-
question_answer1)
Acid present in soured curd is
A)
Lactic acid done
clear
B)
Acetic acid done
clear
C)
Tartaric acid done
clear
D)
Oxalic add done
clear
View Solution play_arrow
-
question_answer2)
Which of the following acid is used by gold smith for dissolving gold?
A)
Nitric acid done
clear
B)
Sulphuric acid done
clear
C)
Aqua regia done
clear
D)
Hydrochloric acid done
clear
View Solution play_arrow
-
question_answer3)
Which of the following substance present in turmeric changes its colour on reaction with base?
A)
Lichen done
clear
B)
Magnesia done
clear
C)
Curcumin done
clear
D)
None of these done
clear
View Solution play_arrow
-
question_answer4)
Which of the following is not an indicator?
A)
China rose done
clear
B)
Onion done
clear
C)
Clove done
clear
D)
Lemon done
clear
View Solution play_arrow
-
question_answer5)
Sodium bicarbonate is commonly called
A)
Lime water done
clear
B)
Slaked lime done
clear
C)
Washing soda done
clear
D)
Baking soda done
clear
View Solution play_arrow
-
question_answer6)
Complete the following analogy: Turmeric: Indicator:: Milk of magnesia:
A)
Strong acid done
clear
B)
Antacid done
clear
C)
Antidote for sting done
clear
D)
Hydrated salt done
clear
View Solution play_arrow
-
question_answer7)
Complete the following analogy: Sea water : Salty:: ______: Soap
A)
Bitter done
clear
B)
Slippery done
clear
C)
Sour done
clear
D)
Both (a) and (b) done
clear
View Solution play_arrow
-
question_answer8)
Which one of the following salts is used for making detergent powder?
A)
Washing soda done
clear
B)
Potassium nitrate done
clear
C)
Sodium bicarbonate done
clear
D)
Common salt done
clear
View Solution play_arrow
-
question_answer9)
Which of the following is not a base?
A)
Magnesium hydroxide done
clear
B)
Calcium hydroxide done
clear
C)
Calcium carbonate done
clear
D)
Sodium hydroxide done
clear
View Solution play_arrow
-
question_answer10)
Antacids contain -
A)
Strong acid done
clear
B)
Weak base done
clear
C)
Strong base done
clear
D)
Weak acid done
clear
View Solution play_arrow
-
question_answer11)
Usefulness of an indicator depends upon change in:
A)
its colour done
clear
B)
its physical state done
clear
C)
its temperature done
clear
D)
its pressure done
clear
View Solution play_arrow
-
question_answer12)
Acid present in vinegar is:
A)
formic acid done
clear
B)
acetic acid done
clear
C)
sulphuric acid done
clear
D)
nitric acid done
clear
View Solution play_arrow
-
question_answer13)
Reaction of an acid with a base is:
A)
decomposition done
clear
B)
combination done
clear
C)
redox done
clear
D)
neutralization done
clear
View Solution play_arrow
-
question_answer14)
Antacids contain:
A)
weak base done
clear
B)
weak acid done
clear
C)
strong base done
clear
D)
strong acid done
clear
View Solution play_arrow
-
question_answer15)
Which medicines are used for treating indigestion?
A)
Antibiotic done
clear
B)
Analgesic done
clear
C)
Antacids done
clear
D)
Antiseptic done
clear
View Solution play_arrow
-
question_answer16)
Which of the following is acidic in nature?
A)
Apple juice done
clear
B)
Soap solution done
clear
C)
Slaked lime done
clear
D)
None of these done
clear
View Solution play_arrow
-
question_answer17)
.................. is present in sour milk-
A)
Glycolic add done
clear
B)
Lactic acid done
clear
C)
Citrus add done
clear
D)
Tartaric acid done
clear
View Solution play_arrow
-
question_answer18)
.................. is the strongest add:
A)
Nitric add done
clear
B)
Malic add done
clear
C)
Formic add done
clear
D)
Lactic add done
clear
View Solution play_arrow
-
question_answer19)
Sharp pain caused by an ant bite is due to:
A)
malic acid done
clear
B)
nitric acid done
clear
C)
formic add done
clear
D)
lactic add done
clear
View Solution play_arrow
-
question_answer20)
Acid turns:
A)
blue litmus red done
clear
B)
red litmus blue done
clear
C)
blue litmus green done
clear
D)
red litmus yellow done
clear
View Solution play_arrow
-
question_answer21)
Which is not a property of an acid?
A)
All acids have sour taste. done
clear
B)
All acids turn blue litmus red. done
clear
C)
All acids turn red litmus blue. done
clear
D)
All the acids give neutralisation reaction. done
clear
View Solution play_arrow
-
question_answer22)
Bases:
A)
have slippery feel done
clear
B)
tastes sour done
clear
C)
turn blue litmus red done
clear
D)
All these are correct done
clear
View Solution play_arrow
-
question_answer23)
Acids are stored in glass container and not in metallic containers. Why?
A)
Glass containers are transparent done
clear
B)
Glass containers are cheaper done
clear
C)
Metal containers are not easily available. done
clear
D)
Metals react with acids if acids are stored in metallic containers. done
clear
View Solution play_arrow
-
question_answer24)
Which of the following is a neutralization reaction?
A)
Sodium chloride + Water\[\to \] Sodium hydroxide + Hydrochloric acid done
clear
B)
Calcium carbonate + Water\[\to \] Calcium hydroxide + Carbon dioxide done
clear
C)
Sodium hydroxide + Hydrochloric add \[\to \]Sodium chloride + Water done
clear
D)
Copper sulphate + Zinc\[\to \] Zinc sulphate + Copper done
clear
View Solution play_arrow
-
question_answer25)
Select the one that should be used for treating a person stung by an ant.
A)
Sodium carbonate solution done
clear
B)
Water done
clear
C)
Vinegar done
clear
D)
None of these done
clear
View Solution play_arrow
-
question_answer26)
................ is present in grapes.
A)
Malic add done
clear
B)
Tartaric acid done
clear
C)
Acetic acid done
clear
D)
Citric acid done
clear
View Solution play_arrow
-
question_answer27)
Disposal of factory wastes in water bodies has been found to be harmful to water plants and animals. To prevent this the factory owner should:
A)
neutralize waste before disposal done
clear
B)
make the waste acidic before disposal done
clear
C)
make the waste basic before disposal done
clear
D)
All of the above are correct done
clear
View Solution play_arrow
-
question_answer28)
Which of the following statements is correct?
A)
Products of neutralization reaction are salt and water. done
clear
B)
Reactants of neutralization reaction are acid and alkali. done
clear
C)
Any neutralization reaction is an exothermic reaction. done
clear
D)
All the above are correct. done
clear
View Solution play_arrow
-
question_answer29)
Which of the following statements is correct?
A)
Hydrochloric acid turns red litmus to blue. done
clear
B)
Sodium hydroxide turns blue litmus to red. done
clear
C)
Both the above are correct. done
clear
D)
None of these is correct. done
clear
View Solution play_arrow
-
question_answer30)
Which of the following statements is correct?
A)
Indicator is a substance that shows different colors in acidic and basic solution. done
clear
B)
Turmeric stain on a white shut turns to red when it is washed with soap. done
clear
C)
Lime water is a solution of calcium hydroxide. done
clear
D)
All the above are correct. done
clear
View Solution play_arrow
-
question_answer31)
Which of the following statements is correct?
A)
Acids are corrosive in nature. done
clear
B)
Excess of carbon dioxide in air can cause acid rain. done
clear
C)
Excess of hydrochloric acid in stomach causes indigestion. done
clear
D)
All the above are correct. done
clear
View Solution play_arrow
-
question_answer32)
A natural dye extracted from lichens and most commonly used as an indicator. What is its colour in distilled water?
A)
Blue (b) done
clear
B)
Red done
clear
C)
Mauve done
clear
D)
Colourless done
clear
View Solution play_arrow
-
question_answer33)
............... changes odour in acidic and basic medium and is used as an olfactory indicator.
A)
Onion done
clear
B)
Vanilla done
clear
C)
Clove oil done
clear
D)
All of these done
clear
View Solution play_arrow
-
question_answer34)
Neutral substances are:
A)
those substances which are slightly acidic or slightly basic in solution. done
clear
B)
those substances which do not change the colour of either red or blue litmus done
clear
C)
Both the above are correct done
clear
D)
None of the above is correct done
clear
View Solution play_arrow
-
question_answer35)
Rain becomes acidic:
A)
because excess of carbon dioxide present in air get dissolved in rain drops to form carbonic acid. done
clear
B)
oxides of nitrogen and sulphur present in air dissolve to form respective acids. done
clear
C)
Both the above are correct done
clear
D)
None of these is correct done
clear
View Solution play_arrow
-
question_answer36)
A base is a substance that
A)
feels slippery done
clear
B)
tastes sour done
clear
C)
reacts with metals to produce hydrogen gas done
clear
D)
and (c) done
clear
View Solution play_arrow
-
question_answer37)
A neutralization reaction
A)
includes an acid and a base done
clear
B)
produces a salt done
clear
C)
forms water done
clear
D)
All of the above done
clear
View Solution play_arrow
-
question_answer38)
Which among the following is organic weak acid?
A)
Formic acid done
clear
B)
Sulphuric acid done
clear
C)
Nitric acid done
clear
D)
Hydrochloric acid done
clear
View Solution play_arrow
-
question_answer39)
Which among the given acids is the strongest acid?
A)
Acetic acid done
clear
B)
Sulphuric acid done
clear
C)
Formic acid done
clear
D)
Tannic acid done
clear
View Solution play_arrow
-
question_answer40)
Which of the following is the weakest acid?
A)
Citric done
clear
B)
Sulphuric done
clear
C)
Hydrochloric done
clear
D)
Nitric done
clear
View Solution play_arrow
-
question_answer41)
DIRECTION: Match Column-I with Column-II and select the correct answer using the codes given below the columns.
| Column - I (Acid) | Column-II (Natural Source of acid) |
| (A) Acetic acid | (p) Spinach |
| (B) Citric acid | (q) Tamarind |
| (C) Tartaric acid | (r) Orange |
| (D) Oxalic acid | (s) Vinegar |
A)
A\[\to \] (p), B\[\to \] (q), C\[\to \] (r), D\[\to \] (s) done
clear
B)
A\[\to \] (q), B\[\to \] (s), C\[\to \] (p), D\[\to \] (r) done
clear
C)
A\[\to \] (r), B\[\to \] (p), C\[\to \] (s), D\[\to \] (q) done
clear
D)
A\[\to \] (s), B\[\to \] (r), C\[\to \] (q), D\[\to \] (p) done
clear
View Solution play_arrow
-
question_answer42)
DIRECTION: Match Column-I with Column-II and select the correct answer using the codes given below the columns.
| Column - I | Column - II |
| (A) Malic acid | (p) Window cleaner |
| (B) Zinc carbonate | (q) Milk |
| (C) Ammonium hydroxide | (r) Calamine |
| (D) Lactice acid | (s) Apple |
A)
A\[\to \] (s), B\[\to \] (q), C\[\to \] (p), D\[\to \] (r) done
clear
B)
A\[\to \] (r), B\[\to \] (s), C\[\to \] (q), D\[\to \] (p) done
clear
C)
A\[\to \] (s), B\[\to \] (r), C\[\to \] (p), D\[\to \] (q) done
clear
D)
A\[\to \] (p), B\[\to \] (q), C\[\to \] (r), D\[\to \] (s) done
clear
View Solution play_arrow
-
question_answer43)
DIRECTION: Match Column-I with Column-II and select the correct answer using the codes given below the columns.
| Column - I | Column - II |
| (A) Sting of an ant | (p) Litmus |
| (B) Alkali | (q) Soluble base |
| (C) Indicator | (r) Calamine |
| (D) Bitter | (s) Formic acid |
A)
A\[\to \] (s), B\[\to \] (p), C\[\to \] (q), D\[\to \] (r) done
clear
B)
A\[\to \] (s), B\[\to \] (q), C\[\to \] (r), D\[\to \] (p) done
clear
C)
A\[\to \] (s), B\[\to \] (q), C\[\to \] (p), D\[\to \] (r) done
clear
D)
A\[\to \] (r), B\[\to \] (q), C\[\to \] (p), D\[\to \] (s) done
clear
View Solution play_arrow
-
question_answer44)
DIRECTION: Match Column-I with Column-II and select the correct answer using the codes given below the columns.
| Column - I | Column - II |
| (A) Slaked lime | (p) Used in batteries |
| (B) Vinegar | (q) Sour in taste |
| (C) Sulphuric acid | (r) Apply on bee?s sting |
| (D) Turmeric indicator | (s) Turns brown with the application of soap |
A)
A\[\to \] (s), B\[\to \] (q), C\[\to \] (p), D\[\to \] (r) done
clear
B)
A\[\to \] (r), B\[\to \] (q), C\[\to \] (p), D\[\to \] (s) done
clear
C)
A\[\to \] (q), B\[\to \] (r), C\[\to \] (p), D\[\to \] (s) done
clear
D)
A\[\to \] (r), B\[\to \] (q), C\[\to \] (s), D\[\to \] (p) done
clear
View Solution play_arrow
-
question_answer45)
DIRECTIONS: Read the passage given below and answer the questions that follow. Passage ? 1 Observe the flow chart given below and answer the questions that follow: \[\text{Magnesium+Oxygen}\xrightarrow{\text{heat}}\text{''A''}\xrightarrow{\text{Water}}\text{''B''}\xrightarrow[\text{solution}]{\text{Red litmus}}\text{''C''}\] "A" in this represents
A)
magnesium oxide done
clear
B)
magnesium hydroxide done
clear
C)
both magnesium oxide and magnesium hydroxide done
clear
D)
none of these done
clear
View Solution play_arrow
-
question_answer46)
DIRECTIONS: Read the passage given below and answer the questions that follow. Passage ? 1 Observe the flow chart given below and answer the questions that follow: \[\text{Magnesium+Oxygen}\xrightarrow{\text{heat}}\text{''A''}\xrightarrow{\text{Water}}\text{''B''}\xrightarrow[\text{solution}]{\text{Red litmus}}\text{''C''}\] "B" in this represents
A)
Magnesium done
clear
B)
Magnesium oxide done
clear
C)
Magnesium hydroxide done
clear
D)
None of these done
clear
View Solution play_arrow
-
question_answer47)
DIRECTIONS: Read the passage given below and answer the questions that follow. Passage ? 1 Observe the flow chart given below and answer the questions that follow: \[\text{Magnesium+Oxygen}\xrightarrow{\text{heat}}\text{''A''}\xrightarrow{\text{Water}}\text{''B''}\xrightarrow[\text{solution}]{\text{Red litmus}}\text{''C''}\] "C" in it indicates that
A)
Red litmus when added to "B", no change in colour occurs. done
clear
B)
Red litmus when added to "B" changes to blue. done
clear
C)
Red litmus when added to "B" changes to green. done
clear
D)
Red litmus when added to "B" it gives a colorless solution done
clear
View Solution play_arrow
-
question_answer48)
DIRECTIONS: Read the passage given below and answer the questions that follow. Passage ? 2 The reaction between an acid and a base is known as neutralization reaction. In this reaction salt and water are produced with evolution of heat. Salt is a product formed as a result of neutralization reaction. Name the salt formed by reaction of sodium hydroxide and hydrochloric acid.
A)
sodium carbonate done
clear
B)
sodium bicarbonate done
clear
C)
sodium chloride done
clear
D)
sodium sulphate done
clear
View Solution play_arrow
-
question_answer49)
DIRECTIONS: Read the passage given below and answer the questions that follow. Passage ? 2 The reaction between an acid and a base is known as neutralization reaction. In this reaction salt and water are produced with evolution of heat. A neutralization reaction is:
A)
an exothermic reaction done
clear
B)
endothermic reaction done
clear
C)
accompained by absorption of heat done
clear
D)
None of these is correct done
clear
View Solution play_arrow
-
question_answer50)
DIRECTIONS: Read the passage given below and answer the questions that follow. Passage ? 2 The reaction between an acid and a base is known as neutralization reaction. In this reaction salt and water are produced with evolution of heat. Which of the following is not an example of neutralization?
A)
Taking antacid in case of acidity done
clear
B)
Treating acidic soil with quick lime done
clear
C)
Change in colour of blue litmus paper to red with acidic solution done
clear
D)
Both (a) and (b) done
clear
View Solution play_arrow
-
question_answer51)
DIRECTIONS: Read the passage given below and answer the questions that follow. Passage - 3 Rahul and Manish went to a party Rahul ate too much fast food in the party and after returning home was complaining about the stomachache. Manish was worried about his condition. They went to doctor. Doctor told them not to get worried as this is due to indigestion of food and gave him some medicines. Which of the following acid present in our stomach helps in digestion?
A)
Acetic acid done
clear
B)
Oxalic acid done
clear
C)
Hydrochloric acid done
clear
D)
None of these done
clear
View Solution play_arrow
-
question_answer52)
DIRECTIONS: Read the passage given below and answer the questions that follow. Passage - 3 Rahul and Manish went to a party Rahul ate too much fast food in the party and after returning home was complaining about the stomachache. Manish was worried about his condition. They went to doctor. Doctor told them not to get worried as this is due to indigestion of food and gave him some medicines. Indigestion is caused by presence of excess of ........................... in the stomach?
A)
food material done
clear
B)
hydrochloric acid done
clear
C)
baking soda done
clear
D)
milk of magnesia done
clear
View Solution play_arrow
-
question_answer53)
DIRECTIONS: Read the passage given below and answer the questions that follow. Passage - 3 Rahul and Manish went to a party Rahul ate too much fast food in the party and after returning home was complaining about the stomachache. Manish was worried about his condition. They went to doctor. Doctor told them not to get worried as this is due to indigestion of food and gave him some medicines. The medicines that are used to relieve us of indigestion are generally called
A)
antibiotic done
clear
B)
analgesic done
clear
C)
antacid done
clear
D)
antipyretic done
clear
View Solution play_arrow
-
question_answer54)
DIRECTIONS: Read the passage given below and answer the questions that follow. Passage - 3 Rahul and Manish went to a party Rahul ate too much fast food in the party and after returning home was complaining about the stomachache. Manish was worried about his condition. They went to doctor. Doctor told them not to get worried as this is due to indigestion of food and gave him some medicines. Which of the following is used as an antacid?
A)
Acetic acid done
clear
B)
Sodium hydroxide done
clear
C)
Magnesium hydroxide done
clear
D)
Hydrochloric acid done
clear
View Solution play_arrow
-
question_answer55)
DIRECTIONS: Read the passage given below and answer the questions that follow. Passage - 4 Indicators change their colour when added to a solution containing an acidic or a basic substance. Turmeric, litmus, China rose petals (Gudhal), etc., are some naturally occurring indicators. Naturally occurring indicator litmus
A)
is extracted from lichens done
clear
B)
has available as red and blue litmus paper done
clear
C)
has purple colour in distilled water done
clear
D)
All the above are correct done
clear
View Solution play_arrow
-
question_answer56)
DIRECTIONS: Read the passage given below and answer the questions that follow. Passage - 4 Indicators change their colour when added to a solution containing an acidic or a basic substance. Turmeric, litmus, China rose petals (Gudhal), etc., are some naturally occurring indicators. Which of the following statements is correct?
A)
Colour of litmus changes from blue to red in an acidic solution. done
clear
B)
Colour of litmus changes from red to blue in an acidic solution. done
clear
C)
Colour of litmus changes from pink to colourless in an acidic solution. done
clear
D)
Colour of litmus changes from colourless to pink in a basic solution. done
clear
View Solution play_arrow
-
question_answer57)
DIRECTIONS: Read the passage given below and answer the questions that follow. Passage - 4 Indicators change their colour when added to a solution containing an acidic or a basic substance. Turmeric, litmus, China rose petals (Gudhal), etc., are some naturally occurring indicators. Which of the following is correct about the indicator China rose?
A)
It is a natural indicator. done
clear
B)
It gives green colour in an acidic solution. done
clear
C)
It gives magenta colour in a basic solution. done
clear
D)
All the above are correct. done
clear
View Solution play_arrow
-
question_answer58)
DIRECTIONS: Read the passage given below and answer the questions that follow. Passage - 5 Consider the following substances and on that basis answer the following questions: Lemon juice, tomato juice, lime water, vinegar, sugar solution, solution of common salt. The number of acidic substances is:
A)
1 done
clear
B)
2 done
clear
C)
3 done
clear
D)
4 done
clear
View Solution play_arrow
-
question_answer59)
DIRECTIONS: Read the passage given below and answer the questions that follow. Passage - 5 Consider the following substances and on that basis answer the following questions: Lemon juice, tomato juice, lime water, vinegar, sugar solution, solution of common salt. The number of basic substances is:
A)
1 done
clear
B)
2 done
clear
C)
3 done
clear
D)
4 done
clear
View Solution play_arrow
-
question_answer60)
DIRECTIONS: Read the passage given below and answer the questions that follow. Passage - 5 Consider the following substances and on that basis answer the following questions: Lemon juice, tomato juice, lime water, vinegar, sugar solution, solution of common salt. The number of neutral substances:
A)
1 done
clear
B)
2 done
clear
C)
3 done
clear
D)
4 done
clear
View Solution play_arrow
-
question_answer61)
DIRECTIONS: The questions in this segment consists of two statements, one labeled as "Assertion A" and the other labeled as "Reason R". You are to examine these two statements carefully and decide if the Assertion A and Reason R are individually true and if so, whether the reason is a correct explanation of the assertion. Select your answers to these items using codes given below. Assertion (A): Sulphuric acid is also known as oil of vitriol. Reason (R): It has oily appearance.
A)
Both A and R are true and R is the correct explanation of A. done
clear
B)
Both A and R are true but R is not the correct explanation of A. done
clear
C)
A is true but R is false. done
clear
D)
A is false but R is true. done
clear
View Solution play_arrow
-
question_answer62)
DIRECTIONS: The questions in this segment consists of two statements, one labeled as "Assertion A" and the other labeled as "Reason R". You are to examine these two statements carefully and decide if the Assertion A and Reason R are individually true and if so, whether the reason is a correct explanation of the assertion. Select your answers to these items using codes given below. Assertion (A): Rain containing excess amount of acid is called acid rain. Reason (R): Air pollutants like carbon dioxide, sulphur dioxide, nitrogen oxides dissolve in water to form carbonic acid, sulphuric acid and nitric acid.
A)
Both A and R are true and R is the correct explanation of A. done
clear
B)
Both A and R are true but R is not the correct explanation of A. done
clear
C)
A is true but R is false. done
clear
D)
A is false but R is true. done
clear
View Solution play_arrow
-
question_answer63)
DIRECTIONS: The questions in this segment consists of two statements, one labeled as "Assertion A" and the other labeled as "Reason R". You are to examine these two statements carefully and decide if the Assertion A and Reason R are individually true and if so, whether the reason is a correct explanation of the assertion. Select your answers to these items using codes given below. Assertion (A): Sodium hydroxide is an alkali. Reason (R): All bases are alkalies.
A)
Both A and R are true and R is the correct explanation of A. done
clear
B)
Both A and R are true but R is not the correct explanation of A. done
clear
C)
A is true but R is false. done
clear
D)
A is false but R is true. done
clear
View Solution play_arrow
-
question_answer64)
DIRECTIONS: The questions in this segment consists of two statements, one labeled as "Assertion A" and the other labeled as "Reason R". You are to examine these two statements carefully and decide if the Assertion A and Reason R are individually true and if so, whether the reason is a correct explanation of the assertion. Select your answers to these items using codes given below. Assertion (A): Toothpaste neutralised the acid produced by fermentation of food particles in mouth. Reason (R): Toothpaste is alkaline in nature.
A)
Both A and R are true and R is the correct explanation of A. done
clear
B)
Both A and R are true but R is not the correct explanation of A. done
clear
C)
A is true but R is false. done
clear
D)
A is false but R is true. done
clear
View Solution play_arrow
-
question_answer65)
DIRECTIONS: The questions in this segment consists of two statements, one labeled as "Assertion A" and the other labeled as "Reason R". You are to examine these two statements carefully and decide if the Assertion A and Reason R are individually true and if so, whether the reason is a correct explanation of the assertion. Select your answers to these items using codes given below. Assertion (A): Sugar is a neutral substance. Reason (R): A sugar solution turns red litmus blue.
A)
Both A and R are true and R is the correct explanation of A. done
clear
B)
Both A and R are true but R is not the correct explanation of A. done
clear
C)
A is true but R is false. done
clear
D)
A is false but R is true. done
clear
View Solution play_arrow
-
question_answer66)
DIRECTIONS: The questions in this segment consists of two statements, one labeled as "Assertion A" and the other labeled as "Reason R". You are to examine these two statements carefully and decide if the Assertion A and Reason R are individually true and if so, whether the reason is a correct explanation of the assertion. Select your answers to these items using codes given below. Assertion (A): Acids are sour in taste. Reason (R): Bases are soapy to touch and bitter in taste.
A)
Both A and R are true and R is the correct explanation of A. done
clear
B)
Both A and R are true but R is not the correct explanation of A. done
clear
C)
A is true but R is false. done
clear
D)
A is false but R is true. done
clear
View Solution play_arrow
-
question_answer67)
DIRECTIONS: Read the following two statements carefully and choose the correct options. (i) Orange juice turns blue litmus red (ii) Sting of an ant contains acetic acid
A)
Statement (i) is correct while statement (ii) is incorrect done
clear
B)
Statement (ii) is correct while statement (i) is incorrect. done
clear
C)
Both statements are correct done
clear
D)
Both statements are incorrect. done
clear
View Solution play_arrow
-
question_answer68)
DIRECTIONS: Read the following two statements carefully and choose the correct options. (i) Baking soda is a mild base that is used in toothpastes and mouthwashes to neutralize acids, which can produce unpleasant odours. (ii) An alkali is a water soluble acid.
A)
Statement (i) is correct while statement (ii) is incorrect done
clear
B)
Statement (ii) is correct while statement (i) is incorrect. done
clear
C)
Both statements are correct done
clear
D)
Both statements are incorrect. done
clear
View Solution play_arrow
-
question_answer69)
DIRECTIONS: Read the following two statements carefully and choose the correct options. (i) All salts are neutral in nature. (ii) A solution of sodium chloride salt in water does not change the colour of any litmus paper.
A)
Statement (i) is correct while statement (ii) is incorrect done
clear
B)
Statement (ii) is correct while statement (i) is incorrect. done
clear
C)
Both statements are correct done
clear
D)
Both statements are incorrect. done
clear
View Solution play_arrow
-
question_answer70)
DIRECTIONS: Read the following two statements carefully and choose the correct options. (i) Alkalies are sour to taste. (ii) Mineral acids change to organic acids on addition of water.
A)
Statement (i) is correct while statement (ii) is incorrect done
clear
B)
Statement (ii) is correct while statement (i) is incorrect. done
clear
C)
Both statements are correct done
clear
D)
Both statements are incorrect. done
clear
View Solution play_arrow
-
question_answer71)
DIRECTIONS: On the basis of following diagram/ picture answer the questions given below: Which of the following is the source of tartaric acid.
A)
B)
C)
D)
View Solution play_arrow
-
question_answer72)
DIRECTIONS: On the basis of following diagram/ picture answer the questions given below: Which of the following will change red litmus paper to blue?
A)
B)
C)
D)
Both (a) and (b) done
clear
View Solution play_arrow
-
question_answer73)
A blue litmus paper was first dipped I dilute hydrochloric acid and then in dilute sodium hydroxide solution. It was observed that the colour of the litmus paper:
A)
changed to red done
clear
B)
changed first to red and then to blue done
clear
C)
changed blue to colourless done
clear
D)
remained blue in both solutions. done
clear
View Solution play_arrow
-
question_answer74)
If you perform an experiment bare handed and you realise that your palm has become slippery and slimy. The most probable reason for this is that you have dropped:
A)
sodium hydroxide on your hands. done
clear
B)
hydrochloric acid on your hands. done
clear
C)
sodium chloride on your hands. done
clear
D)
None of these done
clear
View Solution play_arrow
-
question_answer75)
To test a solution in container 'A.' You dipped the filter paper in this solution and then in lime water. The colour of filter paper after dipping in lime water becomes brownish red. It shows that the solution in container 'A' was:
A)
Red litmus solution done
clear
B)
Methyl red solution done
clear
C)
Turmeric solution done
clear
D)
Phenolphthalein solution done
clear
View Solution play_arrow
-
question_answer76)
Which is responsible for the damaging effects caused by acid rain?
A)
Carbonic acid done
clear
B)
Sulphuric acid done
clear
C)
Nitric acid done
clear
D)
All of these done
clear
View Solution play_arrow
-
question_answer77)
You dissolved a sample, in distilled water and then added a drop of this solution to a test-tube containing blue litmus solution which changes to red. It shows that the sample given to you is of:
A)
soap done
clear
B)
tomato juice done
clear
C)
baking soda done
clear
D)
sugar done
clear
View Solution play_arrow
-
question_answer78)
When few drops of phenolphthalein are added to sample 'A' it turned pink. To this pink coloured solution we added a few drops of sample 'B'. With continuous stirring the pink colour disappears. Samples 'A' and 'B' are:
A)
sample 'A' is acidic and sample 'B' is basic. done
clear
B)
sample 'A' is basic and sample 'B' is acidic. done
clear
C)
sample 'A' is basic and sample 'B' is neutral. done
clear
D)
sample 'A' is acidic and sample 'B' is neutral. done
clear
View Solution play_arrow
-
question_answer79)
You are provided with two solutions 'X' and 'Y?. The colour of turmeric stains on your clothes on coming in contact with solution 'X' changes to brick red and when in contact with solution 'Y' changes to yellow. The solution 'X' and 'Y' provided to you are respectively:
A)
Soap solution and lemon juice done
clear
B)
Lemon juice and soap solution done
clear
C)
Lemon juice and vinegar done
clear
D)
Soap solution and sugar solution done
clear
View Solution play_arrow
-
question_answer80)
Following observation were recorded upon performing experiments with sample 'A' and sample 'B'. (i) When 2-3 drops of phenolphthalein are added to sample 'A' it becomes pink. (ii) When equal quantities of two samples are added in a test-tube, the test-tube becomes hot.
A)
sample 'A' is basic done
clear
B)
sample 'B' is acidic done
clear
C)
(ii) observation confirms neutralisation done
clear
D)
All the above are correct done
clear
View Solution play_arrow
-
question_answer81)
............... is an acid-base indicator, its colour is yellow in basic medium and pink in acidic medium.
A)
Phenolphthalein done
clear
B)
Methyl orange done
clear
C)
Litmus done
clear
D)
Both (a) and (b) are correct done
clear
View Solution play_arrow
-
question_answer82)
............... is used to neutralize the acidic effect of ant bite by rubbing it at the point of ant bite.
A)
Moist baking soda done
clear
B)
Calamine solution done
clear
C)
Both (a) and (b) done
clear
D)
Calcium carbonate done
clear
View Solution play_arrow
-
question_answer83)
............... is used to treat a soil that is acidic and min which plants do not grow well.
A)
Quicklime done
clear
B)
Slaked lime done
clear
C)
Calcium oxide done
clear
D)
Any one of (a), (b) or (c) done
clear
View Solution play_arrow
-
question_answer84)
Salts are compounds:
A)
having a bitter taste done
clear
B)
having a sour taste done
clear
C)
obtained as a result of neutralization reaction between acid and base. done
clear
D)
All the above are correct done
clear
View Solution play_arrow
-
question_answer85)
Many salts absorb water (moisture) from the atmosphere. This property is called
A)
hydration done
clear
B)
dehydration done
clear
C)
crystallization done
clear
D)
decantation done
clear
View Solution play_arrow
-
question_answer86)
The substances which form hydronium ions \[\left( {{\text{H}}_{\text{3}}}{{\text{O}}^{\text{+}}} \right)\] in water are called
A)
acids done
clear
B)
bases done
clear
C)
alkalies done
clear
D)
None of these done
clear
View Solution play_arrow
-
question_answer87)
The compounds which produce hydroxyl ions\[\text{(O}{{\text{H}}^{\text{-}}}\text{)}\] in water are called
A)
alkalies done
clear
B)
bases done
clear
C)
acids done
clear
D)
None of these done
clear
View Solution play_arrow
-
question_answer88)
Metallic oxides dissolve in water to form
A)
acidic solution done
clear
B)
neutral solution done
clear
C)
basic solution done
clear
D)
None of these done
clear
View Solution play_arrow
-
question_answer89)
Non-metallic oxides react with water to form
A)
alkaline solution done
clear
B)
acidic solution done
clear
C)
neutral solution done
clear
D)
None of these done
clear
View Solution play_arrow
-
question_answer90)
A solution turns red litmus blue, its pH is likely to be
A)
1 done
clear
B)
4 done
clear
C)
5 done
clear
D)
10 done
clear
View Solution play_arrow
-
question_answer91)
The acid present in tea is:
A)
tannic done
clear
B)
lactic done
clear
C)
tartaric done
clear
D)
citric done
clear
View Solution play_arrow
-
question_answer92)
Acid reacts with metal to form:
A)
\[\text{salt+C}{{\text{O}}_{\text{2}}}\] done
clear
B)
salt + water done
clear
C)
\[\text{salt+}{{\text{O}}_{\text{2}}}\] done
clear
D)
\[\text{salt+}{{\text{H}}_{\text{2}}}\] done
clear
View Solution play_arrow
-
question_answer93)
Manisha took a little bit of soil from her garden and mixed it with water. When she dipped a blue litmus in it, the litmus turned red- By adding which of the following to her garden will she get better plant growth?
A)
Hydrochloric acid done
clear
B)
Slaked lime done
clear
C)
Water done
clear
D)
Salt done
clear
View Solution play_arrow
-
question_answer94)
Common name of \[{{\text{H}}_{\text{2}}}\text{S}{{\text{O}}_{\text{4}}}\] is:
A)
oil of vitriol done
clear
B)
muriatic acid done
clear
C)
blue vitriol done
clear
D)
green vitriol done
clear
View Solution play_arrow
-
question_answer95)
Common name of copper sulphate is:
A)
chalk done
clear
B)
quicklime done
clear
C)
nitre done
clear
D)
blue vitriol done
clear
View Solution play_arrow
-
question_answer96)
The waste from a paper factory contains high amount of hydrochloric acid. How should we treat the waste, before disposal, to make it safe?
A)
Treat it with chemicals containing sodium hydroxide done
clear
B)
Treat it with chemicals containing sulphuric acid done
clear
C)
Treat it with chemicals containing sodium chloride done
clear
D)
Treat it with pure hydrochloric acid done
clear
View Solution play_arrow
-
question_answer97)
Read the following statements: (i) It is a reaction between an acid and a base. (ii) Salt and water are produced in this reaction. (iii) It is an exothermic reaction. Which reaction is being referred to?
A)
Displacement reaction done
clear
B)
Neutralization reaction done
clear
C)
Redox reaction done
clear
D)
Decomposition reaction done
clear
View Solution play_arrow
-
question_answer98)
Study the table carefully
| | Sample | Blue litmus to red | Red litmus to blue |
| (i) | Tamarind juice | √ | × |
| (ii) | Sugar syrup | × | √ |
| (iii) | Lime water | × | √ |
| (iv) | Soap solution | √ | × |
Which of the above are correctly matched?
A)
(i) & (iii) done
clear
B)
(ii) & (iv) done
clear
C)
(i), (ii) & (iii) done
clear
D)
(i), (iii) & (iv) done
clear
View Solution play_arrow
-
question_answer99)
The two types of litmus paper are:
A)
blue and red done
clear
B)
blue and yellow done
clear
C)
red and green done
clear
D)
yellow and red done
clear
View Solution play_arrow
-
question_answer100)
In acids, methyl orange turns:
A)
yellow done
clear
B)
green done
clear
C)
red done
clear
D)
white done
clear
View Solution play_arrow
-
question_answer101)
In bases, methyl orange turns:
A)
green done
clear
B)
black done
clear
C)
red done
clear
D)
yellow done
clear
View Solution play_arrow
-
question_answer102)
Phenolphthalein turns_______ in acidic and neutral solutions.
A)
colourless done
clear
B)
pink done
clear
C)
red done
clear
D)
green done
clear
View Solution play_arrow
-
question_answer103)
Lime water is a solution of:
A)
\[\text{Ca(OH}{{\text{)}}_{\text{2}}}\]in water done
clear
B)
\[\text{CaC}{{\text{l}}_{\text{2}}}\] in water done
clear
C)
\[\text{NaOH}\] in water done
clear
D)
\[\text{NaCl}\] in water done
clear
View Solution play_arrow
-
question_answer104)
Silver and gold are purified with:
A)
nitric acid done
clear
B)
\[\text{HCl}\] done
clear
C)
acetic acid done
clear
D)
sulphuric acid done
clear
View Solution play_arrow
-
question_answer105)
Soluble bases are called:
A)
salts done
clear
B)
acids done
clear
C)
alkalies done
clear
D)
All the three done
clear
View Solution play_arrow
-
question_answer106)
Caustic soda is the common name for:
A)
\[\text{Mg(OH}{{\text{)}}_{\text{2}}}\] done
clear
B)
\[\text{KOH}\] done
clear
C)
\[\text{Ca(OH}{{\text{)}}_{\text{2}}}\] done
clear
D)
\[\text{NaOH}\] done
clear
View Solution play_arrow
-
question_answer107)
Calcium hydroxide (slaked lime) is used in:
A)
plastics and dyes done
clear
B)
fertilizers done
clear
C)
antacid done
clear
D)
whitewashing done
clear
View Solution play_arrow
-
question_answer108)
DIRECTIONS: Match Column-I with Column-II and select the correct answer using the codes given below the columns.
| Column - I (Common name) | Column - II (Chemical name/ Chemical present) |
| (A) Slaked lime | (p) Zinc carbonate |
| (B) Baking soda | (q) Calcium hydroxide |
| (C) Calamine solution | (r) Sodium bicarbonate |
| (D) Milk of Magnesia | (s) Magnesium hydroxide |
A)
(A) \[\to \] (p), (B) \[\to \] (q), (C) \[\to \] (r), (D) \[\to \] (s) done
clear
B)
(A) \[\to \] (q), (B) \[\to \] (r), (C) \[\to \] (p), (D) \[\to \] (s) done
clear
C)
(A) \[\to \] (r), (B) \[\to \] (s), (C) \[\to \] (q), (D) \[\to \] (p) done
clear
D)
(A) \[\to \] (s), (B) \[\to \] (p), (C) \[\to \] (q), (D) \[\to \] (r) done
clear
View Solution play_arrow
-
question_answer109)
DIRECTIONS: Match Column-I with Column-II and select the correct answer using the codes given below the columns.
| Column - I | Column - II |
| (A) Oxalic acid | (p) As an eye-wash, antiseptic and grain preservation |
| (B) Nitric acid | (q) For making explosives |
| (C) Boric acid | (r) In food preservation |
| (D) Benzoic acid | (s) As a constituent of ink stain remover |
A)
(A) \[\to \] (s), (B) \[\to \] (q), (C) \[\to \] (r), (D) \[\to \] (p) done
clear
B)
(A) \[\to \] (q), (B) \[\to \] (s), (C) \[\to \] (p), (D) \[\to \] (r) done
clear
C)
(A) \[\to \] (r), (B) \[\to \] (q), (C) \[\to \] (p), (D) \[\to \] (s) done
clear
D)
(A) \[\to \] (s), (B) \[\to \] (q), (C) \[\to \] (p), (D) \[\to \] (r) done
clear
View Solution play_arrow
-
question_answer110)
DIRECTIONS: Match Column-I with Column-II and select the correct answer using the codes given below the columns.
| Column - I | Column - II |
| (A) Fertilizer | (p) King of chemicals |
| (B) Sulphuric acid | (q) Basic |
| (C) Lime water | (r) Magnesium hydroxide |
| (D) Milk of magnesia | (s) Potassium nitrate |
A)
(A) \[\to \] (s), (B) \[\to \] (p), (C) \[\to \] (q), (D) \[\to \] (r) done
clear
B)
(A) \[\to \] (q), (B) \[\to \] (s), (C) \[\to \] (p), (D) \[\to \] (r) done
clear
C)
(A) \[\to \] (r), (B) \[\to \] (p), (C) \[\to \] (s), (D) \[\to \] (q) done
clear
D)
(A) \[\to \] (s), (B) \[\to \] (r), (C) \[\to \] (q), (D) \[\to \] (p) done
clear
View Solution play_arrow
-
question_answer111)
DIRECTIONS: Read the passage given below and answer the questions that follow. Passage - 1 The rain which contains a higher level of acid than normal is called acid rain. Acidic gases like sulphur dioxide, nitrogen dioxide and carbon dioxide which are released as pollutants during the burning of various types of faels dissolve in rain drops and when these rain drops fall as rain on earth, we call it acid rain. Nonmetallic oxides react with water to form:
A)
acids done
clear
B)
bases done
clear
C)
salt done
clear
D)
None of these done
clear
View Solution play_arrow
-
question_answer112)
DIRECTIONS: Read the passage given below and answer the questions that follow. Passage - 1 The rain which contains a higher level of acid than normal is called acid rain. Acidic gases like sulphur dioxide, nitrogen dioxide and carbon dioxide which are released as pollutants during the burning of various types of faels dissolve in rain drops and when these rain drops fall as rain on earth, we call it acid rain. \[\text{C}{{\text{O}}_{\text{2}}}\] dissolves in a water to form:
A)
\[\text{C+}{{\text{H}}_{\text{2}}}+{{\text{O}}_{\text{2}}}\] done
clear
B)
\[\text{C}{{\text{H}}_{\text{4}}}\] done
clear
C)
\[\text{CO}\] done
clear
D)
carbonic acid done
clear
View Solution play_arrow
-
question_answer113)
DIRECTIONS: Read the passage given below and answer the questions that follow. Passage - 1 The rain which contains a higher level of acid than normal is called acid rain. Acidic gases like sulphur dioxide, nitrogen dioxide and carbon dioxide which are released as pollutants during the burning of various types of faels dissolve in rain drops and when these rain drops fall as rain on earth, we call it acid rain. Sulphuric acid is formed when ____ reacts with water.
A)
\[\text{S}{{\text{O}}_{\text{2}}}\] done
clear
B)
S done
clear
C)
\[\text{S}{{\text{O}}_{\text{3}}}\] done
clear
D)
\[{{\text{O}}_{\text{2}}}\] done
clear
View Solution play_arrow
-
question_answer114)
DIRECTIONS: Read the passage given below and answer the questions that follow. Passage - 1 The rain which contains a higher level of acid than normal is called acid rain. Acidic gases like sulphur dioxide, nitrogen dioxide and carbon dioxide which are released as pollutants during the burning of various types of faels dissolve in rain drops and when these rain drops fall as rain on earth, we call it acid rain. What is the main cause of acid rain?
A)
Pollution done
clear
B)
Deforestation done
clear
C)
Excessive rains done
clear
D)
All factors are equally effective done
clear
View Solution play_arrow
-
question_answer115)
DIRECTIONS: Read the passage given below and answer the questions that follow. Passage - 2 Three different substances were taken and tested with litmus paper. The results are given below. Based on the results answer the questions 43 to 48.
| Type of Litmus | X | Y | Z |
| Red litmus | turns blue | no change | no change |
| Blue litmus | no litmus | turns red | no change |
What could be the substance X?
A)
An acid done
clear
B)
A base done
clear
C)
Water done
clear
D)
Salt done
clear
View Solution play_arrow
-
question_answer116)
DIRECTIONS: Read the passage given below and answer the questions that follow. Passage - 2 Three different substances were taken and tested with litmus paper. The results are given below. Based on the results answer the questions 43 to 48.
| Type of Litmus | X | Y | Z |
| Red litmus | turns blue | no change | no change |
| Blue litmus | no litmus | turns red | no change |
What could be the substance Y?
A)
An acid done
clear
B)
A base done
clear
C)
Water done
clear
D)
Salt done
clear
View Solution play_arrow
-
question_answer117)
DIRECTIONS: Read the passage given below and answer the questions that follow. Passage - 2 Three different substances were taken and tested with litmus paper. The results are given below. Based on the results answer the questions 43 to 48.
| Type of Litmus | X | Y | Z |
| Red litmus | turns blue | no change | no change |
| Blue litmus | no litmus | turns red | no change |
What could be the substance Z?
A)
Water done
clear
B)
Salt done
clear
C)
Either A or B done
clear
D)
Either an acid or a base done
clear
View Solution play_arrow
-
question_answer118)
DIRECTIONS: Read the passage given below and answer the questions that follow. Passage - 2 Three different substances were taken and tested with litmus paper. The results are given below. Based on the results answer the questions 43 to 48.
| Type of Litmus | X | Y | Z |
| Red litmus | turns blue | no change | no change |
| Blue litmus | no litmus | turns red | no change |
How can you obtain the substance Z?
A)
By the reaction of X and Y done
clear
B)
By dissolving X in water done
clear
C)
By dissolving Y in water done
clear
D)
Cannot be said done
clear
View Solution play_arrow
-
question_answer119)
DIRECTIONS: Read the passage given below and answer the questions that follow. Passage - 2 Three different substances were taken and tested with litmus paper. The results are given below. Based on the results answer the questions 43 to 48.
| Type of Litmus | X | Y | Z |
| Red litmus | turns blue | no change | no change |
| Blue litmus | no litmus | turns red | no change |
Which of the following could be the substance Z?
A)
\[{{\text{H}}_{\text{2}}}\text{S}{{\text{O}}_{\text{4}}}\] done
clear
B)
\[\text{HN}{{\text{O}}_{\text{3}}}\] done
clear
C)
\[\text{KOH}\] done
clear
D)
\[{{\text{H}}_{\text{2}}}\text{O}\] done
clear
View Solution play_arrow
-
question_answer120)
DIRECTIONS: Read the passage given below and answer the questions that follow. Passage - 2 Three different substances were taken and tested with litmus paper. The results are given below. Based on the results answer the questions 43 to 48.
| Type of Litmus | X | Y | Z |
| Red litmus | turns blue | no change | no change |
| Blue litmus | no litmus | turns red | no change |
How will the substance X behave with phenolphthalein?
A)
It turns pink done
clear
B)
It remains colorless done
clear
C)
It turns blue done
clear
D)
It turns red done
clear
View Solution play_arrow
-
question_answer121)
DIRECTIONS: The questions in this segment consists of two statements, one labelled as ?Assertion A? and the other labelled as ?Reason R?. You are to examine these two statements carefully and decide if the Assertion A and Reason R are individually true and if so, whether the reason is a correct explanation of the assertion. Select your answers to these items using codes given below. Assertion (A): Turmeric is a neutral indicator. Reason (R): Turmeric acquires orange colour in alkaline solution.
A)
Both A and R are true and R is the correct explanation of A. done
clear
B)
Both A and R are true but R is not the correct explanation of A. done
clear
C)
A is true but R is false. done
clear
D)
A is false but R is true. done
clear
View Solution play_arrow
-
question_answer122)
DIRECTIONS: The questions in this segment consists of two statements, one labelled as ?Assertion A? and the other labelled as ?Reason R?. You are to examine these two statements carefully and decide if the Assertion A and Reason R are individually true and if so, whether the reason is a correct explanation of the assertion. Select your answers to these items using codes given below. Assertion (A): Salts are the products of an acid- base reaction. Reason (R): Salt may be acidic or basic.
A)
Both A and R are true and R is the correct explanation of A. done
clear
B)
Both A and R are true but R is not the correct explanation of A. done
clear
C)
A is true but R is false. done
clear
D)
A is false but R is true. done
clear
View Solution play_arrow
-
question_answer123)
DIRECTIONS: The questions in this segment consists of two statements, one labelled as ?Assertion A? and the other labelled as ?Reason R?. You are to examine these two statements carefully and decide if the Assertion A and Reason R are individually true and if so, whether the reason is a correct explanation of the assertion. Select your answers to these items using codes given below. Assertion (A): Neutralization reaction is accompanied by evolution of heat. Reason (R): Neutralization reaction is a reaction between an acid and a base to form salt and water.
A)
Both A and R are true and R is the correct explanation of A. done
clear
B)
Both A and R are true but R is not the correct explanation of A. done
clear
C)
A is true but R is false. done
clear
D)
A is false but R is true. done
clear
View Solution play_arrow
-
question_answer124)
DIRECTIONS: The questions in this segment consists of two statements, one labelled as ?Assertion A? and the other labelled as ?Reason R?. You are to examine these two statements carefully and decide if the Assertion A and Reason R are individually true and if so, whether the reason is a correct explanation of the assertion. Select your answers to these items using codes given below. Assertion (A): The substance which shows the nature of a solution as acidic or basic with the change in colour are called indicators. Reason (R): Red litmus turns blue in a solution containing vitamin 'C'.
A)
Both A and R are true and R is the correct explanation of A. done
clear
B)
Both A and R are true but R is not the correct explanation of A. done
clear
C)
A is true but R is false. done
clear
D)
A is false but R is true. done
clear
View Solution play_arrow
-
question_answer125)
DIRECTIONS: Read the following three statements and choose the correct answer. (i) The salt formed by the neutralization of \[\text{NaOH}\] with \[\text{HCl}\] is basic in nature. (ii) Milk of magnesia contains magnesium hydroxide. (iii) Bases have pH from 1 to 7.
A)
Statement (i) and (iii) are incorrect but (ii) is correct done
clear
B)
Statement (i) and (ii) are incorrect but (iii) is correct done
clear
C)
All statements are correct done
clear
D)
All statements are incorrect done
clear
View Solution play_arrow
-
question_answer126)
DIRECTIONS: Read the following three statements and choose the correct answer. (i) Phenolphthalein is a natural indicator. (ii) Sodium carbonate is commonly known as caustic soda. (iii) Universal indicator is made up of a mixture of different dyes.
A)
Statement (i) and (iii) are incorrect but (ii) is correct done
clear
B)
Statement (i) and (ii) are incorrect but (iii) is correct done
clear
C)
All statements are correct done
clear
D)
All statements are incorrect done
clear
View Solution play_arrow
-
question_answer127)
DIRECTIONS: Read the following three statements and choose the correct answer. (i) If the soil is basic then compost is added to it to neutralize the basic nature of soil. (ii) China rose indicator changes colour to dark pink in acidic media and green in basic media. (iii) Tomato contains acetic acid.
A)
Statement (i) and (iii) are incorrect but (ii) is correct done
clear
B)
Statement (i) and (ii) are incorrect but (iii) is correct done
clear
C)
All statements are correct done
clear
D)
All statements are incorrect done
clear
View Solution play_arrow
-
question_answer128)
DIRECTIONS: On the basis of following diagram/picture answer the questions given below: In an experiment about 10 ml of sodium hydroxide solution was taken in a beaker 'A'. To it was added a few drops of red litmus solution which turned blue. This solution was then taken in beaker 'B' and vinegar was added to it drop by drop with continuous stirring. It was observed that the colour of solution in beaker 'B' has changed to red. This experiment shows:

A)
Saponification done
clear
B)
Acidification done
clear
C)
Neutralization done
clear
D)
Both (b) and (c) done
clear
View Solution play_arrow
-
question_answer129)
DIRECTIONS: On the basis of following diagram/picture answer the questions given below: Study the following chart and Identify 'A' and 'B'.
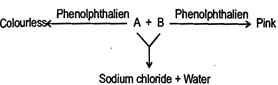
A B
A)
Sodium hydroxide Sodium chloride done
clear
B)
Hydrochloric acid Sodium hydroxide done
clear
C)
Sodium hydroxide acid Aluminum hydroxide done
clear
D)
Hydrochloric acid Sodium chloride done
clear
View Solution play_arrow
-
question_answer130)
DIRECTIONS: On the basis of following diagram/picture answer the questions given below: In three test- tubes 'A', 'B', 'C' with solutions of different substances were added a few drops of China rose indicator. The colours shows in these test-tubes were magenta, colourless and green respectively Select the correct set indicating the substance whose solution are there in these test-tubes.

A)
B)
C)
D)
View Solution play_arrow
 done
clear
done
clear
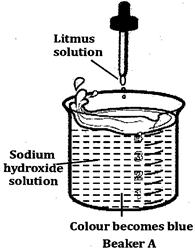
 done
clear
done
clear
 done
clear
done
clear
 done
clear
done
clear
 done
clear
done
clear
 done
clear
done
clear
 done
clear
done
clear
