-
question_answer1)
Different substances are classified as:
| I Pure substance |
| II Homogeneous mixture |
| III Heterogeneous mixture |
A few common substances are listed below.
|
(i) Air,
|
(ii) Graphite,
|
(iii) Gasoline,
|
(iv) Diamond,
|
(v) Tap water,
|
|
(vi) Iron,
|
(vii) Sodium chloride,
|
(viii) Iodised salt,
|
(ix) Brass,
|
(x) Oil and water
|
Which of the following represents the correct classification of the given substances?
A)
B)
C)
D)
None of these represents the correct match. done
clear
View Solution play_arrow
-
question_answer2)
|
Nidhi took two China dishes and marked them as I and II.
|
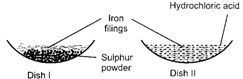
|
|
In China dish I, she mixed iron filings and sulphur powder. In China dish II, she mixed iron filings and dilute hydrochloric acid. What were her observations in both the dishes?
|
A)
In China dish I, a mixture is formed, where iron filings and sulphur powder can be seen separately. done
clear
B)
In China dish II, a new compound is formed, which is different from iron filings and dilute hydrochloric acid. done
clear
C)
A colourless and odourless gas comes out from China dish II. done
clear
D)
All the above observations are correct. done
clear
View Solution play_arrow
-
question_answer3)
Which of the following statements is/are incorrect?
| I Centrifugation method can be used to separate butter from cream. |
| II Coloured component of blue ink can be separated by evaporation. |
| III Chromatography can be used to detect drugs in blood. |
| IV Camphor can be separated from salt by crystallisation. |
A)
II only done
clear
B)
II and III only done
clear
C)
IV only done
clear
D)
II and IV only done
clear
View Solution play_arrow
-
question_answer4)
The boiling points of a few gases found in air are given below:
| Element | Krypton | Neon | Nitrogen | Oxygen |
| Boiling point \[\mathbf{({}^\circ C)}\] | -152 | -246 | -196 | -183 |
If liquid mixture is fractionally distilled, the order of gases distilling out is
A)
Krypton, Neon, Nitrogen, Oxygen done
clear
B)
Neon, Nitrogen, Oxygen, Krypton done
clear
C)
Nitrogen, Neon, Oxygen, Krypton done
clear
D)
Oxygen, Neon, Nitrogen, Krypton. done
clear
View Solution play_arrow
-
question_answer5)
|
Soham, a class 9 student mixed some iron filings and sulphur in a China dish and heated them strongly to obtain a residue.
|
|
Which of the following is not a characteristic property of the residue?
|
A)
It cannot be separated into sulphur and iron filings by normal physical methods. done
clear
B)
Its properties are completely different from iron and sulphur. done
clear
C)
Its composition is same throughout. done
clear
D)
Its appearance is different from iron and sulphur but it shows chemical properties of both iron and sulphur. done
clear
View Solution play_arrow
-
question_answer6)
Sheela heated a mixture of iodine and common salt by keeping an inverted funnel on it. After some time, she observed that
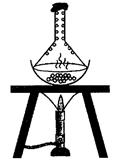
A)
White fumes come out from the mixture done
clear
B)
Violet particles deposit on the neck of the funnel done
clear
C)
White particles deposit on the neck of the funnel done
clear
D)
A gas with popping sound comes out. done
clear
View Solution play_arrow
-
question_answer7)
Geetika poured 20 g of salt into 200 mL of water in a beaker. She stirred the water to dissolve the salt completely. Then she heated the solution until it was reduced to half. How many grams of salt can be recovered from the remaining solution?
A)
\[\text{0 g}\] done
clear
B)
\[\text{10 g}\] done
clear
C)
\[\text{20 g}\] done
clear
D)
\[\text{40 g}\] done
clear
View Solution play_arrow
-
question_answer8)
|
The given chromatogram was obtained for proteins extracted from soya milk.
|

|
|
The components present in protein extract are
|
A)
W and X done
clear
B)
X and Y done
clear
C)
W and Y done
clear
D)
Y and Z done
clear
View Solution play_arrow
-
question_answer9)
|
Direction: Read the given passage carefully and answer the following questions.
|
|
The constituents of a heterogeneous mixture are X, Y and Z. If mixture containing X and Y is taken, X can be separated using magnetic separation from Y. If mixture containing Y and Z is taken, the two can be separated using evaporation method.
|
The different states of X, Y and Z respectively are
A)
Solid, Solid, Liquid done
clear
B)
Solid, Liquid, Solid done
clear
C)
All are liquids done
clear
D)
All are solids. done
clear
View Solution play_arrow
-
question_answer10)
|
Direction: Read the given passage carefully and answer the following questions.
|
|
The constituents of a heterogeneous mixture are X, Y and Z. If mixture containing X and Y is taken, X can be separated using magnetic separation from Y. If mixture containing Y and Z is taken, the two can be separated using evaporation method.
|
Which of the following identifies X, Y and Z correctly?
A)
Iron, salt, water done
clear
B)
Sulphur, carbon disulphide, water done
clear
C)
Gun powder, common salt, water done
clear
D)
Iodine, sand, alcohol done
clear
View Solution play_arrow
-
question_answer11)
What is the function of X in the given experimental set-up?
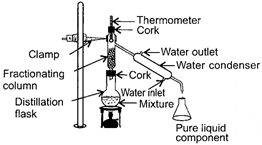
A)
To apply pressure to the mixture in distillation flask done
clear
B)
To allow only lower boiling component to escape done
clear
C)
To cool and condense the vapours done
clear
D)
To allow the water from the mixture to move out done
clear
View Solution play_arrow
-
question_answer12)
Sahil took a white solid from his teacher to find out its melting point. During the experiment, he found that the melting point was \[\text{160 }\!\!{}^\circ\!\!\text{ C}\]. But, the teacher told him that actual melting point of the solid is \[\text{150 }\!\!{}^\circ\!\!\text{ C}\]. What could be the reason for Sahil's observation?
A)
Melting point of a solid depends upon the procedure and the amount taken. done
clear
B)
Solids can have different melting points depending upon the method of storage. done
clear
C)
The solid contains some impurities mixed with the pure compound. done
clear
D)
If solid is heated strongly, the melting point increases. done
clear
View Solution play_arrow
-
question_answer13)
Natural water is impure and needs to be purified before this water can be supplied to homes for drinking purposes. The sequence of steps involved in purification of water is
A)
Filtration, sedimentation, decantation and aeration done
clear
B)
Sedimentation, loading, filtration and chlorination done
clear
C)
Chlorination, aeration, sedimentation and filtration done
clear
D)
Chlorination, filtration and distillation. done
clear
View Solution play_arrow
-
question_answer14)
Ms Mehta, a science teacher gave different mixtures to four groups of students to separate their components. Which group was not following the correct method?
A)
Group 1 was separating a mixture of ethyl alcohol and water by using separating funnel. done
clear
B)
Group 2 was separating a mixture of ammonium chloride and sodium chloride using sublimation. done
clear
C)
Group 3 was separating a mixture of iron pins and sand by using a magnet. done
clear
D)
Group 4 was separating mud particles suspended in water using sedimentation and decantation. done
clear
View Solution play_arrow
-
question_answer15)
|
Read the given passage and fill in the blanks by choosing an appropriate option.
|
|
When a mixture of two ___i___ liquids having a difference of \[25{}^\circ C\] in their boiling points is to be separated, a ___ii___ is used which increases the ___iii___ surface area. As a result, when the vapours of both the liquids rise, the ___iv___ volatile liquid will condense while the vapours of ___v___ volatile liquid will rise.
|
A)
B)
C)
D)
View Solution play_arrow
-
question_answer16)
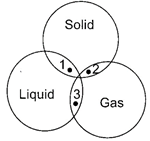
|
Study the given Venn diagram carefully.
|
|
|
Which of the following colloidal systems correctly represent points 1, 2 and 3?
|
A)
B)
C)
D)
View Solution play_arrow
-
question_answer17)
Match column I with column II and select the correct option from the codes given below.
| Column I | Column II |
| (a) Sublimation | (i) Separation of water and \[\text{CC}{{\text{l}}_{4}}\] |
| (b) Magnetic separation | (ii) Separation of \[\text{NaCl}\] from \[\text{KN}{{\text{O}}_{3}}\] |
| (c) Distillation | (iii) Separation of benzene and aniline |
| (d) Separating funnel | (iv) Separation of iron and sand |
| (e) Fractional Crystallisation | (v) Separation of iodine and sand |
A)
(a)-(i), (b)-(ii), (c)-(iii), (d)-(iv), (e)-(v) done
clear
B)
(a)-(v), (b)-(iv), (c)-(iii), (d)-(i), (e)-(ii) done
clear
C)
(a)-(iv), (b)-(v), (c)-(ii), (d)-(i), (e)-(iii) done
clear
D)
(a)-(i), (b)-(ii), (c)-(iv), (d)-(v), (e)-(iii) done
clear
View Solution play_arrow
-
question_answer18)
Study the given flow chart and choose the correct statements.
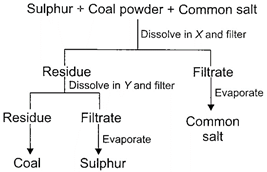
| (i) The property used to separate the mixture is solubility of different components in different solvents. |
| (ii) Sulphur is soluble in carbon disulphide, salt in water and coal is insoluble in both. |
| (iii) X is water and Y is carbon disulphide. |
A)
(i) and (ii) only done
clear
B)
(ii) and (iii) only done
clear
C)
(i) and (iii) only done
clear
D)
All are correct. done
clear
View Solution play_arrow
-
question_answer19)
Siddharth dissolved a mixture into water and then filtered it. Solid X was left behind as residue in the filter paper and solid Y was obtained by evaporating the filtrate to dryness. X and Y could be respectively
A)
Sodium hydroxide and calcium carbonate done
clear
B)
Calcium chloride and sodium carbonate done
clear
C)
Calcium carbonate and sodium chloride done
clear
D)
Sodium carbonate and calcium chloride. done
clear
View Solution play_arrow
-
question_answer20)
Which of the following statements about the given experimental set-up is/are incorrect?
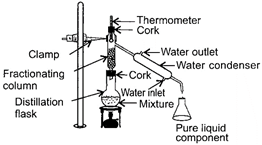
| I. It is used to separate a mixture of two or more miscible liquids for which the difference in boiling points is more than 25 K. |
| II. It is used for the separation of different gases from air. |
| III. It is used to separate acetone and water from their mixture. |
| IV. It is based on the principle of repeated cooling and condensation of vapours by providing surface for the vapours. |
A)
I only done
clear
B)
III and IV only done
clear
C)
I and II only done
clear
D)
I and III only done
clear
View Solution play_arrow

