of \[n\] done
clear
View Solution play_arrow
-
question_answer85)
The electronic configuration of chromium is [MP PMT 1993; MP PET 1995; BHU 2001; BCECE 2005]
A)
\[[Ne]3{{s}^{2}}3{{p}^{6}}3{{d}^{4}},4{{s}^{2}}\] done
clear
B)
\[[Ne]3{{s}^{2}}3{{p}^{6}}3{{d}^{5}},4{{s}^{1}}\] done
clear
C)
\[[Ne]3{{s}^{2}}3{{p}^{6}},4{{s}^{2}}4{{p}^{4}}\] done
clear
D)
\[[Ne]3{{s}^{2}}3{{p}^{6}}3{{d}^{1}},4{{s}^{2}}4{{p}^{3}}\] done
clear
View Solution play_arrow
-
question_answer86)
The shape of \[p\]-orbital is [MP PMT 1993]
A)
Elliptical done
clear
B)
Spherical done
clear
C)
Dumb-bell done
clear
D)
Complex geometrical done
clear
View Solution play_arrow
-
question_answer87)
The electronic configuration (outermost) of \[M{{n}^{+2}}\] ion (atomic number of \[Mn=25\]) in its ground state is [MP PET 1993]
A)
\[3{{d}^{5}},4{{s}^{0}}\] done
clear
B)
\[3{{d}^{4}},4{{s}^{1}}\] done
clear
C)
\[3{{d}^{3}},4{{s}^{2}}\] done
clear
D)
\[3{{d}^{2}},4{{s}^{2}}4{{p}^{2}}\] done
clear
View Solution play_arrow
-
question_answer88)
The principal quantum number represents [CPMT 1991]
A)
Shape of an orbital done
clear
B)
Distance of electron from nucleus done
clear
C)
Number of electrons in an orbit done
clear
D)
Number of orbitals in an orbit done
clear
View Solution play_arrow
-
question_answer89)
When the azimuthal quantum number has a value of \[l=1\], the shape of the orbital is [MP PET 1993]
A)
Unsymmetrical done
clear
B)
Spherically symmetrical done
clear
C)
Dumb-bell done
clear
D)
Complicated done
clear
View Solution play_arrow
-
question_answer90)
How many electrons can be accommodated in a sub-shell for which \[n=3,\,l=1\] [CBSE PMT 1990]
A)
8 done
clear
B)
6 done
clear
C)
18 done
clear
D)
32 done
clear
View Solution play_arrow
-
question_answer91)
For azimuthal quantum number \[l=3\], the maximum number of electrons will be [CBSE PMT 1991; EAMCET 1991; RPMT 2002; CBSE PMT 2002]
A)
2 done
clear
B)
6 done
clear
C)
0 done
clear
D)
14 done
clear
View Solution play_arrow
-
question_answer92)
An ion has 18 electrons in the outermost shell, it is [CBSE PMT 1990]
A)
\[C{{u}^{+}}\] done
clear
B)
\[T{{h}^{4+}}\] done
clear
C)
\[C{{s}^{+}}\] done
clear
D)
\[{{K}^{+}}\] done
clear
View Solution play_arrow
-
question_answer93)
The order of filling of electrons in the orbitals of an atom will be [CBSE PMT 1991]
A)
\[3d,\,4s,\,4p,\,4d,\,5s\] done
clear
B)
\[4s,\,3d,\,4p,\,5s,\,4d\] done
clear
C)
\[5s,\,4p,\,3d,\,4d,\,5s\] done
clear
D)
\[3d,\,4p,\,4s,\,4d,\,5s\] done
clear
View Solution play_arrow
-
question_answer94)
The quantum number which may be designated by \[s,\,p,\,d\] and \[f\] instead of number is BHU 1980]
A)
\[n\] done
clear
B)
\[l\] done
clear
C)
\[{{m}_{l}}\] done
clear
D)
\[{{m}_{s}}\] done
clear
View Solution play_arrow
-
question_answer95)
Which of the following represents the correct sets of the four quantum numbers of a \[4d\] electron [MNR 1992; UPSEAT 2001; J&K CET 2005]
A)
\[4,\,3,\,2,\,\frac{1}{2}\] done
clear
B)
4, 2, 1, 0 done
clear
C)
\[4,\,3,\,-2,\,+\frac{1}{2}\] done
clear
D)
\[4,\,2,\,1,\,-\frac{1}{2}\] done
clear
View Solution play_arrow
-
question_answer96)
Which of the following statements is not correct for an electron that has the quantum numbers \[n=4\] and \[m=2\] [MNR 1993]
A)
The electron may have the quantum number \[s=+\frac{1}{2}\] done
clear
B)
The electron may have the quantum number \[l=2\] done
clear
C)
The electron may have the quantum number \[l=3\] done
clear
D)
The electron may have the quantum number \[l=0,\,1,\,2,\,3\] done
clear
View Solution play_arrow
-
question_answer97)
The set of quantum numbers not applicable for an electron in an atom is [MNR 1994]
A)
\[n=1,\,l=1,\,{{m}_{l}}=1,\,{{m}_{s}}=+1/2\] done
clear
B)
\[n=1,\,l=0,\,{{m}_{l}}=0,\,{{m}_{s}}=+1/2\] done
clear
C)
\[n=1,\,l=0,\,{{m}_{l}}=0,\,{{m}_{s}}=-1/2\] done
clear
D)
\[n=2,\,l=0,\,{{m}_{l}}=0,\,{{m}_{s}}=+1/2\] done
clear
View Solution play_arrow
-
question_answer98)
Correct configuration of \[F{{e}^{+3}}\] [26] is [CPMT 1994; BHU 1995; KCET 1992]
A)
\[1{{s}^{2}},2{{s}^{2}}2{{p}^{6}},3{{s}^{2}}3{{p}^{6}}3{{d}^{5}}\] done
clear
B)
\[1{{s}^{2}},2{{s}^{2}}s{{p}^{6}},3{{s}^{2}}3{{p}^{6}}3{{d}^{3}},4{{s}^{2}}\] done
clear
C)
\[1{{s}^{2}},2{{s}^{2}}2{{p}^{6}},3{{s}^{2}}3{{p}^{6}}3{{d}^{6}},4{{s}^{2}}\] done
clear
D)
\[1{{s}^{2}},2{{s}^{2}}2{{p}^{6}},3{{s}^{2}}3{{p}^{6}}3{{d}^{5}},4{{s}^{1}}\] done
clear
View Solution play_arrow
-
question_answer99)
Azimuthal quantum number for last electron of \[Na\] atom is [BHU 1995]
A)
1 done
clear
B)
2 done
clear
C)
3 done
clear
D)
0 done
clear
View Solution play_arrow
-
question_answer100)
A \[3p\]orbital has [IIT 1995]
A)
Two spherical nodes done
clear
B)
Two non-spherical nodes done
clear
C)
One spherical and one non-spherical nodes done
clear
D)
One spherical and two non-spherical nodes done
clear
View Solution play_arrow
-
question_answer101)
All electrons on the \[4p\] sub-shell must be characterized by the quantum number(s) [MP PET 1996]
A)
\[n=4,\,m=0,\,s=\pm \frac{1}{2}\] done
clear
B)
\[l=1\] done
clear
C)
\[l=0,\,s=\pm \frac{1}{2}\] done
clear
D)
\[s=\pm \frac{1}{2}\] done
clear
View Solution play_arrow
-
question_answer102)
The electronic configuration of the element of atomic number 27 is
A)
\[1{{s}^{2}},\ 2{{s}^{2}}2{{p}^{6}},\ 3{{s}^{2}}3{{p}^{6}},\ 4s\ (\uparrow \downarrow )\ 4p\ (\uparrow \downarrow )(\uparrow \downarrow )(\uparrow \downarrow )\ 5s\ (\uparrow )\] done
clear
B)
\[1{{s}^{2}},\ 2{{s}^{2}}2{{p}^{6}},\ 3{{s}^{2}}3{{p}^{6}}3d\ (\uparrow \downarrow )(\uparrow \downarrow )(\uparrow \downarrow ),\ 4s\ (\uparrow \downarrow )\ 4p\ (\uparrow )\] done
clear
C)
\[1{{s}^{2}},\ 2{{s}^{2}}2{{p}^{6}},\ 3{{s}^{2}}3{{p}^{6}},\ 3d\ (\uparrow \downarrow )(\uparrow \downarrow )(\uparrow \downarrow )(\uparrow \downarrow ),\ 4s\ (\uparrow )\] done
clear
D)
\[1{{s}^{2}},\ 2{{s}^{2}}2{{p}^{6}},\ 3{{s}^{2}}3{{p}^{6}},\ 3d\ (\uparrow \downarrow )(\uparrow \downarrow )(\uparrow )(\uparrow )(\uparrow )\ 4s\ (\uparrow \downarrow )\] done
clear
View Solution play_arrow
-
question_answer103)
When the value of the principal quantum number \[n\] is 3, the permitted values of the azimuthal quantum numbers \[l\] and the magnetic quantum numbers \[m\] , are
A)
\[l\]-\[\begin{matrix} 0 \\ 1 \\ 2 \\ \end{matrix}\] \[m\]-\[\begin{matrix} 0 \\ +1,\,0,\,-1 \\ +2,+1,\,\,0,\,-1,-2 \\ \end{matrix}\] done
clear
B)
\[l\]-\[\begin{matrix} 1 \\ 2 \\ 3 \\ \end{matrix}\] \[m\]-\[\begin{matrix} 1 \\ +2,\,1,\,-1 \\ +3,+2,\,\,1,\,-2,-3 \\ \end{matrix}\] done
clear
C)
\[l\]-\[\begin{matrix} 0 \\ 1 \\ 2 \\ \end{matrix}\] \[m\]-\[\begin{matrix} 0 \\ 1,\,\,2,\,3 \\ +3,\,+2,\,1,\,-2,-3 \\ \end{matrix}\] done
clear
D)
\[l\]-\[\begin{matrix} 1 \\ 2 \\ 3 \\ \end{matrix}\] \[m\]-\[\begin{matrix} 0,\,\,1 \\ 0,\,\,1,\,\,2 \\ 0,\,\,1,\,\,2,\,\,3 \\ \end{matrix}\] done
clear
View Solution play_arrow
-
question_answer104)
The number of possible spatial orientations of an electron in an atom is given by its
A)
Spin quantum number done
clear
B)
Spin angular momentum done
clear
C)
Magnetic quantum number done
clear
D)
Orbital angular momentum done
clear
View Solution play_arrow
-
question_answer105)
Which of the following sets of orbitals may degenerate
A)
\[2s,\,2{{p}_{x}},\,2{{p}_{y}}\] done
clear
B)
\[3s,\,3{{p}_{x}},3{{d}_{xy}}\] done
clear
C)
\[1s,\,2s,\,3s\] done
clear
D)
\[2{{p}_{x}},\,2{{p}_{y}},\,2{{p}_{z}}\] done
clear
View Solution play_arrow
-
question_answer106)
The set of quantum numbers \[n=3,\,l=0,\,m=0,\,s=-1/2\] belongs to the element
A)
\[Mg\] done
clear
B)
\[Na\] done
clear
C)
\[Ne\] done
clear
D)
\[F\] done
clear
View Solution play_arrow
-
question_answer107)
An electron has principal quantum number 3. The number of its (i) sub-shells and (ii) orbitals would be respectively [MP PET 1997]
A)
3 and 5 done
clear
B)
3 and 7 done
clear
C)
3 and 9 done
clear
D)
2 and 5 done
clear
View Solution play_arrow
-
question_answer108)
What is the electronic configuration of \[C{{u}^{2+}}(Z=29)\] of least position [MP PET/PMT 1998; MP PET 2001]
A)
\[[Ar]\,4{{s}^{1}}3{{d}^{8}}\] done
clear
B)
\[[Ar]\,4{{s}^{2}}3{{d}^{10}}4{{p}^{1}}\] done
clear
C)
\[[Ar]\,4{{s}^{1}}3{{d}^{10}}\] done
clear
D)
\[[Ar]\,3{{d}^{9}}\] done
clear
View Solution play_arrow
-
question_answer109)
The correct electronic configuration of \[Ti(Z=22)\] atom is [MP PMT 1999]
A)
\[1{{s}^{2}}2{{s}^{2}}2{{p}^{6}}3{{s}^{2}}3{{p}^{6}}4{{s}^{2}}3{{d}^{2}}\] done
clear
B)
\[1{{s}^{2}}2{{s}^{2}}2{{p}^{6}}3{{s}^{2}}3{{p}^{6}}3{{d}^{4}}\] done
clear
C)
\[1{{s}^{2}}2{{s}^{2}}2{{p}^{6}}3{{s}^{2}}3{{p}^{6}}3{{d}^{4}}\] done
clear
D)
\[1{{s}^{2}}2{{s}^{2}}2{{p}^{6}}3{{s}^{2}}3{{p}^{6}}4{{s}^{1}}3{{d}^{3}}\] done
clear
View Solution play_arrow
-
question_answer110)
Which of the following configuration is correct for iron [CBSE PMT 1999]
A)
\[1{{s}^{2}}2{{s}^{2}}2{{p}^{6}}3{{s}^{2}}3{{p}^{6}}3{{d}^{5}}\] done
clear
B)
\[1{{s}^{2}}2{{s}^{2}}2{{p}^{6}}3{{s}^{2}}3{{p}^{6}}4{{s}^{2}}3{{d}^{5}}\] done
clear
C)
\[1{{s}^{2}}2{{s}^{2}}2{{p}^{6}}3{{s}^{2}}3{{p}^{6}}4{{s}^{2}}3{{d}^{7}}\] done
clear
D)
\[1{{s}^{2}}2{{s}^{2}}2{{p}^{6}}3{{s}^{2}}3{{p}^{6}}4{{s}^{2}}3{{d}^{6}}\] done
clear
View Solution play_arrow
-
question_answer111)
Which of the following set of quantum numbers belong to highest energy [CPMT 1999]
A)
\[n=4,\,l=0,\,m=0,\,s=+\frac{1}{2}\] done
clear
B)
\[n=3,\,l=0,\,m=0,\,s=+\frac{1}{2}\] done
clear
C)
\[n=3,\,l=1,\,m=1,\,s=+\frac{1}{2}\] done
clear
D)
\[n=3,\,l=2,\,m=1,\,s=+\frac{1}{2}\] done
clear
View Solution play_arrow
-
question_answer112)
Which quantum number will determine the shape of the subshell [CPMT 1999; Pb. PMT 1998]
A)
Principal quantum number done
clear
B)
Azimuthal quantum number done
clear
C)
Magnetic quantum number done
clear
D)
Spin quantum number done
clear
View Solution play_arrow
-
question_answer113)
For the \[n=2\] energy level, how many orbitals of all kinds are possible [Bihar CEE 1995]
A)
2 done
clear
B)
3 done
clear
C)
4 done
clear
D)
5 done
clear
View Solution play_arrow
-
question_answer114)
Which one is in the ground state [DPMT 1996]
A)
B)
C)
D)
View Solution play_arrow
-
question_answer115)
When the principal quantum number \[(n=3)\], the possible values of azimuthal quantum number (\[l\]) is [Bihar MEE 1996; KCET 2000]
A)
0, 1, 2, 3 done
clear
B)
0, 1, 2 done
clear
C)
? 2, ? 1, 0, 1, 2 done
clear
D)
1, 2, 3 done
clear
E)
0, 1 done
clear
View Solution play_arrow
-
question_answer116)
Which statement is not correct for \[n=5\], \[m=3\] [CPMT 1996]
A)
\[l=4\] done
clear
B)
\[l=0,\,1,\,3;\,s=+\frac{1}{2}\] done
clear
C)
\[l=3\] done
clear
D)
All are correct done
clear
View Solution play_arrow
-
question_answer117)
\[1{{s}^{2}}2{{s}^{2}}2{{p}^{6}}3{{s}^{1}}\] shows configuration of [CPMT 1996]
A)
\[A{{l}^{3+}}\] in ground state done
clear
B)
\[Ne\] in excited state done
clear
C)
\[M{{g}^{+}}\] in excited state done
clear
D)
None of these done
clear
View Solution play_arrow
-
question_answer118)
Five valence electrons of \[{{p}^{15}}\] are labelled as

If the spin quantum of \[B\] and \[Z\] is \[+\frac{1}{2}\], the group of electrons with three of the quantum number same are [JIPMER 1997]
A)
\[AB,\,XYZ,\,BY\] done
clear
B)
\[AB\] done
clear
C)
\[XYZ,\,AZ\] done
clear
D)
\[AB,\,XYZ\] done
clear
View Solution play_arrow
-
question_answer119)
Electronic configuration of \[S{{c}^{21}}\] is [BHU 1997]
A)
\[1{{s}^{2}}2{{s}^{2}}2{{p}^{6}}3{{s}^{2}}3{{p}^{6}}4{{s}^{2}}3{{d}^{1}}\] done
clear
B)
\[1{{s}^{2}}2{{s}^{2}}2{{p}^{6}}3{{s}^{2}}3{{p}^{6}}4{{s}^{1}}3{{d}^{2}}\] done
clear
C)
\[1{{s}^{2}}2{{s}^{2}}2{{p}^{6}}3{{s}^{2}}3{{p}^{6}}4{{s}^{0}}3{{d}^{3}}\] done
clear
D)
\[1{{s}^{2}}2{{s}^{2}}2{{p}^{6}}3{{s}^{2}}3{{p}^{2}}4{{s}^{2}}3{{d}^{2}}\] done
clear
View Solution play_arrow
-
question_answer120)
If \[n+l=6\], then total possible number of subshells would be [RPMT 1997]
A)
3 done
clear
B)
4 done
clear
C)
2 done
clear
D)
5 done
clear
View Solution play_arrow
-
question_answer121)
An electron having the quantum numbers \[n=4,\,l=3,\,m=0\], \[s=-\frac{1}{2}\] would be in the orbital [Orissa JEE 1997]
A)
\[3s\] done
clear
B)
\[3p\] done
clear
C)
\[4d\] done
clear
D)
\[4f\] done
clear
View Solution play_arrow
-
question_answer122)
Which of the following sets of quantum numbers is not allowed [Orissa JEE 1997]
A)
\[n=1,\,l=0,\,m=0,\,s=+\frac{1}{2}\] done
clear
B)
\[n=1,\,l=1,\,m=0,\,s=-\frac{1}{2}\] done
clear
C)
\[n=2,\,l=1,\,m=1,\,s=+\frac{1}{2}\] done
clear
D)
\[n=2,\,l=1,\,m=0,\,s=-\frac{1}{2}\] done
clear
View Solution play_arrow
-
question_answer123)
For which of the following sets of four quantum numbers, an electron will have the highest energy [CBSE PMT 1994]
A)
\[n\]-3 \[l\]-2 \[m\]-1 \[s\]- +1/2 done
clear
B)
\[n\]-4 \[l\]-2 \[m\]-1 \[s\]- +1/2 done
clear
C)
\[n\]-4 \[l\]-1 \[m\]- 0 \[s\]- -1/2 done
clear
D)
\[n\]-5 \[l\]-0 \[m\]-0 \[s\]- -1/2 done
clear
View Solution play_arrow
-
question_answer124)
The electronic configuration of gadolinium (atomic no. 64) is [CBSE PMT 1997]
A)
\[[Xe]4{{s}^{8}}5{{d}^{9}}6{{s}^{2}}\] done
clear
B)
\[[Xe]4{{s}^{7}}5{{d}^{1}}6{{s}^{2}}\] done
clear
C)
\[[Xe]4{{s}^{3}}5{{d}^{5}}6{{s}^{2}}\] done
clear
D)
\[[Xe]4{{f}^{6}}5{{d}^{2}}6{{s}^{2}}\] done
clear
View Solution play_arrow
-
question_answer125)
An \[{{e}^{-}}\] has magnetic quantum number as \[-3\], what is its principal quantum number [BHU 1998]
A)
1 done
clear
B)
2 done
clear
C)
3 done
clear
D)
4 done
clear
View Solution play_arrow
-
question_answer126)
The number of quantum numbers required to describe an electron in an atom completely is [CET Pune 1998]
A)
1 done
clear
B)
2 done
clear
C)
3 done
clear
D)
4 done
clear
View Solution play_arrow
-
question_answer127)
The electronic configuration \[1{{s}^{2}}2{{s}^{2}}2p_{x}^{1}2p_{y}^{1}2p_{z}^{1}\] [AFMC 1997; Pb. PMT 1999; CBSE PMT 2001; AIIMS 2001]
A)
Oxygen done
clear
B)
Nitrogen done
clear
C)
Hydrogen done
clear
D)
Fluorine done
clear
View Solution play_arrow
-
question_answer128)
Which one of the following set of quantum numbers is not possible for \[4p\] electron [EAMCET 1998]
A)
\[n=4,\,l=1,\,m=-1,\,s=+\frac{1}{2}\] done
clear
B)
\[n=4,\,l=1,\,m=0,\,s=+\frac{1}{2}\] done
clear
C)
\[n=4,\,l=1,\,m=2,\,s=+\frac{1}{2}\] done
clear
D)
\[n=4,\,l=1,\,m=-1,\,s=+\frac{1}{2}\] done
clear
View Solution play_arrow
-
question_answer129)
Which of the following orbital is not possible [RPMT 1999]
A)
3 f done
clear
B)
4 f done
clear
C)
5 f done
clear
D)
6 f done
clear
View Solution play_arrow
-
question_answer130)
Which set of quantum numbers for an electron of an atom is not possible [RPMT; DCE 1999]
A)
\[n=1,\,l=0,\,m=0,\,s=+1/2\] done
clear
B)
\[n=1,\,l=1,\,m=1,\,s=+1/2\] done
clear
C)
\[n=1,l=0,\,m=0,\,s=-1/2\] done
clear
D)
\[n=2,\,l=1,\,m=-1,\,s=+1/2\] done
clear
View Solution play_arrow
-
question_answer131)
Electronic configuration of ferric ion is [RPET 2000]
A)
\[[Ar]\,3{{d}^{5}}\] done
clear
B)
\[[Ar]\,3{{d}^{7}}\] done
clear
C)
\[[Ar]\,3{{d}^{3}}\] done
clear
D)
\[[Ar]\,3{{d}^{8}}\] done
clear
View Solution play_arrow
-
question_answer132)
What is the maximum number of electrons which can be accommodated in an atom in which the highest principal quantum number value is 4 [MP PMT 2000]
A)
10 done
clear
B)
18 done
clear
C)
32 done
clear
D)
54 done
clear
View Solution play_arrow
-
question_answer133)
Which of the following electronic configurations is not possible [CPMT 2000]
A)
\[1{{s}^{2}}\,\,2{{s}^{2}}\] done
clear
B)
\[1{{s}^{2}}2{{s}^{2}}2{{p}^{6}}\] done
clear
C)
\[3{{d}^{10}}4{{s}^{2}}4{{p}^{2}}\] done
clear
D)
\[1{{s}^{2}}2{{s}^{2}}\,2{{p}^{2}}3{{s}^{1}}\] done
clear
View Solution play_arrow
-
question_answer134)
The electronic configuration of an element is \[1{{s}^{2}}2{{s}^{2}}2{{p}^{6}}3{{s}^{2}}3{{p}^{6}}3{{d}^{5}}4{{s}^{1}}\]. This represents its [IIT Screening 2000]
A)
Excited state done
clear
B)
Ground state done
clear
C)
Cationic form done
clear
D)
Anionic form done
clear
View Solution play_arrow
-
question_answer135)
Which of the following set of quantum numbers is possible [AIIMS 2001]
A)
\[n=3;\,\,l=2;\,\,m=2\] and \[s=+\frac{1}{2}\] done
clear
B)
\[n=3;\,\,l=4;\,\,m=0\] and \[s=-\frac{1}{2}\] done
clear
C)
\[n=4;\,l=0;\,\,m=2\] and \[s=+\frac{1}{2}\] done
clear
D)
\[n=4;\,\,l=4;\,\,m=3\] and \[s=+\frac{1}{2}\] done
clear
View Solution play_arrow
-
question_answer136)
Which of the following set of quantum number is not valid [AIIMS 2001]
A)
\[n=1,\,\,l=2\] done
clear
B)
\[3=2,\,\,m=1\] done
clear
C)
\[m=3,\,\,l=0\] done
clear
D)
\[3=4,\,\,l=2\] done
clear
View Solution play_arrow
-
question_answer137)
Which one pair of atoms or ions will have same configuration [JIPMER 2001]
A)
\[{{F}^{+}}\] and \[Ne\] done
clear
B)
\[L{{i}^{+}}\] and \[H{{e}^{-}}\] done
clear
C)
\[C{{l}^{-}}\] and \[Ar\] done
clear
D)
\[Na\] and K done
clear
View Solution play_arrow
-
question_answer138)
Which of the following sets of quantum number is not possible [MP PET 2001]
A)
\[n=3;\,l=+2;\,m=0;\,s=+\frac{1}{2}\] done
clear
B)
\[n=3;\,l=0;\,m=0;\,s=-\frac{1}{2}\] done
clear
C)
\[n=3;\,l=0;\,m=-1;\,s=+\frac{1}{2}\] done
clear
D)
\[n=3;\,l=1;\,m=0;\,s=-\frac{1}{2}\] done
clear
View Solution play_arrow
-
question_answer139)
Which of the following set of quantum numbers is correct for the 19th electron of chromium [DCE 2001]
A)
n-3 l-0 m-0 s- 1/2 done
clear
B)
n-3 l-2 m- ? 2 s- 1/2 done
clear
C)
n-4 l-0 m- 0 s- ½ done
clear
D)
n-4 l-1 m- ?1 s- ½ done
clear
View Solution play_arrow
-
question_answer140)
When the value of azimuthal quantum number is 3, magnetic quantum number can have values [DPMT 2001]
A)
+ 1, 0, ? 1 done
clear
B)
+ 2, + 1, 0, ? 1, ? 2 done
clear
C)
? 3, ? 2, ? 1, ? 0, + 1, + 2, + 3 done
clear
D)
+ 1, ? 1 done
clear
View Solution play_arrow
-
question_answer141)
The quantum numbers \[n=2,\,\,l=1\] represent [AFMC 2002]
A)
1s orbital done
clear
B)
2s orbital done
clear
C)
2p orbital done
clear
D)
3d orbital done
clear
View Solution play_arrow
-
question_answer142)
The magnetic quantum number of valence electron of sodium (Na) is [RPMT 2002]
A)
3 done
clear
B)
2 done
clear
C)
1 done
clear
D)
0 done
clear
View Solution play_arrow
-
question_answer143)
Azimuthal quantum number defines [AIIMS 2002]
A)
e/m ratio of electron done
clear
B)
Spin of electron done
clear
C)
Angular momentum of electron done
clear
D)
Magnetic momentum of electron done
clear
View Solution play_arrow
-
question_answer144)
Quantum numbers of an atom can be defined on the basis of [AIIMS 2002]
A)
Hund?s rule done
clear
B)
Aufbau?s principle done
clear
C)
Pauli?s exclusion principle done
clear
D)
Heisenberg?s uncertainty principle done
clear
View Solution play_arrow
-
question_answer145)
Which of the following has maximum energy [AIIMS 2002]
A)
B)
C)
D)
View Solution play_arrow
-
question_answer146)
The total magnetic quantum numbers for d-orbital is given by [Orissa JEE 2002]
A)
2 done
clear
B)
0, ± 1, ± 2 done
clear
C)
0, 1, 2 done
clear
D)
5 done
clear
View Solution play_arrow
-
question_answer147)
The outer electronic structure \[3{{s}^{2}}3{{p}^{5}}\] is possessed by [Pb. PMT 2002; Pb. CET 2001]
A)
Cl done
clear
B)
O done
clear
C)
Ar done
clear
D)
Br done
clear
View Solution play_arrow
-
question_answer148)
Which of the following set of quantum number is not possible [Pb. PMT 2002]
A)
n-3 l-2 \[{{m}_{1}}\]-1 \[{{m}_{2}}\]- +1/2 done
clear
B)
n ? 3 l ? 2 \[{{m}_{1}}\]-1 \[{{m}_{2}}\]- ? 1/2 done
clear
C)
n ? 3 l ? 2 \[{{m}_{1}}\]-1 \[{{m}_{2}}\]- 0 done
clear
D)
n ? 5 l ? 2 \[{{m}_{1}}\]- ? 1 \[{{m}_{2}}\]- + 1/2 done
clear
View Solution play_arrow
-
question_answer149)
The configuration \[1{{s}^{2}},\,2{{s}^{2}}2{{p}^{5}},\,3{{s}^{1}}\] shows [Pb. PMT 2002]
A)
Excited state of \[O_{2}^{-}\] done
clear
B)
Excited state of neon done
clear
C)
Excited state of fluorine done
clear
D)
Ground state of fluorine atom done
clear
View Solution play_arrow
-
question_answer150)
The quantum number ?m? of a free gaseous atom is associated with [AIIMS 2003]
A)
The effective volume of the orbital done
clear
B)
The shape of the orbital done
clear
C)
The spatial orientation of the orbital done
clear
D)
The energy of the orbital in the absence of a magnetic field done
clear
View Solution play_arrow
-
question_answer151)
Correct statement is [BHU 2003]
A)
\[K=4{{s}^{1}},\,\,Cr=3{{d}^{4}}4{{s}^{2}},\,\,Cu=3{{d}^{10}}4{{s}^{2}}\] done
clear
B)
\[K=4{{s}^{2}},\,\,Cr=3{{d}^{4}}4{{s}^{2}},\,\,Cu=3{{d}^{10}}4{{s}^{2}}\] done
clear
C)
\[K=4{{s}^{2}},\,\,Cr=3{{d}^{5}}4{{s}^{1}},\,\,Cu=3{{d}^{10}}4{{s}^{2}}\] done
clear
D)
\[K=4{{s}^{1}},\,\,Cr=3{{d}^{5}}4{{s}^{1}},\,\,Cu=3{{d}^{10}}4{{s}^{1}}\] done
clear
View Solution play_arrow
-
question_answer152)
Number of orbitats in h sub-shell is [BHU 2003]
A)
11 done
clear
B)
15 done
clear
C)
17 done
clear
D)
19 done
clear
View Solution play_arrow
-
question_answer153)
Electronic configuration \[1{{s}^{2}},\,2{{s}^{2}}2{{p}^{6}},\,3{{s}^{2}}3{{p}^{6}}3{{d}^{5}},\,4{{s}^{1}}\] represents [CPMT 2003]
A)
Ground state done
clear
B)
Excited state done
clear
C)
Anionic state done
clear
D)
All of these done
clear
View Solution play_arrow
-
question_answer154)
Which of the following sets is possible for quantum numbers [RPET 2003]
A)
\[n=4,\,l=3,\,m=-2,\,s=0\] done
clear
B)
\[n=4,\,l=4,\,m=+2,\,s=-\frac{1}{2}\] done
clear
C)
\[n=4,\,l=4,\,m=-2,\,s=+\frac{1}{2}\] done
clear
D)
\[n=4,\,l=3,\,m=-2,\,s=+\frac{1}{2}\] done
clear
View Solution play_arrow
-
question_answer155)
For principle quantum number \[n=4\] the total number of orbitals having \[l=3\] [AIIMS 2004]
A)
3 done
clear
B)
7 done
clear
C)
5 done
clear
D)
9 done
clear
View Solution play_arrow
-
question_answer156)
The number of \[2p\] electrons having spin quantum number \[s=-1/2\] are [KCET 2004]
A)
6 done
clear
B)
0 done
clear
C)
2 done
clear
D)
3 done
clear
View Solution play_arrow
-
question_answer157)
Which of the following sets of quantum numbers is correct for an electron in \[4f\] orbital [AIEEE 2004]
A)
\[n=4,\,l=3,\,m=+1,\,s=+\frac{1}{2}\] done
clear
B)
\[n=4,\,l=4,\,m=-4,\,s=-\frac{1}{2}\] done
clear
C)
\[n=4,\,l=3,\,m=+4,\,s=+\frac{1}{2}\] done
clear
D)
\[n=3,\,l=2,\,m=-2,\,s=+\frac{1}{2}\] done
clear
View Solution play_arrow
-
question_answer158)
Consider the ground state of \[(Z=24)\]. The numbers of electrons with the azimuthal quantum numbers, \[l=1\] and 2 are, respectively [AIEEE 2004]
A)
16 and 4 done
clear
B)
12 and 5 done
clear
C)
12 and 4 done
clear
D)
16 and 5 done
clear
View Solution play_arrow
-
question_answer159)
The four quantum numbers of the valence electron of potassium are [DPMT 2004]
A)
4, 1, 0 and \[\frac{1}{2}\] done
clear
B)
4, 0, 1 and \[\frac{1}{2}\] done
clear
C)
4, 0, 0 and \[+\frac{1}{2}\] done
clear
D)
4, 1, 1 and \[\frac{1}{2}\] done
clear
View Solution play_arrow
-
question_answer160)
Which of the following electronic configuration is not possible according to Hund?s rule [Kerala PMT 2004]
A)
\[1{{s}^{2}}2{{s}^{2}}\] done
clear
B)
\[1{{s}^{2}}2{{s}^{1}}\] done
clear
C)
\[1{{s}^{2}}2{{s}^{2}}2p_{x}^{1}2p_{y}^{1}2p_{x}^{1}\] done
clear
D)
\[1{{s}^{2}}2{{s}^{2}}2p_{x}^{2}\] done
clear
E)
\[1{{s}^{2}}2{{s}^{2}}2p_{x}^{2}2p_{y}^{1}2p_{z}^{1}\] done
clear
View Solution play_arrow
-
question_answer161)
The ground state term symbol for an electronic state is governed by [UPSEAT 2004]
A)
Heisenberg?s principle done
clear
B)
Hund?s rule done
clear
C)
Aufbau principle done
clear
D)
Pauli exclusion principle done
clear
View Solution play_arrow
-
question_answer162)
The electronic configuration of element with atomic number 24 is [Pb. CET 2004]
A)
\[1{{s}^{2}},2{{s}^{2}}2{{p}^{6}},3{{s}^{2}}3{{p}^{6}}3{{d}^{4}},4{{s}^{2}}\] done
clear
B)
\[1{{s}^{2}},2{{s}^{2}}2{{p}^{6}},3{{s}^{2}}3{{p}^{6}}3{{d}^{10}}\] done
clear
C)
\[1{{s}^{2}},2{{s}^{2}}2{{p}^{6}},3{{s}^{2}}3{{p}^{6}}3{{d}^{6}}\] done
clear
D)
\[1{{s}^{2}},2{{s}^{2}}2{{p}^{6}},3{{s}^{2}}3{{p}^{6}}3{{d}^{5}}4{{s}^{1}}\] done
clear
View Solution play_arrow
-
question_answer163)
The maximum number of electrons in \[p\]-orbital with \[n=5,\,m=1\] is [Pb. CET 2003]
A)
6 done
clear
B)
2 done
clear
C)
14 done
clear
D)
10 done
clear
View Solution play_arrow
-
question_answer164)
Number of two electron can have the same values of ?? quantum numbers [UPSEAT 2004]
A)
One done
clear
B)
Two done
clear
C)
Three done
clear
D)
Four done
clear
View Solution play_arrow
-
question_answer165)
The number of orbitals present in the shell with \[n=4\] is [UPSEAT 2004]
A)
16 done
clear
B)
8 done
clear
C)
18 done
clear
D)
32 done
clear
View Solution play_arrow
-
question_answer166)
Which of the following electronic configuration is not possible [MHCET 2003]
A)
\[1{{s}^{2}}2{{s}^{2}}\] done
clear
B)
\[1{{s}^{2}},2{{s}^{2}}2{{p}^{6}}\] done
clear
C)
\[[Ar]\,3{{d}^{10}},4{{s}^{2}}4{{p}^{2}}\] done
clear
D)
\[1{{s}^{2}},2{{s}^{2}}2{{p}^{2}},3{{s}^{1}}\] done
clear
View Solution play_arrow
-
question_answer167)
\[{{p}_{x}}\] orbital can accommodate [MNR 1990; IIT 1983; MADT Bihar 1995; BCECE 2005]
A)
4 electrons done
clear
B)
6 electrons done
clear
C)
2 electrons with parallel spins done
clear
D)
2 electrons with opposite spins done
clear
View Solution play_arrow
-
question_answer168)
The maximum number of electrons that can be accommodated in \['f'\] sub shell is [CPMT 1983, 84; MP PET/PMT 1988; BITS 1988]
A)
2 done
clear
B)
8 done
clear
C)
32 done
clear
D)
14 done
clear
View Solution play_arrow
-
question_answer169)
The number of electrons which can be accommodated in an orbital is [DPMT 1981; AFMC 1988]
A)
One done
clear
B)
Two done
clear
C)
Three done
clear
D)
Four done
clear
View Solution play_arrow
-
question_answer170)
The number of electrons in the atom which has 20 protons in the nucleus [CPMT 1981, 93; CBSE PMT 1989]
A)
20 done
clear
B)
10 done
clear
C)
30 done
clear
D)
40 done
clear
View Solution play_arrow
-
question_answer171)
The maximum number of electrons accommodated in \[5f\] orbitals are [MP PET 1996]
A)
5 done
clear
B)
10 done
clear
C)
14 done
clear
D)
18 done
clear
View Solution play_arrow
-
question_answer172)
The maximum number of electrons in an atom with \[l=2\] and \[n=3\] is [MP PET/PMT 1998]
A)
2 done
clear
B)
6 done
clear
C)
12 done
clear
D)
10 done
clear
View Solution play_arrow
-
question_answer173)
The configuration \[1{{s}^{2}}2{{s}^{2}}2{{p}^{5}}3{{s}^{1}}\] shows [AIIMS 1997]
A)
Ground state of fluorine atom done
clear
B)
Excited state of fluorine atom done
clear
C)
Excited state of neon atom done
clear
D)
Excited state of ion \[O_{2}^{-}\] done
clear
View Solution play_arrow
-
question_answer174)
For sodium atom the number of electrons with \[m=0\] will be [RPMT 1999]
A)
2 done
clear
B)
7 done
clear
C)
9 done
clear
D)
8 done
clear
View Solution play_arrow
-
question_answer175)
The number of electrons that can be accommodated in \[d{{z}^{2}}\] orbital is [Kurukshetra CEE 2002]
A)
10 done
clear
B)
1 done
clear
C)
4 done
clear
D)
2 done
clear
View Solution play_arrow
-
question_answer176)
Number of unpaired electrons in \[1{{s}^{2}}2{{s}^{2}}2{{p}^{3}}\] is [CPMT 1982; MP PMT 1987; BHU 1987; CBSE PMT 1990; CET Pune 1998; AIIMS 2000]
A)
2 done
clear
B)
0 done
clear
C)
3 done
clear
D)
1 done
clear
View Solution play_arrow
-
question_answer177)
Total number of unpaired electrons in an atom of atomic number 29 is [CPMT 1984, 93]
A)
1 done
clear
B)
3 done
clear
C)
4 done
clear
D)
2 done
clear
View Solution play_arrow
-
question_answer178)
The number of unpaired electrons in \[1{{s}^{2}},\,2{{s}^{2}}2{{p}^{4}}\] is [NCERT 1984; CPMT 1991; MP PMT 1996, 2002]
A)
4 done
clear
B)
2 done
clear
C)
0 done
clear
D)
1 done
clear
View Solution play_arrow
-
question_answer179)
The maximum number of electrons that can be accommodated in a \[3d\] subshell is
A)
2 done
clear
B)
10 done
clear
C)
6 done
clear
D)
14 done
clear
View Solution play_arrow
-
question_answer180)
The maximum number of electrons which each sub-shell can occupy is [Pb. CET 1989]
A)
\[2{{n}^{2}}\] done
clear
B)
\[2n\] done
clear
C)
\[2(2l+1)\] done
clear
D)
\[(2l+1)\] done
clear
View Solution play_arrow
-
question_answer181)
Number of unpaired electrons in the ground state of beryllium atom is
A)
2 done
clear
B)
1 done
clear
C)
0 done
clear
D)
All the above done
clear
View Solution play_arrow
-
question_answer182)
How many unpaired electrons are present in \[N{{i}^{2+}}\] cation (atomic number = 28) [IIT 1981; MNR 1984; MP PMT 1995; Kerala PMT 2003]
A)
0 done
clear
B)
2 done
clear
C)
4 done
clear
D)
6 done
clear
View Solution play_arrow
-
question_answer183)
The number of unpaired electrons in an \[{{O}_{2}}\] molecule is [MNR 1983]
A)
0 done
clear
B)
1 done
clear
C)
2 done
clear
D)
3 done
clear
View Solution play_arrow
-
question_answer184)
The number of unpaired electrons in a chromic ion \[C{{r}^{3+}}\] (atomic number = 24) is [MNR 1986; CPMT 1992]
A)
6 done
clear
B)
4 done
clear
C)
3 done
clear
D)
1 done
clear
View Solution play_arrow
-
question_answer185)
\[3{{d}^{10}}4{{s}^{0}}\] electronic configuration exhibits by
A)
\[Z{{n}^{++}}\] done
clear
B)
\[C{{u}^{++}}\] done
clear
C)
\[C{{d}^{++}}\] done
clear
D)
\[H{{g}^{++}}\] done
clear
View Solution play_arrow
-
question_answer186)
Which of the following metal ions will have maximum number of unpaired electrons [CPMT 1996]
A)
\[F{{e}^{+2}}\] done
clear
B)
\[C{{O}^{+2}}\] done
clear
C)
\[N{{i}^{+2}}\] done
clear
D)
\[M{{n}^{+2}}\] done
clear
View Solution play_arrow
-
question_answer187)
Which of the metal ion will have highest number of unpaired electrons
A)
\[C{{u}^{+}}\] done
clear
B)
\[F{{e}^{2+}}\] done
clear
C)
\[F{{e}^{3+}}\] done
clear
D)
\[C{{o}^{2+}}\] done
clear
View Solution play_arrow
-
question_answer188)
The maximum number of unpaired electron can be present in \[d\]orbitals are
A)
1 done
clear
B)
3 done
clear
C)
5 done
clear
D)
7 done
clear
View Solution play_arrow
-
question_answer189)
The molecule having one unpaired electron is
A)
\[NO\] done
clear
B)
\[CO\] done
clear
C)
\[C{{N}^{-}}\] done
clear
D)
\[{{O}_{2}}\] done
clear
View Solution play_arrow
-
question_answer190)
A filled or half-filled set of \[p\] or \[d\]-orbitals is spherically symmetric. Point out the species which has spherical symmetry [NCERT 1983]
A)
\[Na\] done
clear
B)
\[C\] done
clear
C)
\[C{{l}^{-}}\] done
clear
D)
\[Fe\] done
clear
View Solution play_arrow
-
question_answer191)
The atom of the element having atomic number 14 should have [AMU 1984]
A)
One unpaired electron done
clear
B)
Two unpaired electrons done
clear
C)
Three unpaired electrons done
clear
D)
Four unpaired electrons done
clear
View Solution play_arrow
-
question_answer192)
An atom has 2 electrons in \[K\] shell, 8 electrons in \[L\] shell and 6 electrons in \[M\] shell. The number of \[s\]-electrons present in that element is [CPMT 1989]
A)
6 done
clear
B)
5 done
clear
C)
7 done
clear
D)
10 done
clear
View Solution play_arrow
-
question_answer193)
The number of unpaired electrons in carbon atom in excited state is [MNR 1987]
A)
One done
clear
B)
Two done
clear
C)
Three done
clear
D)
Four done
clear
View Solution play_arrow
-
question_answer194)
Maximum number of electrons present in \['N'\] shell is [EAMCET 1984]
A)
18 done
clear
B)
32 done
clear
C)
2 done
clear
D)
8 done
clear
View Solution play_arrow
-
question_answer195)
The number of \[d\] electrons in \[F{{e}^{+2}}\] (atomic number of \[Fe=26\]) is not equal to that of the [MNR 1993]
A)
\[p\]-electrons in \[Ne\] (At. No.= 10) done
clear
B)
\[s\]-electrons in \[Mg\] (At. No.= 12) done
clear
C)
\[d\]-electrons in \[Fe\] done
clear
D)
\[p\]-electrons in \[C{{l}^{-}}\] (At. No. of \[Cl\] = 17) done
clear
View Solution play_arrow
-
question_answer196)
A transition metal \[X\] has a configuration \[[Ar]3{{d}^{4}}\] in its \[+3\] oxidation state. Its atomic number is [EAMCET 1990]
A)
25 done
clear
B)
26 done
clear
C)
22 done
clear
D)
19 done
clear
View Solution play_arrow
-
question_answer197)
The total number of electrons present in all the \[p\]-orbitals of bromine are [MP PET 1994]
A)
Five done
clear
B)
Eighteen done
clear
C)
Seventeen done
clear
D)
Thirty five done
clear
View Solution play_arrow
-
question_answer198)
Which of the following has the maximum number of unpaired electrons [IIT 1996]
A)
\[M{{g}^{2+}}\] done
clear
B)
\[T{{i}^{3+}}\] done
clear
C)
\[{{V}^{3+}}\] done
clear
D)
\[F{{e}^{2+}}\] done
clear
View Solution play_arrow
-
question_answer199)
Which of the following has more unpaired \[d\]-electrons [CBSE PMT 1999]
A)
\[Z{{n}^{+}}\] done
clear
B)
\[F{{e}^{2+}}\] done
clear
C)
\[{{N}^{3+}}\] done
clear
D)
\[C{{u}^{+}}\] done
clear
View Solution play_arrow
-
question_answer200)
Maximum electrons in a \[d\]-orbital are [CPMT 1999]
A)
2 done
clear
B)
10 done
clear
C)
6 done
clear
D)
14 done
clear
View Solution play_arrow
-
question_answer201)
The number of unpaired electrons in \[F{{e}^{3+}}(Z=26)\] are [KCET 2000]
A)
5 done
clear
B)
6 done
clear
C)
3 done
clear
D)
4 done
clear
View Solution play_arrow
-
question_answer202)
How many unpaired electrons are present in cobalt [Co] metal [RPMT 2002]
A)
2 done
clear
B)
3 done
clear
C)
4 done
clear
D)
7 done
clear
View Solution play_arrow
-
question_answer203)
The number of unpaired electrons in nitrogen is [Pb. CET 2002]
A)
1 done
clear
B)
3 done
clear
C)
2 done
clear
D)
None of these done
clear
View Solution play_arrow
-
question_answer204)
Which of the following has the least energy
A)
\[2p\] done
clear
B)
\[3p\] done
clear
C)
\[2s\] done
clear
D)
\[4d\] done
clear
View Solution play_arrow
-
question_answer205)
Pauli's exclusion principle states that [CPMT 1983, 84]
A)
Nucleus of an atom contains no negative charge done
clear
B)
Electrons move in circular orbits around the nucleus done
clear
C)
Electrons occupy orbitals of lowest energy done
clear
D)
All the four quantum numbers of two electrons in an atom cannot be equal done
clear
View Solution play_arrow
-
question_answer206)
For the energy levels in an atom, which one of the following statements is correct [AIIMS 1983]
A)
There are seven principal electron energy levels done
clear
B)
The second principal energy level can have four sub-energy levels and contains a maximum of eight electrons done
clear
C)
The \[M\]energy level can have maximum of 32 electrons done
clear
D)
The \[4s\] sub-energy level is at a higher energy than the \[3d\] sub-energy level done
clear
View Solution play_arrow
-
question_answer207)
The statements [AIIMS 1982] (i) In filling a group of orbitals of equal energy, it is energetically preferable to assign electrons to empty orbitals rather than pair them into a particular orbital. (ii) When two electrons are placed in two different orbitals, energy is lower if the spins are parallelare valid for
A)
Aufbau principle done
clear
B)
Hund's rule done
clear
C)
Pauli's exclusion principle done
clear
D)
Uncertainty principle done
clear
View Solution play_arrow
-
question_answer208)
According to Aufbau's principle, which of the three \[4d,\,5p\] and \[5s\]will be filled with electrons first [MADT Bihar 1984]
A)
\[4d\] done
clear
B)
\[5p\] done
clear
C)
\[5s\] done
clear
D)
\[4d\] and \[5s\] will be filled simultaneously done
clear
View Solution play_arrow
-
question_answer209)
The energy of an electron of \[2{{p}_{y}}\] orbital is [AMU 1984]
A)
Greater than that of \[2{{p}_{x}}\] orbital done
clear
B)
Less than that of \[2{{p}_{x}}\] orbital done
clear
C)
Equal to that of \[2s\] orbital done
clear
D)
Same as that of \[2{{p}_{z}}\] orbital done
clear
View Solution play_arrow
-
question_answer210)
Which of the following principles/rules limits the maximum number of electrons in an orbital to two [CBSE PMT 1989]
A)
Aufbau principle done
clear
B)
Pauli's exclusion principle done
clear
C)
Hund's rule of maximum multiplicity done
clear
D)
Heisenberg's uncertainty principle done
clear
View Solution play_arrow
-
question_answer211)
The electrons would go to lower energy levels first and then to higher energy levels according to which of the following [BHU 1990; MP PMT 1993]
A)
Aufbau principle done
clear
B)
Pauli's exclusion principle done
clear
C)
Hund's rule of maximum multiplicity done
clear
D)
Heisenberg's uncertainty principle done
clear
View Solution play_arrow
-
question_answer212)
Energy of atomic orbitals in a particular shell is in the order [AFMC 1990]
A)
\[s<p<d<f\] done
clear
B)
\[s>p>d>f\] done
clear
C)
\[p<d<f<s\] done
clear
D)
\[f>d>s>p\] done
clear
View Solution play_arrow
-
question_answer213)
Aufbau principle is not satisfied by [MP PMT 1997]
A)
\[Cr\] and \[Cl\] done
clear
B)
\[Cu\] and \[Ag\] done
clear
C)
\[Cr\] and \[Mg\] done
clear
D)
\[Cu\] and \[Na\] done
clear
View Solution play_arrow
-
question_answer214)
Which of the following explains the sequence of filling the electrons in different shells [AIIMS 1998; BHU 1999]
A)
Hund's rule done
clear
B)
Octet rule done
clear
C)
Aufbau principle done
clear
D)
All of these done
clear
View Solution play_arrow
-
question_answer215)
Aufbau principle is obeyed in which of the following electronic configurations [AFMC 1999]
A)
\[1{{s}^{2}}2{{s}^{2}}2{{p}^{6}}\] done
clear
B)
\[1{{s}^{2}}3{{p}^{3}}3{{s}^{2}}\] done
clear
C)
\[1{{s}^{2}}3{{s}^{2}}3{{p}^{6}}\] done
clear
D)
\[1{{s}^{2}}2{{s}^{2}}3{{s}^{2}}\] done
clear
View Solution play_arrow
-
question_answer216)
Following Hund?s rule which element contains six unpaired electron [RPET 2000]
A)
Fe done
clear
B)
Co done
clear
C)
Ni done
clear
D)
Cr done
clear
View Solution play_arrow
-
question_answer217)
Electron enters the sub-shell for which \[(n+l)\] value is minimum. This is enunciated as [RPMT 2000]
A)
Hund?s rule done
clear
B)
Aufbau principle done
clear
C)
Heisenberg uncertainty principle done
clear
D)
Pauli?s exclusion principle done
clear
View Solution play_arrow
-
question_answer218)
The atomic orbitals are progressively filled in order of increasing energy. This principle is called as [MP PET 2001]
A)
Hund?s rule done
clear
B)
Aufbau principle done
clear
C)
Exclusion principle done
clear
D)
de-Broglie rule done
clear
View Solution play_arrow
-
question_answer219)
The correct order of increasing energy of atomic orbitals is [MP PET 2002]
A)
\[5p<4f<6s<5d\] done
clear
B)
\[5p<6s<4f<5d\] done
clear
C)
\[4f<5p<5d<6s\] done
clear
D)
\[5p<5d<4f<6s\] done
clear
View Solution play_arrow
-
question_answer220)
The orbital with maximum energy is [CPMT 2002]
A)
3d done
clear
B)
5p done
clear
C)
4s done
clear
D)
6d done
clear
View Solution play_arrow
-
question_answer221)
p-orbitals of an atom in presence of magnetic field are [Pb. PMT 2002]
A)
Two fold degenerate done
clear
B)
Non degenerate done
clear
C)
Three fold degenerate done
clear
D)
None of these done
clear
View Solution play_arrow
-
question_answer222)
Orbital angular momentum for a d-electron is [MP PET 2003]
A)
\[\frac{6h}{2\pi }\] done
clear
B)
\[\frac{\sqrt{6}\,h}{2\pi }\] done
clear
C)
\[\frac{12h}{2\pi }\] done
clear
D)
\[\frac{\sqrt{12}\,h}{2\pi }\] done
clear
View Solution play_arrow
-
question_answer223)
Number of nodal centres for 2s orbital [RPET 2003]
A)
1 done
clear
B)
0 done
clear
C)
4 done
clear
D)
3 done
clear
View Solution play_arrow
-
question_answer224)
The orbital angular momentum of an electron in \[2s\]-orbital is [MP PET 2004]
A)
\[\frac{1}{2}\frac{h}{2\pi }\] done
clear
B)
\[\frac{h}{2\pi }\] done
clear
C)
\[\sqrt{2}\frac{h}{2\pi }\] done
clear
D)
Zero done
clear
View Solution play_arrow
-
question_answer225)
The maximum number of electrons present in an orbit \[l=3\], is [Pb. PMT 2004]
A)
6 done
clear
B)
8 done
clear
C)
10 done
clear
D)
14 done
clear
View Solution play_arrow
-
question_answer226)
Number of unpaired electrons in \[M{{n}^{4+}}\] is [DPMT 2005]
A)
3 done
clear
B)
5 done
clear
C)
6 done
clear
D)
4 done
clear
View Solution play_arrow
-
question_answer227)
Which of the following sequence is correct as per Aufbau principle [DPMT 2005]
A)
\[3s<3d<4s<4p\] done
clear
B)
\[1s<2p<4s<3d\] done
clear
C)
\[2s<5s<4p<5d\] done
clear
D)
\[2s<2p<3d<3p\] done
clear
View Solution play_arrow
-
question_answer228)
Electronic configuration of deuterium atom is [J&K CET 2005]
A)
\[1{{s}^{1}}\] done
clear
B)
\[2{{s}^{2}}\] done
clear
C)
\[2{{s}^{1}}\] done
clear
D)
\[1{{s}^{2}}\] done
clear
View Solution play_arrow
 done
clear
done
clear
 done
clear
done
clear
 done
clear
done
clear
