-
question_answer1)
| Direction: Q. 1 to 5 |
| Read the following and answer questions from [1] to [5]. |
| Pure substances are classified as elements or compounds. An element is a substance that contains only one type of atom. Also, an element cannot be broken down or transformed into a new substance even by using some physical or chemical methods. |
| A compound is a substance composed of two or more different types of elements, chemically combined in a fixed proportion. |
| The constitutents cannot be separated by simple physical methods. |
| However, these substances can be broken down into separate elements by chemical methods. |
| On the other hand, mixtures are different from pure substances. They are called impure substances. They have different characteristics than elements and compounds. |
| They are composed of different elements or compounds mixed together or more than one pure substance that combined together in any composition. |
| When, mixture forms, there is only a little or no energy change. Mixtures are further classified into homogeneous and heterogeneous mixture. |
The organic material, wood is
A)
an element done
clear
B)
a compound done
clear
C)
a mixture done
clear
D)
a solution done
clear
View Solution play_arrow
-
question_answer2)
Which of the following substances is a compound?
A)
Oxygen done
clear
B)
Common salt done
clear
C)
Gold done
clear
D)
Air done
clear
View Solution play_arrow
-
question_answer3)
The substance formed by mixing, crushing and heating iron filings sulphur powder is
A)
an element done
clear
B)
a compound done
clear
C)
a mixture done
clear
D)
a solution done
clear
View Solution play_arrow
-
question_answer4)
Which of the following property does not prove that water is a compound?
A)
Water is made up of two different elements done
clear
B)
Water has fixed boiling point done
clear
C)
The constituents of water cannot be separated by simple physical methods done
clear
D)
Distilled water and tap water have same taste and constituents done
clear
View Solution play_arrow
-
question_answer5)
Blood is considered as:
A)
an element done
clear
B)
a compound done
clear
C)
a mixture done
clear
D)
a solution done
clear
View Solution play_arrow
-
question_answer6)
| Direction: Q. 6 to 10 |
| Read the following and answer questions from [6] to [10]. |
| Abhinav tested the solubility of four different salts, i.e. potassium nitrate, sodium chloride, potassium chloride and ammonium chloride at different temperature. |
| He took four beakers and marked as (I) Potassium nitrate, (II) Sodium chloride, (III) Potassium chloride, (IV) Ammonium chloride and then he dissolved each salt in 100 g of water and placed a thermometer in it. Heated the contents of the beaker and recorded the following observations. |
| Substance dissolved |
Temperature (in K) |
| 283 |
293 |
313 |
333 |
353 |
| Solubility |
| I. Potassium nitrate |
21 |
32 |
62 |
106 |
167 |
| II. Sodium chloride |
36 |
36 |
36 |
37 |
37 |
| III. Potassium chloride |
35 |
35 |
40 |
46 |
54 |
| IV. Ammonium chloride |
24 |
37 |
41 |
55 |
66 |
Solubility of which substance is highest at 283 K?
A)
I done
clear
B)
III done
clear
C)
IV done
clear
D)
II done
clear
View Solution play_arrow
-
question_answer7)
Which substance show maximum change in its solubility, when the temperature is raised from 313 K to 333 K.
A)
I done
clear
B)
II done
clear
C)
III done
clear
D)
IV done
clear
View Solution play_arrow
-
question_answer8)
In the above question, if the amount of water taken is reduced by 20 per cent, what amount of ammonium chloride would be required to prepare its saturated solution at 353 K?
A)
86g done
clear
B)
53g done
clear
C)
50g done
clear
D)
36g done
clear
View Solution play_arrow
-
question_answer9)
What is the effect of temperature on the solubility of a salt?
A)
It increases with decrease in temperature done
clear
B)
It decreases with increase in temperature done
clear
C)
It increases with increase in temperature done
clear
D)
It does not depend on temperature done
clear
View Solution play_arrow
-
question_answer10)
To make lemonade, salts is added in water. The addition of salt in water is a.......
A)
suspension done
clear
B)
colloid done
clear
C)
heterogeneous solution done
clear
D)
homogeneous solution done
clear
View Solution play_arrow
-
question_answer11)
| Direction: Q.11 to 15 |
| Read the following and answer questions from [11] to [15]. |
| The scattering of a beam of light by colloidal particles is called Tyndall effect. |
| Due to scattering of light, the path of the light becomes visible. The source of light may be explored completely. The size of the scattering particles determines the colour of scattered light. The colloidal particles may be seen moving as points of light moving against a black backdrop. |
| Ravi took some amount of substance X and add it into a transparent beaker containing water. He mixed the solution very well and then passed light through this solution by using a torch. The result observed by him is shown below : |
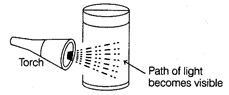 |
| Ravi wants to show this experiment to his younger brother. He kept the solution for 10 minutes and calls his brother and further repeat the experiment, but results are different at this time. The path is not visible. |
What was the reason for change in result?
A)
In first case, the path is visible because of the presence of impurities done
clear
B)
In second case, the particles settle down done
clear
C)
In second case, the concentration increases done
clear
D)
In second case, the impurities dissolve in the solution done
clear
View Solution play_arrow
-
question_answer12)
What is the nature of solution obtained, when X is added to water?
A)
Colloid done
clear
B)
True solution done
clear
C)
Suspension done
clear
D)
Data insufficient done
clear
View Solution play_arrow
-
question_answer13)
Which of the following show Tyndall effect?
A)
Sugar solution done
clear
B)
Salt solution done
clear
C)
Starch solution done
clear
D)
Copper sulphate solution done
clear
View Solution play_arrow
-
question_answer14)
Which of the following is an example of colloidal solution?
A)
Milk done
clear
B)
Urea done
clear
C)
Sugar in water done
clear
D)
Common salt in water done
clear
View Solution play_arrow
-
question_answer15)
When the light passes through the solution in this experiment, then
A)
scattering of light is observed done
clear
B)
path of light becomes visible done
clear
C)
Tyndall effect is observed done
clear
D)
AII of the above done
clear
View Solution play_arrow
-
question_answer16)
| Direction: Q. 16 to 20 |
| Read the following and answer questions from [16] to [20]. |
| A mixture is composed of two or more types of matter that can be present in different amounts. Mixtures are either homogeneous or heterogeneous. Homogeneous mixtures are uniform in composition but heterogeneous mixtures are not uniform in composition. |
| Different methods of separation are used to get individual components from a mixture. |
| Separation makes it possible to study and use the individual components of mixture. Heterogeneous mixtures can be separated into their respective constitutents by simple physical methods like hand picking, sieving filtration that we use in our day-to-day life. |
| Sometimes special techniques have to be used for the separation of the components of a mixture for examples, distillation, centrifugation, chromatography etc. |
The separation of a mixture is done to
A)
maintain equal composition of substance done
clear
B)
obtain a pure sample of a substance done
clear
C)
make it homogeneous mixture done
clear
D)
AII of the above done
clear
View Solution play_arrow
-
question_answer17)
The mixture of ethyl alcohol and water can be separated by
A)
distillation done
clear
B)
centrifugation done
clear
C)
filtration done
clear
D)
chromatography done
clear
View Solution play_arrow
-
question_answer18)
Which of the following technique is used by farmers in village to purify food grains?
A)
Sieving done
clear
B)
Hand picking done
clear
C)
Winnowing done
clear
D)
AII of these done
clear
View Solution play_arrow
-
question_answer19)
The principle of separation based on
A)
the difference in sizes of constituents done
clear
B)
the difference in physical or chemical properties of constituents done
clear
C)
the different techniques done
clear
D)
AII of the above done
clear
View Solution play_arrow
-
question_answer20)
By filtration, which of the following mixture can be separated?
A)
Sugar in water done
clear
B)
Milk in water done
clear
C)
Sand in water done
clear
D)
Oil in water done
clear
View Solution play_arrow
-
question_answer21)
| Direction: Q. 21 to 25 |
| Read the following and answer questions from [21] to [25]. |
| Cooking oil and water both are liquid but their chemical properties are different. They differ in odour and inflammability. Oil bums in air whereas water extinguishes fire. It is the chemical property of oil that makes it different from water. |
| During a chemical change, one substance reacts with other to undergo a change in chemical compositions. A chemical change as also called a chemical reaction. |
| Sometimes both physical and chemical changes take place together. |
| Generally, physical changes do not involve the production of energy while chemical changes usually involve the production of energy. Ecosystems have many chemical and physical changes happening all at once and matter is conserved in each and everyone. In any physical or chemical change, matter does not appear or disappear. |
The chemical change brings change in ....... of matter.
A)
size done
clear
B)
temperature done
clear
C)
phase done
clear
D)
proportions done
clear
View Solution play_arrow
-
question_answer22)
The interconversion of solid, liquid and gas state is a
A)
physical change done
clear
B)
chemical change done
clear
C)
Both [a] and [b] done
clear
D)
no change done
clear
View Solution play_arrow
-
question_answer23)
Burning is a chemical change and burning of candle is
A)
physical change done
clear
B)
chemical change done
clear
C)
Both [a] and [b] done
clear
D)
no change done
clear
View Solution play_arrow
-
question_answer24)
Colour, hardness, melting points, boiling points, odour, etc., are
A)
chemical properties done
clear
B)
physical properties done
clear
C)
Both [a] and [b] done
clear
D)
example of chemical reactions done
clear
View Solution play_arrow
-
question_answer25)
Chemical changes are
A)
temporary and irreversible done
clear
B)
permanent and irreversible done
clear
C)
permanent and reversible done
clear
D)
temporary and reversible done
clear
View Solution play_arrow
-
question_answer26)
| Direction: Q. 26 to 30 |
| Read the following and answer any four questions from [26] to [30]. |
| Different methods of separation are used to get individual components from a mixture. |
| In order to separate components of a mixture, single or a combination of methods are used. |
| The method of selection depends upon the nature of the components present in the mixture. |
| If liquids in a mixture are miscible and have different boiling points, they can be separated by distillation. |
| Distillation involves conversation of a liquid into vapours and then condensing the vapours back into liquid. |
| Distillation is used only if the liquids have a difference in boiling point of more than 25 K. |
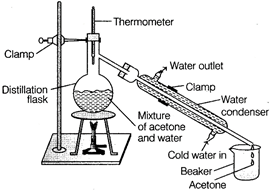 |
Which of the following is not separated by distillation?
A)
Chloroform and water done
clear
B)
Milk and water done
clear
C)
Acetone and ethanol done
clear
D)
Impurities in sea water done
clear
View Solution play_arrow
-
question_answer27)
The distillation is the best technique to separate liquids having different.
A)
Solubility done
clear
B)
Melting points done
clear
C)
Boiling point done
clear
D)
AII of these done
clear
View Solution play_arrow
-
question_answer28)
The residue left in the round bottom flask in the distillation process is a liquid having
A)
high boiling point done
clear
B)
low boiling point done
clear
C)
impurity done
clear
D)
high solubility done
clear
View Solution play_arrow
-
question_answer29)
The observation made from distillation process is
A)
acetone boils first done
clear
B)
water boils first done
clear
C)
impurity evaporates done
clear
D)
water boils at 363 K done
clear
View Solution play_arrow
-
question_answer30)
The distillation process involves the
A)
decomposition and condensation processes done
clear
B)
evaporation and condensation processes done
clear
C)
heating and sublimation processes done
clear
D)
only evaporation process done
clear
View Solution play_arrow
