question_answer 1)
Properties of substances W, X, Y and Z are listed below:
W: A liquid metal
X: A liquid non-metal
Y: Gaseous non-metal
Z: A non-metal which conducts electricity.
W, X, Y and Z are respectively
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Solution play_arrow
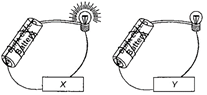
question_answer 2)
Materials 'X and 'Y' in the figure are
A)
B)
C)
D)
View Solution play_arrow
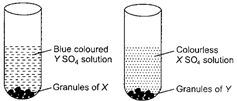
question_answer 3)
An unknown metal X when placed in copper sulphate solution, red brown pieces of copper fall to the bottom of the test tube.
When metal X is placed in aluminium sulphate solution, no change is observed. Metal X can be
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Solution play_arrow
question_answer 4)
Substance X reacts with dilute HCI violently, V reacts slowly while Z does not react at all. X, Y and Z could be respectively
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Solution play_arrow
question_answer 5)
Which of the following reactions are possible?
(i) \[\text{Mg+CuS}{{\text{O}}_{\text{4}}}\to \text{MgS}{{\text{O}}_{\text{4}}}\text{+Cu}\]
(ii) \[\text{Ca+MgS}{{\text{O}}_{\text{4}}}\to \text{CaS}{{\text{O}}_{\text{4}}}\text{+Mg}\]
(iii) \[\text{Cu+ZnS}{{\text{O}}_{\text{4}}}\to \text{CuS}{{\text{O}}_{\text{4}}}\text{+Zn}\]
(iv) \[\text{Fe+MgS}{{\text{O}}_{\text{4}}}\to \text{FeS}{{\text{O}}_{\text{4}}}\text{+Mg}\]
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Solution play_arrow
question_answer 6)
Match column I with column II and select the correct option from the codes given below.
Column I Column II A non-metal, essential for respiration (i) Graphite A non-metal, the source of energy in Sun (ii) Silicon A non-metal, good conductor of electricity (iii) Oxygen A non-metal, used in semiconductors (iv) Hydrogen
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Solution play_arrow
question_answer 7)
Which of the following statements is incorrect?
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Solution play_arrow
question_answer 8)
What will happen if you keep a solution of copper sulphate in an iron container for a few days? What is the reason for your observation?
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Solution play_arrow
question_answer 9)
Kanika takes a burning substance and collects the evolved vapours in a test tube, making sure that they do not escape.
A small amount of water is added to the test tube. To the solution obtained, a few drops of blue litmus solution are added. The litmus solution turns red. Which of the following could be the burning substance?
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Solution play_arrow
question_answer 10)
Vidisha placed a copper wire in silver nitrate solution as shown in the figure.
Which of the following represents the correct observation?
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Solution play_arrow
question_answer 11)
Read the following statements carefully and identify X, Y and Z respectively.
(i) X is stored under kerosene. (ii) Y catches fire on exposure and stored in water. (iii) Z reacts with steam.
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Solution play_arrow
question_answer 12)
Read the given statements and select the correct option.
Statement 1: Silver objects become green and lose their shine with the passage of time. Statement 2: Silver reacts with carbon dioxide and moisture present in the atmosphere.
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Solution play_arrow
question_answer 13)
Mr. Ramakant, a science teacher organised a quiz in the class. He stated few applications of non-metals and asked students to guess the names of the non-metals. Mark the correct option.
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Solution play_arrow
question_answer 14)
Read the given passage and fill in the blanks by choosing an appropriate option.
Non-metals find wide applications in industries. For example ___i___ is used in electrodes, ___ii___ is used in welding metals, ___iii___ is used to make antiseptic creams while ___iv___ is used as a preservative due to its inert nature.
A)
B)
C)
D)
View Solution play_arrow
question_answer 15)
The oxide of sulphur (which can be further oxidised) when dissolved in water gives
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Solution play_arrow
question_answer 16)
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Solution play_arrow
question_answer 17)
Observe the given figure carefully.
X and Y respectively are
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Solution play_arrow
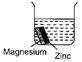
question_answer 18)
Aditya, a class 8 student investigated the reactivity of four metals, iron, copper, zinc and an unknown metal X. He arranged three experimental set-ups as shown in the diagram and observed the changes carefully.
If reactions occur in all the three beakers then, the metal X is
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Solution play_arrow
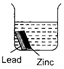
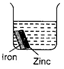
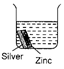
question_answer 19)
Each beaker contains two metal strips of same size fastened together and immersed in hydrochloric acid. After 5 minutes, which beaker will contain the least amount of zinc ions?
A)
B)
C)
D)
View Solution play_arrow
question_answer 20)
Fill in the blanks with the most appropriate option.
'W' is very reactive metal, reacts vigorously with oxygen and water therefore, stored in ___(1)___.
'X' is non-metal, soft and dull and forms ___(2)___ oxides with oxygen.
'Y' is very reactive ___(3)___, catches fire if exposed to air, therefore, stored in water.
'Z' does not react with dilute hydrochloric acid even on heating but it reacts with sulphuric acid. When it is exposed to moist air for long, it acquires a dull ___(4)___ coating.
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Solution play_arrow
 done
clear
done
clear
 done
clear
done
clear
 done
clear
done
clear
 done
clear
done
clear
