-
question_answer1)
Which of the following does not represent characteristic of a good fuel?
A)
It should have a high calorific value. done
clear
B)
Its ignition temperature should be low but well above the room temperature. done
clear
C)
It should have a high rate of combustion. done
clear
D)
It should be fairly cheap and safe to handle and transport. done
clear
View Solution play_arrow
-
question_answer2)
How has the use of CNG in automobiles reduced the pollution in cities?
A)
It produces less amount of sulphur dioxide and chlorine. done
clear
B)
It does not leave any residue or smoke after burning in the engine. done
clear
C)
It produces large amount of carbon dioxide. done
clear
D)
It is used in compressed form. done
clear
View Solution play_arrow
-
question_answer3)
Read the given statements and select the correct option.
| Statement 1: Phosphorus burns in air at room temperature. |
| Statement 2: It is an example of spontaneous combustion. |
A)
Both statements 1 and 2 are true and statement 2 is the correct explanation of statement 1. done
clear
B)
Both statements 1 and 2 are true but statement 2 is not the correct explanation of statement 1. done
clear
C)
Statement 1 is true and statement 2 is false. done
clear
D)
Both statements 1 and 2 are false. done
clear
View Solution play_arrow
-
question_answer4)
Colours of flame produced by kerosene lamp, candle and Bunsen burner are respectively
A)
Blue, yellow and yellow done
clear
B)
Yellow, yellow and blue done
clear
C)
Orange, blue and yellow done
clear
D)
Yellow, orange and yellow. done
clear
View Solution play_arrow
-
question_answer5)
Global warming leads to a rise in the sea level causing floods in the coastal areas. One of the main reasons of global warming is
A)
Combustion of fuels releases sulphur dioxide which increases the temperature done
clear
B)
Combustion of fuels releases carbon monoxide which decreases the temperature done
clear
C)
Combustion of fuels releases unburnt carbon particles which causes decrease in temperature done
clear
D)
Combustion of fuels releases carbon dioxide which causes increase in temperature. done
clear
View Solution play_arrow
-
question_answer6)
Read the given statements and select the correct options.
| Statement 1: Kerosene oil and wood do not catch fire on their own at room temperature. |
| Statement 2: A combustible material cannot catch fire as long as its temperature is lower than its ignition temperature. |
A)
Both statements 1 and 2 are true and statement 2 is the correct explanation of statement 1. done
clear
B)
Both statements 1 and 2 are true but statement 2 is not the correct explanation of statement 1. done
clear
C)
Statement 1 is true and statement 2 is false. done
clear
D)
Both statements 1 and 2 are false. done
clear
View Solution play_arrow
-
question_answer7)
In an experiment, 4.5 kg of a fuel was completely burnt. The heat produced was 180,000 kJ. The calorific value of the fuel is
A)
180,000 kJ done
clear
B)
40,000 kJ done
clear
C)
40,000 kJ/kg done
clear
D)
180,000 kJ/kg done
clear
View Solution play_arrow
-
question_answer8)
What is the principle of soda fire extinguisher?
A)
Carbon dioxide produced cuts off the supply of oxygen by covering the fire like a blanket. done
clear
B)
Water produced cuts off the supply of oxygen by covering the fire. done
clear
C)
Acid present in the container reacts with fire. done
clear
D)
Nitrogen gas produced cuts off the supply of oxygen by covering the fire. done
clear
View Solution play_arrow
-
question_answer9)
Which of the following reactions takes place in soda-acid type fire extinguisher?
A)
\[\text{CaC}{{\text{O}}_{3}}\xrightarrow{\Delta }CaO+C{{O}_{2}}\] done
clear
B)
\[2NaHC{{O}_{3}}\xrightarrow{\Delta }N{{a}_{2}}C{{O}_{3}}+{{H}_{2}}O+C{{O}_{2}}\] done
clear
C)
\[\begin{align} & 2NaHC{{O}_{3}}+{{H}_{2}}S{{O}_{4}}\xrightarrow{{}}N{{a}_{2}}S{{O}_{4}} \\ & +2{{H}_{2}}O+2C{{O}_{2}} \\ \end{align}\] done
clear
D)
\[\begin{align} & \text{CaC}{{\text{O}}_{\text{3}}}\text{+}{{\text{H}}_{\text{2}}}\text{S}{{\text{O}}_{\text{4}}}\xrightarrow{{}}\text{CaS}{{\text{O}}_{\text{4}}} \\ & \text{+}{{\text{H}}_{\text{2}}}\text{O+C}{{\text{O}}_{\text{2}}} \\ \end{align}\] done
clear
View Solution play_arrow
-
question_answer10)
Match column I with column II and select the correct option from the codes given below.
| Column I | Column II |
| P. Rusting | (i) Explosion |
| Q. Burning of Paper | (ii) Spontaneous combustion |
| R. Sodium | (iii) Rapid combustion |
| S. Fire crackers | (iv) Slow combustion |
A)
P-(ii), Q-(iii), R-(i), S-(iv) done
clear
B)
P-(i), Q-(iii), R-(ii), S-(iv) done
clear
C)
P-(iii), Q-(ii), R-(i), S-(ii) done
clear
D)
P-(iv), Q-(iii), R-(ii), S-(i) done
clear
View Solution play_arrow
-
question_answer11)
Which of the following represents the correct increasing order of calorific value of the given fuels?
A)
Hydrogen < LPG < Biogas < Cow dung cake < coal done
clear
B)
Cow dung cake < Biogas < LPG < Coal < Hydrogen done
clear
C)
Cow dung cake < Coal < Biogas < LPG < Hydrogen done
clear
D)
Hydrogen < Cow dung cake < LPG < Biogas < Coal done
clear
View Solution play_arrow
-
question_answer12)
Liquids and gaseous fuels have more advantages over solid fuels. Some of the advantages are
| (i) Calorific value of liquid and gaseous fuels is higher than the solid fuels. |
| (ii) Liquids and gaseous fuels have higher ignition temperatures than the solid fuels. |
| (iii) Liquids and gaseous fuels are easier to store since solid fuels occupy lot of space. |
| (iv) Liquids and gaseous fuels burn completely, leaving no residue. |
The correct advantages are
A)
(i), (ii) and (iii) done
clear
B)
(i), (iii) and (iv) done
clear
C)
(ii), (iii) and (iv) done
clear
D)
(i), (ii) and (iv). done
clear
View Solution play_arrow
-
question_answer13)
A spoon is kept in contact with ice cubes for some time. Later, the same spoon was held over the flame of a small candle. The observation is shown in the figure.
|
What do you infer from the given figure?
|
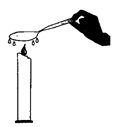
|
A)
Spoon is an inflammable substance. done
clear
B)
Burning of candle is a spontaneous process. done
clear
C)
Carbon dioxide is a product of combustion. done
clear
D)
Water vapour is a product of combustion. done
clear
View Solution play_arrow
-
question_answer14)
Which of the following statements are true?
| I. Yellow flames are ideal for heating. |
| II. The substances which vaporise during burning give flames. |
| III. Luminous zone contains unburnt carbon particles. |
| IV. The non-luminous zone has highest temperature. |
A)
I, II and III done
clear
B)
I, II and IV done
clear
C)
II, III and IV done
clear
D)
I, III and IV done
clear
View Solution play_arrow
-
question_answer15)
Calorific values of some fuels are given:
| Fuel | Calorific value (kJ/kg) |
| Coal | 25000-33000 |
| Diesel | 45000 |
| LPG | 55000 |
| CNG | 50000 |
On the basis of this data, find out the correct order of efficiency of fuels.
A)
LPG > CNG > Diesel > Coal done
clear
B)
Coal > Diesel > LPG > CNG done
clear
C)
Diesel > CNG > Coal > LPG done
clear
D)
CNG > LPG > Diesel > Coal done
clear
View Solution play_arrow
-
question_answer16)
|
Different zones of a candle flame are marked by the letters P, Q, R and S.
|
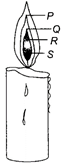
|
Which of the following statements are correct?
| (i) P is the luminous zone and is the hottest part of candle flame. |
| (ii) In zone Q, there is inadequate supply of oxygen. |
| (in) Zone R contains unburnt wax vapours produced by melting of wax. |
| (iv) In S zone, carbon monoxide burns with a blue flame. |
A)
(i) and (ii) only done
clear
B)
(ii), (iii) and (iv) only done
clear
C)
(ii) and (iv) only done
clear
D)
(i), (ii) and (iii) only done
clear
View Solution play_arrow
-
question_answer17)
Mark the correct statements) among the following.
| I. Water is a very good fire extinguisher as it cools down the fuels such as wood below its ignition temperature. |
| II. Water is a very good fire extinguisher for burning oils and electrical equipment?s. |
| III. Sand and soil may act as a good fire extinguisher for burning oils. |
A)
I only done
clear
B)
II only done
clear
C)
III only done
clear
D)
I and III only done
clear
View Solution play_arrow
-
question_answer18)
Different methods of extinguishing a fire involve the following :
| I. Removing the combustible substance. |
| II. Cooling the substance to a temperature below its ignition temperature. |
| III. Cutting off the supply of air. |
Which of the following statements is correct?
A)
Method I is suitable in case of a building on fire. done
clear
B)
Method II involves water which works when wood or paper are on fire. done
clear
C)
Method III involves carbon dioxide gas which is lighter than oxygen and forms a blanket around the air. done
clear
D)
All of these done
clear
View Solution play_arrow
-
question_answer19)
Neha took a burning candle and covered it with an empty glass by putting it upside down. She observed that the candle burns for some time before it finally goes off. What is the reason for this?

A)
The candle keeps burning for some time as long as the air present in the glass is available to it and then it goes off. done
clear
B)
The ignition temperature of the candle becomes low due to glass cover hence it goes off. done
clear
C)
The candle keeps burning as long as the complete wax melts away and then goes off. done
clear
D)
Oxygen is produced when the candle burns. This oxygen keeps the candle burning for some time and then it goes off. done
clear
View Solution play_arrow
-
question_answer20)
Natural resources are classified as:
| P. Exhaustible |
| Q. Inexhaustible |
Some natural resources are
|
(i) Petrol, diesel
|
|
(ii) Coal, petroleum
|
|
(iii) Air, water
|
|
(iv) Cow dung cakes, sunlight
|
|
(v) Forests, minerals
|
Which of the following represents the correct match?
A)
B)
C)
D)
None of these is a correct match. done
clear
View Solution play_arrow
