-
question_answer1)
Alcohols have higher boiling point than parent hydrocarbons because of
A)
higher molecular mass done
clear
B)
presence of hydrogen bonding done
clear
C)
presence of carbon chain done
clear
D)
None of the above done
clear
View Solution play_arrow
-
question_answer2)
| \[{{C}_{6}}{{H}_{5}}CH=CHCHO\xrightarrow{x}{{C}_{6}}{{H}_{5}}CH=CHC{{H}_{2}}OH\] |
| In the above sequence X can be |
A)
\[{{K}_{2}}C{{r}_{2}}{{O}_{5}}/{{H}^{+}}\] done
clear
B)
\[NaB{{H}_{4}}\] done
clear
C)
\[{{N}_{2}}/Ni\] done
clear
D)
Both (b) and (c) done
clear
View Solution play_arrow
-
question_answer3)
Which of the following is a tertiary alcohol?
A)
B)
C)
\[C{{H}_{3}}\_\_C{{H}_{2}}\_\_OH\] done
clear
D)
View Solution play_arrow
-
question_answer4)
Which enzyme converts glucose and fructose both into ethanol?
A)
Maltase done
clear
B)
Diastase done
clear
C)
Invertase done
clear
D)
Zymase done
clear
View Solution play_arrow
-
question_answer5)
The reagent which easily reacts with ethanol and propanol is
A)
Grignard reagent done
clear
B)
Fehling reagent done
clear
C)
aqua-regia done
clear
D)
Tollen's reagent done
clear
View Solution play_arrow
-
question_answer6)
| \[A\xleftarrow[\Delta ]{Cu}C{{H}_{3}}C{{H}_{2}}OH\xrightarrow[\Delta ]{A{{l}_{2}}{{O}_{2}}}B\] |
| A and B respectively are |
A)
alkene, alkyne done
clear
B)
alkanal, alkene done
clear
C)
alkyne, alkanal done
clear
D)
alkene, alkanal done
clear
View Solution play_arrow
-
question_answer7)
| The order of reactivity of the following alcohols towards cone. HCl is |
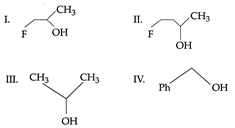 |
A)
\[IV>III>II>I\] done
clear
B)
\[I>II>III>IV\] done
clear
C)
\[IV>III>I>II\] done
clear
D)
\[I>III>II>IV\] done
clear
View Solution play_arrow
-
question_answer8)
1-propanal and 2-propanal can differentiated by
A)
oxidation with acidic \[{{K}_{2}}C{{r}_{2}}O\] followed by reaction with Fehling solution done
clear
B)
oxidation with \[KMn{{O}_{4}}\] followed by reaction with Fehling solution done
clear
C)
oxidation with cone. \[{{H}_{2}}S{{O}_{4}}\] followed by reaction with Fehling solution done
clear
D)
oxidation by heating with copper followed by reaction with Fehling solution done
clear
View Solution play_arrow
-
question_answer9)
| Which of the following in correct regarding the change in compound given below? |
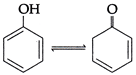 |
A)
Former is more stable then latter due to more resonance done
clear
B)
Former is enol form, latter is keto form done
clear
C)
Two compounds exhibits keto-enol isomerism done
clear
D)
All of the above done
clear
View Solution play_arrow
-
question_answer10)
| Identify the reagent(s) needed to carry out the following reaction. |
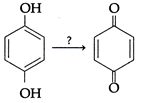 |
A)
\[LiAl{{H}_{4}}\] done
clear
B)
NaOH done
clear
C)
\[N{{a}_{2}}C{{r}_{2}}{{O}_{7}},{{H}_{2}}S{{O}_{4}}/{{H}_{2}}O\] done
clear
D)
Na done
clear
View Solution play_arrow
-
question_answer11)
| In the following compound given below the correct order of decreasing acidic strength is |
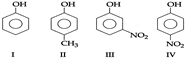 |
A)
\[IV>III>I>II\] done
clear
B)
\[II>I>III>IV\] done
clear
C)
\[I>IV>III>II\] done
clear
D)
\[III>IV>I>III\] done
clear
View Solution play_arrow
-
question_answer12)
A compound that easily undergoes bromination is
A)
benzene done
clear
B)
toluene done
clear
C)
phenol done
clear
D)
benzoic acid done
clear
View Solution play_arrow
-
question_answer13)
The reagent used for dehydration of an alcohol is
A)
phosphorus pentachloride done
clear
B)
aluminium oxide done
clear
C)
calcium chloride done
clear
D)
sodium chloride done
clear
View Solution play_arrow
-
question_answer14)
| Complete the analogy regarding type of reaction. |
| Williamson ether synthesis : A : Aryl ether : B |
A)
A : Nucleophilic substitution, B : Electrophilic substitution done
clear
B)
A : Electrophilic substitution, B : Electrophilic substitution done
clear
C)
A : Nucleophilic addition, B : Nucleophilic substitution done
clear
D)
A : Electrophilic substitution, B : Nucleophilic addition done
clear
View Solution play_arrow
-
question_answer15)
| Complete the reaction by identifying |
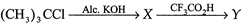 |
A)
\[C{{H}_{2}}=CH\_\_C{{H}_{2}}OH\] done
clear
B)
C)
\[C{{H}_{3}}COC{{H}_{2}}C{{H}_{3}}\] done
clear
D)
\[{{(C{{H}_{3}})}_{2}}CHC{{H}_{2}}OH\] done
clear
View Solution play_arrow
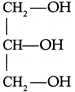 done
clear
done
clear
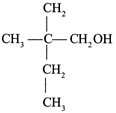 done
clear
done
clear
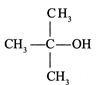 done
clear
done
clear



![]()
 done
clear
done
clear
