![]() gives aldehyde and
ketone.
gives aldehyde and
ketone.
![]()
![]()
![]() in
in
![]()
![]()

![]() or
or ![]() ?
?
![]() or diborane?
or diborane?
![]() reacts with bromine in the
presence of red phosphorus.
reacts with bromine in the
presence of red phosphorus.
![]() group.
group.
![]() (ii)
(ii)![]()
![]()
![]()
![]()
 (ii)
(ii) ![]() (iii)
(iii)
![]()
![]()
![]() (ii)
(ii)
![]()
![]() value than acetic
acid.
value than acetic
acid.
![]() .
.
![]()
![]() in the addition of alcohol to carbonyl
group?
in the addition of alcohol to carbonyl
group?
![]() hydrogen
of aldehydes and ketones are acidic. Why?
hydrogen
of aldehydes and ketones are acidic. Why?
![]()
![]()
![]()
![]()
![]()
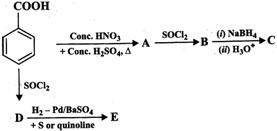
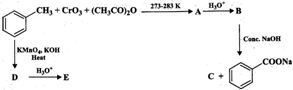
You need to login to perform this action.
You will be redirected in
3 sec
