-
question_answer1)
Which of the following will fail to react with potassium dichromate and dilute sulphuric acid
A)
Ethyl alcohol (ethanol) done
clear
B)
Acetaldehyde (ethanal) done
clear
C)
Secondary propyl alcohol (2-propanol) done
clear
D)
Acetone (propanone) done
clear
View Solution play_arrow
-
question_answer2)
Acetone and acetaldehyde are differentiated by [CPMT 1987, 93]
A)
\[NaOH+{{I}_{2}}\] done
clear
B)
\[Ag(N{{H}_{3}})_{2}^{+}\] done
clear
C)
\[HN{{O}_{2}}\] done
clear
D)
\[{{I}_{2}}\] done
clear
View Solution play_arrow
-
question_answer3)
Which of the following will react with water [IIT 1998]
A)
\[CHC{{l}_{3}}\] done
clear
B)
\[C{{l}_{3}}CCHO\] done
clear
C)
\[CC{{l}_{4}}\] done
clear
D)
\[ClC{{H}_{2}}C{{H}_{2}}Cl\] done
clear
View Solution play_arrow
-
question_answer4)
An organic compound 'A' has the molecular formula \[{{C}_{3}}{{H}_{6}}O,\] it undergoes iodoform test. When saturated with dil. \[HCl\] is gives 'B' of molecular formula \[{{C}_{9}}{{H}_{14}}O\]. A and B respectively are [Tamil Nadu CET 2002]
A)
Propanal and mesitylene done
clear
B)
Propanone and mesityl oxide done
clear
C)
Propanone and 2,6-dimethyl -2, 5-heptadien-4-one done
clear
D)
Propanone and mesitylene oxide done
clear
View Solution play_arrow
-
question_answer5)
Which alkene is formed from the following reaction \[C{{H}_{3}}C{{H}_{2}}C{{H}_{2}}CH=PP{{h}_{3}}+2-\,\text{Butanone}\] [Manipal 2001]
A)
3- Methyl-3-heptene done
clear
B)
4-Methyl-3-heptene done
clear
C)
5-Methyl-3-heptene done
clear
D)
1-Methyl-5- methane done
clear
View Solution play_arrow
-
question_answer6)
Compound ?A? (molecular formula \[{{C}_{3}}{{H}_{8}}O)\] is treated with acidified potassium dichromate to form a product ?B? (molecular formula \[{{C}_{3}}{{H}_{6}}O).\] ?B? forms a shining silver mirror on warming with ammoniacal silver nitrate. ?B? when treated with an aqueous solution of \[{{H}_{2}}NCONHN{{H}_{2}}.HCl\] and sodium acetate gives a product ?C?. Identify the structure of ?C? [IIT-JEE (Screening) 2002]
A)
\[C{{H}_{3}}C{{H}_{2}}CH=NNHCON{{H}_{2}}\] done
clear
B)
\[C{{H}_{3}}-CH=\underset{C{{H}_{3}}}{\mathop{\underset{|\,\,\,\,\,\,\,\,\,\,}{\mathop{NNH}}\,}}\,CON{{H}_{2}}\] done
clear
C)
\[C{{H}_{3}}CH=\underset{C{{H}_{3}}}{\mathop{\underset{|\,\,\,\,\,\,\,\,\,\,}{\mathop{NCO}}\,}}\,NHN{{H}_{2}}\] done
clear
D)
\[C{{H}_{3}}C{{H}_{2}}CH-NCONHN{{H}_{2}}\] done
clear
View Solution play_arrow
-
question_answer7)
Which is not true about acetophenone [Manipal 2002]
A)
Reacts to form 2, 4-dinitorphenyl hydrazine done
clear
B)
Reacts with Tollen?s reagent to form silver mirror done
clear
C)
Reacts with \[{{I}_{2}}/NaOH\] to form iodoform done
clear
D)
On oxidation with alkaline \[KMn{{O}_{4}}\] followed by hydrolysis gives benzoic acid done
clear
View Solution play_arrow
-
question_answer8)
The enol form of acetone, after treatment with \[{{D}_{2}}O,\] gives [IIT-JEE (Screening) 1999]
A)
\[C{{H}_{3}}-\overset{OD\,\,\,\,\,\,\,\,\,\,}{\mathop{\overset{|\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,}{\mathop{C=C{{H}_{2}}}}\,}}\,\] done
clear
B)
\[C{{D}_{3}}-\overset{O\,\,}{\mathop{\overset{||\,\,\,\,}{\mathop{C-}}\,}}\,C{{D}_{3}}\] done
clear
C)
\[C{{H}_{2}}=\overset{\,OH}{\mathop{\overset{|\,\,\,\,\,}{\mathop{C-}}\,}}\,C{{H}_{2}}D\] done
clear
D)
\[C{{D}_{2}}=\overset{OD}{\mathop{\overset{|\,\,\,\,\,}{\mathop{C-}}\,}}\,CD\] done
clear
View Solution play_arrow
-
question_answer9)
The appropriate reagent for the transformation
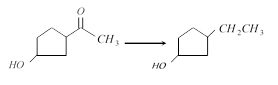
[IIT-JEE (Screening) 2000]
A)
\[Zn(Hg),\,HCl\] done
clear
B)
\[N{{H}_{2}}N{{H}_{2}}O{{H}^{-}}\] done
clear
C)
\[{{H}_{2}}/Ni\] done
clear
D)
\[NaB{{H}_{4}}\] done
clear
View Solution play_arrow
-
question_answer10)
Which of the following has the most acidic hydrogen [IIT-JEE (Screening) 2000]
A)
3-hexanone done
clear
B)
2, 4-hexanedione done
clear
C)
2, 5-hexanedione done
clear
D)
2, 3-hexanedione done
clear
View Solution play_arrow
-
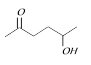
question_answer11)
Which of the following will be most readily dehydrated in acidic conditions [IIT-JEE (Screening) 2000]
A)
B)
C)
D)
View Solution play_arrow
-
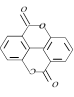
question_answer12)
Major Product is [IIT-JEE (Screening) 2003]
A)
B)
C)
D)
View Solution play_arrow
-
question_answer13)
Among the given compounds, the most susceptible to nucleophilic attack at the carbonyl group is [IIT 1997]
A)
MeCOCl done
clear
B)
MeCHO done
clear
C)
MeCOOMe done
clear
D)
MeCOOCOMe done
clear
View Solution play_arrow
-
question_answer14)
Which of the following will give yellow precipitate with \[{{I}_{2}}/NaOH\] [IIT 1997]
A)
\[IC{{H}_{2}}COC{{H}_{2}}C{{H}_{3}}\] done
clear
B)
\[C{{H}_{3}}COOCOC{{H}_{3}}\] done
clear
C)
\[C{{H}_{3}}CON{{H}_{2}}\] done
clear
D)
\[C{{H}_{3}}CH(OH)C{{H}_{2}}C{{H}_{3}}\] done
clear
View Solution play_arrow
-
question_answer15)
The product of acid hydrolysis of P and Q can be distinguished by [IIT-JEE (Screening) 2003]
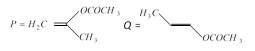
A)
Lucas Reagent done
clear
B)
2,4-DNP done
clear
C)
Fehling's Solution done
clear
D)
\[NaHS{{O}_{3}}\] done
clear
View Solution play_arrow
-
question_answer16)
On vigorous oxidation by permanganate solution \[{{(C{{H}_{3}})}_{2}}C=CH-C{{H}_{2}}C{{H}_{2}}C{{H}_{3}}\] gives [AIEEE 2002]
A)
\[C{{H}_{3}}\underset{C{{H}_{3}}}{\overset{OH}{\mathop{\underset{|\,\,\,\,\,\,\,}{\overset{|\,\,\,\,\,\,\,\,}{\mathop{-C-\,\,\,}}}\,}}}\,\overset{OH\,\,}{\mathop{\overset{|\,\,\,\,\,\,\,\,\,\,\,\,}{\mathop{CH-}}\,}}\,C{{H}_{2}}C{{H}_{3}}\] done
clear
B)
\[\begin{matrix} C{{H}_{3}} & {} & {} \\ {} & > & CHC{{O}_{2}}H+C{{H}_{3}}COOH \\ C{{H}_{3}} & {} & {} \\ \end{matrix}\] done
clear
C)
\[\begin{matrix} C{{H}_{3}} & {} & {} \\ {} & > & CHOH+C{{H}_{3}}C{{H}_{2}}C{{H}_{2}}OH \\ C{{H}_{3}} & {} & {} \\ \end{matrix}\] done
clear
D)
\[\begin{matrix} C{{H}_{3}} & {} & {} \\ {} & > & C=O+C{{H}_{3}}C{{H}_{2}}COOH \\ C{{H}_{3}} & {} & {} \\ \end{matrix}\] done
clear
View Solution play_arrow
-
question_answer17)
Which of the following reactions give benzo phenone [Roorkee Qualifying 1998]
A)
\[2{{C}_{6}}{{H}_{6}}+CC{{l}_{4}}\underset{\text{(ii) }{{H}_{2}}O}{\mathop{\xrightarrow{\text{(i) }AlC{{l}_{3}}}}}\,\] done
clear
B)
\[{{C}_{6}}{{H}_{6}}+{{C}_{6}}{{H}_{5}}COCl\underset{{}}{\mathop{\xrightarrow{AlC{{l}_{3}}}}}\,\] done
clear
C)
\[o-C{{H}_{3}}{{C}_{6}}{{H}_{4}}CO{{C}_{6}}{{H}_{5}}\xrightarrow{\text{Heat}}\] done
clear
D)
\[o-HOOC-{{C}_{6}}{{H}_{4}}-CO{{C}_{6}}{{H}_{5}}\underset{{{260}^{o}}C}{\mathop{\xrightarrow{Cu}}}\,\] done
clear
View Solution play_arrow
-
question_answer18)
Aldehyde and ketones can decolourize by [CPMT 2003]
A)
Bromine water done
clear
B)
Quick lime done
clear
C)
dil. \[{{H}_{2}}S{{O}_{4}}\] done
clear
D)
None of these done
clear
View Solution play_arrow
-
question_answer19)
Which of the following statements regarding chemical properties of acetophenone are wrong (1) It is reduced to methyl phenyl carbinol by sodium and ethanol (2) It is oxidised to benzoic acid with acidified \[KMn{{O}_{4}}\] (3) It does not undergo iodoform electrophilic substitution like nitration at meta position (4) It does not undergo iodoform reaction with iodine and alkali [Tamil Nadu CET 2001]
A)
1 and 2 done
clear
B)
2 and 4 done
clear
C)
3 and 4 done
clear
D)
1 and 3 done
clear
View Solution play_arrow
-
question_answer20)
The product(s) obtained via oxymercuration \[(HgS{{O}_{4}}+{{H}_{2}}S{{O}_{4}})\] of 1-butyne would be [IIT-JEE 1999]
A)
\[C{{H}_{3}}C{{H}_{2}}COC{{H}_{3}}\] done
clear
B)
\[C{{H}_{3}}C{{H}_{2}}C{{H}_{2}}CHO\] done
clear
C)
\[C{{H}_{3}}C{{H}_{2}}CHO+HCHO\] done
clear
D)
\[C{{H}_{3}}C{{H}_{2}}COOH+HCOOH\] done
clear
View Solution play_arrow
-
question_answer21)
The most reactive compound towards formation of cyanohydrin on treatment with KCN followed by acidification is [GATE 2001]
A)
Benzaldehyde done
clear
B)
p-Nitrobenzaldehyde done
clear
C)
Phenyl acetaldehyde done
clear
D)
p-Hydroxybenzaldehyde done
clear
View Solution play_arrow
-
question_answer22)
The key step in cannizzaro?s reaction is the intermolecular shift of [Orissa JEE 2003]
A)
Proton done
clear
B)
Hydride ion done
clear
C)
Hydronium ion done
clear
D)
Hydrogen bond done
clear
View Solution play_arrow
-
question_answer23)
Benzophenone does not react with [BHU 2003]
A)
\[RN{{H}_{2}}\] done
clear
B)
\[S{{O}_{3}}\] done
clear
C)
\[NaOH\] done
clear
D)
\[N{{a}_{2}}C{{O}_{3}}\] done
clear
View Solution play_arrow
-
question_answer24)
The most suitable reagent for the conversion of \[RC{{H}_{2}}OH\to RCHO\] is [AIIMS 2004]
A)
\[KMn{{O}_{4}}\] done
clear
B)
\[{{K}_{2}}C{{r}_{2}}{{O}_{7}}\] done
clear
C)
\[Cr{{O}_{3}}\] done
clear
D)
PCC (Pyridine chloro chromate) done
clear
View Solution play_arrow
-
question_answer25)
The conversion of acetophenone to acetanilide is best accomplished by using : [UPSEAT 2004]
A)
Backmann rearrangement done
clear
B)
Curtius rearrangement done
clear
C)
Lossen rearrangement done
clear
D)
Hofmann rearrangement done
clear
View Solution play_arrow
-
question_answer26)
Which of the following will not give iodoform test? [UPSEAT 2004]
A)
Isopropyl alcohol done
clear
B)
Ethanol done
clear
C)
Ethanal done
clear
D)
Benzyl alcohol done
clear
View Solution play_arrow
-
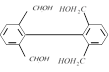
question_answer27)
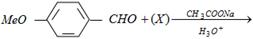
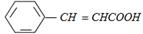
The compound (X) is [IIT-JEE 2005]
A)
\[C{{H}_{3}}COOH\] done
clear
B)
\[BrC{{H}_{2}}-COOH\] done
clear
C)
\[{{(C{{H}_{3}}CO)}_{2}}O\] done
clear
D)
\[CHO-COOH\] done
clear
View Solution play_arrow
-
question_answer28)
The major organic product formed from the following reaction [CBSE PMT 2005]
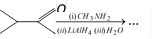
A)
B)
C)
D)
View Solution play_arrow
-
question_answer29)
Products of the following reaction \[C{{H}_{3}}C\equiv C\,C{{H}_{2}}C{{H}_{3}}\underset{(2)\,Hydrolysis}{\mathop{\xrightarrow{(1)\,{{O}_{3}}}}}\,\,\,\,...\text{are}\] [CBSE PMT 2005]
A)
\[C{{H}_{3}}CHO+C{{H}_{3}}C{{H}_{2}}CHO\] done
clear
B)
\[C{{H}_{3}}COOH+C{{H}_{3}}C{{H}_{2}}CHO\] done
clear
C)
\[C{{H}_{3}}COOH+HOOCC{{H}_{2}}C{{H}_{3}}\] done
clear
D)
\[C{{H}_{3}}COOH+C{{O}_{2}}\] done
clear
View Solution play_arrow
-
question_answer30)
A compound, containing only carbon, hydrogen and oxygen, has a molecular weight of 44. On complete oxidation it is converted into a compound of molecular weight 60. The original compound is [KCET 2005]
A)
An aldehyde done
clear
B)
An acid done
clear
C)
An alcohol done
clear
D)
an ether done
clear
View Solution play_arrow
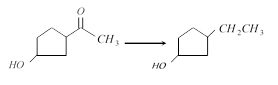 [IIT-JEE (Screening) 2000]
[IIT-JEE (Screening) 2000]  done
clear
done
clear
 done
clear
done
clear
 Major Product is [IIT-JEE (Screening) 2003]
Major Product is [IIT-JEE (Screening) 2003]  done
clear
done
clear
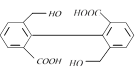 done
clear
done
clear
 done
clear
done
clear
 done
clear
done
clear
![]()
![]() The compound (X) is [IIT-JEE 2005]
The compound (X) is [IIT-JEE 2005] ![]()
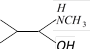 done
clear
done
clear
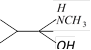 done
clear
done
clear
